Abstract
Bovine viral diarrhea virus (BVDV) is an emerging infectious pathogen of concern to the alpaca industry. A 4-month-old, intact, male alpaca cria was diagnosed as persistently infected with BVDV on the basis of repeated positive antemortem polymerase chain reaction (PCR) and virus isolation (VI) assays and negative serologic titers to BVDV. Immunohistochemistry, real-time reverse transcription PCR, and VI performed on tissues collected at necropsy demonstrated disseminated BVDV-1b infection. Virus was detected in multiple tissues, including parotid salivary gland, testes, prostate, kidneys, skin, and gastrointestinal tract. Demonstration of BVDV in previously unreported tissues suggests additional potential routes of BVDV transmission in alpacas.
Keywords
A 4-month-old, intact, male alpaca (Vicugna pacos) cria presented to the Washington State University Veterinary Teaching Hospital (Pullman, WA) with a history of decreased weight gain, ill thrift, and anorexia. The cria was from a herd of 45 animals and was diagnosed as persistently infected (PI) with Bovine viral diarrhea virus type 1 (BVDV-1) on the basis of 3 repeated positive tests by polymerase chain reaction (PCR) and virus isolation on ethylenediamine tetra-acetic acid (EDTA) blood samples obtained at 3, 4, and 5 months of age and negative serologic titers to BVDV on serum samples. On presentation, the cria was bright, alert, and responsive with normal temperature, pulse, and respiration. The cria was deemed to be significantly underweight (13.6 kg) compared with his herd mates. All other findings on physical examination were within normal limits. Complete blood cell count revealed a mild left shift (band neutrophils: 309 neutrophils/μl, reference [ref.] range: <200 neutrophils/μl), high normal fibrinogen (400 mg/dl, ref. range: 100–400 mg/dl), anemia (PCV 21%, ref. range: 27–45%), decreased hemoglobin (9.4 g/dl, ref. range: 11.9–19.4 g/dl), and marked anisocytosis. Serum biochemical profile was within normal limits.
Therapy with tulathromycin (5.5 mg/kg intramuscularly) and a nutritional supplement had been initiated approximately 1 week earlier because of a suspected upper respiratory tract infection. Because of the grave prognosis due to PI status and general poor health of the cria, as well as financial considerations, the owners declined further antemortem diagnostic tests or treatment and elected to have the cria hospitalized for temporary monitoring. Over the next 8 days, the cria was observed to be ambulating, eating, drinking, urinating, and defecating normally. Florfenicol therapy (22 mg/kg, subcutaneous, q48hr) was initiated on day 4 because of a suspected respiratory infection. On the morning of day 9, the cria was found dead in its stall and transferred to the Washington Animal Disease Diagnostic Laboratory (Pullman, WA) for complete postmortem examination.
Gross postmortem examination revealed acute, locally extensive bronchopneumonia of the cranial lung lobes; caudal lung lobes were mottled red to pink and moderately firm. Several well-delineated (0.5–1.5 cm in diameter) ulcers were observed on the mucosal surface of gastric compartments 1 (C1), 2 (C2), and 3 (C3). Numerous petechiae were noted on the surface of the spleen. No other gross lesions were identified. Tissues were collected in 10% neutral buffered formalin, routinely processed, and embedded in paraffin blocks. From the paraffin blocks, 4-μm sections were cut and stained with hematoxylin and eosin. Lung, liver, and a tracheal swab submitted for aerobic culture revealed mixed bacterial growth; similar samples were negative for Mycoplasma culture.
Histologic examination of tissue from the cranial lung lobes revealed moderate, multifocal, suppurative broncho-pneumonia. Sections from the remaining lung lobes were characterized by patchy infiltrates of minimal to low numbers of lymphocytes, plasma cells, and rare neutrophils within the interlobular and alveolar septa. Gastric compartments 1 and 2 had multifocal gastritis, characterized by a mild to moderate infiltration of the lamina propria by variable admixtures of lymphocytes, plasma cells, eosinophils, neutrophils, and macrophages. No fungi were identified. In several sections of both large and small intestine, the lamina propria was mildly to moderately expanded by neutrophils, lymphocytes, and plasma cells. The liver exhibited moderate, multifocal, random hepatocellular necrosis consistent with bacteremia. Submandibular, inguinal, and mesenteric lymph nodes had mildly to markedly reduced numbers of lymphocytes within the cortex and medulla; mesenteric lymph nodes were most severely affected. The cortex and medulla of the thymic lobules and the white pulp of the spleen had moderately to severely reduced lymphocyte numbers; the thymic corticomedullary junction was indistinct.
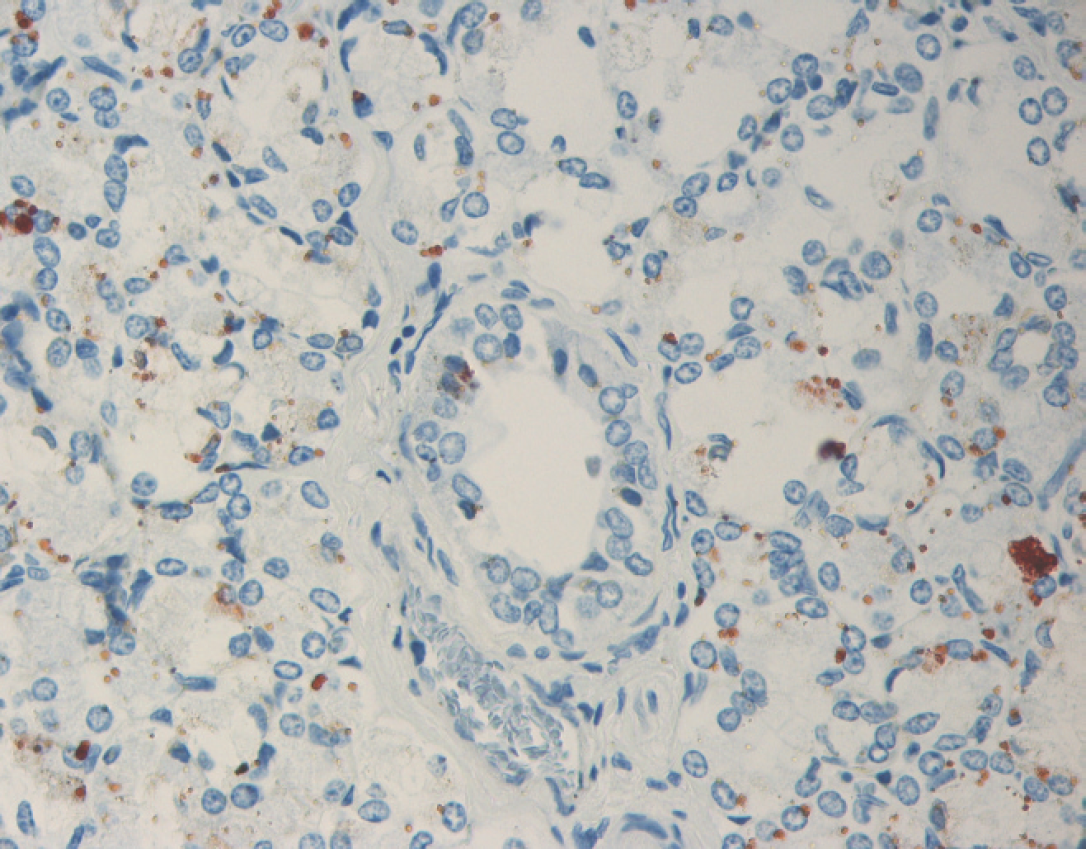
Parotid salivary gland with immunohistochemical staining for Bovine viral diarrhea virus (BVDV) antigen. BVDV immunoreactivity within the cytoplasm of acinar epithelial cells and rare ductular epithelial cells.
Immunohistochemical detection of BVDV antigen was performed on sections of formalin-fixed, paraffin-embedded tissues with the use of monoclonal antibody 15c5 a at a 1:1,000 dilution using a streptavidin-biotin-immunoperoxi-dase b technique, with diaminobenzidine b as a chromogen. 11 Large amounts of BVDV antigen were detected in the following tissues: parotid salivary gland, testis, prostate, esophagus, C1, C2, C3, right kidney, bone marrow, liver, lung, spleen, thymus, and the mesenteric and submandibular lymph nodes. In sections of parotid salivary gland, intracytoplasmic immunoreactivity was strong within acinar epithelial cells and occasional ductular epithelial cells (Fig. 1). Similarly, a section of testis revealed frequent cells within the seminiferous tubules that had strong intracytoplasmic immunoreactivity for BVDV antigen (Fig. 2). Scattered groups of epithelial cells within the convoluted tubules of the kidney had mild to moderate intracytoplasmic immunoreactivity for BVDV, whereas rare macrophages within the interstitium had intracytoplasmic immunoreactivity for BVDV. Within the esophagus and C1, BVDV had immunoreactivity within the cytoplasm of basaloid epithelial cells. Compartment 2 had immunoreactivity of both the basaloid epithelial cells within the areas of stratified squamous epithelium and within the glandular epithelium of the saccules. In C3, frequent areas were immunoreactive within the cytoplasm of glandular epithelial cells and within some of the gastric ulcers. Within the lung, thymus (Fig. 3), spleen, and lymph nodes, rare to frequent cells (presumptive macrophages) had intracytoplasmic immunoreactivity for BVDV antigen, whereas scattered Kupffer cells within the liver had similar immunoreactivity. Frequent myeloid precursor cells and presumptive macrophages within the bone marrow had similar intracytoplasmic immunoreactivity for BVDV antigen.
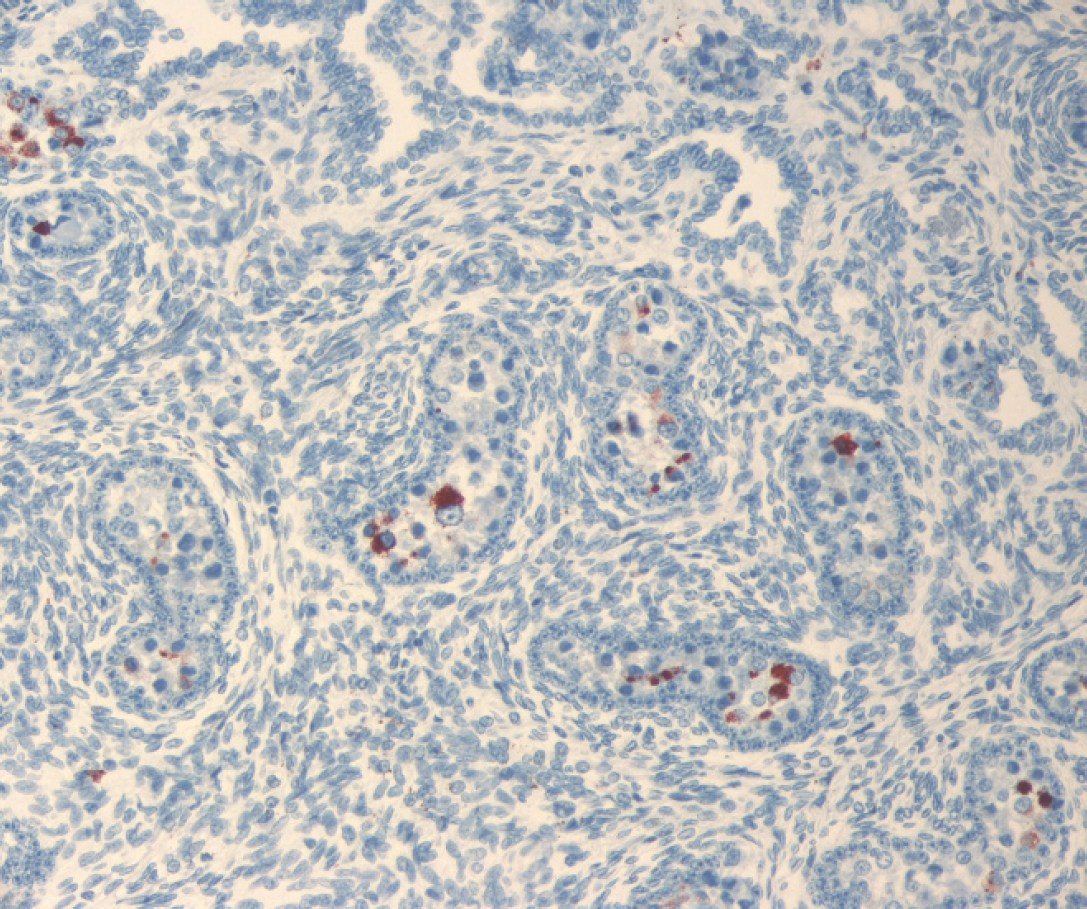
Immature testis with immunohistochemical staining for Bovine viral diarrhea virus (BVDV) antigen. BVDV immunoreactivity within the cytoplasm of cells of the seminiferous tubules.
Real-time reverse transcription PCR (real-time RT-PCR) to detect and subtype BVDV in tissues was performed according to a duplex TaqMan real-time PCR procedure as previously described. 3 After extraction of total RNA with TRIzol c , BVDV-1 was detected in kidney, liver, spleen, lung, and thymus. Bovine viral diarrhea virus was isolated from the same tissues in BVDV-free bovine turbinate cells. 7 Bovine viral diarrhea virus RNA was extracted from the supernatants of virus-infected cells with the use of a commercial RNA extraction kit d and detected via TaqMan real-time PCR procedure as previously described. 3 To obtain a longer amplicon for sequencing, a separate RT-PCR reaction was run with the use of recombinant Thermus thermophilus (rTth) DNA polymerase e to amplify a highly conserved 290-bp portion of the 5′ untranslated region (5′ UTR) of the BVDV genome as previously described. 20 Polymerase chain reaction amplicons were visualized on a 1.5% agarose gel containing ethidium bromide, excised with a sterile scalpel blade under ultraviolet illumination, and purified with the use of a nucleic acid purification kit f according to the manufacturer's directions. Amplicon DNA was sequenced directly on both strands by a local vendor g using a commercial sequencing kit, h with analysis on a DNA sequencer. e Sequencing reactions were done in duplicate, and sequences were confirmed by sequencing of both strands. Forward and reverse sequences were aligned by the ClustalW algorithm. 17 Each sequence was compared with the GenBank nucleotide sequence database for similarity with BLASTn (nucleotide Basic Local Alignment Search Tool; http://www.ncbi.nlm.nih.gov/blast/Blast.cgi). The sequence most closely matched that of BVDV-1b (98% sequence identity to GenBank AY159530 and other type 1b accessions) when compared with sequences in GenBank.
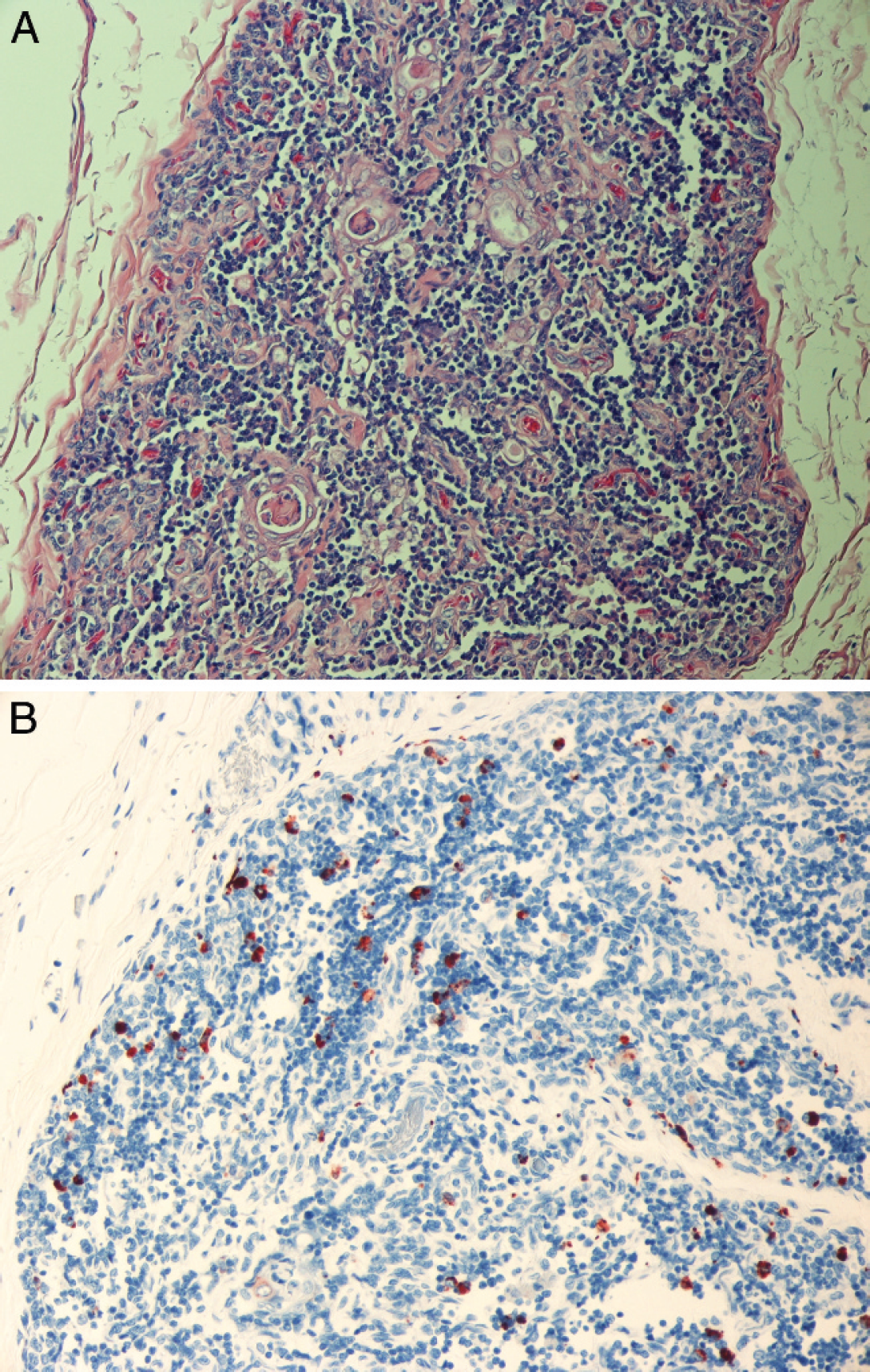
Hematoxylin and eosin–stained section of thymus (
Bovine viral diarrhea virus is considered to be primarily an infectious pathogen in cattle, but other even-toed ungulates, including camelids, have been reported to be susceptible. 1 Early studies in camelids suggested either resistance or decreased ability to transmit BVDV because of low seroprevalence, minimal clinical disease, and lack of pathology after experimental infections. 14,21 However, current evidence suggests that BVDV might be an emerging and significant disease of alpacas. 2,5,8,10,15 The recent increase in cases within alpacas is likely multifactorial and might involve the emergence of a novel viral strain, differences in animal management practices, or increased awareness of BVDV by alpaca owners. The BVDV genome is known to be dynamic, with replication of any isolate giving rise to a viral swarm until a predominant strain emerges, and one of these could have an increased predilection for alpacas. 6,15 Preliminary research indicates the recent BVDV infections in alpacas have been primarily noncytopathic BVDV-1b strain. 2,5,8,10
Research on PI bovine fetuses and calves has identified virus in all tissues and shedding occurring from multiple sites. 4,9 Previous reports on 4 confirmed PI alpacas identified virus in various organs but did not mention reproductive tissues or salivary gland. 5,8,15 Unlike those cases, the cria in this report had disseminated BVDV infections that included parotid salivary gland, esophagus, C1–C3, testes, prostate, and kidneys. Identification of virus in these organs indicates potential routes for transmission through communicative and reproductive behaviors. Alpacas spit as a communication tool within the herd, and breeders frequently use the behavior to evaluate sexual receptivity of the female. 19 Breeding involves cervical penetration by the penis and deposition of semen deep inside both uterine horns. The uterine trauma occurring during the breeding process may increase the risk of BVDV transmission. 18
Bovine viral diarrhea virus infection in calves is also associated with “weak calf syndrome” and “shipping fever,” in part because of viral immunosuppressive effects. Clinically, poor growth and recurrent infection in the animal in this study mimicked that seen with many PI calves. Similar signs have also been reported in other PI alpacas. 5,8,15 The similarities between the disease in cattle and alpacas suggest that the viral pathogenesis could be comparable.
The fetal outcome of BVDV infection in naive, pregnant cattle is variable and dependent on the stage of gestation at which infection occurs. It is believed that cattle become persistently infected when fetal infection occurs before immunologic development, and viral proteins are recognized as “self antigens” and therefore tolerated. 13 The gestational exposure time for BVDV immunotolerance in alpacas has not been fully identified but appears to occur in the first trimester. The dam of the PI cria in this study was exposed to a suspected nonclinical PI or transiently infected alpaca while located at the breeding facility during the first 90 days of her pregnancy. It was unknown whether the female was exposed before pregnancy or continually during this time period. The animal was not exposed to cattle during her pregnancy.
Research in the cattle industry has identified fetal infection rates of over 10%, even in vaccinated herds, with the rate of PI calves approximately 0.5%. 16 Fetal infection rates are suspected to be lower within the alpaca industry because of the lower population and the recent emergence of the virus within the species; however, because potential PI crias are typically euthanatized to prevent herd exposure and limit farm reputation damage, this cannot be confirmed.
The alpaca cria in this case had a disseminated BVDV infection, which included the reproductive, salivary, and upper gastrointestinal tract tissues. These findings suggest a significant risk for BVDV transmission because of alpaca behavior and management practices. Subclinical to acute infections have been estimated to cost the cattle industry $50 to $400 per head, for all animals in the herd, not just those suffering disease. 12 With the current value of many female alpacas beginning at approximately $10,000 per animal, the potential economic losses for alpaca producers because of BVDV are significant; therefore, continued testing and biosecurity efforts are warranted.
Footnotes
a.
IDEXX MoAB 15c-5 (anti-BVDV EO), IDEXX Laboratories, Westbrook, ME.
b.
LSAB™2 Kit, Dako North America Inc., Carpinteria, CA.
c.
Invitrogen Corp., Carlsbad, CA.
d.
QIAamp® Viral RNA Mini Kit, Qiagen Inc., Valencia, CA.
e.
Applied Biosystems, Foster City, CA.
f.
Bio-Rad Freeze 'N Squeeze™ Kit, Bio-Rad Laboratories, Hercules, CA.
g.
Amplicon Express Inc., Pullman, WA.
h.
Amersham DYEnamic ET Terminator Cycle Sequencing Kit, Amersham Biosciences, Piscataway, NJ.
