Abstract
In the United States, tuberculosis of captive cervids, caused by Mycobacterium bovis, attracted attention in 1991 when investigations, prompted by the identification of a tuberculous elk (Cervus elaphus nelsoni) of U.S. origin exported to Canada, revealed tuberculosis in 10 different elk herds in 8 different states. Based on methods used in cattle, official regulations pertaining to testing and eradication of tuberculosis in captive cervids were added to the U.S. Department of Agriculture bovine tuberculosis eradication effort in 1994. However, little published information exists on the accuracy of intradermal tuberculin testing in naturally infected cervids. Evaluation of a captive herd of 71 animals in Wisconsin included postmortem examination and tissue sample collection from both tuberculin test responders and nonresponders. Within this captive herd, of admittedly small size, results showed the single cervical test to have a sensitivity of 88% and a specificity of 69%. Evaluation of diagnostic tests in the species of interest is important, as extrapolation of data obtained from other species may not be appropriate.
In the United States, tuberculosis of captive cervids, caused by Mycobacterium bovis, attracted attention in 1991 when investigations were prompted by the identification of a tuberculous elk (Cervus elaphus nelsoni) in Canada that was imported from the United States in 1988. Testing of U.S. elk herds with links to the Canadian elk revealed tuberculosis in 10 different herds in 8 different states. 4 The discovery of tuberculosis in captive elk in the United States resulted in the addition of captive cervids to the 1994 U.S. Department of Agriculture (USDA) uniform methods and rules (UMR) for eradication of bovine tuberculosis. 21
Summary results of single cervical intradermal tuberculin test (SCT) and comparative cervical test (CCT) testing of an elk herd (n = 71) naturally infected with Mycobacterium bovis. *
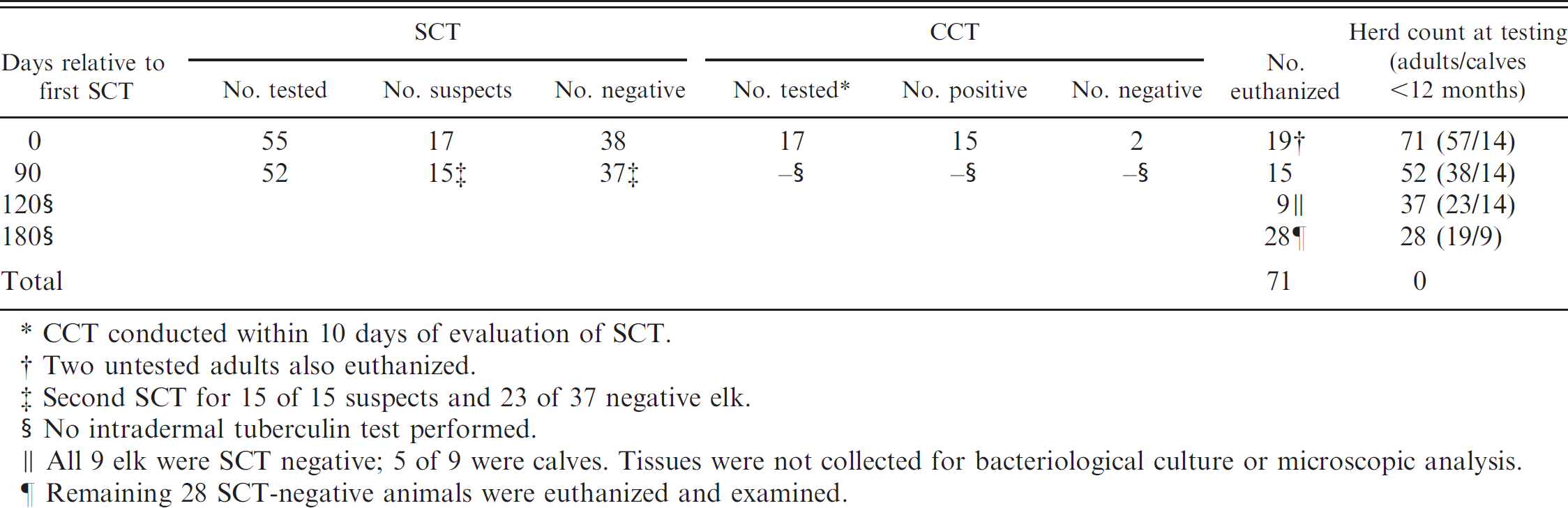
CCT conducted within 10 days of evaluation of SCT.
Two untested adults also euthanized.
Second SCT for 15 of 15 suspects and 23 of 37 negative elk.
No intradermal tuberculin test performed.
All 9 elk were SCT negative; 5 of 9 were calves. Tissues were not collected for bacteriological culture or microscopic analysis.
Remaining 28 SCT-negative animals were euthanized and examined.
Antemortem diagnosis of tuberculosis relies on measurement of delayed type hypersensitivity (DTH) by intradermal injection of mycobacterial extracts, the most common of which is purified protein derivative (PPD). Intradermal injection of M. bovis PPD in the caudal fold region at the base of the tail became the accepted official test for cattle in the United States in 1920. 20 In New Zealand, an early leader in deer farming as an industry, investigation of intradermal tuberculin testing of cervids showed the caudal fold test, as performed in cattle, to be unreliable in red deer (Cervus elaphus). 8 In red deer, caudal fold testing had a sensitivity of approximately 21%. 7 Testing in the lateral neck region proved more reliable, and in 1979, the cervical intradermal tuberculin test replaced the caudal fold test for cervids in New Zealand. 1 Presently, in the United States, only the cervical intradermal tuberculin test and the blood tuberculosis test are approved by the USDA for antemortem diagnosis of tuberculosis in cervids. 21 However, the blood tuberculosis test is no longer available in the United States.
Cervids in a herd of unknown infection status are tested by the single cervical intradermal tuberculin test (SCT), and results are categorized as negative or suspect. Suspected cases may be reevaluated using the comparative cervical tuberculin test (CCT). The CCT requires injection of PPD from M. bovis (bovine PPD) and Mycobacterium avium (avian PPD) at 2 different sites on the lateral neck. The addition of avian PPD increases specificity of the CCT as compared with the SCT. Cervids infected with nontuberculous mycobacteria may be suspects using the SCT; however, by using the CCT, these cervids can often be identified due to a greater change in skin thickness at the avian PPD injection site compared with the change in skin thickness at the bovine PPD injection site. Results of the CCT are then used to categorize SCT suspects as negative, suspect, or reactor.
In New Zealand, the sensitivity of the SCT in naturally or experimentally infected red deer has been reported to be 82% and 86%, respectively. 3,6 Specificity has varied from 46% to 76% in herds of tuberculosis-negative red deer. 11 Little published information exists as to the sensitivity or specificity of intradermal tuberculin testing of cervids in the United States. The USDA reports a combined herd specificity of the SCT in all species of cervids between 71% and 86%. 11 Sensitivity has not been reported since SCT-negative animals are rarely euthanized and examined; thus, the true infection status of these animals is not known. 11 In the United States, where numerous species of cervids are raised for commercial purposes, few studies have investigated the accuracy of intradermal tuberculin testing in selected species of the family Cervidae. The objective of the current study was to evaluate the SCT in a herd of naturally infected elk, in which both SCT suspects and nonresponding negative elk would be examined.
As part of a statewide testing program, the SCT was performed on elk from 3 different captive elk herds in Wisconsin. The combined population of the 3 herds was 71 elk, including 14 calves too young to be tested (<12 months of age). Because of the close proximity and sharing of animals and facilities among the 3 herds, elk were grouped together and considered a single herd for testing purposes. Fifty-five of 71 elk were first tested. Seventeen of the 55 elk tested by the SCT were classified as suspect and retested with the CCT within 10 days of the SCT (Table 1). Fifteen of the 17 suspect elk were categorized as reactors by the CCT; however, all 17 elk were euthanized. Two untested elk were also euthanized, for a total of 19 elk examined. Tissues were collected for microscopic analysis and bacteriological isolation of M. bovis as described previously. 15 Mycobacterium bovis was isolated, and histocompatible lesions (granulomas with intralesional acid-fast bacteria) were seen in 14 of 15 CCT reactor elk and 1 CCT-negative elk.
Three months later, the remaining 52 elk, including 14 calves not previously tested, now >12 months of age, were tested using the SCT. Fifteen of the remaining 38 elk tested were categorized as reactors by the SCT and were euthanized (UMR guidelines allow the use of the SCT as a single and confirmatory test in herds known to be infected with M. bovis). All 15 elk had tested negative by SCT 3 months earlier. Mycobacterium bovis was isolated, and histocompatible lesions were seen in 7 of 15 reactors. Over the next 3 months, 9 of the remaining 37 elk were euthanized; however, samples were not collected due to logistical complications. Six months after initial testing, the remaining 28 elk, all of which had tested negative by SCT, were euthanized and examined. Two of 28 elk had histocompatible lesions, and M. bovis was isolated from a third elk. Polymerase chain reaction (PCR) of formalin-fixed sections, as described previously, 10 amplified a product specific for members of the Mycobacterium tuberculosis complex from both histocompatible lesions. At no time were any of the calves categorized as reactors.
Summary of single cervical intradermal tuberculin test (SCT) results and infection status.
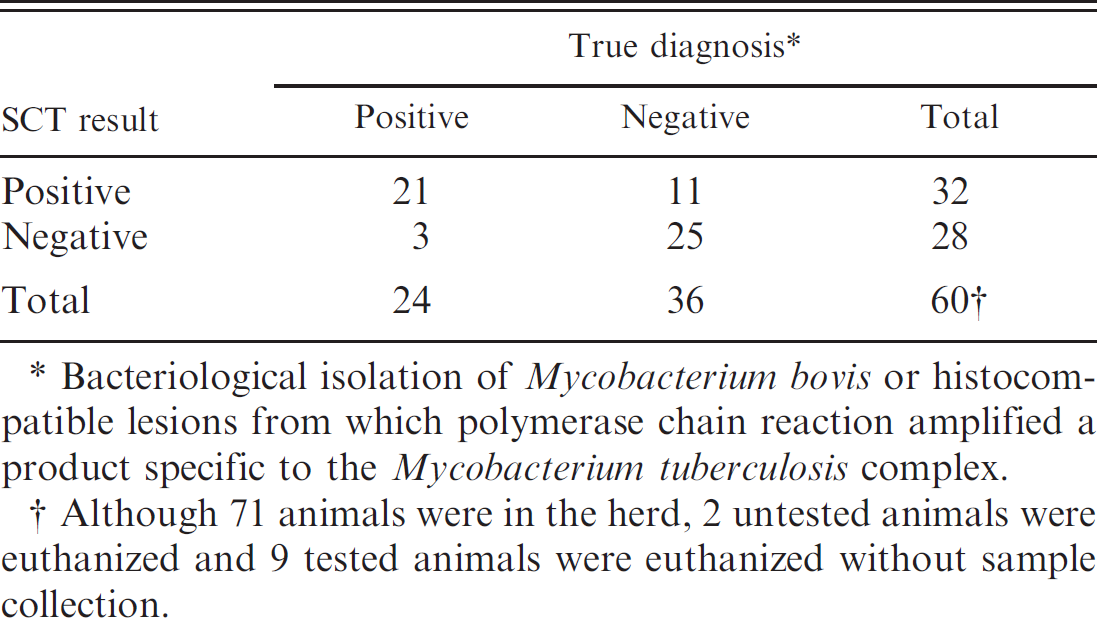
Bacteriological isolation of Mycobacterium bovis or histocompatible lesions from which polymerase chain reaction amplified a product specific to the Mycobacterium tuberculosis complex.
Although 71 animals were in the herd, 2 untested animals were euthanized and 9 tested animals were euthanized without sample collection.
In total, 107 SCTs were conducted on 69 different elk. Samples were collected for analysis from 60 of 69 elk. Of 24 elk confirmed to be tuberculous by bacteriological culture or histopathology and PCR, 21 (88%) were SCT positive (Table 1). Thirty-eight of 69 elk were tested twice by the SCT. Fifteen elk that were negative on the first SCT were found to be SCT positive 3 months later. Seven of these 15 (47%) elk were confirmed to be tuberculous by bacteriological culture or histopathology and PCR. Reasons for conversion of SCT status from negative to positive over the 3-month interval could be due to an early state of disease. In experimental infection studies using cattle, DTH responses, as measured by intradermal tuberculin testing, do not develop for 20–30 days after inoculation. 5 Likewise, gross lesions are not present until approximately 28 days after inoculation. 14 Other reasons for false-negative results may include general animal health status or human error. Repeated skin testing, over short time intervals, is known to suppress DTH responses to tuberculin in cattle. 2 It is unclear if similar desensitization occurs in cervids. Previous skin testing of this herd occurred more than 12 months earlier, when all tests were interpreted as negative.
Mycobacterium bovis could not be isolated, and histocompatible lesions were not seen in 25 of 36 (69%) elk that tested negative by SCT. Correspondingly, 11 elk that tested SCT positive could not be confirmed as tuberculous by bacteriological culture, histopathology, or PCR. Although true false-positives are expected with any test displaying less than 100% specificity, exposure to nontuberculous mycobacteria can also result in a DTH response to PPD. Deer species are exposed to and colonized by numerous nontuberculous mycobacterial species. Indeed, in the present report, Mycobacterium fortuitum and M. avium were isolated from 3 of 11 and 1 of 11 false-positive SCT reactors, respectively. A previous study using white-tailed deer (Odocoileus virginianus) showed that careful bacteriological isolation techniques often recover numerous nontuberculous species of mycobacteria. 13
Gross and microscopic lesions of tuberculosis in the elk herd in the present study were similar to those described previously in captive elk. 19 Gross lesions were most common in the medial retropharyngeal lymph node (16/21, 76%). Tracheobronchial or mediastinal lymph node lesions were seen in 3 of 21 (14%) elk. Gross lesions were also seen in the mandibular lymph node (1) and tonsil (1), whereas generalized tuberculosis involving tissues in the head, thorax, and abdomen was seen in 3 of 21 elk. The nature of gross lesions ranged from abscess-like lesions with liquefactive necrosis to gritty, partially mineralized granulomas with caseous necrosis.
Microscopically, lesions were similar to those previously described in elk and red deer. 18 However, central but not peripheral mineralization 18 was present, and unlike lesions in cattle, circumferential fibrosis was minimal. 16 Acid-fast bacteria, as identified by Ziehl–Neelsen staining, were infrequent. Individual lesions often contained only one to several acid-fast bacteria, most often present within the necrotic caseum.
Although using terms such as test sensitivity and specificity with a small sample size, such as that provided by the herd in the present study, is vulnerable to criticism, for comparative purposes it could be concluded that in this herd, the sensitivity of the SCT was 88% and the specificity 69% (Table 2). Sensitivity and specificity of the SCT in this herd is similar to that which has been reported in naturally infected red deer in New Zealand. However, the specificity of the SCT in this herd is lower than that reported in experimentally infected red deer or other deer species. 3,6,9,11,17
It is important to evaluate diagnostic tests, such as intradermal tuberculin testing in the species of interest, as extrapolation of results obtained in other species may not be appropriate. The application and interpretation of intradermal tuberculin testing in cervids is heavily influenced by regulations governing intradermal tuberculin testing of cattle. Assumptions are sometimes made using data obtained from cattle or other species of the Cervidae family. However, it is known that species differences in immune function exist. Indeed, studies in elk, examining immune responses to intracellular pathogens such as Brucella abortus, demonstrate a response biased toward antibody production, while cell-mediated responses are weak compared with other ruminants. 12 This may explain the failure in elk of some brucella vaccines that are effective in cattle or bison. 12 Likewise, species, as well as individual, animal differences in cell-mediated immune response to mitogens have been described in white-tailed deer, elk, fallow deer (Dama dama), and reindeer (Rangifer tarandus). 22 It is important to evaluate diagnostic tests such as intradermal tuberculin testing in the species of interest and to recognize that a single set of rules or methods may not be appropriate for all species.
