Abstract
A real-time reverse transcription polymerase chain reaction test for the identification of the H7 subtype in North American Avian influenza viruses (AIVs) was first reported in 2002; however, recent AIV surveillance efforts in wild birds and H7 outbreaks in poultry demonstrated that the 2002 test did not detect all H7 AIVs present in North and South America. Therefore, a new test, the 2008 Pan-American H7 test, was developed by using recently available H7 nucleotide sequences. The analytical specificity of the new assay was characterized with an RNA panel composed of 19 H7 viruses from around the world and RNA from all hemagglutinin subtypes except H16. Specificity for North and South American lineage H7 viruses was observed. Assay limits of detection were determined to be between 103 and 104 gene copies per reaction with in vitro transcribed RNA, and 100.0 and 100.8 50% egg infectious doses per reaction. The 2008 Pan-American H7 test also was shown to perform similarly to the 2002 test with specimens from chickens experimentally exposed to A/Chicken/BritishColumbia/314514-2/04 H7N3 highly pathogenic AIV. Furthermore, the 2008 test was able to detect 100% (n = 27) of the H7 AIV isolates recovered from North American wild birds in a 2006–2007 sample set (none of which were detected by the 2002 H7 test).
Keywords
Real-time reverse transcription polymerase chain reaction (real-time RT-PCR) is frequently used to test type A influenza positive avian-origin specimens for the H5 and H7 hemagglutinin (HA) subtypes because of regulatory recommendations. One of the most widely used tests for H7 HA subtype identification in the Americas is the validated United States Department of Agriculture, National Animal Health Laboratory Network (USDA-NAHLN) H7 HA subtype real-time RT-PCR test. 8 The USDA-NAHLN test, initially reported in 2002 (2002 H7 test, hereafter), was designed to identify North American H7 Avian influenza virus (AIV) isolates and was developed based on the sequence data available at the time, which primarily consisted of a sequence from isolates recovered from the northeast U.S. live bird market system and poultry outbreaks in North America. 1,3,7,9
Since 2002, H7 AIV recovered from poultry in Chile 5 and some wild bird isolates from North and South America 6 were not identified by the 2002 H7 test because of sequence mismatches in the primer or probe binding sites 10 (these isolates were subsequently identified as H7 by the routine testing of AIV isolates by a hemagglutination inhibition (HI) assay, which is done regardless of the real-time RT-PCR result). Therefore, a new H7 assay, the 2008 Pan-American H7 real-time RT-PCR test (2008 H7 test, hereafter), was developed by using the new H7 sequence data from these isolates. This new test was developed to detect more H7 viruses from the Americas (sometimes referred to as the “New World” lineage in influenza virus A genetics) 1 and is meant to be an improvement of a previous test, not a definitive subtyping method.
The primers and probes for the 2008 H7 test (H7 PanAm F1515 5′-ATGGAGAGCATAAGGAAC-3′, H7 PanAm R1628 5′-CCGAAGCTAAACCATAAG-3′, H7 PanAm F1585 probe 5′-[6-FAM]-CAGATAGACCCAGTGAAA TTGAGT-[BHQ1]-3′) were designed to target the conserved sequence in the HA gene (which does not overlap the regions targeted by the previous test) by using all currently available avian origin H7 sequence, including H7 viruses from recent outbreaks in poultry in North and South America and newly available sequences from North and South American wild bird AIV isolates (GenBank accession numbers EU684257-EU684262; although there were wild bird H7 isolates collected before 2002 [Table 1], which were not detected by the 2002 test, neither the sequence nor the RNA was available at the time that the 2002 test was developed). Australian and Eurasian virus sequences were included in initial alignments but were not targeted with the primers and probe because of sequence variation, and the number of mismatches was considered too high for degenerate or mixed bases to compensate (Fig. 1).
In addition, because of the high variation in sequence, it was not possible to develop a primer and probe set that was a 100% match to all known North and South American H7 virus sequences. Mismatches were minimized so that no isolate had more than 2 nucleotide differences with either a primer or the probe, and mismatches were placed near the middle of the primers or probe (Fig. 1). One or 2 mismatches in a primer or probe were shown to be tolerated by real-time RT-PCR tests with no loss of sensitivity. 2 Overall, 3 patterns of mismatches were observed; because they differed in location, it was expected that the addition of mixed bases or degenerate bases would not provide a clear advantage.
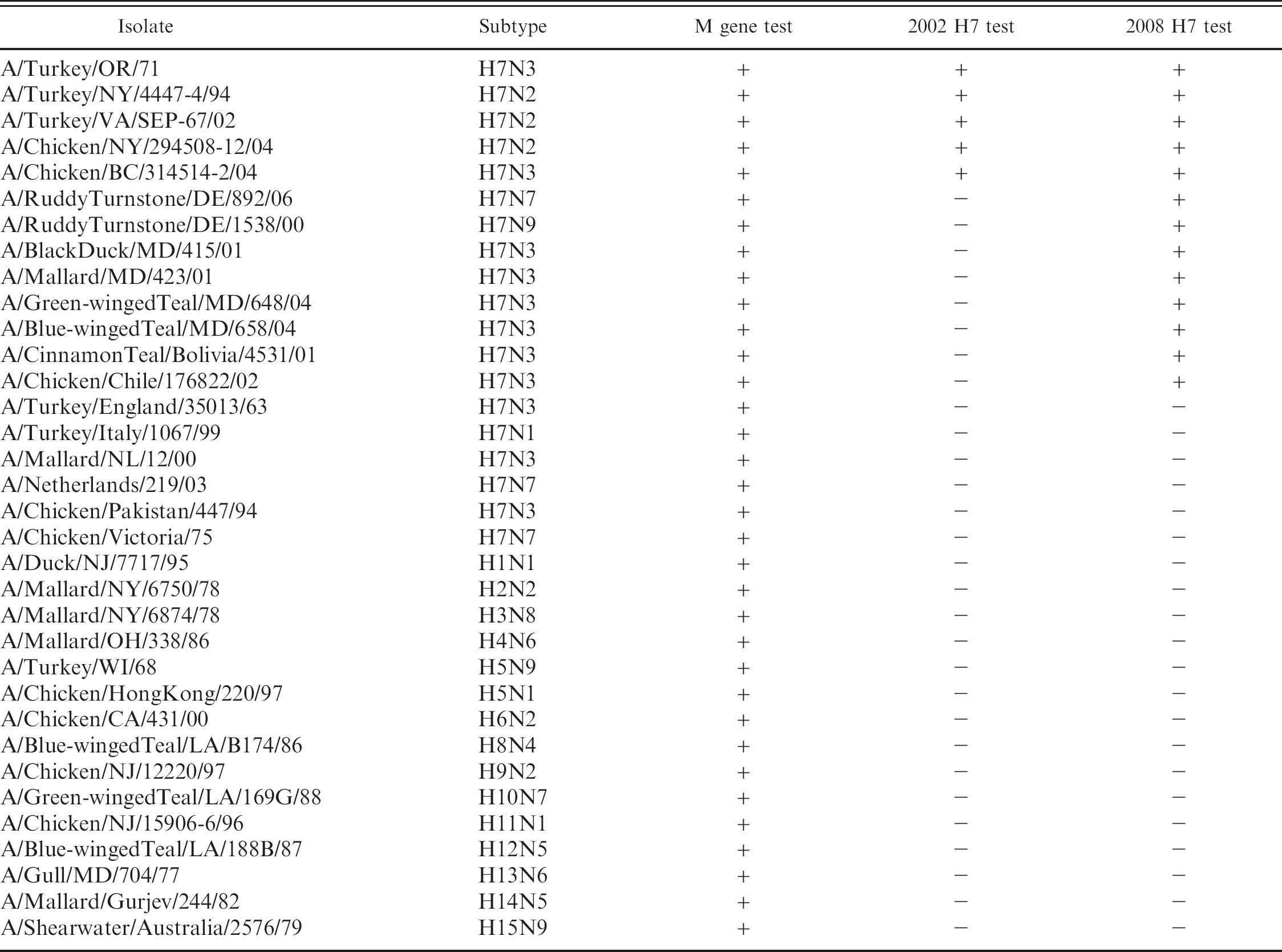
Detection of selected avian influenza virus isolates with the real-time reverse transcription polymerase chain reaction (real-time RT-PCR) for the influenza matrix (M) gene, the 2002 United States Department of Agriculture, National Animal Health Laboratory Network H7 hemagglutinin real-time RT-PCR test, and the 2008 Pan-American H7 real-time RT-PCR test.
The real-time RT-PCR reaction was optimized with a commercial 1-step RT-PCR kit a for a 25-μl reaction that contained: 1.1 μl of nuclease free water, 12.5-μl kit supplied 2 x buffer (containing MgCl2 and deoxynucleotide-triphosphates [dNTPs]), 1.7 μl kit supplied detection enhancer, 5 pmol of each primer and probe, 1-μl 25 x kit supplied enzyme mix, and 8 μl of template. Reactions were run on the real-time PCR instrument b in standard mode as follows: RT stage: 45°C for 10 min, 95°C for 10 min; PCR stage: 40 cycles of 95°C for 5 sec, 54°C for 30 sec, 72°C for 15 sec. Data were collected at the 54°C step. Results were interpreted by using the “auto” threshold calculations of the real-time PCR system b software.
Assay limits of detection (LOD) were determined in relation to 50% egg infectious doses (EID50). For each virus tested, 10-fold serial dilutions were tested in a minimum of triplicate, and the last concentration where the majority of the replicates tested positive was considered the end point. Furthermore, to determine whether known sequence mismatches would effect the LOD, 4 viruses were selected for LOD testing (Fig. 1): A/Turkey/VA/SEP-67/02 (100% sequence match), which had an LOD of 100.8 EID50/reaction; A/RuddyTurnstone/DE/892/06 (2 mismatches in the probe), with an LOD of 100.5 EID50/reaction; A/Turkey/OR/71 (2 mismatches in the forward primer and 1 mismatch in the probe) with an LOD of 100.1 EID50/reaction, and A/Mallard/MD/423/01 (1 mismatch the reverse primer) with an LOD of 100.0 EID50/reaction. The gene copy LOD was also calculated for A/Turkey/VA/SEP-67/02 and was between 103 and 104 gene copies per reaction, by using in vitro transcribed RNA produced as previously described. 8
Test sensitivity was within 1 log10 concentration among all the isolates tested, and the detection limit based on EID50 was about a log10 higher than what was reported for the 2002 H7 test. 8 This discrepancy was probably because of a different platform and reaction chemistry because, when the 2002 and 2008 H7 tests were run side by side on the same platform b with the same reaction chemistry a when using A/Turkey/VA/SEP-67/02 RNA, the LOD by both EID50 and gene copy numbers were the same.
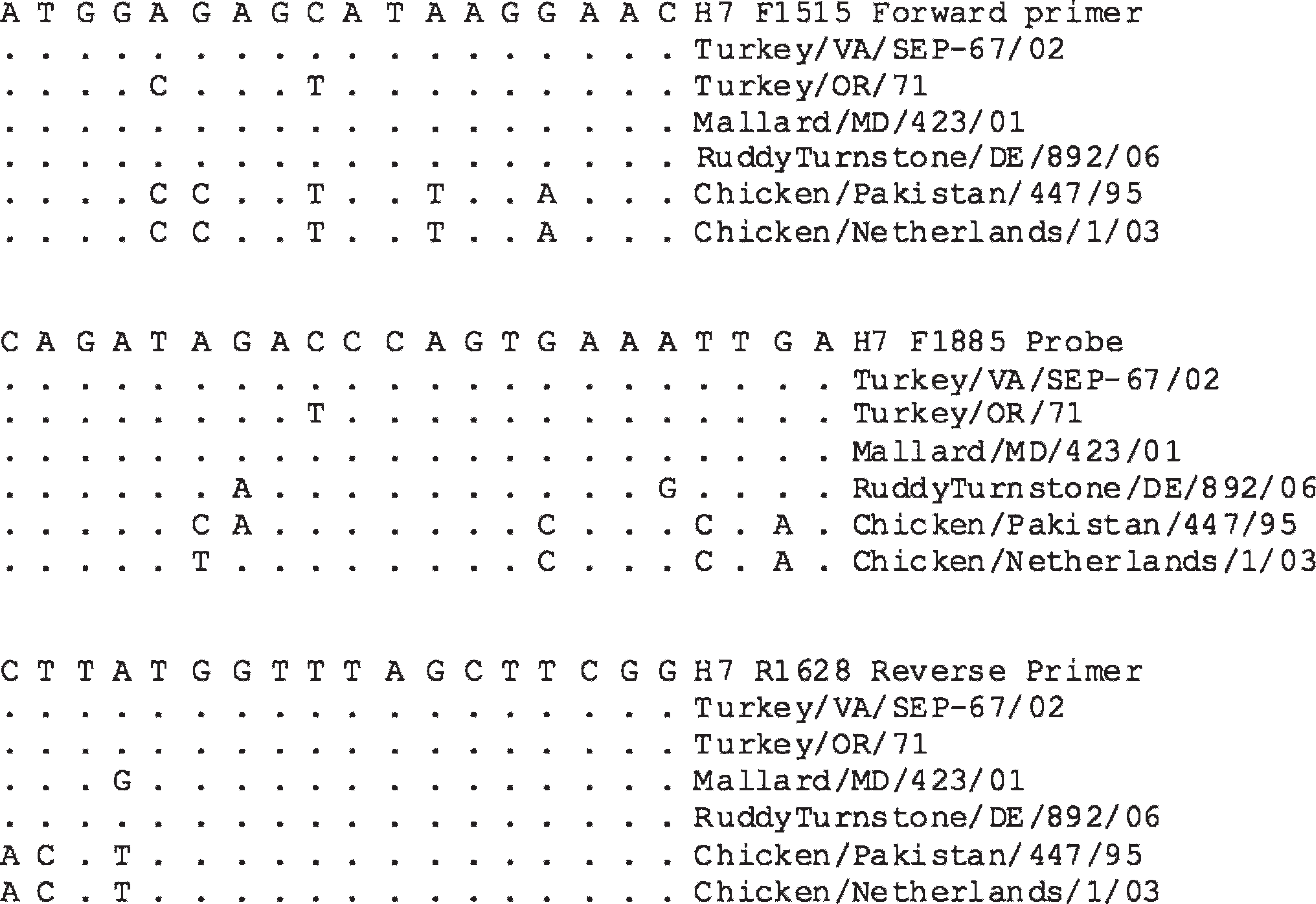
Alignment of selected H7 viruses in the primer and probe regions. All sequence is shown in positive sense from 5′ to 3′. A “•” indicates a match with the primer or probe sequence.
Assay specificity was evaluated by performing the test as described above with RNA extracted infectious amnioallantoic fluid (AAF) with a commercial reagent, c from a total of 34 influenza virus isolates, which included 19 H7 influenza viruses from poultry and wild birds from North and South America, Europe, Asia, and Australia, and 15 AIVs from all other HA subtypes except H16 (Table 1). The 2008 H7 test detected all H7 AIVs from North and South America included in the panel (Table 1), and the test was negative for the other 14 subtypes tested. No H7 AIVs with Eurasian lineage HA genes were detected with the 2008 H7 test.
To evaluate assay performance with simulated clinical specimens, 9, 10-week-old, specific pathogen–free, white leghorn chickens from the Southeast Poultry Research Laboratory in-house flocks were inoculated by the intranasal route with 106EID/50 per bird of A/Chicken/ BritishColumbia/314514-2/04 H7N3 high pathogenicity pathogenicity AIV. 4 Cloacal and oral-pharyngeal (OP) swabs were collected at 2 days postinoculation (PI) and from surviving birds at 7 days PI (n = 12 OP swabs, n = 12 cloacal swabs). The chickens were housed in custom designed and manufactured, modified Horsfall isolators in a biosafety level 3Ag facility, with ad libitum access to feed and water. Animals were cared for and euthanized in accordance with institutional animal care and use procedures.
Swab material was processed for RNA with a commercial RNA isolation kit d by using a commercial magnetic particle processor e and was tested with the USDA-NAHLN influenza A matrix (M) gene real-time RT-PCR test 8 (M gene test, hereafter) and with both the 2002 and 2008 H7 tests. The M gene test was run with the addition of a standard curve (a new standard curve was run with each set of reactions to account for between-run variation). The standard curve was established by running RNA extracted from 10-fold serial dilutions of titrated AAF stocks of the challenge virus in triplicate, and the commercial real-time PCR system b software was used to calculate the standard curve and quantities per reaction.
The 2008 H7 test detected virus in similar numbers of swabs as the 2002 H7 test; however, there was one cloacal swab that was positive with the 2002 H7 test only (Table 2). All samples that were positive with the M gene test were positive with the 2008 H7 test (Table 3). Three samples were positive with the 2008 H7 test only, this may suggest marginally better sensitivity with the 2008 H7 test versus the M gene test, because all samples contained RNA from the same isolate specificity differences would not account for the discrepancy. Virus titers in the swab samples based on quantitative real-time RT-PCR for the M gene ranged from 100.1 to 102.3 EID50/ml. Although the number of swabs from experimentally infected birds was small, the 2008 H7 test appeared to be at least similar in sensitivity to the M gene test and was shown to work with poultry OP and cloacal swabs.
Detection of matrix (M) gene and H7 viral RNA in oral-pharyngeal and cloacal swabs collected from chickens experimentally infected with A/Chicken/BritishColumbia/314514-2/04 H7N3 highly pathogenic avian influenza at 2 and 7 days postinoculation by the M gene test, 2002 United States Department of Agriculture, National Animal Health Laboratory Network H7 hemagglutinin real-time reverse transcription polymerase chain reaction (real-time RT-PCR) test, and 2008 Pan-American H7 real-time RT-PCR test.
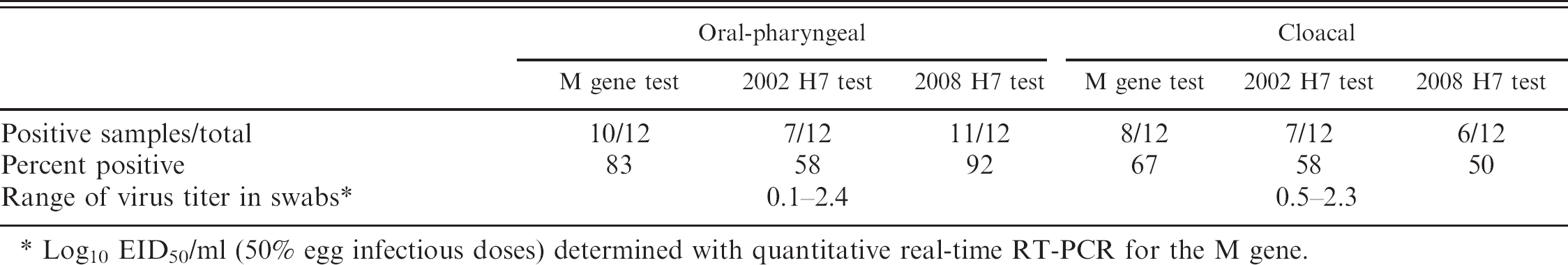
Log10 EID50/ml (50% egg infectious doses) determined with quantitative real-time RT-PCR for the M gene.
Finally, the test was evaluated with wild bird surveillance specimens collected in the United States from 2006 to 2007. Samples were processed as follows: RNA from the swab samples was tested with the M gene test and virus isolation (VI) was attempted on real-time RT-PCR positive swab material (n = 918). The AAF from VI attempts was then tested for AIV by the M gene test. Samples that were positive by the M gene test (n = 468) were then tested to identify the H and N subtype of the isolate. The initial subtype testing for H7 was done with the 2002 H7 test, which detected no H7 viruses in the 468 positive samples. The subtypes of all isolates, regardless of real-time RT-PCR results, were determined by HI assay, which is the reference standard subtyping test by using the standard procedure (Alexander DJ: 2005, Avian influenza. In: Manual of diagnostic tests and vaccines for terrestrial animals. World Organization for Animal Health, Paris, France. Available at: http://www.oie.int/eng/normes/mmanual/A_00037.htm. Accessed April 11, 2008). The 2008 H7 test was performed as an additional test on the 468 VI positive samples, of which there were a total of 27 isolates that were positive for H7, all of which were confirmed to be the H7 subtype by HI assay. No H7 viruses were identified by HI that were not detected by the 2008 H7 test.
Detection of A/Chicken/BritishColumbia/314514-2/04 H7N3 from oral-pharyngeal and cloacal swabs collected from experimentally inoculated chickens 2 and 7 days after inoculation with the influenza matrix (M) gene real-time reverse transcription polymerase chain reaction (real-time RT-PCR) test and the 2008 Pan-American H7 real-time RT-PCR test.

Real-time RT-PCR has been used extensively in recent years for the rapid detection of AIV and for the subsequent identification of the most economically important HA subtypes for poultry, H5 and H7. The 2002 H7 test does not have adequate specificity, because of sequence mismatches with some New World AIVs; therefore, because H7 real-time RT-PCR testing of M gene test positive specimens is required for NAHLN laboratories, the development and validation of an improved test is warranted. The H7 real-time RT-PCR test reported here has broader specificity than the previous test, and, although additional field testing needs to be done to adequately validate this test, the sensitivity and performance with field samples was acceptable. Finally, it is critical to note that RT-PCR based methods for identifying AIV HA subtypes are not definitive, because there is always a possibility that, for example, an H7 virus collected in North or South America will not be identified by the 2008 H7 test either because of novel sequences or because it contains Eurasian lineage H7 genes; therefore, users should interpret results carefully.
Acknowledgements. The authors gratefully thank Scott Lee, Kimberley Hill, Patti Miller, Suzanne DeBlois, and the SAA sequencing facility for technical assistance with this work. The work was supported by USDA/ARS CRIS project 6612-32000-048. Mention of trade names or commercial products in this article is solely for the purpose of providing specific information and does not imply recommendation or endorsement by the U.S. Department of Agriculture.
Footnotes
a.
AgPath-IDTM One-Step RT-PCR Kit, Ambion Inc., Austin, TX.
b.
7500 Fast Real-time PCR System, Applied Biosystems, Foster City, CA.
c.
Trizol LS reagent, Invitrogen Corp., Carlsbad, CA.
d.
MagMAXTM-96 AI/ND viral RNA Isolation Kit, Ambion Inc., Austin, TX.
e.
KingFisher® magnetic particle processor, Thermo Fisher Scientific Inc., Waltham, MA.
