Abstract
Canine parvovirus (CPV) has been evolving, generating new genetic and antigenic variants throughout the world. This study was conducted to determine the types of CPV circulating in dogs in Figueira da Foz, Portugal. Thirty fecal samples, collected between 2006 and 2007 from dogs with clinical signs of CPV infection, were tested for CPV by a rapid, in-clinic, enzyme-linked immunosorbent assay (ELISA)/immunomigration test, by conventional real-time polymerase chain reaction (PCR), and by minor-groove binding TaqMan PCR. Of the 29 PCR-positive samples, 15 were identified as CPV-2b and 14 as CPV-2c. No CPV-2a was detected. The sensitivity of the ELISA test was 82.76% compared with the PCR assays. No significant associations were found between CPV type, clinical outcome, breed, vaccination status, or age.
Keywords
Canine parvovirus (CPV) was first isolated in 1978. 1 Over time, the original CPV-2 isolates were replaced by new antigenic types, termed CPV-2a, and a virus type displaying a single mutation that was termed CPV-2b. 9,11 Since 1996, a number of additional mutations have been described, some of which resulted in antigenic changes in the virus. 18 Recent studies on CPV strains in Italy revealed the appearance of a CPV-2 mutant with an amino acid substitution at position 426 (Asp to Glu). 2,6,9 Currently, this CPV-2 type, previously referred to as Glu-426 mutant and now named CPV-2c, co-circulates with CPV-2a and CPV-2b. Since its discovery, CPV-2c has also been detected in Vietnam, 10 Europe, 3 the United States, 7,8 and South America. 13 Canine parvovirus appears to be undergoing continuous evolution, and the new viruses show antigenic differences that may influence the susceptibility of young animals to infection at the time when the level of maternally derived antibody decreases to the minimum protective titer. 3 This variation could, potentially, negatively affect vaccine efficacy, although it has been shown recently that dogs vaccinated once with a particular live, attenuated CPV-2 vaccine were protected from challenge with a type 2c field isolate. 17 Similarly, the performance of diagnostic testing could be affected by viral variation; although to date, there has been no evidence for missed diagnoses because of antigenic variation, despite the observed differences between the assays. 6
Rapid enzyme-linked immunosorbent assays (ELISAs) that detect CPV antigen in feces using monoclonal antibody (mAb) or polyclonal antisera are frequently used in the diagnosis of parvovirus infections in veterinary practice. 6 The ELISA broadly recognizes CPV-2 viruses and does not distinguish between antigenic types. To determine antigenic differences between CPV-2 types, panels of mAbs are tested for their reactivity to each virus in hemagglutination inhibition assays. 5,10 However, mAb characterization requires samples with high hemagglutination titers (≥1:64). 6 A polymerase chain reaction (PCR)-based method was developed to identify CPV-2a and CPV-2b, 11 but that assay does not differentiate between types 2b and 2c. 2 A PCR restriction fragment-length polymorphism assay using the enzyme MboII was developed, but it only differentiated type 2c and not other CPV types. 2 To distinguish between CPV types, a real-time PCR assay, based on minor groove binder (MGB) probe technology was developed. 5,6 This assay takes advantage of a single nucleotide polymorphism that occurs at position 426 in the viral VP2 gene to identify and characterize antigenic variants. An additional probe was also designed to discriminate between vaccine and field strains of CPV. 5
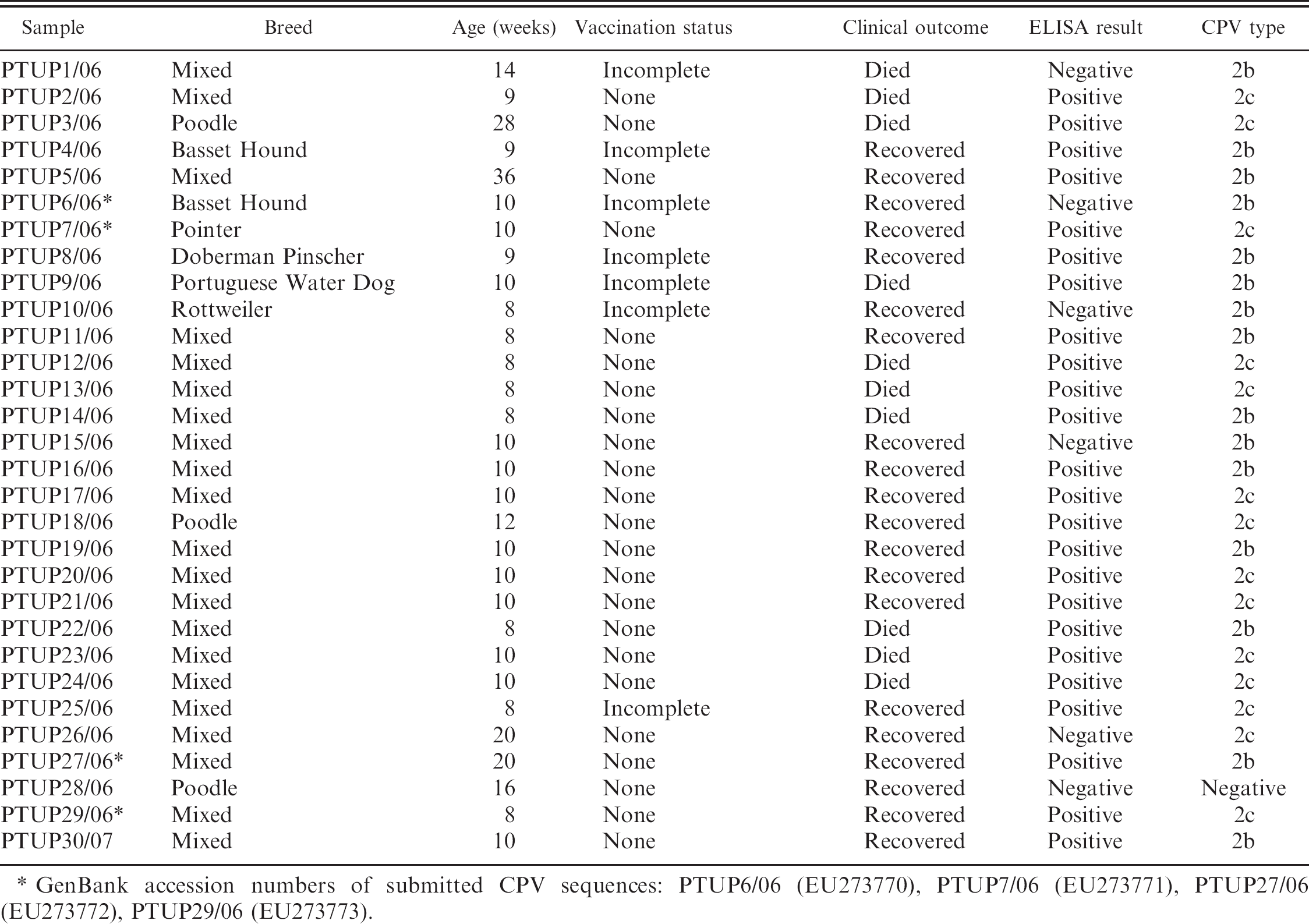
The objective of the current study was to determine and identify the CPV variants present in the area of Figueira da Foz in central Portugal. The study sample comprised 30 dogs, less than 36 weeks of age, with clinical signs suggestive of parvovirus infection (lethargy, anorexia, vomiting, or diarrhea), and with a history of incomplete or no vaccinations. The animals had been admitted to a local veterinary clinic between April 2006 and January 2007. Fecal samples were collected from these dogs, and samples were screened for CPV with a commercial rapid antigen-detection ELISA. a The associations of risk factors, such as breed, age, vaccination status, clinical outcome, or CPV-2b or CPV-2c, were determined by chi-square testing using 1 degree of freedom at the 5% level of significance.
For the molecular characterization, all fecal samples were homogenized (10% w/v) in phosphate-buffered saline and centrifuged for 15 min at 1,500 X g. The viral DNA was extracted from the supernatant of each specimen by boiling for 10 min and chilling on ice and then diluting 1:10 in distilled water. 6,15 This method has been described to be as efficient as phenol-chloroform extraction methods and is less laborious than the latter methods. 15
For the generic detection of CPV DNA, the nucleic acid extracts of all samples were tested by real-time PCR as previously described. 6 For further characterization of the CPV-2 variants, the MGB probe technology 5 was applied to all the samples positive by real-time PCR. The fecal samples from 2 animals, PTUP06/06 and PTUP25/06 (Table 1), that were vaccinated less than 10 days before sample collection, were further tested with the MGB probe to discriminate vaccine virus from field strains. Conventional PCR was performed to amplify a fragment with 583–base pair (bp) of the VP2 gene, a region surrounding the 426 site, using primers 555f and 555r. 2 Sequence analysis was carried out by direct sequencing of the amplicons of 4 selected isolates of CPV-2b (PTUP6/06 and PTUP27/06) and CPV-2c (PTUP7/06 and PTUP29/06) described in this study. The amino acid sequences were deduced and aligned.
Twenty-four of 30 fecal samples tested by rapid ELISA were positive for CPV. Using real-time PCR and MGB probe assays, CPV was detected in 29 of 30 samples (Table 1). The rapid ELISA detected 11 type 2b and 13 type 2c viruses, but failed to detect 4 type 2b and 1 type 2c viruses; therefore, the sensitivity of the ELISA relative to real-time PCR was 82.76%. The feces of 1 animal (PTUP28/06) were negative for CPV by all assays, suggesting that the cause of its clinical condition may not have been CPV infection. Retrospective determination of seroconversion that could clarify the case in this unvaccinated animal was not performed.
Together with the CPV test results, breed, age, and vaccination status were recorded for each animal (Table 1). Of the 30 dogs with clinical signs, 20 recovered and 10 died. Twenty-one dogs were of undefined or mixed breed, and 9 dogs belonged to 6 different breeds. Ages ranged from 8 to 36 weeks. Seven animals were incompletely vaccinated, and the rest were unvaccinated. Significant associations (P < 0.05) between clinical outcome and breed, vaccination status, age category (younger or older than 12 weeks), or CPV type were not found.
Real-time PCR and MGB probe analysis of the 29 samples identified 15 (52%) CPV type 2b and 14 (48%) type CPV type 2c (Table 1). The specificity of these results was confirmed by conventional PCR and sequencing of 2 selected CPV-2b isolates, PTUP6/06 (GenBank EU273770) and PTUP27/06 (GenBank EU273772), and 2 selected CPV-2c isolates, PTUP7/06 (GenBank EU273771) and PTUP29/06 (GenBank EU272773). The nucleotide and deduced amino-cid polymorphic positions were compared with the most similar CPV sequences obtained through a BLAST search of GenBank database (Table 2). The comparative sequence analyses showed that sample EU273770 shared 99% identity with the North American CPV strain M74849, as a result of single nucleotide polymorphisms in positions 430 (G → A) and 580 (T → A), and only differed at position 580 (T → A) from the Brazilian isolate DQ340409. The other CPV-2b isolate analyzed in this study, EU273772, showed only 1 nucleotide polymorphism at position 580 (T → A). Both CPV-2c sequences, EU273771 and EU273773, had 100% nucleotide identity with that of CPV-2c isolate AY380577 from Italy (Table 2). The 2 sequences, PTUP06/06 and PTUP25/06, were from dogs that had been recently vaccinated; however, the viruses were determined to be CPV field strains of types 2b and 2c and not the vaccine virus.
Canine parvovirus 2a and 2b are present in the canine population worldwide, replacing the original strain CPV-2. The recent appearance of CPV-2c variants in the field is evidence of a high mutation rate for this DNA virus equivalent to that observed in RNA viruses. 3,7,13,14,16 Although modified-live vaccines based on CPV-2 protected puppies against challenge with a recent isolate of CPV-2c, 17 and modified-live vaccines based on CPV-2b protected puppies against infections with CPV-2a and CPV-2b, 2 the rapid evolution of this virus warrants vigilance for the epidemiological consequences and for the potential need for new vaccines and diagnostic tools.
Breed, age, vaccination status, clinical outcome, and Canine parvovirus 2 (CPV-2) type detected in dogs with clinical signs suggestive of parvovirus infection.
GenBank accession numbers of submitted CPV sequences: PTUP6/06 (EU273770), PTUP7/06 (EU273771), PTUP27/06 (EU273772), PTUP29/06 (EU273773).
A sensitivity of 82.76% was calculated for the rapid ELISA compared with real-time PCR. This means that approximately 17% of dogs with clinical signs suggestive of CPV infection may not be positive for CPV by the ELISA. More sensitive in-clinic tests are needed to aid CPV diagnosis in practice. On the other hand, the ELISA was able to detect both CPV-2b and CPV-2c, indicating that CPV-2c will not go undetected in the field. Interestingly, however, the test failed to detect 4 type 2b viruses. This could very likely have been due to the reduced amount of free virus available for the ELISA because the rapidly developing intestinal immune response to CPV-2 results in the formation of undetectable immune complexes.
Clinicians worry about the emergence of new CPV-2 strains that may be responsible for outbreaks of virulent disease. Some studies suggest that CPV-2c may be more pathogenic than types 2a or 2b, 2,5 whereas others describe less-severe clinical symptoms and lower mortality rates in CPV-2c–infected dogs. 4 The results presented in the current study did not confirm either hypothesis. Both, CPV-2b and CPV-2c were found in severe disease, and significant differences were not found in the clinical outcome of dogs infected with CPV-2b versus CPV-2c. Further, significant associations were not found between breed, age category (<12 weeks, >14 weeks), vaccination status, or clinical outcome.
Nucleotide and amino acid polymorphic positions in the VP2 sequences of Canine parvovirus 2 (CPV-2) isolated from canine feces from clinical cases of CPV enteritis in Portugal.

The present findings revealed that of the 29 analyzed CPV-2 strains obtained in central Portugal, nearly half were of type 2c, suggesting that this variant is replacing CPV-2a and CPV-2b. Type 2a viruses were not detected in this group of dogs, but more samples should be tested to assess the actual prevalence of this CPV type. The sequence analysis of field isolates in this study also indicated a single nucleotide polymorphism in the VP2 gene, similar to that found in Brazil. 12 To the authors' knowledge, the single nucleotide polymorphism found in codon 580 (T → A) has not been previously reported. This mutation does not result in an amino-acid change in the viral capsid, and the epidemiological consequences of this replacement are yet to be determined. Canine parvovirus 2c sequences obtained from 2 dogs from Portugal were identical to the strain AY380577 from Italy. However, more samples should be analyzed to investigate whether mutations (e.g., those observed in North American CPV-2c isolates 7,8 ) are also present in Europe and whether an international trend in viral evolution can be observed. It is, therefore, important to assess the current prevalence of CPV-2 variants in domestic dog populations, as well as the diagnostic sensitivity of current in-clinic tests and vaccine efficacy.
Acknowledgements. This work was supported by the Foundation for Science and Technology Portugal (FCT) grant SFRH/BD/ 23101/2005 to M. J. Vieira and by Intervet Portugal. The authors thank the National Laboratory for Veterinary Investigation (LNIV, Delegação do Porto) for infrastructural support.
Footnotes
a.
Witness® Parvo, Symbyotics Corp., Lyon, France.
