Abstract
Canine parvovirus 2 (CPV-2) is considered the main pathogen responsible for acute gastroenteritis in dogs, causing vomiting and hemorrhagic enteritis mainly. However, infection in cats by CPV variants causes clinical signs similar to Feline panleukopenia virus. The current study reports a case of CPV-2c in a domestic cat, in Portugal. The findings suggest that more surveys are needed to know the true prevalence and significance of cats in CPV epidemiology worldwide.
Canine parvovirus 2 (CPV-2) emerged as a dog pathogen in the late 1970s and rapidly spread worldwide.2,10,15,18,23 The virus originated as a host range variant from Feline panleukopenia virus (FPV) that adapted to the canine host via wild carnivores such as minks and foxes.16,21 In dogs, CPV-2 causes a highly contagious and often fatal disease characterized by vomiting and hemorrhagic enteritis. 2 Over time, the original CPV-2 isolates have been replaced by 3 mutants with changes within the VP2 gene, including the 4 substitutions that define the CPV-2a variant, as well as the change of VP2 residue 426 to Asp or to Glu (the so-called CPV-2b and CPV-2c variants). Overall, the virus has shown relatively rapid evolution and high genomic substitution rates. 7
In cats, the diseases caused by CPV generally appear to be much milder than those seen in dogs infected with the virus, or those caused by FPV. 19 The CPV-2 isolates do not replicate in cats, but the CPV-2a–derived variants do. 21 A previous study 17 reported the detection of CPV at high frequency in samples collected from domestic and wild cats in Japan and Vietnam. Canine parvovirus has also been isolated from the feces of clinically healthy domestic and wild felines, 19 suggesting that the virus was either shed long after infection, or that it causes very mild or subclinical disease. 14 Coinfection by multiple CPV variants has also been described in a cat,5,6 when CPV-2a and the 426 Glu variants were detected in the same animal. Mixed infections by FPV and CPV-2 or FPV and CPV-2a have also been reported.3,22 These type infections could potentially facilitate recombinations and high genetic heterogeneity. 4 The current study reports a case of CPV-2c in a female cat with severe disease, in Portugal.
A 5-month-old female cat was presented to a veterinary clinic in Porto, Portugal, in February 2008, suspected of FPV infection. The cat was a mixed breed and unvaccinated. At physical examination, the cat was depressed, in lateral recumbence, with 7% dehydration, fever, and low body condition. A blood sample was collected, and the following parameters were measured: red blood cell count (10.66 × 106/μl), hemoglobin concentration (14.1 g/dl), mean corpuscular volume (41.3 fl), hematocrit (44%), and white blood cell count (1.9 × 103/μl); results were indicative of panleukopenia. A fecal sample was collected from the animal and sent to the Laboratory of Microbiology and Infectious Diseases of the Veterinary Clinics Department at the Abel Salazar Biomedical Sciences Institute (ICBAS; University of Porto, Portugal) for virus detection.
The sample was screened by conventional polymerase chain reaction (PCR) assay for FPV and CPV, simultaneously. Viral DNA extraction from the fecal sample was as previously described.12,13 The PCR assay was carried out using primer pair 555 for (5′-CAGGAAGATATCCAGAAGGA-3′) and 555rev (5′-GGTGCTAGTTGATATGTAATAAACA-3′), which amplify a fragment with 583 base pairs (bps) of the VP2 gene (nucleotide [nt] position 4003–4585), a region that includes residue 426. 7 The PCR amplification reaction was performed as described, 13 in a total volume of 25 μl. The amplified products were analyzed on a 1.2% (weight/volume) agarose gel stained with GelRed a and visualized under ultraviolet light. b The sample in the study tested positive by PCR assay. The PCR product obtained from the amplification of the fecal sample was sequenced at a commercial facility c with the use of the same primer pair. Alignments and sequence analysis were performed using commercial software, d and parvovirus reference strains obtained from the GenBank database were used for comparison. The sample sequence was identified as CPV-2c showing an identity that varied between 99.8% and 100%, when compared with other CPV-2a–derived reference strains (Table 1). Further comparisons with reference strain sequences showed a synonymous substitution in the codon for residue 430 (nt 4074–4076; Fig. 1), as previously reported (accession nos. EU273770 and JF906814, respectively) in dogs. The sequence obtained in the current study was deposited in GenBank as accession number KF824851.
Nucleotide and amino acid polymorphic positions in the VP2 sequences of Canine parvovirus (CPV) isolates and Portuguese feline fecal sample, with reference strains.
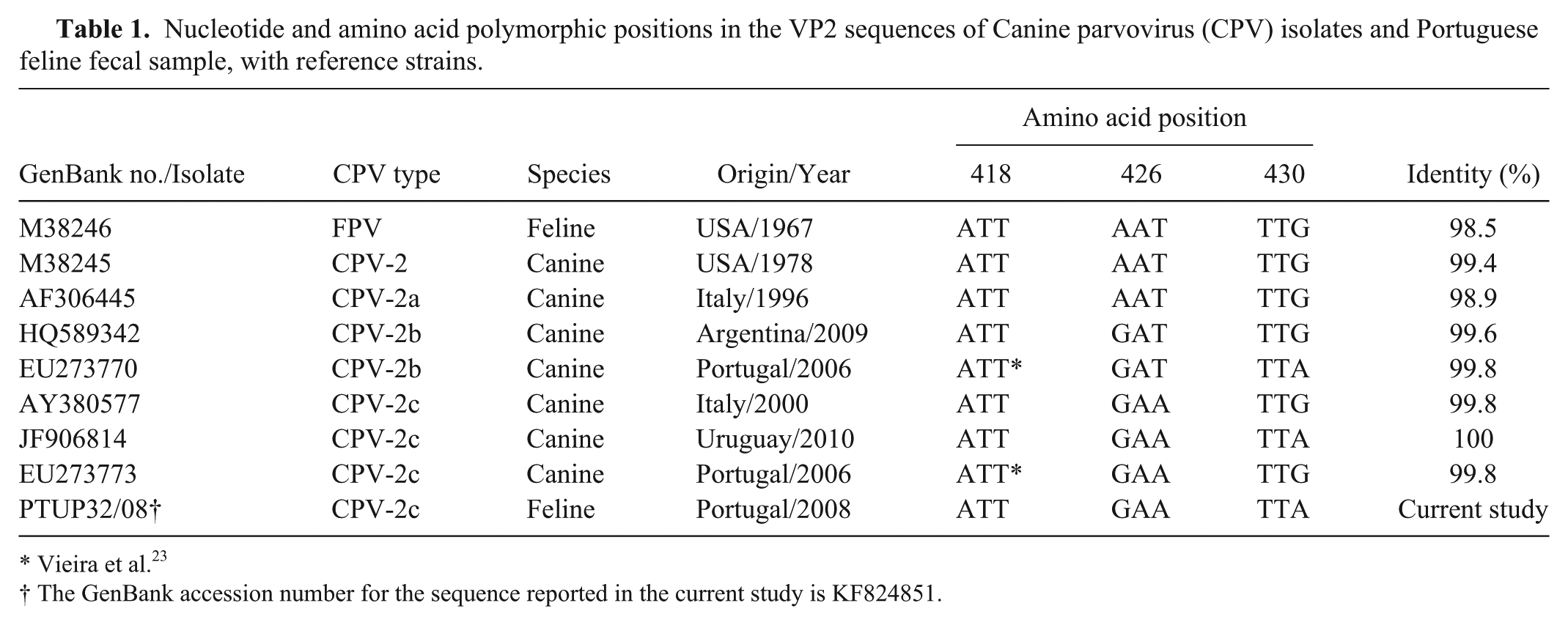
Vieira et al. 23
The GenBank accession number for the sequence reported in the current study is KF824851.
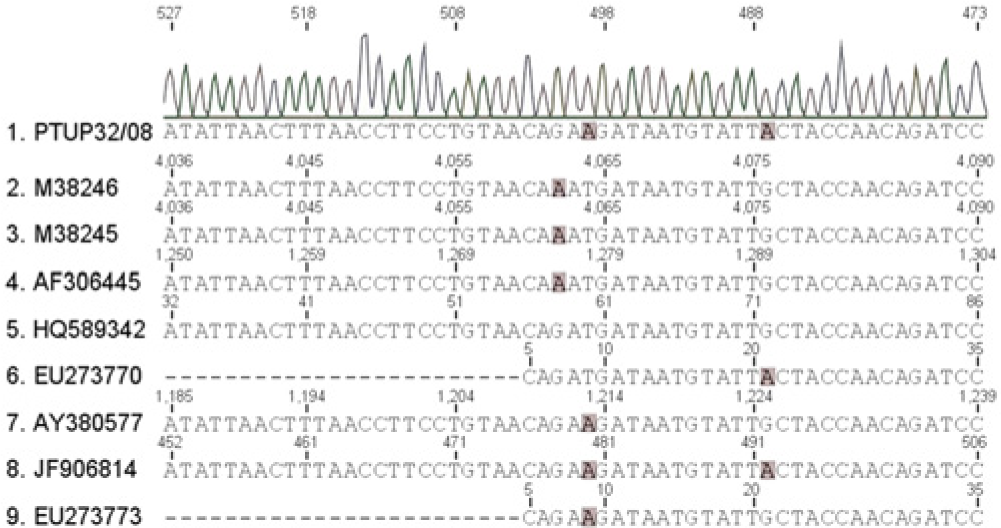
Representative schema of partial VP2 sequence of Canine parvovirus and the feline fecal sample in the study, with shading indicating the last base of 2 amino acid positions (426 and 430, respectively).
Further testing on this sample consisted of restriction fragment length polymorphism (RFLP) of the PCR product. The PCR products obtained were digested with an adapted method previously described, 7 using the enzyme MboII. e The GAA codon in this restriction site (GAAGA) codes for glutamate at position 426. The codon at position 426 is replaced by AAT (asparagine) in the CPV-2/2a variants and by GAT (aspartate). 20 Only PCR products obtained from 426 Glu mutant strains are cut by MboII, generating 2 fragments of approximately 500 bps and 80 bps, respectively. 7 Two positive controls were used with the 426 Asp and Glu variants, as well as a negative control. After digestion, products were run in agarose gel, which showed the generation of 2 fragments in the tested fecal sample.
The present study reports the circulation of CPV, with the change of residue 426 to Glu, in a domestic cat. Canine parvovirus has been described as an example of how a virus can successfully cross species barriers and become established in a new host, 15 including ancestral. Phylogenetic analysis of parvoviruses in nondomestic animals revealed a significant biodiversity, which is clearly the result of multiple cross-species transmission events. 1 Further evidence of a synonymous substitution in residue 430 found in the cat in the current study has been previously described in 2 dogs.20,23 The finding shows that these parvoviruses can readily infect and cause disease in cats, through transfer from dogs, although cat-to-cat transfer is also possible. However, the evolutionary mechanisms that allow this virus to infect new hosts and the emergence of new strains in “real time” are complex and not yet understood. 15
The new variants of CPV appear to be able to infect cats and other hosts (puma, coyote, gray wolf, bobcat, raccoon, and striped skink) because the variants can bind the feline transferrin receptor as well as the receptors of other hosts,1,16 while still remaining a severe threat to dogs. The role of these variants in cats still remains somewhat uncertain. 8 Most reports describing CPV in cats describe clinical signs similar to those seen in feline panleukopenia3,5,14 or much milder than those seen in dogs. 19 Other variants of CPV resulting from CPV-2a–derived viruses have also been isolated from the feces of apparently healthy cats.8,19 A previous study 8 demonstrated that the virus was likely being shed for periods of 4–6 weeks as an asymptomatic infection. Cats could potentially be a risk factor for infecting other dogs and cats with parvoviruses. 8 In the current study, the cat presented severe clinical signs and survived after 3 days of treatment. In contrast, another study 9 also detected the CPV 426 Glu variant in a kitten but without hemorrhagic diarrhea; instead, that kitten had normal white blood cell counts and had been administered multivalent vaccines against FPV and other pathogens. The CPV-2a variant with the 426 Glu variation has already been reported in cats, in Italy.4,6,9,11 In one case, the same cat was reported to have a coinfection with multiple CPV variants. 6 Multiple infections may increase the epidemiologic role of the cat as a reservoir and as a source of new variants of parvovirus. 3 Reports on CPV prevalence in the dog population in Portugal showed that the prevalent antigenic variants were the 426 Asp and Glu replacements.10,23 However, there is no information about the prevalence of CPV and FPV in the cat population until now. Canine parvovirus 2a with the change of 426 Glu was first described in 2000, in Italy, 7 and rapidly spread worldwide.10,18 The presence of this variant in cats may reflect the transfer of viruses from dogs, confirming the diffusion and evolution of virus variants of CPV-2 in nature. 4
In conclusion, the current study reports the detection of the 426 Glu variant of CPV in a domestic cat in Portugal. The pathogenicity of extant CPV strains in cats and the relative prevalence of CPV and FPV in cat populations is currently very limited. As most diagnostic tests do not discriminate between CPV and FPV infections, and only clinically infected animals are tested in most of the cases, 15 more studies should be conducted on a large scale to explore the true prevalence and significance of CPV in cats worldwide.
Footnotes
Acknowledgements
The authors thank Dr. Maria João Vieira for contribution on animal sample collection and Dr. Eliane Silva for technical assistance.
a.
GelRed nucleic acid gel stain, Biotium Inc., Hayward, CA.
b.
Bio-Rad Laboratories, Hercules, CA.
c.
Macrogen Inc., Amsterdam, The Netherlands.
d.
Geneious software package version 6.0.6, Biomatters, Auckland, New Zealand.
e.
FastDigest MboII, Fermentas, St. Leon-Rot, Germany.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
This work was supported by the Foundation for Science and Technology (FCT) Portugal grant SFRH/BD/76291/2011 to C. Miranda.
