Abstract
Previously published studies have neither used nor reported the results of an indirect enzyme-linked immunosorbent assay (iELISA) to measure serologic responses in natural outbreaks of strangles. The concept of using serologic responses to identify persistent carriers of Streptococcus equi has been proposed but not scientifically evaluated. The specific aims of the current study were to determine the duration and level of truncated fibrinogen-binding protein-specific (SeM allele 1) antibody production in ponies involved in a natural outbreak of strangles and to determine if test results from this serologic iELISA could predict persistent carrier status. Serologic samples were obtained before and after an outbreak of naturally occurring strangles infection. Persistent carriers of S. equi were identified via culture and polymerase chain reaction (PCR) testing of lavage fluid collected from the guttural pouches and nasopharynx or swabs of the nasopharynx after recovery from acute disease and at postmortem examination. Logistic regression analysis was used to determine if an association existed between serologic response and persistent carrier state. The ELISA reported in the current study definitively confirmed a recent exposure to S. equi. However, the measured serologic response did not predict carrier status in this strangles outbreak. Therefore, a guttural-pouch endoscopy with subsequent culture or PCR testing to detect S. equi remains the most accurate method available for the identification of persistent carriers.
Introduction
Streptococcus equi subspecies equi (S. equi), the etiologic agent of strangles in equids, is highly contagious and is spread through aerosol and oral routes by exposure to nasal secretions or contaminated fomites. Clinical signs associated with S. equi infection include acute onset pyrexia, mucopurulent nasal discharge, lymphadenitis, cough, difficult breathing, and enlargement of submandibular and retropharyngeal lymph nodes with subsequent rupture of abscesses. 9,16 A challenging aspect of disease control for S. equi infections is asymptomatic carriers that persistently shed the bacteria and act as sources of the bacteria for months to years, subsequently exposing susceptible animals. 2,7,11,16 The guttural pouches are a common site of persistence of the organism and can be visibly abnormal when examined via an endoscopy. 7,14
The current recommendation for identification of persistent carriers is to perform culture and/or polymerase chain reaction (PCR) on nasal swabs or lavages on a weekly basis for 3 weeks after clinical-disease resolution. 11 These screening processes are minimally invasive but are expensive and require multiple sampling procedures. A guttural-pouch endoscopy and testing of lavage fluid from the pouch can increase the rate of detection of carriers, 11 but it can be expensive, time consuming, and invasive, and may not always be readily available. In contrast, blood samples can generally be easily obtained and sent to a laboratory for determination of antibody levels against S. equi–specific proteins.
The fibrinogen-binding protein (FgBP) of S. equi, also known as SeM, is highly immunogenic, antiphagocytic, and binds fibrinogen. 3 Indirect enzyme-linked immunosorbent assays (iELISA) that detect total immunoglobulin G (IgG) directed against the FgBP protein of S. equi were proposed for the diagnosis of conditions such as purpura hemorrhagica or abdominal abscessation because of S. equi infection. 11
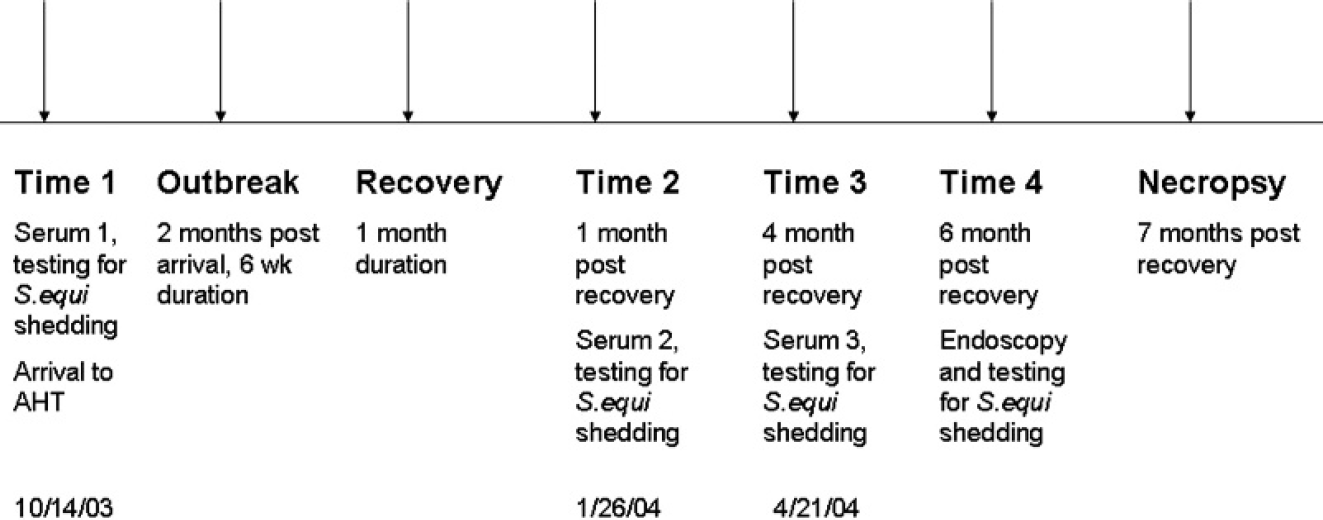
Timeline for sampling surrounding Streptococcus equi outbreak.
In the current study, the antigen used to sensitize the ELISA plates was truncated recombinant fibrinogen-binding protein (FgBP1). 6 The truncated form lacked the C terminal cell-wall anchor domain of FgBP. 6 Fibrinogen-binding protein is essentially identical in nucleotide sequence to that of the SeM gene of S. equi, 12 with only 11 nucleotide differences, which results in a SeM allele change from 2 to 1. 4,15 The concept of determining antibody levels to FgBP and using it to detect persistent carriers in outbreaks of strangles has also previously been suggested 10–14,16 but, to the authors' knowledge, has never been scientifically evaluated. Furthermore, no other published studies of outbreaks of strangles have used an immunoassay to investigate the antibody titer of carrier animals. The aim of this article is to describe the use of an iELISA to follow serologic response in a natural outbreak of strangles within a naïve population of Welsh mountain ponies and to answer the question of whether serology can be used to predict the persistent carrier state.
Materials and methods
Study population and design
The study population consisted of 30 naïve, 4–6 month old, weanling ponies that arrived at the Animal Health Trust (AHT; Newmarket, England) quarantine facility in October 2003. Despite testing negative on arrival for a range of equine pathogens, including S. equi, a strangles outbreak commenced within the herd 2 months later. Clinical signs persisted for 6 weeks and ponies were allowed to recover for 1 month before screening via nasopharyngeal swabs and lavages for detection of S. equi by PCR and culture (Fig. 1).
Time of serum sampling
Serum samples were available from 3 separate time points. These included serum retrieved upon arrival at the AHT before the strangles outbreak (time 1: October 14, 2003), within 1 month of recovery from strangles (time 2: January 26, 2004), and at 4 months after recovery from strangles (time 3: April 21, 2004). A timeline that described sampling intervals can be found in Figure 1.
Methods used to categorize and define persistent carriers of S. equi
In addition to the initial testing on arrival for S. equi shedding, the ponies were also monitored for S. equi shedding within 1 month after recovery and at 4 and 6 months after recovery from strangles. The procedures used at each sampling period included testing of nasopharyngeal swabs and lavages for S. equi by PCR and culture. A guttural-pouch endoscopy and a lavage with culture and PCR test to detect S. equi were performed at 6 months after recovery from strangles (Fig. 1). The ponies were categorized as persistent carriers at the 6-month after-recovery sampling time if one or more of the following occurred: a positive (culture/PCR) nasopharyngeal lavage or swab, marked pathology of the guttural pouches with an endoscopy, or a positive (culture/PCR) guttural pouch lavage for S. equi. Postmortem examinations of the head and neck areas were performed on those ponies identified as or suspected to be persistent carriers approximately 7 months after recovery from clinical disease.
Description of iELISA
The iELISA used in the current study was validated by using sera from 84 horses that were selected based on their status related to exposure to S. equi. Seventy-nine of the 84 horses had known natural exposure to S. equi (history of strangles or exposure to horses with the disease) or a history of vaccination against S. equi. Of the 79 samples, 54 were from horses that had been vaccinated against S. equi. The remaining 25 samples were from horses that were non-vaccinated but were either culture positive for S. equi or had known exposure to the disease. Sera from 5 foals were obtained before colostrum ingestion, and this was used as the negative control. Although the neonatal foal precolostral samples were the definitive negatives with regard to exposure to S. equi used during validation, serum likely to contain a broader array of immunoglobulins that could potentially cross-react in the iELISA were sought. Serum was obtained from 2 juvenile horses from a closed herd at remote premises that had no known exposure to S. equi. During test validation and development, screening of serum samples was performed at dilutions of 1:500, 1:2,000, and 1:4,000.
Testing procedure
FgBP1 was used to sensitize the ELISA plates. 6 Wells of medium-binding polystyrene U-bottom 96-well plates were coated with 60 μl of FgBP1 diluted in carbonate coating buffer (0.2 M, pH 9.4) a to 0.15 μg/ml. To monitor and control for nonspecific binding of test and to control sera to the plate (background), a set of wells on each plate was sensitized with only the carbonate coating buffer. Sensitization was completed after shaking for 60 min at 37°C in an orbital shaker/incubator b at 1,400 oscillations per minute. Wells were washed 4 times c with 350 μl of phosphate buffered saline solution (PBS) with a 10-sec soak between each wash step. To block potential additional protein-binding sites, 75 μl of a blocking solution, which consisted of 1:2 commercial stabilizing reagent d with PBS and 0.1% commercial milk diluent, e was added to each well and incubated for 30 min as above. This solution was then aspirated, and the plates were dried in a desiccating cabinet for 4 hr before being sealed with tape and stored at 4°C in vacuum-sealed bags.
The buffer used for serum and conjugate dilutions consisted of a 1:2 mixture of a commercial stabilizing buffer f and 0.1 M Tris-buffered saline solution, pH 8.0. Serum samples diluted to 1:250 were mixed with an equal volume of standardized (optical density at 600 nm [OD600] = 1.8) heat-killed Streptococcus zooepidemicus and incubated for 30 min at 37°C to remove antibodies that might cross-react with FgBP1 but not be specific for S. equi (e.g., the recently identified SzM protein of S. zooepidemicus shares near-sequence identity with the C-terminal half of FgBP). 4 After centrifugation at 4,330 X g for 10 min at room temperature to pellet cells with adsorbed cross-reacting antibodies, the adsorbed serum was aspirated. This 1:500 dilution was then further diluted to 1:2,000 and 1:4,000 with buffer solution. Fifty microliters of each adsorbed and diluted serum sample were added to duplicate wells of a sensitized and blocked plate. Positive-control serum was from a horse that had been vaccinated against strangles and had a medium-high level of antibody to the FgBP1 during the test validation. This control serum was diluted, adsorbed, and added to each plate in the same manner as the samples. The plates were incubated as above for 30 min and were washed as above. Fifty microliters of recombinant Protein A and Protein G horseradish peroxidase conjugate, a diluted to 0.18 μg/ml, was added to each well and incubated as above. After washing, 50 μl of tetramethylbenzidine substrate solution e was added to each well and incubated as above for 10 min. Reactions were stopped by the addition of 50 μl of 1 N hydrochloric acid per well. The OD450 for each plate was recorded by spectrophotometry g by using equipment-compatible software. h Averages of background (no antigen) wells were subtracted from averages of duplicate readings from test wells to calculate the reported OD450. Variance between duplicate readings averaged less than 5.0%.
Statistical analysis
The geometric mean and standard deviation (SD) were determined i for positive serologic titers (≥1:2,000) for each sampling period. Ponies that had an antibody titer of <1:2,000 were designated as negative. Logistic regression analysis was used to evaluate potential associations between serologic, culture, and PCR results with carrier status. j Predictors evaluated for persistent carrier status included serologic antibody titers at times 2 and 3, a change (increase or decrease) or maintenance of serologic response between times 2 and 3, being positive for S. equi via nasopharyngeal culture (lavage or swab) or nasopharyngeal PCR (lavage or swab) at time 2, or having abnormal guttural-pouch endoscopic findings or being positive for S. equi via guttural-pouch lavage fluid (PCR or culture). Potential predictors were individually screened for association with carrier status by using univariable logistic regression analysis, and were eligible for further analysis if they significantly improved model fit (P ≤ 0.05 on likelihood ratio testing).
Results
iELISA validation results
In the validation, 75 of the 79 samples from horses with known S. equi exposure tested positive during iELISA validation (94.9%). Sera from all presuckle foals tested negative by iELISA, and the average OD450 obtained by iELISA of the presuckle foal sera was 0.002. The serum samples from the 2 horses at the remote premises, at the 1:2,000 dilution, yielded an average OD450 of 0.240, which was chosen as the realistic cutoff point between a positive and a negative test result. To determine the validity of the single cutoff point for each dilution, the OD of the positive control was plotted graphically against each of the chosen dilutions. The positive control data was compiled from 17 separate assays and 100 ELISA plates. The plot of the ODs of the positive control versus the chosen dilutions yielded a line with an R 2 value of 0.977, which indicated a high degree of linearity between the dilutions and the OD of the positive control.
The 1:2,000 serum dilution was chosen as the definitive cutoff dilution, because it yielded the least number of discrepancies when all validation samples were categorized according to OD and S. equi exposure history. Thus, samples with OD450 values <0.240 at the 1:2,000 dilution were considered negative and samples with OD450 values ≥0.240 at the 1:2,000 dilution were considered positive. Samples with OD450 values ≥0.240 at the 1:4,000 dilution were further diluted to the end point of detection of specific antibody to FgBP1.
Serology
All ponies had baseline serum antibody levels to FgBP1 consistent with nonexposure to S. equi at the time of arrival to the AHT (<1:500, time 1). At 1 month after recovery (time 2), serum was available for only 29 of the 30 ponies. At time 2, the geometric mean antibody titer of the 28 ponies (persistent carriers and noncarriers) with positive titers (≥1:2,000) was 1:5,252 (SD: 2.3; range: 1:2,000 to 1:64,000). All but one of the ponies seroconverted from baseline levels at this time. At 4 months after recovery (time 3), the geometric mean antibody titer for the 24 ponies with positive titers (≥1:2,000) was 1:3,175 (SD: 1.9; range: 1:2,000 to 1:32,000). In addition, at time 3, 21 of 30 ponies (67.8%) had lower antibody titers compared with their respective titer at time 2. This was illustrated with 14 of 30 serum sample titers decreasing by 2-fold, 3 of 30 decreasing 4-fold, and 4 of 30 decreasing 8-fold. A comparison of log titers for ponies classified as carriers or noncarriers is presented in Figure 2.
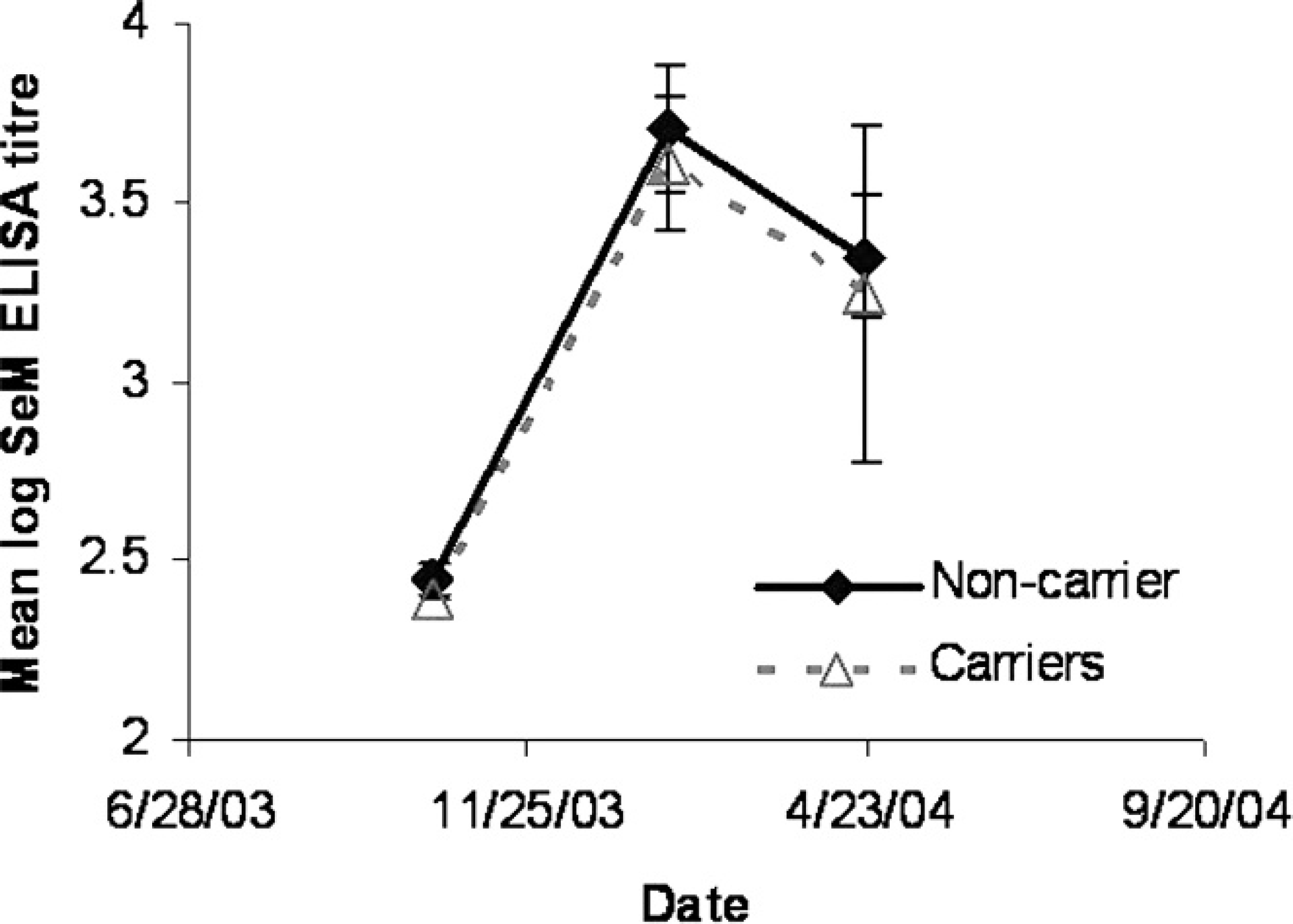
Comparison of anti–truncated recombinant fibrinogen-binding protein titer in carrier and noncarrier ponies at time 1 (October 14, 2003), time 2 (January 26, 2004) and time 3 (April 21, 2004).
Persistent carriers
There were 5 ponies that met the case definition for being a persistent carrier at 6 months after recovery. Several of the carrier ponies were positive for S. equi on more than one test criteria. Only 1 of 5 ponies (20%) was positive for S. equi based on a nasopharyngeal lavage culture. Similarly, 1 of 5 ponies (20%) was categorized as a persistent carrier based on the degree of guttural-pouch pathology visualized in the postrecovery screen. Three of 5 ponies (60%) had positive cultures for S. equi from guttural-pouch lavage samples. And, finally, 3 of 5 ponies (60%) were positive for S. equi via PCR test from guttural-pouch lavage samples.
The majority of the persistent carriers' titers decreased (3/5) between times 2 and 3. The antibody titer as measured with the iELISA described in the current study at both postrecovery times was not associated with carrier status (Table 1). Similarly, the change in titer between times 2 and 3 was not associated with carrier status. None of the risk factors analyzed had a statistically significant association with carrier status, although the data were suggestive (P = 0.09) of an association between testing positive via culture or PCR for S. equi on one or more nasal or guttural-pouch samples at time 2 and becoming a persistent carrier (Table 1).
Postmortem results
Guttural-pouch chondroids were present in 2 of 5 ponies classified as persistent carriers. A guttural-pouch sample from 1 pony was culture positive for both S. zooepidemicus and S. equi. This pony was identified as a persistent carrier based on the degree of pathology in the guttural pouches seen in postrecovery testing protocol. No antemortem samples from this pony were positive for S. equi via PCR or culture at any of the postrecovery testing periods. 7 A second pony was positive for S. zooepidemicus based on culture of the guttural-pouch sample collected at postmortem. The remaining 3 ponies identified as persistent carriers did not have any pathology in their guttural pouches at the time of postmortem, and no S. equi was detected from the guttural-pouch samples collected at postmortem.
Evaluation of univariable logistic regression models for prediction of carrier status.*

PCR = polymerase chain reaction.
Time 2 = 1-month postrecovery phase serum.
Time 3 = 5-month postrecovery phase serum.
Time 2 vs. time 3 = maintenance or change in serum between 2 samplings.
Positive for S. equi shedding during postrecovery samplings.
Discussion
The need for research on the value of serology for identification of carriers of S. equi was previously acknowledged. 1,11–13 Lack of a unique serologic response to various antigens of S. equi by carriers represent potential limitations of using current serologic assays for identification of carriers. However, the ease and practicality of obtaining a blood sample compared with an endoscopy of the guttural pouches or repeated nasal flushes justified the investigation of serology as a potential predictor of persistent carriers of S. equi. To the authors' knowledge, this is the first study to report serial determinations of antibody levels to the FgBP1 protein of S. equi during a natural outbreak of the disease and attempt to correlate the titer to the occurrence of persistent carrier status.
Persistent carriers could theoretically have a different antibody response profile when compared with animals that clear the infection in the initial postrecovery period, either in the amount of antibody produced or the persistence of the antibody response. Therefore, it was hypothesized that the ponies identified as persistent carriers of S. equi via culture, PCR, or guttural-pouch lesions as determined by endoscopic examination in the postrecovery period would have significantly different serologic responses compared with noncarrier ponies.
The ability of the current study to detect statistical associations between serologic, culture, and PCR response and the development of the persistent carrier state was hampered by the sample size. Only 5 ponies were categorized as persistent carriers. With a sample size of 5 carriers and 24 noncarriers, a risk factor would have to be highly correlated with being a persistent carrier to be statistically significant. The power to detect an association between the variable most strongly associated (odds ratio = 7.3 for testing positive via culture or PCR on nasal or guttural-pouch samples) with carrier status was 0.49. k To detect a statistically significant association, the authors would have needed a minimum of 16 persistent carriers and 93 noncarriers. l
Interestingly, 1 pony identified as a persistent carrier did not culture positive for S. equi at any of the screening tests performed on nasal lavages, nasal swabs, or guttural-pouch lavages, but did culture positive for S. equi from chondroids recovered at a postmortem. This pony was identified as a potential carrier based on the degree of guttural-pouch pathology (multiple, smooth, greenish/yellow chondroids) seen during a guttural-pouch endoscopy. The failure to obtain a positive culture or PCR test for S. equi based on repeated sampling of nasal secretions and testing of guttural-pouch fluid reinforces that nasal shedding from guttural pouches can occur intermittently, 2,7 and that the most accurate identification of persistent carriers still requires visualization of the guttural pouches via an endoscopy with a subsequent culture of a guttural-pouch lavage.
Though serologic status at any of the reported time periods did not positively predict persistent carriers in the study's population of ponies, it was demonstrated that all of the ponies mounted a titer to S. equi as part of the outbreak and that antibody levels decreased within 4 months after initial resolution of clinical signs. A pertinent question to address is the degree of nonspecific immunoglobulin binding in the iELISA in the current study. The truncated FgBP1 protein used in the iELISA retains an intact immunoglobulin-binding region. 5 Therefore, it cannot be ruled out that part of the signal detected by the iELISA in the post-outbreak serum samples was because of nonspecific binding of immunoglobulin to FgBP1.
The iELISA presented in the current study can identify equids that were recently exposed to S. equi even in the absence of clinical signs of disease. However, the level or change in titer as measured with the iELISA was unable to identify ponies that were persistently infected with S. equi in its current format. Therefore, improved diagnostic ELISAs that would measure antibody response to alternative S. equi cell-surface proteins and/or measure fractionated immunoglobulin isotype responses are still worth pursuing to determine if they would better predict the persistent carrier of S. equi. Quantification of immunoglobulin isotype-specific responses in addition to total immunoglobulin response may prove to be useful. Levels of anti-FgBP IgGa fall rapidly after resolution of strangles compared with a prolonged IgGb response. 8 Therefore, the quantification of an immunoglobulin isotype–specific response toward some antigens, e.g., FgBP, may enhance the discriminatory power of a diagnostic ELISA for carriers. The recent completion of the S. equi genome sequencing project (www.sanger.ac.uk/Projects/S_equi/) may yield information regarding new surface antigens that could prove to be more suitable for the generation of an ELISA or other serologic tests capable of identifying horses persistently infected with S. equi. 15 Furthermore, recent studies suggested that the FgBP protein of S. equi has undergone immune pressure that led to genetic variation of the FgBP protein recovered from persistently infected horses. 4 These differences in the FgBP protein will likely have implications for improved diagnosis of persistently infected equids and may prove useful for outbreak investigations.
Acknowledgements
Funding for this project was provided by the Program for Economically Important Infectious Animal Diseases (PEIIAD), Animal Population Health Institute, Fort Collins, CO.
Footnotes
a.
Pierce Biotechnology Inc., Rockford, IL.
b.
Thermo LabSystems iEMS, Thermo Electron Corp., Waltham, MS.
c.
BioTek Instruments Inc., Winooski, VT.
d.
StabilGuard®, SurModics, Eden Prairie, MN.
e.
Kirkeguard and Perry Laboratories, Gaithersburg, MD.
f.
StabilZyme SELECT® Stabilizer, SurModics, Inc., Eden Prairie, MN.
g.
SpectraMax®, Molecular Devices Corp., Sunnyvale, CA.
h.
SoftMax® Pro, Molecular Devices Corp., Sunnyvale, CA.
i.
Minitab Inc., State College, PA.
j.
Egret® 2.0, Cytel Inc., Cambridge, MA.
k.
l.
Epi InfoTM 6, Centers for Disease Control and Prevention, Atlanta, GA.
