Abstract
The western barred bandicoot, Perameles bougainville, is an endangered Australian marsupial species. Routine histology of liver samples collected at necropsy from 19 of 20 (95%) western barred bandicoots revealed the sporadic to common occurrence of abnormal hepatocyte nuclei characterized by margination of chromatin and concomitant central pallor. Some abnormal hepatocyte nuclei were mildly to markedly enlarged and irregularly shaped. Periodic acid-Schiff reagent stained 131 of 142 (92%) of these abnormal hepatocyte nuclei. Positive staining was completely eliminated by diastase pretreatment. Transmission electron microscopy revealed that abnormal hepatocyte nuclei with marginated chromatin did not contain viral particles. Rather, glycogen β-particles and α-rosettes were identified within some abnormal hepatocyte nuclei. Glycogen intranuclear inclusions were an incidental finding in western barred bandicoot hepatocytes.
Western barred bandicoots, Perameles bougainville (an endangered Australian marsupial species), can be affected by a papillomatosis and carcinomatosis syndrome. 12,13 This syndrome is characterized by multicentric hyperplastic, dysplastic, and neoplastic lesions of cutaneous and mucocutaneous tissues and has been associated with a novel virus. 12,13 Abnormal keratinocytes containing nuclei with marginated chromatin and large amphophilic intranuclear inclusion bodies have been identified in the stratum granulosum of conjunctival lesions examined by routine histology. 12 Crystalline arrays of spherical viral particles, 45 nm in diameter, have been observed within the nuclei of rare conjunctival keratinocytes within these lesions by transmission electron microscopy. 12
Routine histology of western barred bandicoot livers collected at necropsy typically revealed the presence of abnormal hepatocyte nuclei characterized by margination of chromatin and concomitant central pallor with occasional hepatic nuclear enlargement and asymmetry (Fig. 1). The nature of these hepatic intranuclear inclusions was of particular interest, given the occurrence of intranuclear viral inclusions in keratinocytes from conjunctiva affected by the western barred bandicoot papillomatosis and carcinomatosis syndrome.
Archival hematoxylin and eosin-stained histology slides from 20 western barred bandicoots necropsied between 2000 and 2006 were examined for the occurrence of abnormal hepatocyte nuclei. These individuals all had lesions consistent with western barred bandicoot papillomatosis and carcinomatosis syndrome, and some had incidental Klossiella quimrensis infection of their kidneys. 1 The frequency with which these abnormal nuclei occurred was determined by examination of 10 randomly selected high-power fields (40 × objective) per slide. Of the 20 livers assessed, 19 (95%) contained abnormal hepatic nuclei characterized by intranuclear inclusions. On average, 7.8 hepatocyte intranuclear inclusions were observed per 10 high-power fields examined (range 0–56).
Sections (6-μm thick) of 4 formalin-fixed, paraffin-embedded western barred bandicoot liver samples were treated with diastase a followed by periodic acid-Schiff (PAS) or with PAS alone. Examination of sections treated with PAS alone showed positive staining in 131 of 142 (92%) intranuclear inclusions (Fig. 2). The intensity of PAS-positive staining varied from mild to marked. The PAS-positive staining of abnormal nuclei was completely eliminated (0/143; 0%) in sections pretreated with diastase (Fig. 3), suggesting a glycogen content of the intranuclear inclusions. Glycogen intranuclear inclusions were most prevalent in liver sections containing the highest levels of cytoplasmic glycogen detected using the PAS/diastase method, both in terms of the number of glycogen-rich hepatocytes and the amount of glycogen they contained. Glycogen-containing nuclei were scattered randomly throughout the liver. Rare, profoundly PAS-positive, round intranuclear glycogen bodies were observed in these sections (Fig. 2), as has been previously described. 2 Approximately 8% of abnormal hepatocyte nuclei did not show detectable PAS-positive staining. This result might be due to leaching of glycogen from hepatocytes during fixation and processing. Alternate fixation and processing techniques to improve the retention of glycogen in liver sections have been described 4 ; however, the sections from archival specimens used in the present study were routinely fixed in 10% neutral-buffered formalin and paraffin embedded.
One western barred bandicoot liver sample was fixed in 5% glutaraldehyde; rinsed in Sorensen phosphate buffer; postfixed in Dalton chrome osmic acid; dehydrated through graded alcohols; transferred into propylene oxide or propylene oxide/Epon 812; and embedded in Epon 812. b Ultrathin sections were cut, mounted on grids, and stained with lead citrate and uranyl acetate. These were examined using a Philips CM 100 BioTwin transmission electron microscope at an accelerating voltage of 80 kV. c Abnormal nuclei had peripherally marginated chromatin and nucleoli. No virions were seen in any hepatic nuclei examined. Occasional abnormal nuclei contained a central zone of glycogen β-particles, each with an average diameter of 20 nm, typically arranged in α-rosettes. These glycogen inclusions were in direct contact with the nucleoplasm and appeared to have displaced the chromatin and nucleolus peripherally. No contact was observed between the nuclear membrane and the central glycogen inclusion, nor were cytoplasmic elements, such as mitochondria, ribosomes, and endoplasmic reticulum, observed within the glycogen mass (Figs. 4, 5).
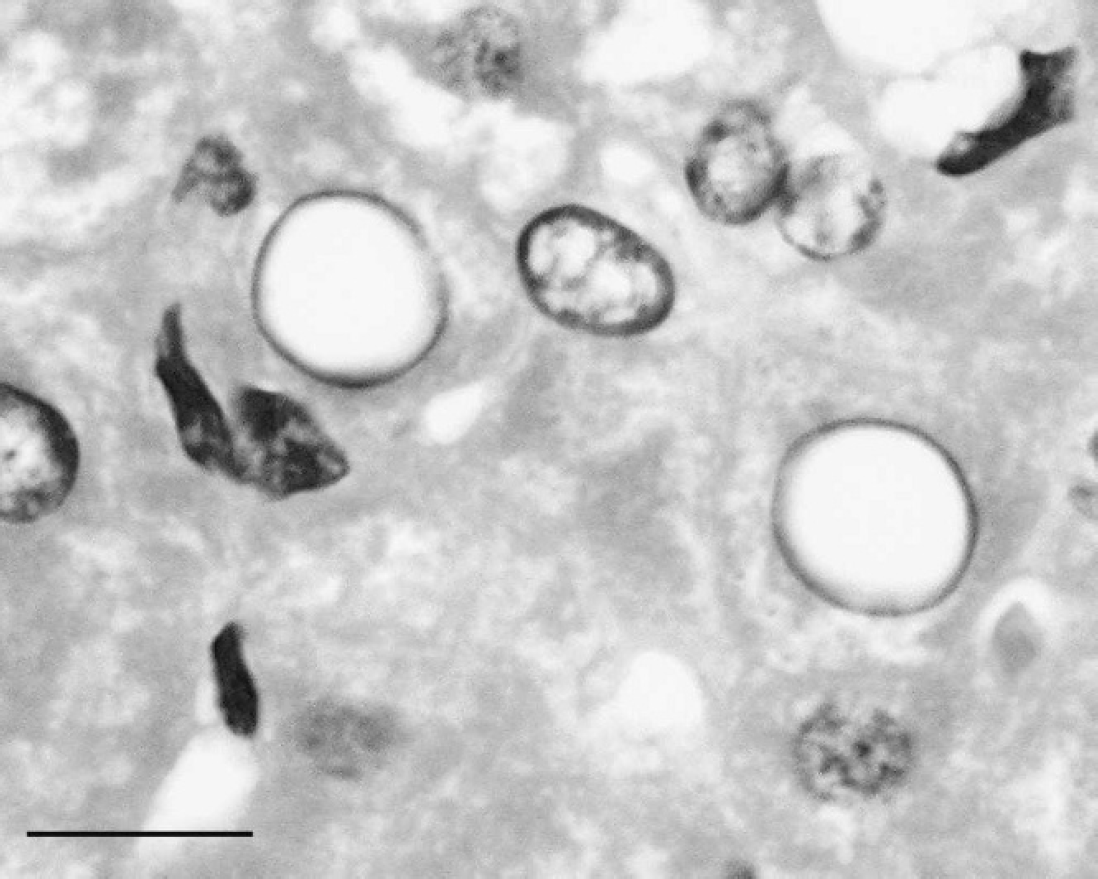
Hematoxylin and eosin-stained liver section from a western barred bandicoot (Perameles bougainville) showing 2 abnormal hepatocyte nuclei. Abnormal hepatocyte nuclei have peripherally distributed chromatin surrounding a zone of central pallor and are enlarged and asymmetrical. Bar = 25 μm.
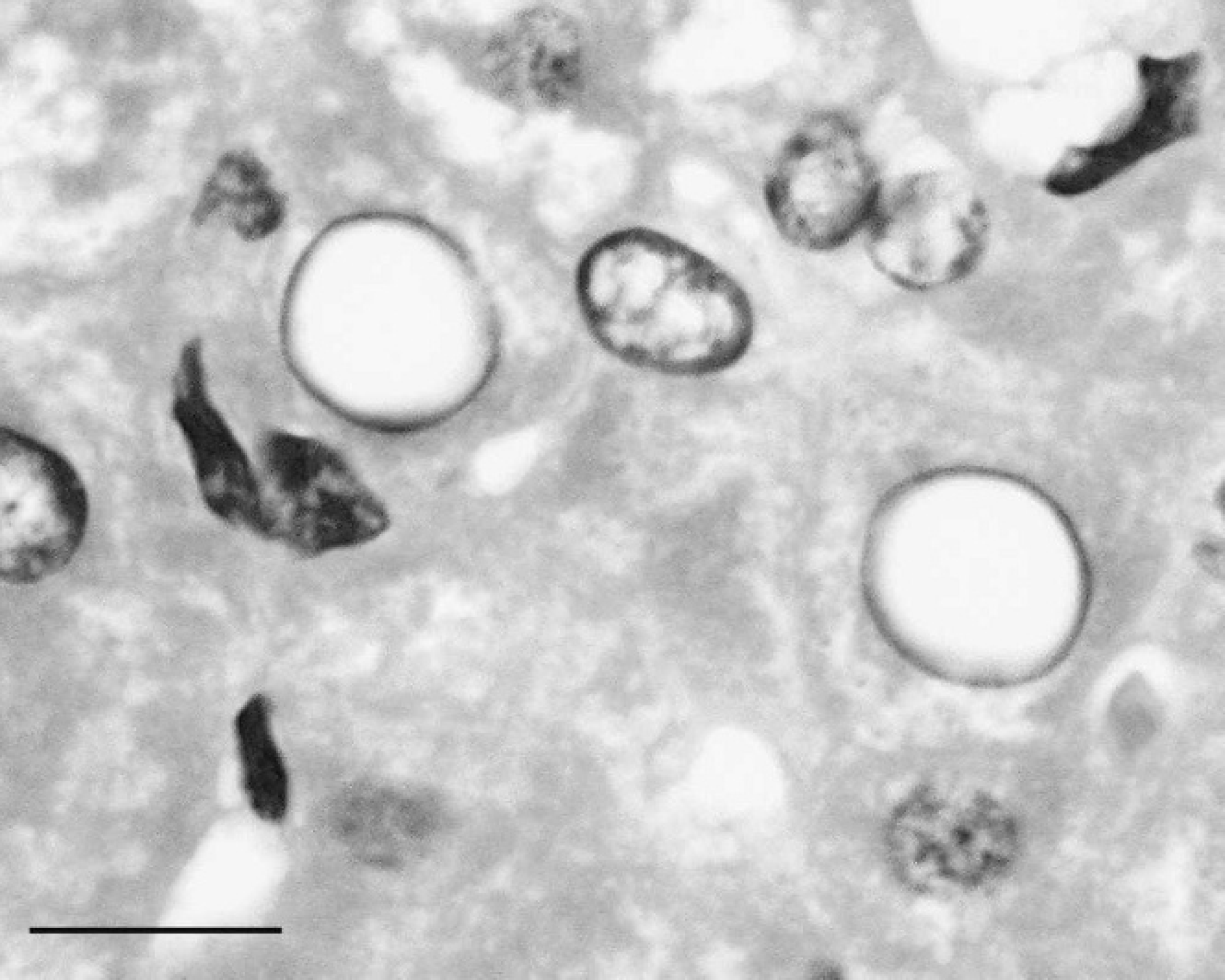
Periodic acid-Schiff (PAS)-stained liver section from a western barred bandicoot (Perameles bougainville) showing positive intranuclear staining of an abnormal hepatocyte nucleus. In this example, numerous PAS-positive, variably sized, round bodies are present within the nucleus. The nuclear material has been displaced peripherally by the presence of intranuclear glycogen. Bar = 25 μm.
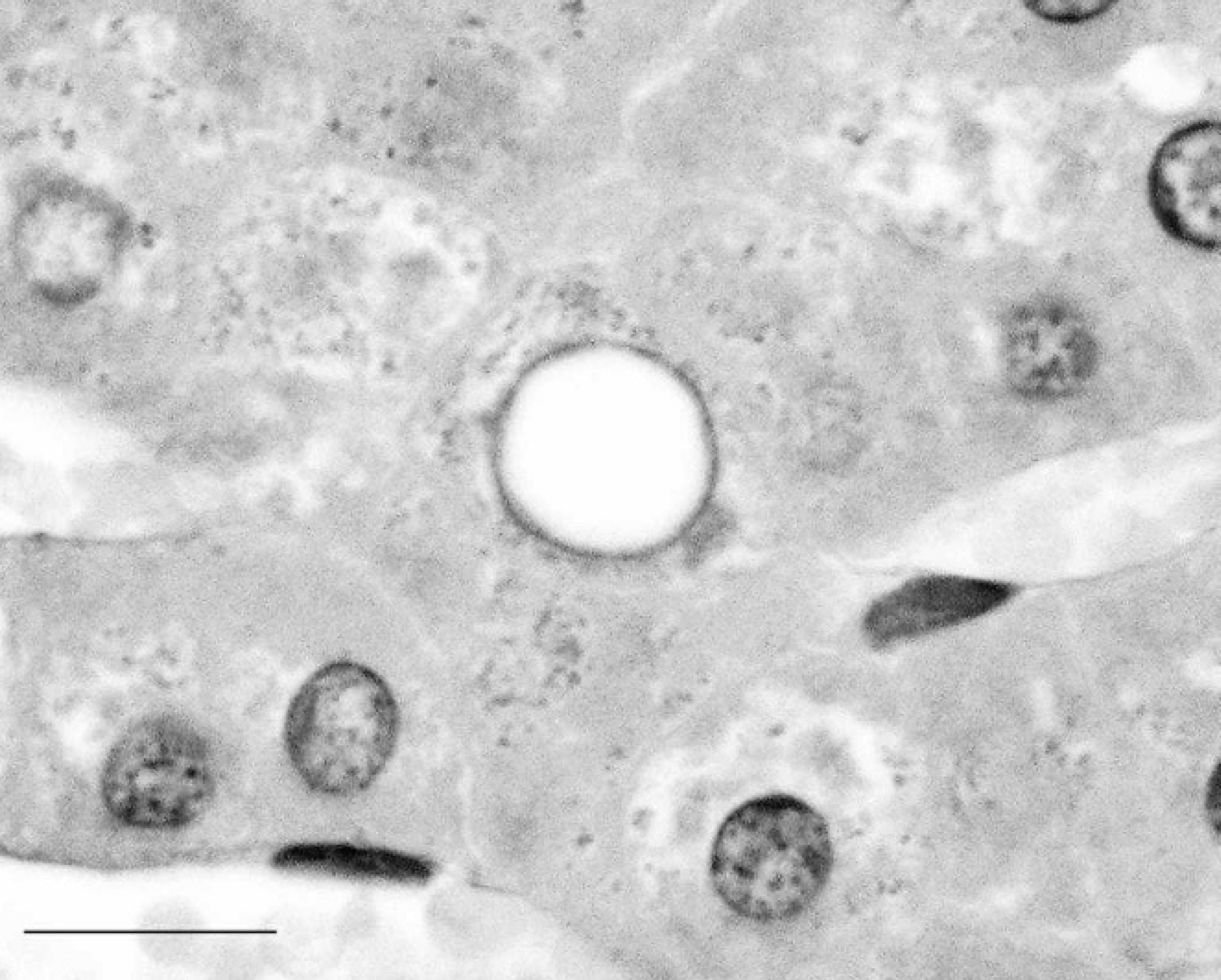
Diastase-pretreated, periodic acid-Schiff (PAS)-stained liver section from a western barred bandicoot (Perameles bougainville) showing abolition of PAS-positive staining of hepatocyte intranuclear inclusions (compared with Fig. 2). Bar = 25 μm.
Glycogen is a branched polymer of glucose that is normally found in the cytoplasm of many cell types. It acts as a depot for intracellular glucose, which can be released from storage by the combined enzymatic activities of phosphorylase, transferase, and α-1,6-glucosidase. 3,11 Occasionally, glycogen may appear within hepatocyte nuclei and has been reported in humans with von Gierke disease 5,9 (glucose-6-phosphatase deficiency), diabetes mellitus, 4,5 Graves disease, 5 Hodgkin disease, 5 hepatitis, 2,5 Wilson disease, 5 Gilbert disease, 5 and carcinoma of the stomach 5 and also after prednisolone treatment for disseminated lupus erythematosus. 5,10 Although intranuclear glycogen is an accompaniment to many disease processes, including inflammation, neoplasia, and necrosis, there has been speculation on a possible association between the occurrence of intranuclear glycogen inclusions and illnesses that lead to relative hypoxia in humans. 4 Therefore, it may be noteworthy that the western barred bandicoot with the most abundant intranuclear glycogen deposition in the present study was concurrently affected by numerous pulmonary metastases of a squamous cell carcinoma. Aged lactating and nonlactating clinically healthy ruminants 8 and well-fed premetamorphic tadpoles 5,6 commonly have hepatic intranuclear glycogen accumulations, highlighting that intranuclear glycogen deposition is not necessarily indicative of a pathologic process.
It is possible for glycogen to be synthesized in situ within the interchromatin regions of the nucleus. 7,14 Some authors suggest that glycogen may be translocated from the cytoplasm across the nuclear membrane and accumulate in the nucleus; however, the experimental evidence to support this hypothesis is lacking. 5 The illusion of intranuclear glycogen inclusions may sometimes be given by invagination of cytoplasmic elements (including glycogen) into the nucleus. 5 These cytoplasmic invaginations typically contain other cytoplasmic elements, such as ribosomes, endoplasmic reticulum, and mitochondria, and are delimited by the nuclear membrane. 5 The present study clearly demonstrates that western barred bandicoot hepatocyte nuclei can contain genuine intranuclear glycogen inclusions. The occurrence of glycogen intranuclear inclusions in hepatocytes does not appear to be related to the papillomatosis and carcinomatosis syndrome present in this species but is, rather, an incidental finding of no apparent clinical significance.
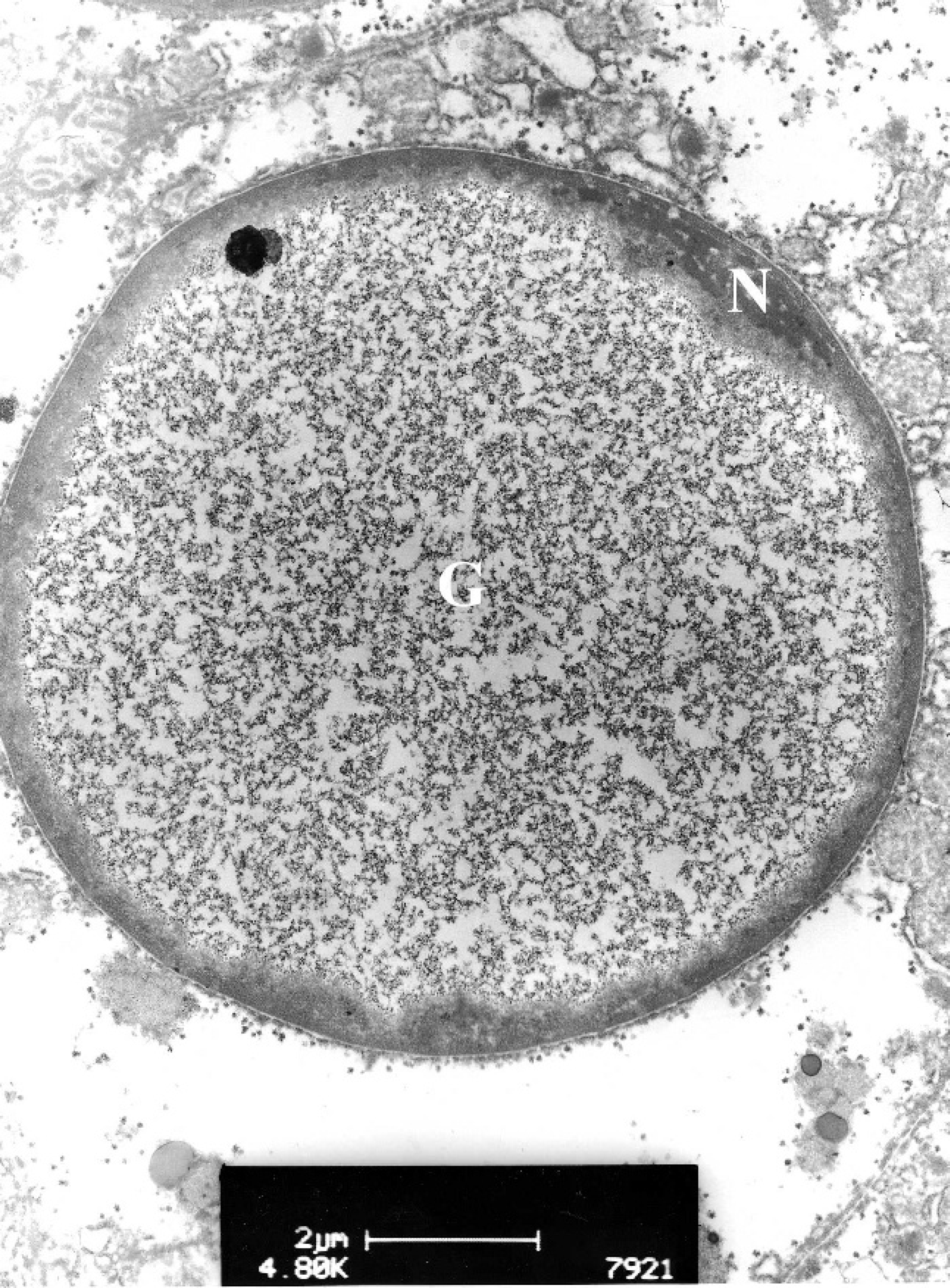
Transmission electron micrograph of a glycogen intranuclear inclusion from a western barred bandicoot (Perameles bougainville) hepatocyte (lead citrate and uranyl acetate stain). The nuclear material (N) has been displaced peripherally. The center of the nucleus is occupied by glycogen (G) arranged in β-particles ∼20 nm in diameter and α-rosettes. The glycogen inclusion body is not part of a cytoplasmic invagination.
Acknowledgements. Thanks to Michael Slaven and Gerard Spoelstra for preparing the histology slides and Peter Fallon for assisting with electron microscopy. This project is a collaboration between Murdoch University and the Western Australian Department of Environment and Conservation, funded through Australian Research Council linkage project LP0455050.
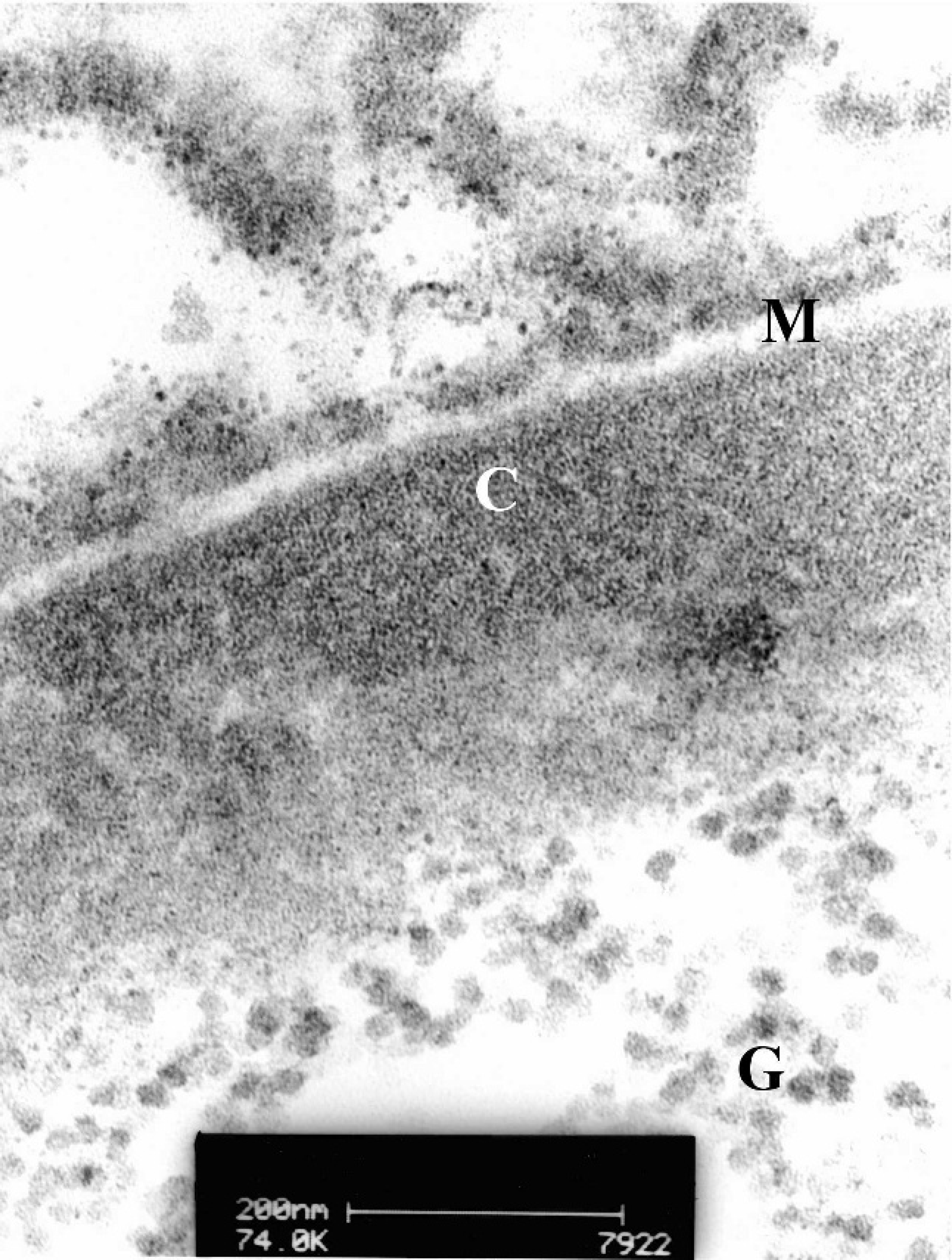
Higher magnification transmission electron micrograph of the glycogen intranuclear inclusion depicted in Figure 4. The fine structure of glycogen α-rosettes (G) is visible within the nucleus. Chromatin (C) has been displaced to the periphery of the nucleus. The nuclear membrane (M) is well demarcated.
Footnotes
a.
Diastase (α-amylase type VI-B from porcine pancreas), Sigma Chemical Co., St. Louis, MO.
b.
TAAB Laboratories Equipment Ltd., Reading, Berkshire, UK.
c.
Philips/FEI Corporation, Eindhoven, Holland.
