Abstract
A 7-y-old, castrated male, leucistic sugar glider (Petaurus breviceps) was presented because of a progressive history of lethargy, ataxia, diarrhea, and anorexia. Abdominal ultrasound revealed fluid in the abdomen and an infiltrative mass in the liver. Due to a poor prognosis, euthanasia was performed. Postmortem examination revealed a focally extensive, infiltrative, off-white, firm mass in the liver with adhesion to the omentum, mesentery, gastric serosa, and diaphragm. The remaining hepatic parenchyma was diffusely yellow. Histologically, the hepatic mass was consistent with metastatic cholangiocarcinoma (cholangiocellular carcinoma) with proliferation of neoplastic epithelial cells surrounded by marked desmoplasia. Neoplastic cells expanded and infiltrated the adjacent omentum, mesentery, and the serosal surfaces of the stomach, kidney, and small and large intestines. To our knowledge, cholangiocarcinoma has not been reported previously in a sugar glider.
A 7-y-old, 140-g, castrated male, leucistic sugar glider (Petaurus breviceps) was presented to a veterinarian because of anorexia, lethargy, ataxia, and diarrhea, and was treated with metronidazole and subcutaneous fluids. Abdominal ultrasound suggested the presence of a peritoneal effusion and a mass in the liver. The sugar glider’s condition deteriorated, the owner elected euthanasia, and an autopsy with histopathology was performed.
Grossly, the haircoat was diffusely pale-yellow, and the abdomen was markedly distended. A 3.0 × 2.0 × 1.0 cm, off-white, irregular, firm mass was expanding and effacing the right medial and lateral liver lobes (Fig. 1). On cut surface, the mass had infiltrated ~60% of the parenchyma of affected liver lobes. The adjacent omentum, mesentery, stomach, and loops of small and large intestine adhered to the mass. The remaining hepatic parenchyma was diffusely yellow, and the liver was firmly adhered to the diaphragm. The pleural cavity organs and the brain were grossly unremarkable.
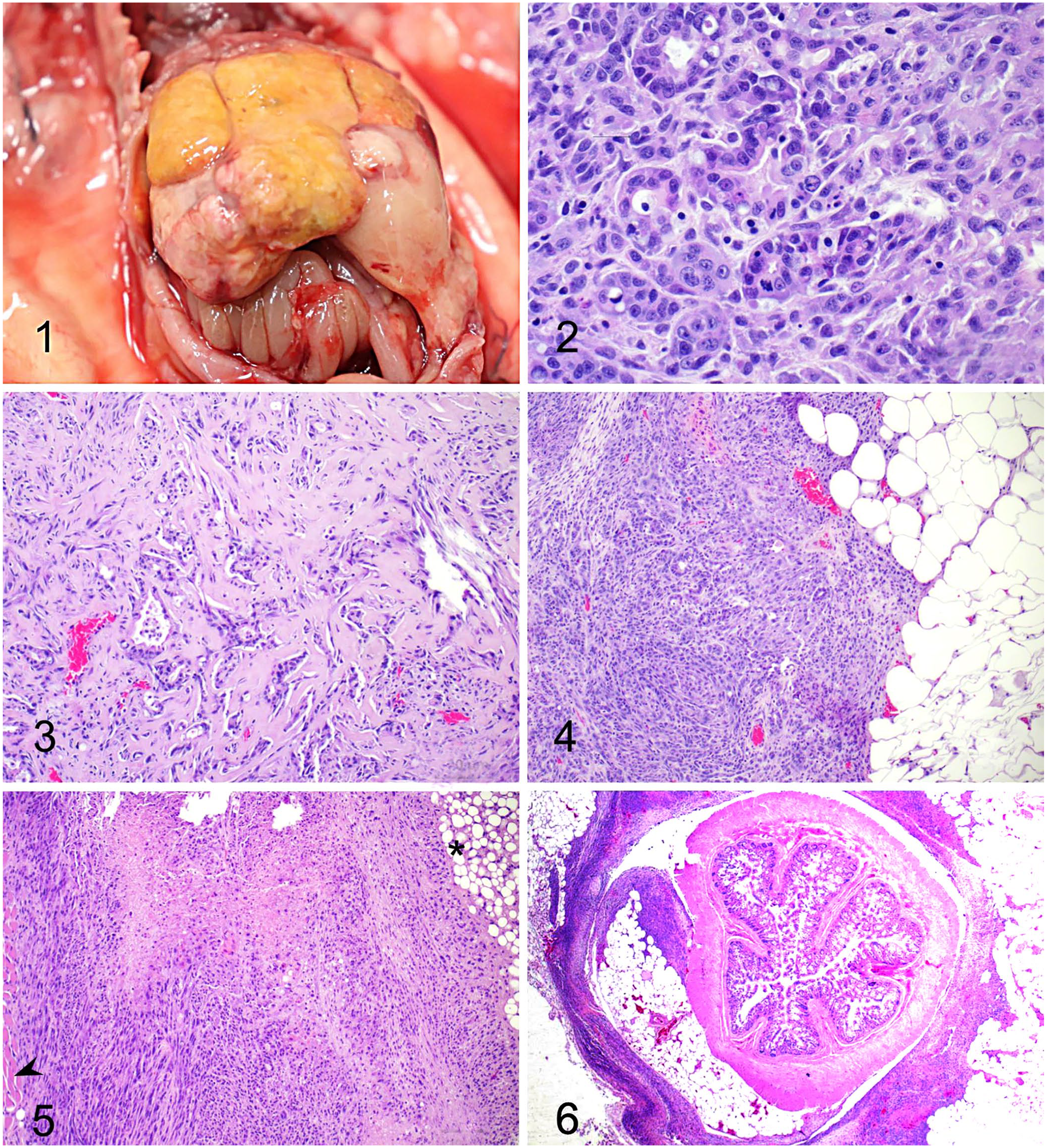
Cholangiocarcinoma in a sugar glider (Petaurus breviceps).
Samples of tissue were fixed in 10% neutral-buffered formalin, processed routinely, and 5-μm tissue sections were stained with H&E. Histologically, the hepatic mass consisted of an unencapsulated, poorly demarcated, infiltrative, densely cellular neoplasm composed of cuboidal-to-columnar cells arranged in irregular tubules and acini, occasionally filled with amorphous pale eosinophilic material, embedded in an abundant scirrhous stroma (Figs. 2, 3). Most neoplastic cells had indistinct cell borders, a moderate amount of pale eosinophilic cytoplasm, and round-to-elongated nuclei with finely stippled chromatin and 1–2 distinct nucleoli. There was moderate anisocytosis and anisokaryosis. The mitotic count was 4 in ten 400× fields (2.37 mm2). Multifocal areas of necrosis were present throughout the neoplasm. The remaining, compressed hepatic parenchyma had markedly swollen hepatocytes characterized by well-defined, clear cytoplasmic vacuoles that displaced the nucleus to the periphery (hepatic steatosis). The omentum (Fig. 4), gastric serosa, renal capsule, diaphragm (Fig. 5), mesentery, and intestinal serosa (Fig. 6) were multifocally expanded and infiltrated by neoplastic cells morphologically similar to those described in the liver. Histologic findings were consistent with a diagnosis of metastatic hepatic cholangiocarcinoma with peritoneal carcinomatosis. To rule out a combined hepatocellular cholangiocarcinoma, immunohistochemistry (IHC; CK7, HepPar1) was performed. On IHC, cells in the neoplastic lesions as well as the inner control were negative for CK7 (mouse monoclonal), which is likely due to nonreactivity against sugar glider biliary epithelium. In addition, neoplastic cells were negative for HepPar1 (mouse monoclonal); however, adjacent normal hepatocytes exhibited strong immunolabeling, which rules out hepatocellular differentiation. These findings, associated with classic microscopic features, support the diagnosis of cholangiocarcinoma.
Sugar gliders are nocturnal, arboreal marsupials native to Indonesia, Australia, and New Guinea. As the most common marsupial species in veterinary clinics, they have become popular pocket pets in the United States from the exotic pet trade, and the inability to accurately simulate their native arboreal environment makes them vulnerable to a number of husbandry-associated diseases. 10 Although sugar gliders are most often presented to veterinarians for diseases related to malnutrition, neoplastic disease does occur. Cases of neoplasia in sugar gliders, as well as exotic animals held in captivity in general, are increasing as they live longer lifespans than in the wild. Their average lifespan is 12 y in captivity, although they are considered geriatric when >8-y-old. 10 Reports of neoplasms in sugar gliders are scarce. Lymphoid neoplasia and adenocarcinoma (chest scent gland, duodenal, lung, mammary) are the most frequently described.3,4,10 One study reported that lymphoma accounts for ~50% of all documented cases of neoplasia in sugar gliders, most often affecting the liver and lymph nodes. 3 Another assessment of neoplasia prevalence in sugar gliders suggested that adenocarcinomas are the most common tumor type, making up 22% of cases, followed by lymphoma at 12% of cases. 14 The skin and subcutaneous tissue were the most common sites for neoplasia, followed by hepatocellular tumors. 14 In addition to lymphoma and adenocarcinoma, other tumors that have been documented in sugar gliders include biliary cystadenoma, cutaneous lymphoma, adrenocortical carcinoma, hepatocellular carcinoma, histiocytic sarcoma, urothelial carcinoma, mammary carcinoma, and dermal hemangiosarcoma.10,11,13,16 We conducted a literature search using Google, Scopus, and PubMed using various search terms, including “cholangiocarcinoma in sugar gliders,” “biliary tract neoplasia in Petaurus breviceps,” and related phrases. We found no documented cases of cholangiocarcinoma, suggesting that this condition has not been reported previously in sugar gliders. Neoplasia is infrequently reported in marsupials. Of all marsupial species, koalas have the most documented cases, with a high prevalence of lymphoma.4,8 There has also been a case of cholangiocarcinoma in a koala. 4 A survey of neoplasia in red kangaroos (Macropus rufus) showed that most tumors were malignant including mammary gland adenocarcinomas, which metastasized to the lymph nodes and lungs, lymphoma, and squamous cell carcinoma. 17
Cholangiocarcinomas are malignant tumors originating from bile duct epithelium and have been documented in dogs, cats, sheep, cattle, horses, and goats. 5 There have also been case reports in non-domestic species, including bats, a sloth bear, and a meerkat.1,6,7 Morphologically, they often occur as a large, single mass, but may also appear as multiple irregular nodules within the liver. Abundance of mitotic figures and fibrous connective tissue is distinctive for these tumors, and aids in its differentiation from biliary adenomas, biliary cysts, and hepatocellular carcinomas. The deposition of fibrous connective tissue observed throughout this neoplasm, referred to as a scirrhous response or desmoplastic reaction, is a feature of this neoplasm, gives it its firm texture, and helps to differentiate it from a hepatocellular carcinoma grossly.2,5 Metastasis to surrounding hepatic parenchyma and tissue, including by peritoneal seeding, is common, as observed in our case. These sites of invasion also involve scirrhous response and seeding of biliary epithelial cells.5,15 Antemortem diagnosis is difficult and uncommon across all species. In humans, dogs, and cats, cholangiocarcinoma has been linked to infectious origin (parasitic), cholelithiasis, and liver tissue damage.5,7,9
A potential predisposition for neoplastic transformation in the liver and bile duct is iron storage disease, which causes toxic hepatocellular changes, 11 although hepatic hemosiderosis was not present in our case. Cholangiocarcinoma in the Egyptian fruit bat (Rousettus aegyptiacus) is prevalent and has been associated with liver damage secondary to dietary excess of iron. Other lesions of chronic iron overload in R. aegyptiacus include portal-to-bridging fibrosis or cirrhosis, icterus, cavitary effusions, and histologic evidence of excess iron stores in other organs. 6 In humans, histologic lesions and genetic changes caused by certain congenital and developmental conditions are associated with an increased risk of developing cholangiocarcinoma. These lesions include nuclear changes, formation of a secondary gland, and positive reaction for carcinoembryonic antigen (CEA). 15 The most common condition that was associated with increased risk of cholangiocarcinoma was chronic cholangitis. These studies show promise for antemortem diagnosis and treatment, but currently, the cascade leading to cholangiocarcinoma formation is largely unknown. 15
Carcinomatosis often occurs with cholangiocarcinomas, but specific cases have not been reported in marsupials. Carcinomatosis has been observed in the western barred bandicoot (Perameles bougainville), secondary to infec-tion with bandicoot papillomatosis carcinomatosis virus 1 (BPCV1).12,18 The western barred bandicoot has a high prevalence of neoplasia, and cases of BPCV1 infection, which causes papillomas or carcinomas, are increasing. Lesions in the western barred bandicoot are multicentric and proliferative, affecting cutaneous and mucocutaneous tissue. 18 Spread of this syndrome has been suggested to be a result of the amount of captive breeding and potential inbreeding being done to help repopulate the species in the wild but which also can cause low genetic diversity. 12 Carcinomatosis with cholangiocellular carcinoma has been seen in a sloth bear, infiltrating to the serosal surface of the diaphragm, parietal tunic of the testicles, and the mesentery. 7
Statistical analysis of neoplasia in wild species is difficult to assess accurately, which can be partially attributed to a lack of postmortem examinations and case reports properly documenting neoplastic diseases. Neoplasia is a main determinant of fitness among all animals, making it essential to consider in conservation efforts. It is important to understand the prevalence of all diseases of wildlife, including neoplastic diseases, to estimate trends in morbidity and mortality. Animals with neoplastic disease are more susceptible to poor body condition, predation, and infectious disease. 12 Cases of neoplasia in captive wild species aid in this attempt by giving a better understanding of which neoplastic diseases affect wildlife, how the neoplastic diseases affect the survival of the animal, and how the conditions differ between captive and wild populations. An increasing prevalence of neoplasia may also be an early indicator of low genetic variability in a population, as seen with the western barred bandicoots. 12
Footnotes
Acknowledgements
We thank Dr. Dalen Agnew (Veterinary Diagnostic Laboratory, Michigan State University, E. Lansing, MI, USA) for performing the IHC as well as providing expertise in interpreting IHC in exotic species.
Declaration of conflicting interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
