Abstract
Confirmation of Borrelia burgdorferi infection in horses has required enzyme-linked immunosorbent assay (ELISA) or Western blot tests performed by reference laboratories. An in-clinic C6 ELISA SNAP kit has been marketed for dogs. This canine kit was evaluated for horses using serum from experimentally infected ponies. Serum samples originated from 2 previous studies. In the first study, 7 ponies were exposed to B. burgdorferi–infected ticks; 4 ponies served as uninfected controls. Serum samples were obtained bimonthly for 9 months. In the second study, 16 ponies were exposed to B. burgdorferi–infected ticks. After confirmation of infection by skin culture, polymerase chain reaction (PCR), and serology, the ponies were allocated to 4 groups that received tetracycline, doxycycline, ceftiofur, or no treatment. Serum samples were obtained monthly, both before and after antibiotic treatments, for 11 months. For the current study, selected samples (n = 220) from both studies were tested with IDEXX SNAP Heartworm Ab/Borrelia burgdorferi Ab/Ehrlichia canis Ab Test Kits. Tested samples included samples taken before infection, from various times postinfection, and after antibiotic treatments. Results from confirmed positive or negative samples were used to determine sensitivity and specificity of the assay. Results indicate that the test kits have fair sensitivity (63%) and very high specificity (100%) for horses recently infected with B. burgdorferi. Validation of this test provides equine practitioners with an inexpensive, in-clinic method to confirm infection, although its moderate sensitivity may result in a moderate chance of a false negative test.
Infection with Borrelia burgdorferi is increasingly recognized in horses and ponies, particularly in certain areas of the country. 12 Clinical signs attributed to B. burgdorferi infection include chronic weight loss, sporadic lameness, stiffness, arthritis, swollen joints, muscle tenderness, hepatitis, laminitis, fever, abortion, uveitis, and encephalitis. 2,3,12 With B. burgdorferi infection, diagnosis of clinical disease (borreliosis or Lyme disease) is challenging because of its variable clinical manifestations and serologic test limitations. Serologic diagnostic testing options include enzyme-linked immunosorbent assay (ELISA), indirect fluorescent antibody test (IFAT), and Western blot tests. These tests are only available at reference laboratories, increasing their cost and processing time, thus delaying final results. Therefore, veterinarians may use response to antibiotic therapy as a means of diagnosis, and many horses with suggestive signs are treated with oxytetracycline or doxycycline. Unfortunately, using the horse's response to treatment as an indicator of disease status is commonly unreliable because tetracycline antibiotics have anti-inflammatory effects in addition to their antibiotic effects. 16
Current research in human and canine serologic testing for Lyme disease is mainly focused on detection of antibodies against the C6 antigen. 1,7–9,11,13–15 This antigen is a short (25-amino acid sequence), synthetic peptide that reproduces the sequence of an immunodominant, conserved region (designated IR6) of a B. burgdorferi surface lipoprotein named Vls-E (Vmp-like sequence, expressed). 13 Assays using the C6 antigen have superior sensitivity and specificity when used for people and dogs. 1,11 A significant advantage of these assays is the ability to obtain accurate results in vaccinated animals because the C6 peptide-based ELISA is reportedly nonreactive with serum from patients vaccinated with the outer-surface protein A (OspA) or whole-cell (bacterin) Lyme disease vaccines. 13
Recently, a simple and quick in-clinic screening test a using the C6 antigen has been marketed for dogs in combination with heartworm antigen and Ehrlichia canis antibody tests. This test has quickly gained favor among small-animal practitioners based on its ease of use and its ability to distinguish vaccinated from infected animals. 8–10 Although marketed for dogs, the test is not species-specific in design and has been validated in cats. 10 The C6 synthetic peptide is conjugated to 2 substances: bovine serum albumin (BSA) and horseradish peroxidase (HRP). The BSA–C6 peptide conjugate is deposited to the porous flow matrix, whereas the HRP–C6 peptide conjugate is included in a conjugate diluent. The test sample is mixed with the conjugate diluent and the solution applied to the flow matrix. If anti-C6 antibodies are present in the sample, they bind to both the HRP–C6 peptide conjugate and the BSA–C6 peptide conjugate. The purpose of this study was to determine whether this screening test is also suitable for equine serum.
Serum samples originated from 2 earlier experimental pony studies as previously described. 4,6 Briefly, in the first study, 6 7 specific pathogen-free (SPF) ponies were exposed to B. burgdorferi–infected Ixodes scapularis ticks. Four ponies were uninfected controls. Serum samples were obtained at the time of tick exposure and then bimonthly for 9 months. In the second study, 4 16 SPF ponies were exposed to B. burgdorferi–infected ticks. Three months after tick exposure, after confirmation of infection, the ponies were allocated to 4 groups of equal size. Group 1 received tetracycline (5 mg/kg IV q 24 hr), group 2 received doxycycline (10 mg/kg PO q 24 hr), group 3 received ceftiofur (2.2 mg/kg IM q 24 hr), and group 4 received no treatment. Antibiotic treatment was maintained for 28 days. Serum samples were obtained at the time of tick exposure and then monthly for 11 months. Testing for B. burgdorferi in both studies included ELISA and Western blot on serum samples plus culture and polymerase chain reaction (PCR) on skin biopsies and postmortem samples. Infected ponies were identified by positive culture, PCR, and serology results, whereas uninfected ponies remained negative on all tests. Seven ponies in the treatment study were cleared of the infection. All serum samples were stored at −70°C after collection.
For the current study, selected samples (n = 220) from both studies were tested in accordance with the manufacturer's instructions using IDEXX SNAP Heartworm antigen/Borrelia burgdorferi antibody/Ehrlichia canis antibody test kits (hereafter SNAP). a One hundred eight samples from the first study were used; 9 different dates were chosen (before exposure to infected ticks and then monthly thereafter), and samples from all 12 ponies were evaluated for each date. One hundred twelve samples from the second study were used; 7 different dates were chosen (before exposure to infected ticks, 1 and 2 months after tick exposure, before initiation of antibiotic treatment, at conclusion of treatment, and 1 and 4 months after conclusion of treatment), and samples from all 16 ponies were evaluated for each date. The serum samples were thawed, and all samples and SNAP test components were at room temperature during the test procedure. The test was performed per manufacturer's instructions, and the device was left on a horizontal surface and read at 8 min. Any color development in the B. burgdorferi antibody-indicator spot constituted a positive result.
All SNAP test kits developed color in the positive control spot. No color development was seen in the heartworm antigen or E. canis antibody spots for any sample. Samples were divided into 3 groups for evaluation: group 1 = samples from uninfected ponies, group 2 = samples from the same ponies taken postinfection but before any treatments, and group 3 = samples from treated ponies taken posttreatment. Because of this allocation, the first 2 groups included samples from both experimental studies, whereas the third group included only samples from the second experimental study.
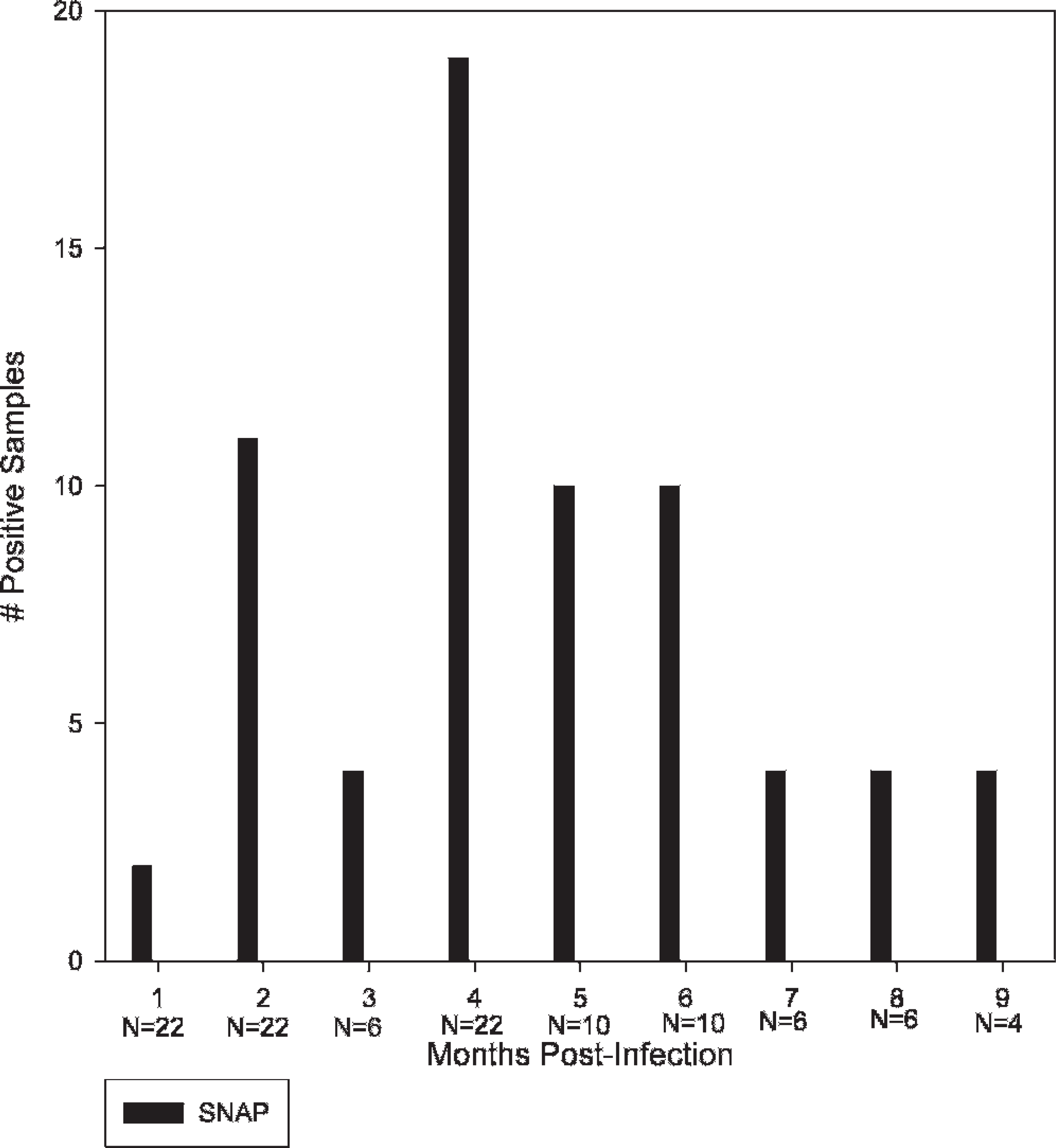
SNAP results on samples from infected ponies. Y-axis shows the number of positive samples. X-axis represents the number of months postinfection with Borrelia burgdorferi. N = number of samples tested for that month. IDEXX SNAP Heartworm Ag/Borrelia burgdorferi Ab/Ehrlichia canis Ab Test Kit–positive results for B. burgdorferi are shown in black.
In group 1, a total of 68 samples taken from 28 uninfected ponies were tested, including 52 samples from the first study (preinfection samples from 7 ponies plus 9 samples each from 5 ponies that were never infected with B. burgdorferi) and 16 preinfection samples from the second study. All 68 samples were SNAP-negative, yielding a specificity of 100%.
In group 2, a total of 108 samples taken from 27 ponies after infection were used in analysis, including 48 samples from the first study and 60 samples from the second study. All infected ponies were confirmed positive by culture and PCR. Sixty-three percent of these samples were SNAP-positive (68/108; Fig. 1). Samples from 2 ponies in the second study were SNAP-positive at earlier sampling times but SNAP-negative by the end of the study.
In group 3, 36 posttreatment samples were tested: 3 each from the 7 ponies that were successfully treated and 3 each from the 5 ponies that were unsuccessfully treated. Ponies successfully treated were negative for B. burgdorferi on PCR and cultures by the end of the study period, whereas unsuccessfully treated ponies remained positive. SNAP was negative for all successfully treated ponies (n = 7) and positive for all unsuccessfully treated ponies (n = 5) at the final test date (Fig. 2).
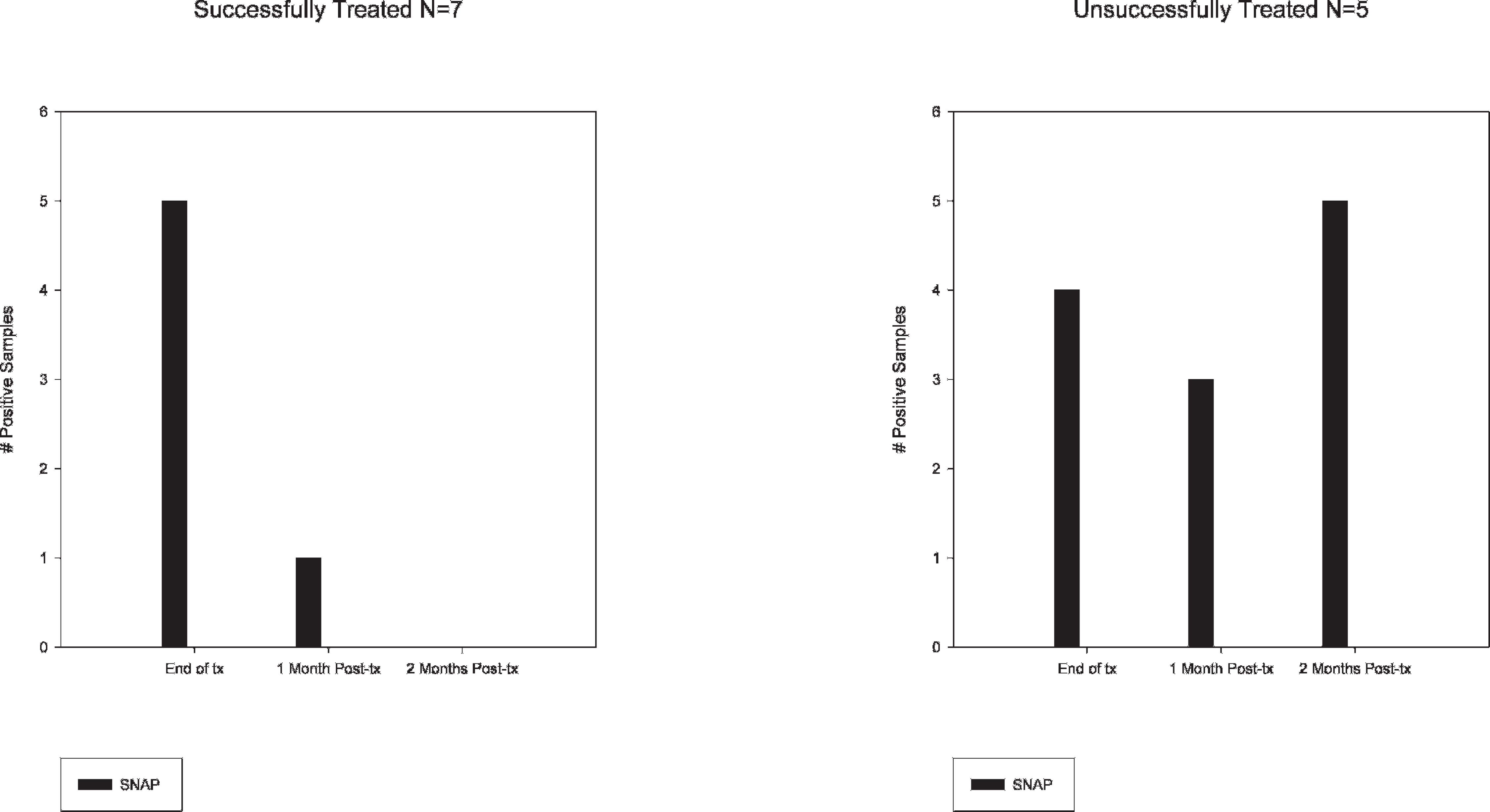
SNAP results on samples from treated ponies. The left graph shows tests performed on successfully treated ponies, whereas the right graph shows tests performed on unsuccessfully treated ponies. Y-axis shows the number of positive samples. X-axis represents the number of months after treatment. IDEXX SNAP Heartworm Ag/Borrelia burgdorferi Ab/Ehrlichia canis Ab Test Kit–positive results for Borrelia burgdorferi are shown in black.
In this experimental model, which used SPF ponies, the SNAP had excellent specificity (100%). Although the SNAP will likely also have a high specificity in naturally infected horses, false-positive results may occur and lower these values. The sensitivity (63%) is not as high as the specificity. The 63% sensitivity was lower than anticipated because results from canine studies indicate a SNAP sensitivity of 94.4%. 13 Possible explanations include the use of samples collected shortly after experimental infection, the assay not being optimized for horse serum, or a species-specific difference in anti-C6 antibody response.
The benefits of using a C6 ELISA as a screening test have been elaborated for dogs and cats. 8–10 In early infection in these species, the anti-C6 antibody response is more sensitive than the antibody response to whole-cell antigen. A study 11 with experimentally infected dogs demonstrated positive C6 ELISA results as early as 3 weeks postinfection, with all dogs demonstrating positive results by 5 weeks. Using visual inspection of the well, the results of the current study did not demonstrate such a rapid anti-C6 antibody response in horses. Once infected, serum samples from the dogs remained positive on the C6 ELISA for the duration of the study (69 weeks). 11 However, the ponies in the current study did not consistently remain SNAP-positive for the duration of the testing period. Two ponies in group 2 (experimentally infected and untreated) were SNAP-negative by the end of the study. These 2 ponies remained culture- and PCR-positive for B. burgdorferi, suggesting that the anti-C6 antibody response ceased despite persistent infection. However, the significance of this finding is unknown because none of the ponies developed clinical signs compatible with Lyme disease.
Another potential advantage of C6 technology is that anti-C6 antibodies appear highly specific for B. burgdorferi infection. In a previous study, 11 dogs infected with leptospirosis, Rocky Mountain spotted fever, babesiosis, ehrlichiosis, and heartworm disease did not have antibodies to C6 antigen. Furthermore, dogs vaccinated with either the OspA vaccine, a whole-spirochete vaccine, or common puppy vaccines had negative C6 ELISA results. 11,13 These findings allow evaluation of vaccine efficacy using the C6 ELISA kit. Horses are not routinely vaccinated against B. burgdorferi because a commercially produced vaccine is not currently available. However, 1 experimental study 5 has shown that vaccination with recombinant OspA protected ponies against infection after experimental challenge with B. burgdorferi–infected ticks. Should equine vaccination become more common, the authors anticipate that vaccinal antibodies will not interfere with SNAP testing.
Finally, recent reports 14,15 indicate that C6 technology may allow evaluation of successful antibiotic treatment of Lyme disease based on a decreasing titer. Human studies indicate that a decreasing titer is usually seen with successful treatment of early infection but not in cases of chronic infection despite extensive antibiotic treatment. 7 SNAP testing of experimentally infected ponies successfully treated with antibiotics (based on negative culture and PCR results by the end of the study) revealed that all ponies became negative on SNAP test. Practitioners in Lymeendemic areas often see horses with persistently positive ELISA results despite long-term antibiotic treatment. It is not known whether those cases represent failure of antibiotic therapy to eliminate the organism, reinfection with B. burgdorferi, or a persistent immune response despite successful treatment. Further research is needed to determine whether C6 technology will better define the infection status of these horses. For example, if a horse becomes negative for anti-C6 antibody on SNAP test but continues to have positive ELISA results, it could indicate that the infection has been successfully eliminated although the horse continues to produce antibodies that are not specific to viable B. burgdorferi. It has been hypothesized that Vls-E is a rapidly turned over antigen that disappears soon after spirochetal death and, therefore, is not well represented in follicular dendritic cells (FDCs). 15 Without Vls-E reaching the FDC reservoir, anti–Vls-E (C6) antibodies would not continue to be produced after spirochetal death. In contrast, whole-cell antigens are stored in FDCs and may continue to elicit antibody production after spirochetal death. Because ELISA measures antibodies against whole-cell antigens whereas SNAP is specific for anti-C6 antibodies, a positive ELISA result could result from B-cell memory, whereas a positive SNAP test should represent active infection. 15
Validation of the C6 SNAP test kits for horses provides practitioners with an inexpensive, in-clinic method to confirm B. burgdorferi infection. Results of the current study indicate that the test kits have a fair sensitivity and very high specificity for horses acutely infected with B. burgdorferi. Further research is needed to elucidate whether C6 technology will help to determine the reason that antibiotic therapy does not result in negative ELISA results in some naturally infected cases.
Acknowledgements. The authors would like to acknowledge the Harry M. Zweig Memorial Fund for Equine Research and the Jack Lowe Equine Research Fund for providing funding for the experimental pony studies; IDEXX Laboratories, Inc., for providing the SNAP tests used in this study; and Dr. Michael Alessi for his help with figures.
Footnotes
a.
SNAP 3Dx (SNAP Heartworm Ag/Borrelia burgdorferi Ab/Ehrlichia canis Ab Test Kit), IDEXX Laboratories Inc., Westbrook, ME.
