Abstract
Haemoproteus spp. are ancient apicomplexan hemoparasites that have undergone extensive coevolution with their natural hosts and are typically species specific, with inapparent or minimal pathogenicity. A promiscuous genotype of Haemoproteus capable of undergoing host switching on a familial level was identified. This protozoan caused severe disease with high mortality in 6 species of exotic passerine birds housed in California at the San Diego Zoo's Wild Animal Park: Surinam crested oropendola (Psarocolius decumanus decumanus), Guianan turquoise tanager (Tangara mexicana mexicana), blue-necked tanager (Tangara cyanicollis caeruleocephala, Guianan red-capped cardinal (Paroaria gularis gularis), magnificent bird of paradise (Diphyllodes magnificus hunsteini), and superb bird of paradise (Lophorina superba). The birds had few or no clinical signs. Necropsy findings consisted of hemocoelom and irregularly scattered areas of hemorrhage and hepatocellular necrosis. Affected areas of liver contained solitary protozoal megaloschizonts in varied states of degeneration and peripheral nonsuppurative inflammation. No other parasite life stages were found in parenchymal organs or blood smears. Polymerase chain reaction using consensus primers for an avian malarial mitochondrial cytochrome B gene segment was positive in all cases. Sequencing and BLAST analysis identified the protozoan as a Haemoproteus sp. related to Haemoproteus spp. found in asymptomatic passerine birds native to North America. In situ hybridization was performed in 3 animals with a mitochondrial cytochrome B probe and was positive only in megaloschizonts. These findings suggest the recognition of a genotype of Haemoproteus that exhibits high levels of host infidelity and causes severe disease in captive birds exotic to North America.
Introduction
Hemoparasitism in birds is typically caused by 3 genera of apicomplexan parasites in the family Plasmodiidae: Leukocytozoon, Haemoproteus, and Plasmodium. 6 These protozoa can cause severe clinical disease, but, in many cases, hosts are asymptomatic. Haemoproteus and Plasmodium are distributed almost worldwide 25 with high species diversity. Over 120 species of Haemoproteus have been reported in birds. 6 Haemoproteus spp. are characterized by schizogony (i.e., merogony) within visceral endothelial cells, typically of the lung, liver, or spleen, and gametocyte development in circulating erythrocytes. 6,7 The organisms are transmitted by biting flies, characteristically louse flies (Hippoboscidae) and biting midges (Ceratopogonidae). Sexual stages of Haemoproteus occur in the intermediate host (i.e., insects) with asexual stages in the bird. 7
Haemoproteus infection is usually subclinical but can cause mild clinical signs. 9,15,16 When clinical disease occurs, it is typically associated with anemia because of erythrocytic parasitism, 8 frequently in immunocompromised hosts. Few species of Haemoproteus are reported to induce clinical disease, including Haemoproteus meleagridis, Haemoproteus nettionis, and Haemoproteus columbae, in turkeys, ducks and geese, and pigeons and doves, respectively. 8,19 However, recent reports suggest that Haemoproteus may be a more significant pathogen than previously considered. Compromised host fitness, myopathy, myositis, myonecrosis, and mortality were reported in poultry, quail, and columbiformes with Haemoproteus infections. 2–4,8,9 Populations of wild passerine birds infected with Haemoproteus had greater infection intensity, significant reductions in fledgling success, and elevated nestling mortality compared with other infected birds that were treated with antimalarial medications. 16
Another manifestation of Haemoproteus-associated disease occurs with the pre-erythrocytic stages, rather than circulating intra-erythrocytic gametocytes, which result in megaloschizont-associated lesions. Megaloschizonts can occur in a variety of cell types, and their presence causes tissue damage. Proposed mechanisms of injury include interference with circulation, host-cell destruction via release of merozoites, or release of toxins. 4 In the current study, the periphery of the megaloschizonts were packeted into structures called cytomeres. Generally, zoites develop from cytomeres, and, as the megaloschizonts become more mature, the zoites are released and infect erythrocytes. It should be noted that endothelial schizonts found with Haemoproteus parasitism are distinct from the megaloschizonts described in the current study.
The current study's gross and histologic findings may represent a disease entity that has been previously described but incompletely understood. The identification of these liver protozoa as Haemoproteus is unique in that both polymerase chain reaction (PCR) and in situ hybridization were performed. These cases have important implications for captive bird populations, particularly in a zoological setting, in which multiple species from different regions of the world are brought to foreign locations and exposed to pathogens not endemic to their habitat.
Materials and methods
Case material
Eleven passerine birds housed at the San Diego Zoo's Wild Animal Park were included in this study. All birds except for 2 Surinam crested oropendolas and 1 Guianan turquoise tanager were housed in the bird breeding center (BBC) located at the periphery of the San Diego Zoo's Wild Animal Park and were not accessible to the public. The breeding center contained indoor/outdoor enclosures and was composed of several buildings. Affected birds from the present study inhabited multiple buildings within the breeding center, with no apparent pattern of building distribution. Many affected birds were housed with several other avian species that did not exhibit any clinical signs. Enclosures at the breeding center contained foliage, were lined with fine dirt, and were equipped with receptacles for food and water. Water was distributed as a fine mist throughout the day and did not accumulate. Approximately 75 yards away from the breeding center was a shoebill enclosure, within which was a man-made pond. Two Surinam crested oropendolas were housed at BOTB (Birds of the Bridge), an enclosure on zoo grounds, far away from the BBC, which housed new world species, and contained vegetation and an irrigation system. One Guianan turquoise tanager was recently relocated (5 days previously) from the BBC but was housed at the Bird Brooding Trailer (BBT), an enclosure on zoo grounds far away from the BBC, when found dead. Lawns were present within the BBT, with no known source of standing water. All birds were hatched in captivity at the San Diego Zoo or the Wild Animal Park, except for the magnificent bird of paradise, hatched in Honolulu, and 1 Guianan red-capped cardinal, which was wild caught from South America.
All birds underwent a thorough necropsy, in which representative tissue samples from the skin, heart, trachea, bronchi, lungs, air sacs, oral cavity, esophagus, crop, proventriculus, ventriculus, small intestine, cecum, colon, cloaca, liver, gall bladder, pancreas, spleen, bone, kidney, gonads, muscles, brain, peripheral nerve, thyroid, adrenal gland, eye, thymus (if present), and bursa (if present) were obtained and fixed in 10% neutral buffered formalin. In all cases, impression smears of the lungs were prepared (as a reflection of peripheral blood), and, in some cases, impression smears of the liver and/or spleen were prepared. When possible, portions of fresh liver, spleen, and kidney were frozen at −70°C. The following bird species were represented: Guianan red-capped cardinal (Paroaria gularis gularis; n = 4), Surinam crested oropendola (Psarocolius decumanus decumanus; n = 2), Guianan turquoise tanager (Tangara mexicana mexicana; n = 2), blue-necked tanager (Tangara cyanicollis caeruleocephala; n = 1), magnificent bird of paradise (Diphyllodes magnificus hunsteini; n = 1), and superb bird of paradise (Lophorina superba; n = 1).
Light microscopy
Fixed tissues from the organs listed above were trimmed, embedded in paraffin, sectioned at 5 μm, and stained with hematoxylin and eosin. Liver sections from all cases were stained with Prussian blue for iron. Histologic findings from each case were described as morphologic diagnoses and are summarized in Table 1.
DNA isolation
DNA was extracted from 20 mg of frozen tissues or formalin-fixed tissues in paraffin blocks by using the Qiagen tissue kit a according to the manufacturer's tissue-sample protocol, except that the recommended amounts of sample were first placed with the lysis buffer in 1.5-ml screw-cap FastPrep vials that contained ceramic beads and were lysed by agitation in a FastPrep shaker b at a speed of 4–5.5 m/sec for 40–60 sec. The lysate was then transferred to a clean Eppendorf tube for continuation of the Qiagen protocol. The DNA was extracted from paraffin-embedded tissue by using the Qiagen DNeasy kit, a except that the tissues were first deparaffinized with octane and xylene followed by washes with 100% ethanol.
Polymerase chain reaction
Three PCR assays were performed on DNA from frozen and formalin-fixed tissues. The 3 assays targeted different genes and were used to increase the sensitivity for detecting all hemoparasites present in the samples. The PCR protocols and expected sizes of the amplicons are listed below. For all reactions, 50–500 ng of DNA was added to a 25-μl reaction mixture that contained 10 mM of Tris (pH 8.0), 50 mM of KCl, 5 mM of MgCl2, 200 μM each of dATP (2'-deoxyadenosine-5'-triphosphate), dCTP (2′-deoxycytidine-5′-triphosphate), dGTP (2′-deoxyguanosine-5′-triphosphate), and dTTP (2′-deoxythymidine-5′-triphosphate), 50 pmol of each primer, and AmpliTaq Gold DNA Polymerase at a final concentration of 0.05 U/μl. The following PCR assays were used: 1) malarial (Haemoproteus and Plasmodium spp.) protozoa cytB gene segment: sense primer (5′-ATGGTGCTTTCGATATATGCATG-3′) and antisense primer (5′-GCATTATCTGGATGTGATAATGGT-3′) were used with cycling conditions of 8 min at 95°C, followed by 35 cycles of 94°C for 30 sec, 55°C for 1 min, and 72°C for 45 sec, followed by a final extension at 72°C for 10 min to generate a 525-bp (base pair) product as described 5 ; and 2) parasite chromosomal small subunit (16s) ribosomal ribonucleic acid (rRNA): sense primer (5′-AAGTATAAGCTTTTATACGGCT-3′) and antisense primer (5′-CACTGCCACGGTAGTCCAATAC-3′) were used at 95°C for 6 min, followed by 35 cycles of 95°C for 45 sec, 54°C for 1 min, 72°C for 1 min, and then 72°C for 8 min to generate a 295-bp product as described 13 ; and 3) parasite apicoplast large subunit (23s) rRNA: sense primer (5′-GCGAAATTCCTGTCGGGTAAGTCC-3′) and antisense primer (5′-TTTYYGRTCCTCTCGTACT-3′) were used at 95°C for 6 min, followed by 35 cycles of 95°C for 45 sec, 50°C for 1 min, 72°C for 1 min, and then 72°C for 8 min to generate a 720-bp product as described. 26
Case summaries.
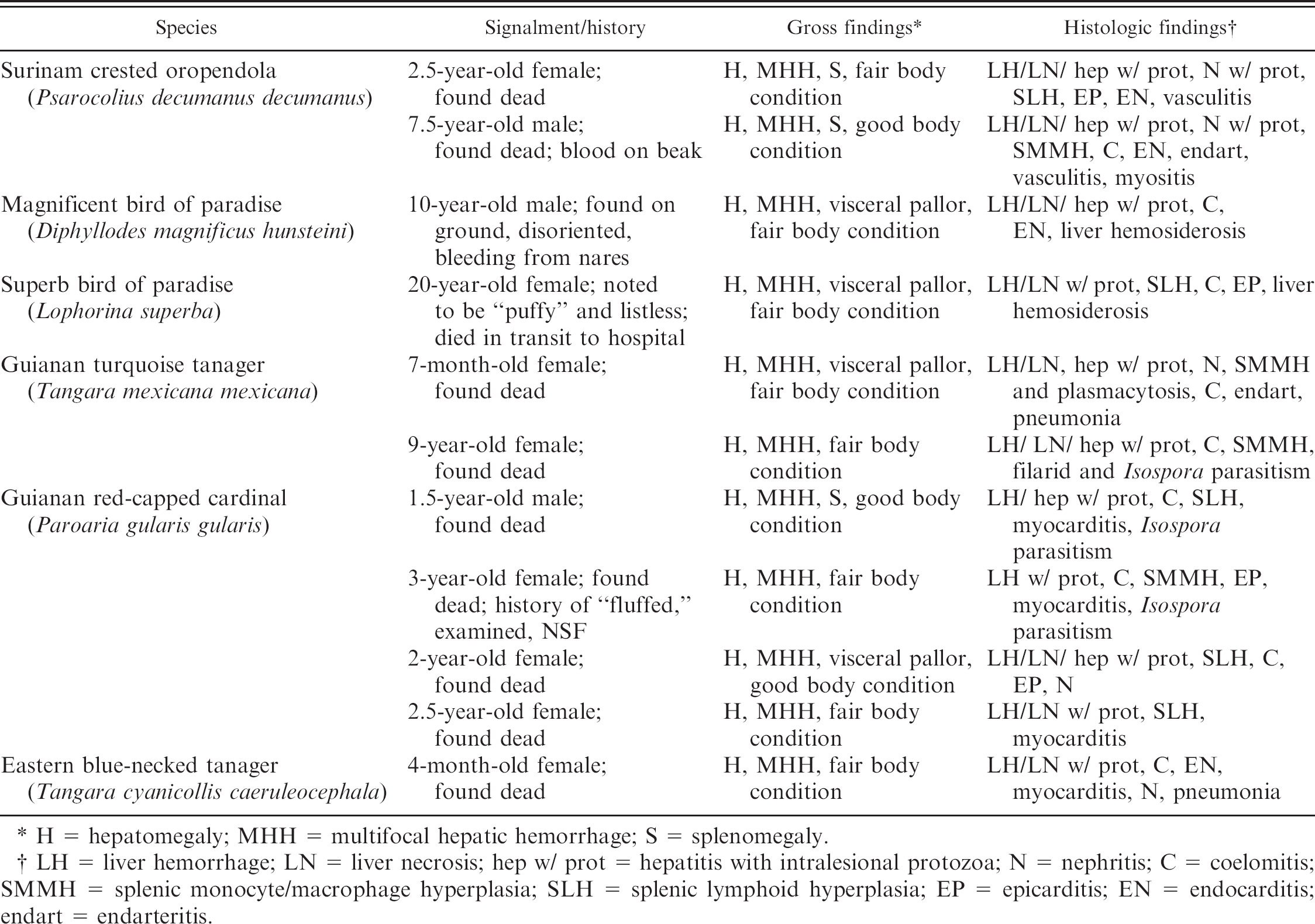
H = hepatomegaly; MHH = multifocal hepatic hemorrhage; S = splenomegaly.
LH = liver hemorrhage; LN = liver necrosis; hep w/ prot = hepatitis with intralesional protozoa; N = nephritis; C = coelomitis; SMMH = splenic monocyte/macrophage hyperplasia; SLH = splenic lymphoid hyperplasia; EP = epicarditis; EN = endocarditis; endart = endarteritis.
DNA manipulation and sequencing
Polymerase chain reaction products of the expected size from all cases were purified and cloned by using the TOPO TA cloning kit. c Sequencing reactions were performed by using the CEQ DTCS (dye terminator cycle sequencing) Quick-Start kit. d Sequences were acquired by using a CEQ 2000XL capillary sequencer d and were analyzed with the MacVector program. e GenBank searches and comparisons were done with the Basic Local Alignment Search Tool (BLAST; http://www.ncbi.nlm.nih.gov/BLAST/). Novel DNA sequences were deposited into GenBank.
Phylogenetic analyses
Sequences were aligned with the ClustalW program. f Phylogenetic relationships were estimated from ClustalW alignments by using the program PAUP* 4.0 g , 23 with neighbor-joining, maximum likelihood (substitution and among-site rate variation models), and parsimony methods. Bootstrap values were counted as percentages over 1,000 replicates.
In situ hybridization
Formalin-fixed, paraffin-embedded tissues were sectioned at 5 μm and mounted on platinum slides. h Sections were deparaffinized, hydrated, treated with Digest-All i at 37°C for 10 min, washed in Tris buffered saline solution (TBS), heated at 98°C for 12 min in TBS, immediately placed in 4°C TBS, and prehybridized with Dig Easy Hyb Granules j at 42°C for 1 hr. A 525-bp segment of the cytochrome B gene was labeled with digoxigenin by using an incorporation method j and used in the Dig Easy Hyb Granule solution for hybridization at 42°C for 16 hr at 50 pmol of labeled probe per milliliter of hybridization solution. Slides were washed in 2X sodium chloride/sodium citrate buffer (SSC) with 0.1% sodium dodecyl sulfate (SDS) 2 times for 5 min at room temperature (RT), followed by 2 washes in 0.5X SSC with 0.1% SDS at 68°C for 15 min. Slides were blocked in blocking solution j for 30 min, washed in TBS, treated with anti-digoxigenin antibody j diluted 1:5000 (150 mU/ml) in TBS for 1 hr at RT, washed in TBS, treated with nitro blue tetrazolium chloride/5-bromo-4-chloro-3-indolyl phosphate (NBT/BCIP), toluidine salt j for 10 min, washed in TBS and distilled water, counter-stained with Gill Hematoxylin k and mounted with Crystal/Mount. l Duplicate control slides received identical treatment, except that no labeled probe was added to the hybridization solution. Additional negative control slides consisted of tissues infected with avian Isospora spp.
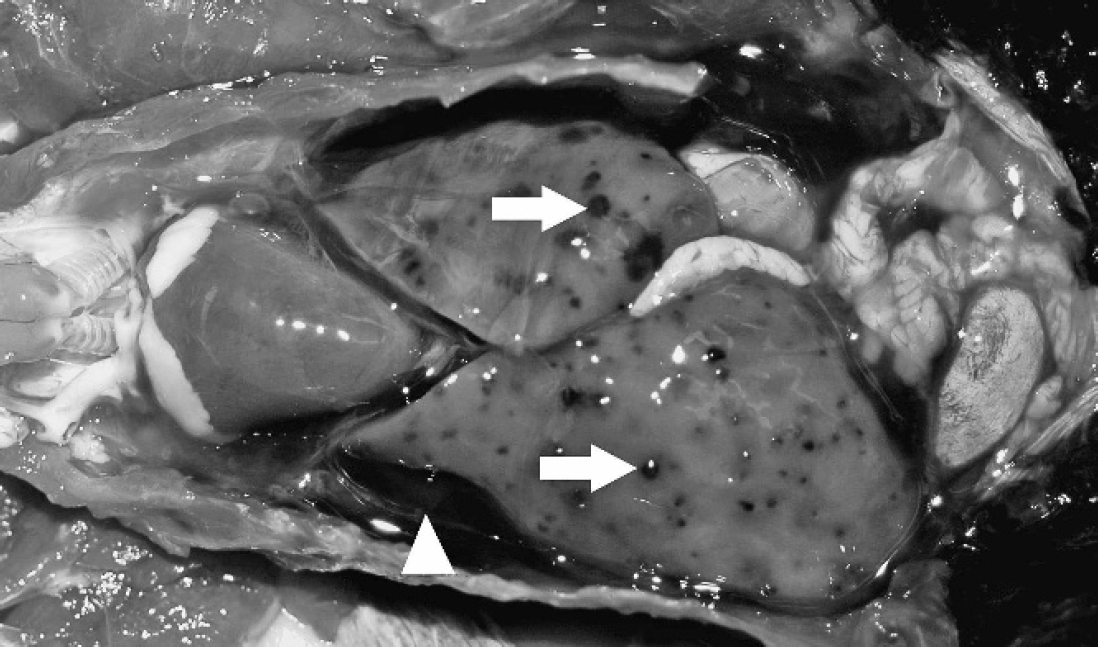
Coelomic viscera from a Surinam crested oropendola. Note free blood in coelom (white arrowhead) and multifocal hemorrhages throughout the liver (white arrows).
Results
Signalment, history, and gross and histologic findings are summarized in Table 1. Of the species affected, 9 of 11 were species from South America. Ages ranged from 4 months to 20 years, with no sex predilection. Historical information was most commonly recorded as “found dead.” One bird (red-capped cardinal) exhibited nonspecific premonitory signs (fluffed), and physical examination was unremarkable. Another bird (magnificent bird of paradise) was found on the ground, was described as disoriented and died a few hours later. The superb bird of paradise was described as listless 12 hr before death. Body condition based on body fat stores was fair (8/11) or good (3/11). In all cases, gross necropsy findings consisted of variable amounts of clotted and unclotted blood in the coelomic cavity (hemocoelom). Livers were discolored tan to red, green to red, or were diffusely pale tan. Multifocally throughout the liver parenchyma, dark red to black foci were randomly scattered, which ranged from pinpoint to 5 mm in diameter (hemorrhages; Fig. 1). In some cases, hematomas were present within liver lobes, which ranged from 3–5 mm in diameter. Splenomegaly was noted in 3 cases, and, in 2 of those cases, the splenic parenchyma was described as dark red to purple. Pallor of viscera was noted in 4 cases. Hemorrhage from the beak or nares was found in 2 birds (Surinam crested oropendola and magnificent bird of paradise).
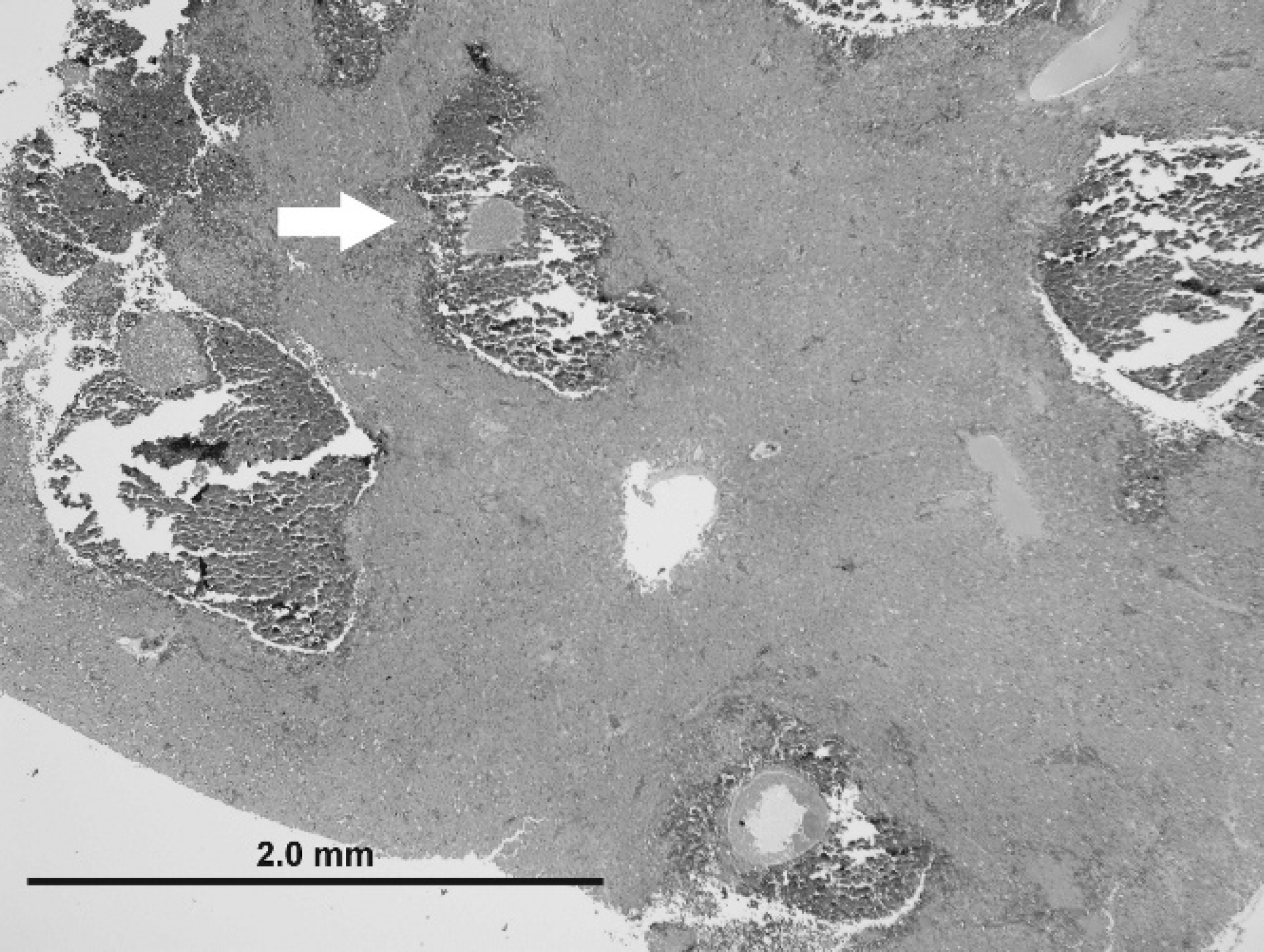
Liver from a magnificent bird of paradise. Low magnification view of protozoal megaloschizonts (white arrow) within regions of hemorrhage and hepatocellular degeneration. Hematoxylin and eosin. Bar = 2 mm, magnification 4X.
On histologic examination, the liver parenchyma in all 11 cases was multifocally disrupted by variably sized poorly demarcated regions of hemorrhage and necrosis, which ranged from 0.5–2 mm in diameter, typically with intralesional protozoal organisms (megaloschizonts; Fig. 2). Protozoal megaloschizonts were spherical and ranged from 200–500 μm in diameter, and consisted of a 1–2-μm-wide basophilic wall bordering a 30–60-μm–thick peripheral basophilic rim. This rim delineated the perimeter of the megaloschizont and was composed of numerous, round-to-oval, often poorly preserved, coalescing basophilic 1–3 μm in diameter merozoites and larger cytomeres (Figs. 3, 4). Cytomeres ranged from 6–15 μm in diameter, averaged 12 μm in diameter, and often contained clear acicular clefts (Fig. 4). Central vacuolated regions of the megaloschizonts contained amorphous, wispy, amphophilic-to-eosinophilic material (Fig. 3). Rare megaloschizonts were composed entirely of well-demarcated cytomeres (Fig. 4). In a few locations, amorphous basophilic-to-amphophilic material that resembled the protozoal zoites conformed to and expanded hepatic sinusoids. Distorted or degenerate megaloschizonts were often only identifiable as serpiginous remnants of megaloschizont walls within a region of hemorrhage or parenchymal disruption. Surrounding and intermixed within the regions of hemorrhage were variable numbers of inflammatory cells in 7 of 11 birds, including lymphocytes, macrophages, plasma cells, few heterophils, and multinucleate giant cells. Hepatocytes that surrounded the parenchymal hemorrhage exhibited degenerative and necrotic features characterized by hypereosinophilia, anisocytosis, anisokaryosis, pyknosis, karyolysis, and karyorrhexis (9/11). In some cases (10/11), fine golden brown anisotropic pigment was commonly noted in regions of hemorrhage but was also found within or adjacent to hepatocytes and macrophages. This pigment did not stain with Prussian blue (interpreted as protozoal pigment or artifactual acid hematin; Fig. 4). Inflammatory cell populations similar to those listed above were randomly disseminated or present at portal regions (7/11).
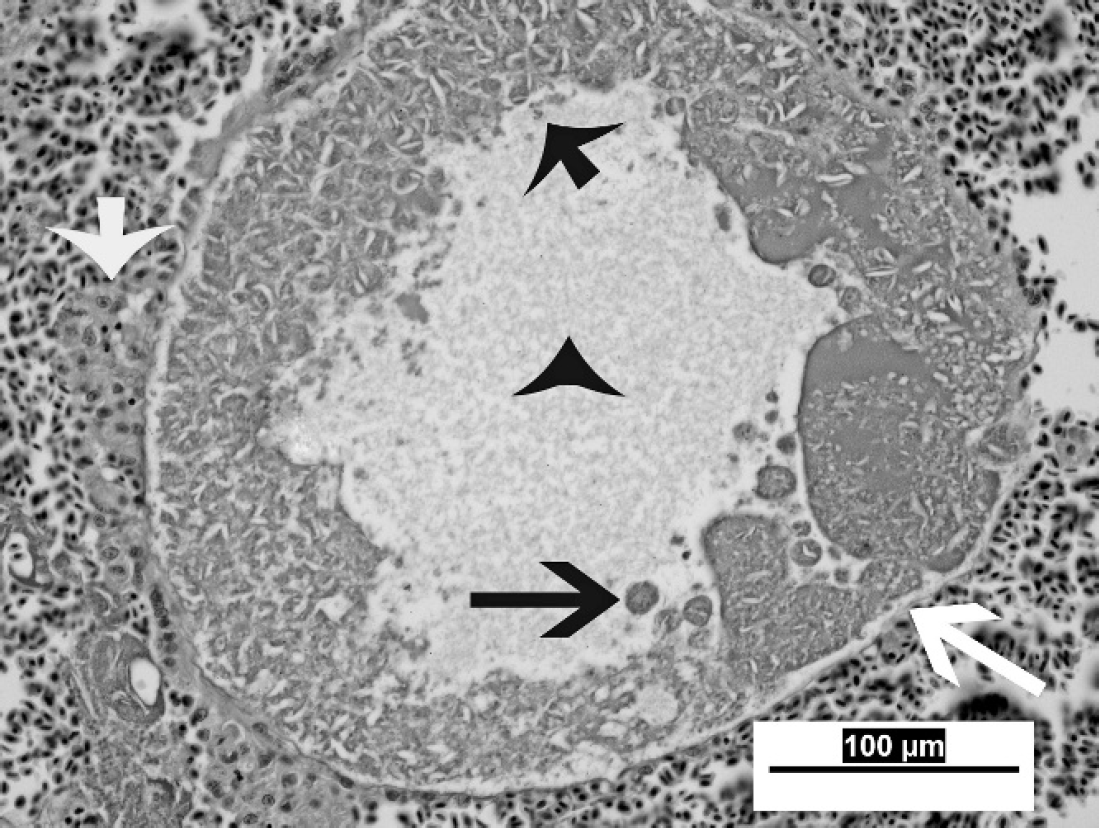
Liver from a magnificent bird of paradise. Higher magnification of a protozoal megaloschizont, approximately 350 μm in diameter. Note the thin, 1–2-μm membrane that surrounds the schizont (long white arrow), the central region of vacuolation (black arrowhead), and nonsuppurative inflammation surrounding the schizont (short white arrow). Merozoites that comprise the schizont wall exhibit variable morphologic preservation, range from 1–3 μm in diameter (short black arrow) and occasionally form 6–15 μm in diameter cytomeres (long black arrow). Hematoxylin and eosin. Bar = 100 μm, magnification 40X.
Occasionally, protozoa were observed in the kidney (2/11) with accompanying lymphoplasmacytic interstitial nephritis. Lymphoplasmacytic heterophilic interstitial nephritis was found in 3 additional cases, and, in one of these 3 cases, tubulointerstitial nephritis, tubular degeneration, and necrosis were noted. Mild-to-moderate nonsuppurative myocarditis, endocarditis, or epicarditis were observed in 9 of 11 cases. Lymphocytic and lymphoplasmacytic endarteritis or vasculitis was observed in 3 of 11 cases, with endothelial hypertrophy noted in the lung of another case (Surinam crested oropendola), with a possible intravascular megaloschizont. Splenic monocyte/macrophage hypertrophy/hyperplasia (4/11) and splenic lymphoid hyperplasia (5/11) were noted. Mild-to-moderate lymphoplasmacytic and histiocytic inflammation of the coelom and associated structures was common (9/11). Lung cytology revealed protozoal organisms consistent with isosporoid coccidia (formerly Atoxoplasma) 22 in 3 cases. Pneumonia, pulmonary edema, and pulmonary hemorrhage were each diagnosed in 2 of 11 cases. Lymphohistiocytic myositis was typically mild but noted in 3 of 11 cases, with focal myodegeneration in 1 case. Both birds of paradise had hepatocellular hemosiderosis (confirmed with Prussian blue stains). Lung impression smears for all 11 birds, liver impression smears for 6 birds, and splenic impression smears for 3 birds were negative for intra-erythrocytic protozoa.
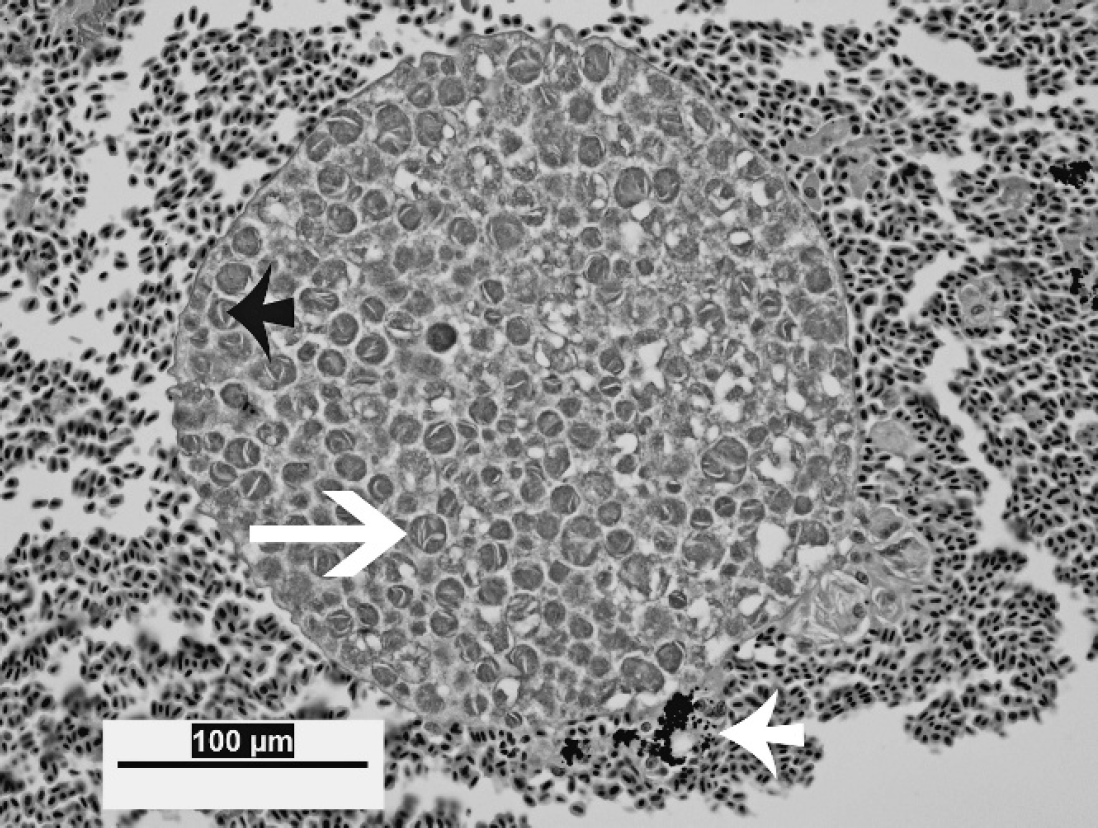
Liver from a magnificent bird of paradise. High magnification view of a megaloschizont, approximately 250 μm in diameter. The schizont is well preserved and composed of cytomeres (long white arrow), which range from 6 to 15 μm in diameter. Acicular clefts are commonly present within cytomeres (short black arrow). Note intracellular and extracellular fine, dark pigment in areas of hemorrhage (short white arrow), which does not stain with Prussian blue. Hematoxylin and eosin. Bar = 100 μm, magnification 40X.
Eleven birds were tested by PCR; 10 of the animals had frozen liver samples available for analysis, and all were positive by PCR for Haemoproteus. The single negative animal, a Surinam crested oropendola, had only formalin-fixed liver and frozen spleen available for evaluation. These samples were negative by PCR. Other organs, including spleen, kidney, heart, brain, intestine, and lung, were tested in several animals, and Haemoproteus organisms were variably detected in spleen and lung. Birds were negative for other hemoparasites by PCR, including Leukocytozoon and Plasmodium. Isospora spp. (junior synonym Atoxoplasma) were found in 2 Guianan red-capped cardinals. Findings are summarized in Table 2.
Polymerase chain reaction (PCR) results.
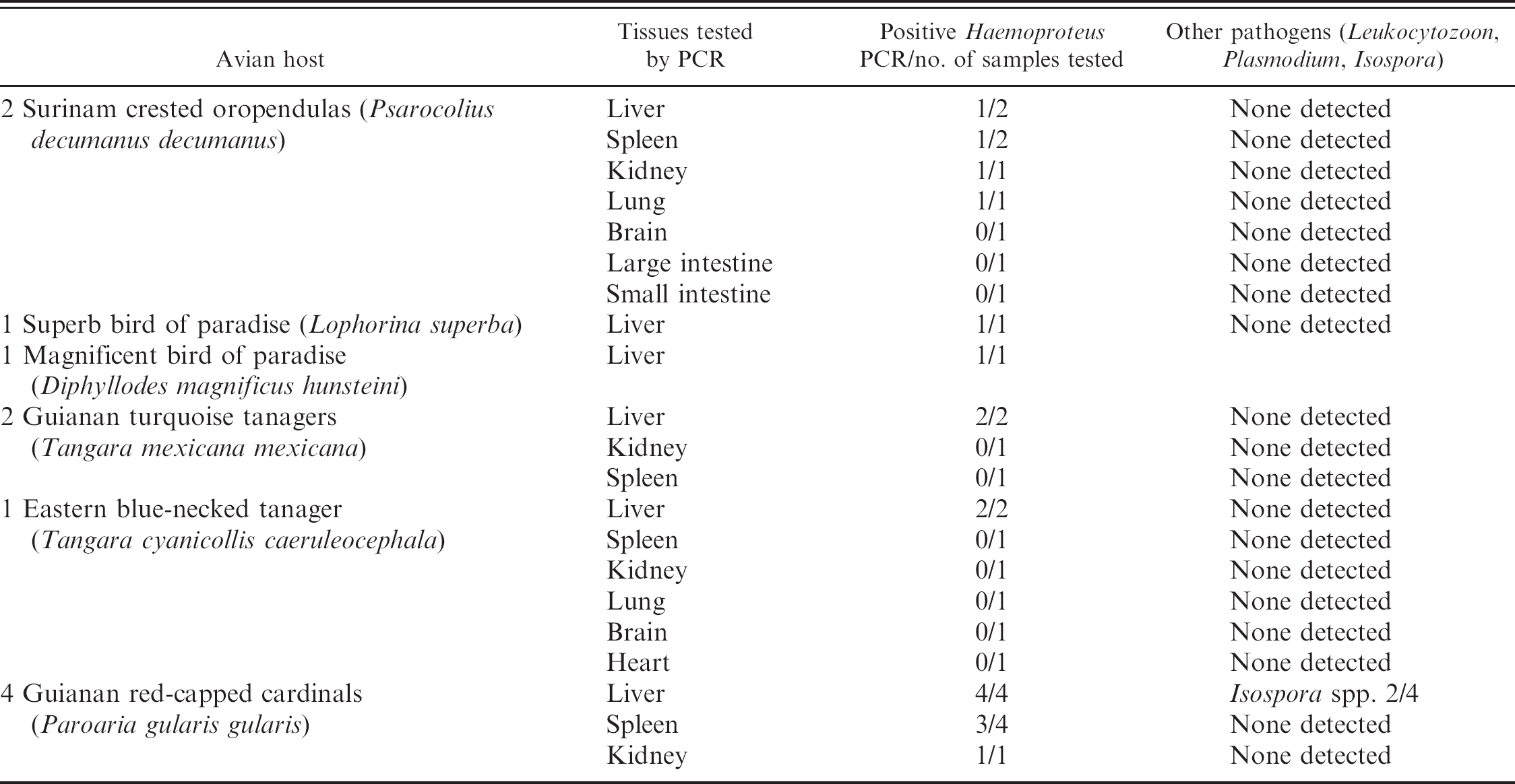
Cloning and DNA sequencing of the Haemoproteus PCR amplicons showed that all 10 positive birds had the same genotype of Haemoproteus (GenBank accession numbers EU327512–EU327517) and that the genotype had 100% identity with a Haemoproteus species recently described from Texas. 10 Phylogenetic analysis grouped the Haemoproteus spp. with other Haemoproteus spp. described in birds native to North America (Fig. 5). Polymerase chain reaction for the apicoplast rRNA gene was also positive in all 10 birds, and all amplicons were identical (GenBank accession numbers EU327518–EU327523).
In situ hybridization was positive in the livers of 3 of 3 (2 Guianan red-capped cardinals and 1 Guianan turquoise tanager) birds tested. Hybridization was specific for cytomeres and merozoites within hepatic lesions (Fig. 6). No hybridization occurred in the negative controls.
Discussion
This report describes 11 cases of protozoal infection in passerine birds that caused sudden death with little or no premonitory signs. Molecular diagnostic tests revealed that the protozoa in 10 cases were consistent with Haemoproteus spp. Haemoproteus is typically considered to be a benign hemoparasite, which causes little or inapparent clinical disease. 9,15 When clinical disease because of Haemoproteus parasitism occurs, it is more commonly associated with anemia because of the erythrocytic stages of parasitism. 8 However, the current study illustrates mortality due to pre-erythrocytic, megaloschizont stages, which resulted in hepatic hemorrhage, necrosis, and hepatitis with hemocoelom. These findings suggest the recognition of a novel, malignant manifestation of Haemoproteus that causes severe disease in captive passerine birds exotic to North America.
The gross lesions in all 11 cases were particularly striking, because foci of hemorrhage were disseminated throughout the hepatic parenchyma with blood in the coelom (Fig. 1). Large protozoal megaloschizonts were present within regions of hepatic hemorrhage (Fig. 2). Severe blood loss into the coelom was the proposed mechanism of sudden death. Other lesions consistently present included coelomitis and airsacculitis, possibly secondary to widespread distribution of the parasite or a reaction to the coelomic hemorrhage. Splenomegaly with macrophage/monocyte hyperplasia and/or lymphoid hyperplasia most likely represents a systemic immune response to the protozoal infection. Also common were nonsuppurative endocarditis, epicarditis, myocarditis, and occasional endarteritis, possibly because of transient, widespread endothelial parasitism. Alternatively, inflammation in the heart may have been an extension of the coelomitis. Concurrent liver disease not associated with protozoa was present in some of the birds, manifest as diffuse hemosiderosis or portal inflammation. Lung impression smears were examined in all cases as a representation of peripheral blood, and liver and spleen impression smears were examined in some cases. No intra-erythrocytic hemoparasites were found. Isosporoid coccidia (formerly Atoxoplasma) 22 were noted in lung impression smears from 3 birds but were interpreted as an incidental finding.
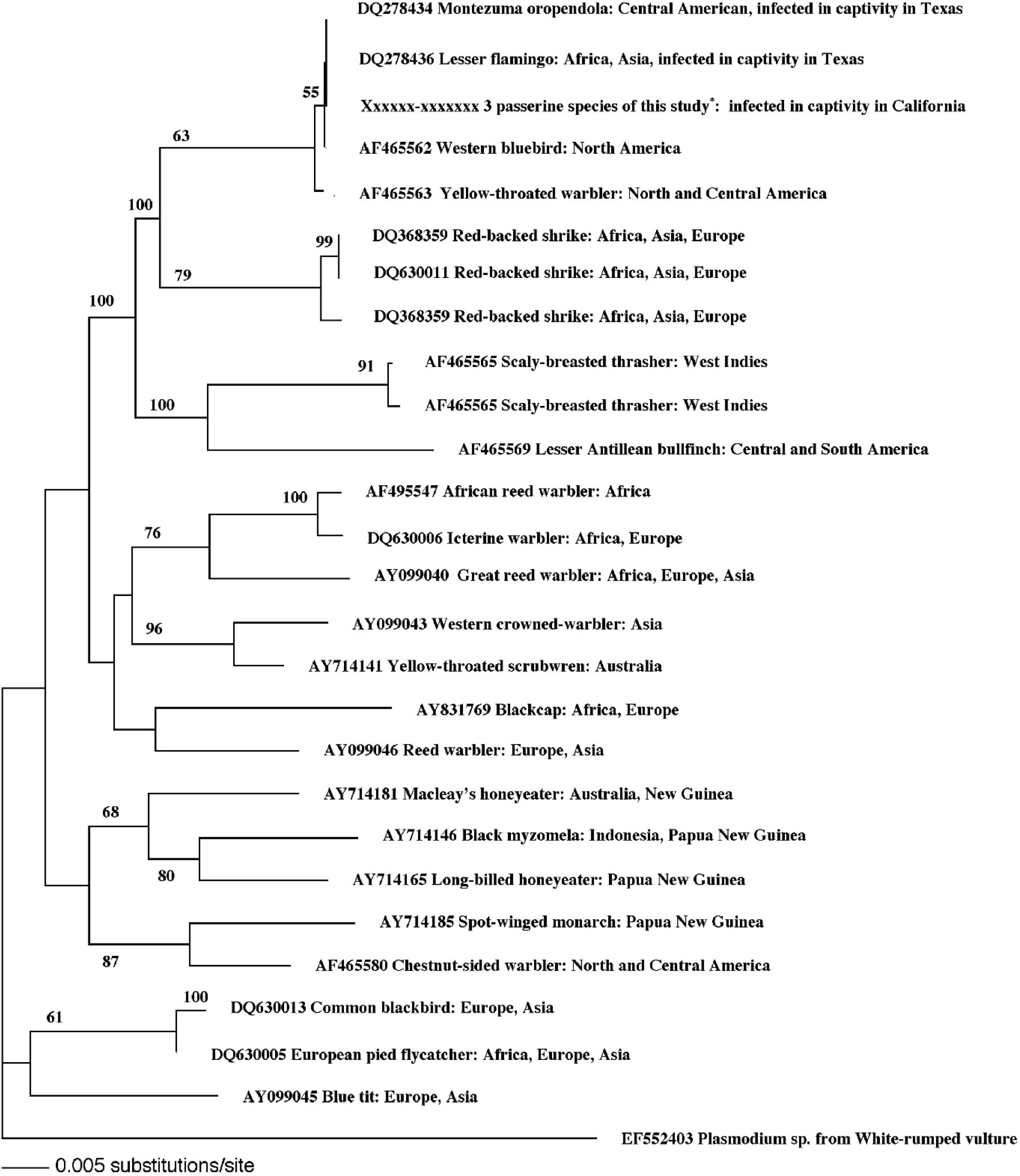
Phylogram of the Haemoproteus sp. identified in 10 of 11 passerine birds from this study compared with other Haemoproteus spp., which shows that it is grouped with Haemoproteus spp. reported from asymptomatic, native North American passerine birds.
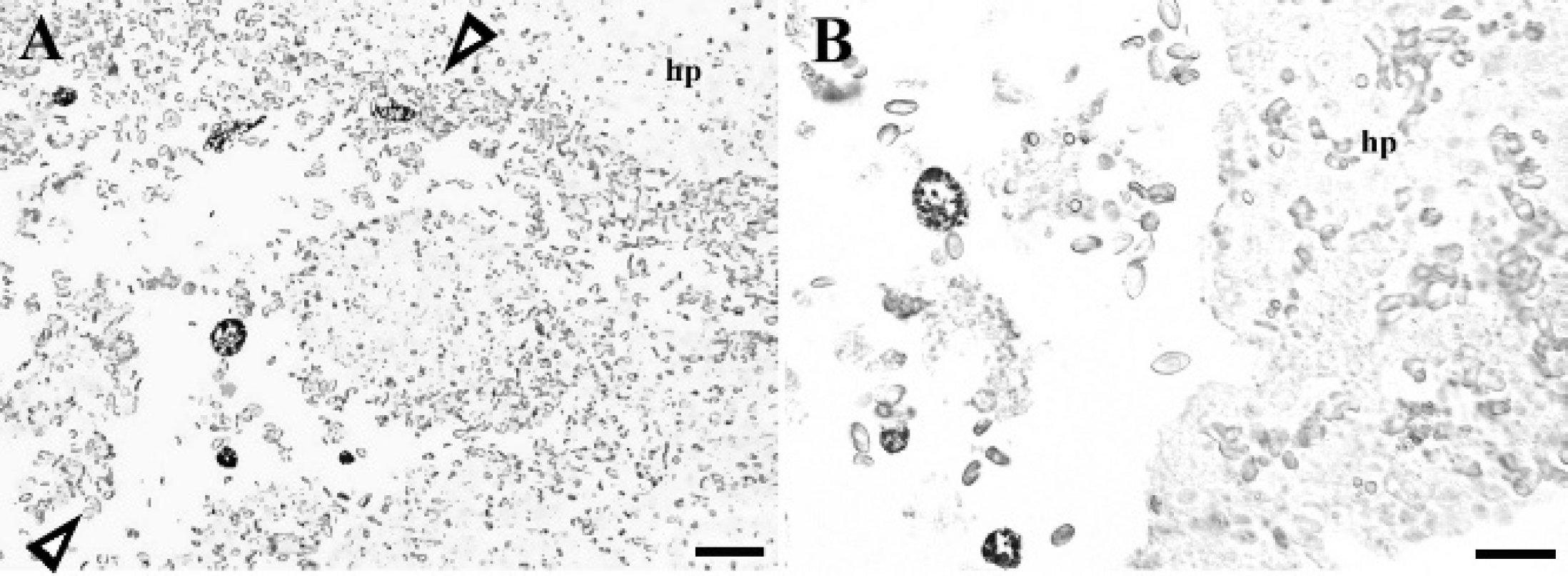
In situ hybridization from a region of Haemoproteus parasitism and hemorrhage in the liver of a Guianan red-capped cardinal. Hybridization signal (nitro blue tetrazolium chloride/5-bromo-4-chloro-3-indolyl phosphate, chromogen) shown as dark staining is specific for cytomeres and merozoites within hepatic lesions. Arrowheads denote areas of hepatocellular disruption and extensive hemorrhage. Hp = hepatocytes.
The megaloschizonts reported in the current study resembled previously described mature second-generation schizonts of Haemoproteus. 4 Megaloschizonts were large (200–500 μm); bounded by a thin membrane; commonly contained clear, central vacuoles; and were composed of small (1–3 μm in diameter), often morphologically indistinct, speck-like merozoites (Fig. 3). These merozoites often formed larger cytomere structures at the periphery of the megaloschizont (Fig. 3). In 1 case (a magnificent bird of paradise), a megaloschizont contained well-defined cytomeres and no central vacuole (Fig. 4), which may represent a younger second-generation schizont. A previous study found that in turkeys, rupture of the first-generation schizont caused intense inflammation, and growth of the second-generation schizont led to necrosis of adjacent muscle fibers. 2,4 These schizonts are thought to rupture and release merozoites into circulation. Development of megaloschizonts in the livers of the affected birds in the current study may have had a direct effect on sinusoidal endothelial cells and an indirect effect on hepatocytes, which resulted in parenchymal hemorrhage, hepatocellular degeneration, and subsequent inflammation. Because of the location and sequelae to hepatic protozoal parasitism (hemorrhage and hemocoelom), it is plausible that the birds die before the erythrocytic forms can be identified. Pre-erythrocytic schizont stages of Haemoproteus spp. have been reported to occur in many organs and are thought to use a variety of host cells, but some cell types may provide a more appealing environment. 4 The reason for the apparent tropism to the liver in the current cases is unknown.
Natural and experimental diseases associated with pre-erythrocytic stages of Haemoproteus have been reported in various bird species. 2–4,8–10,18,20 Although skeletal muscle lesions that result in clinically significant disease is a commonly reported sequela to infection with pre-erythrocytic stages of Haemoproteus spp., muscle necrosis was not a significant feature in any of the 11 cases. Two epizootics of myositis in bobwhite quail caused by protozoal megaloschizonts were reported to result in severe morbidity and mortality. 8,12 In 1 case, Haemoproteus lophortyx was suspected as the cause based upon gametocyte morphology. 8 Natural Haemoproteus infections reported in 2 bleeding heart doves (based on presumptive intraerythrocytic Haemoproteus gametocytes), described megaloschizonts and associated necrosis most prominently in skeletal muscle. 9 In addition, occasional schizonts in other tissues were associated with mild myocardial degeneration, hepatocellular vacuolation, granulation, karyorrhexis, and mild interstitial plasmacytic nephritis. 9 Experimental Haemoproteus infections in domestic turkeys and natural infections in a wild turkey 2–4 resulted in severe myositis with intralesional megaloschizonts, muscle necrosis, and lameness. Molecular confirmation was not available in any of the aforementioned studies. Naturally acquired Haemoproteus megaloschizonts were previously described in the viscera and muscle of various avian species. 18,20 Elongate Haemoproteus schizonts were described in the lung and pulmonary vasculature of a domestic pigeon, causing tissue displacement and blockage of pulmonary vessels, and an ovoid Haemoproteus schizont was found in the skeletal muscle of a sacred kingfisher, with no significant inflammation and circulating gametocytes in both cases. 20 Compartmentalized protozoal schizonts were reported to occur in the lungs, liver, and heart of a sparrow from Israel, similar to those previously described in Baghdad sparrows, 18 and were attributed to Haemoproteus, based upon the morphology of intraerythrocytic gametocytes. Hepatocytes that surrounded the schizonts exhibited fatty degeneration, whereas schizonts in the lungs were surrounded by mild inflammation and hemorrhage. 18 However, these schizonts were not morphologically similar to those in the current cases. Large hepatic protozoal cysts similar to those described in the current study were reported in other avian species 17 (Hall DG, Harmon BG, Howerth EW, et al.: 1998, Sudden death in psittacine and non-psittacine birds associated with hepatic infection with an unclassified haemosporozoan parasite: eight cases (1994–1996) and Hawk MV, Munger LL, Barnes HJ: 1998, An outbreak of fatal disease in peach faced lovebirds [Agapornis roseicollis] attributed to an unidentified protozoan parasite. In: Proceedings of the 1st International Virtual Conference in Veterinary Medicine, Diseases of Psittacine Birds. University of Georgia, Athens, GA), with a designation of haemosporozoa of undetermined taxonomic status. The histologic and cytologic findings in these 11 passerine birds are very similar to a previous characterization, which describes haemosporozoan cysts in the muscle and viscera, with an absence of gamonts and gametes in peripheral blood smears. 11
Identification of protozoa based on megaloschizont forms can be problematic, because many protozoal schizont forms have similar morphologic features. Concurrent mixed infections and taxonomic changes are additional obstacles that may prohibit correct identification of hemoparasites. 10,20 The current study's gross and histologic findings may represent a disease entity that was previously described but incompletely understood. There are many reports of liver hemorrhage and necrosis in passerine and nonpasserine birds of the United States attributed to protozoal schizonts, 11,17 but the protozoal identity was either undetermined or thought to be an aberrant form of Leukocytozoon. It is hypothesized that several reports that designate aberrant leukocytozoonosis as the cause of protozoal megaloschizont–induced injury in various birds are actually consistent with stages of Haemoproteus. 4,9,18
At a zoological institution in Texas, similar liver lesions in passerine and nonpasserine birds, including lesser flamingos and Montezuma oropendolas, were attributed to Haemoproteus spp. by using PCR and gene sequencing. 10 In the current cases, the same techniques were used to identify the protozoan parasites as Haemoproteus spp. and to rule out Leukocytozoon and Plasmodium. In situ hybridization was done in 3 birds with a mitochondrial cytochrome B probe and was positive only in megaloschizonts, which confirmed the presence of these protozoa in the liver (Fig. 6). Cloning and DNA sequencing of the Haemoproteus PCR amplicons from 10 of the 11 cases revealed that all 10 positive birds had the same genotype of Haemoproteus and that the sequences were most consistent with 5 Haemoproteus spp. from North America. Comparison of sequences from these cases to the Haemoproteus sp. found in lesser flamingos and Montezuma oropendolas from a zoological institution in Texas 10 revealed that the 2 Haemoproteus spp. were 100% identical.
The presence of the same Haemoproteus species causing similar hepatic lesions in birds of different families and orders is significant and supports the theory that Haemoproteus is not nearly as host specific as previously thought. 14,21,25 Host switching of protozoal hemoparasites into naïve populations has been linked with changes in virulence and increased morbidity. 1,5,14,21,22 These findings imply that host switching is a plausible mechanism for Haemoproteus infection of naïve birds from different locations assimilated into a novel environment, as is typical in a zoological setting. Although avian species from similar regions of the world are typically grouped together in a zoo, insect vectors can move between groups and may play a role in transmission of infectious organisms. Interestingly, the majority of the birds in the current report are from South America, excluding the magnificent bird of paradise and the superb bird of paradise. In general, prevalence of hemoprotozoa in neotropical birds is low. 24 Therefore, birds from this region of the world may have an increased susceptibility to hemoprotozoal infections. It is possible that infection with North American Haemoproteus in South American birds (i.e., naïve hosts) resulted in an aberrant or more virulent pre-erythrocytic form of the protozoa.
Because the disease process is peracute, prevention by means of vector control is the most promising way to avert further infections. 20 Future studies should include confirmation and identification of vectors, as well as demonstration of the same Haemoproteus spp. in surrounding native bird populations.
Acknowledgements
This work was supported by the Zoological Society of San Diego. Outstanding technical assistance was provided by April Gorow, Julie Albright, Marc Hammond, Yvonne Cates, Patricia M. Gaffney, and Elizabeth Kieran. The authors thank Beckman Coulter Inc. for the donation of the automated capillary DNA sequencer and high-speed centrifuge used in these studies, Charles and Shirley Sykes of San Diego for financial support of the authors' laboratory, and Dr. Chris Gardiner for his thoughtful review of the manuscript.
Footnotes
a.
Qiagen, Inc., Valencia, CA.
b.
Qbiogene, Inc., Carlsbad, CA.
c.
Invitrogen Corp., Carlsbad, CA.
d.
Beckman Coulter, Inc., Fullerton, CA.
e.
MacVector, Inc., Cary, NC.
f.
Accelrys, San Diego, CA.
g.
Sinauer Associates, Inc., Sunderland, MA.
h.
Mercedes Medical, Sarasota, FL.
i.
Zymed Laboratories, Inc., San Francisco, CA.
j.
Roche Diagnostics, Indianapolis, IN.
k.
Surgipath Medical Industries, Inc., Richmond, IL.
l.
Biomeda Corp., Foster City, CA.
