Abstract
The objective of this study was to develop a reliable Taqman® 5′ Nuclease Assay for genotyping sheep for scrapie susceptibility. The sheep prion gene contains 2 single nucleotide olymorphisms (SNPs) that may mediate resistance to classical scrapie, one at codon 136, alanine (A) or valine (V), and another at codon 171, arginine (R) or glutamine (Q). The R allele appears to confer resistance to classical scrapie, with the AA136 RR171 genotype the most resistant to scrapie and QR171 only rarely infected in the US sheep population. The Assays by Design® protocol was used for development of probes and rimers for codon 136 and Primer Express® for codon 171. Commercially available kits were used to isolate genomic DNA from blood or muscle. For validation, 70 SNP determinations for each codon were compared to commercial testing with an error rate of less than 1%. Then, 935 samples from blood (n = 818) and muscle (n = 117) were tested for both codons with 928 successful determinations and only 7 samples (<1% of total samples) that needed repeating. Genotypes were AA QQ (n = 102; 11.0%), AV QQ (n = 28; 3.0%), AA QR (n = 396; 42.7%), AV QR (n = 54; 5.8%), and AA RR (n = 348; 37.5%). Thus, 86% of the sheep tested (n = 798) contained R at codon 171 and were expected to be scrapie-resistant. This new Taqman® 5′ Nuclease SNP genotyping assay is accurate, easy to perform, and useful in the study of classical scrapie in sheep and its prevention through selective breeding programs to eliminate highly susceptible animals.
Scrapie is a fatal and incurable neurological disease of sheep that belongs to a family of prion diseases known as transmissible spongiform encephalopathies (TSE). Other well-known members of the family include bovine spongiform encephalopathy (BSE), variant Creutzfeldt-Jakob disease (vCJD, the disease associated with BSE transmission to humans), and chronic wasting disease of cervids. Although scrapie does not appear to be transmitted to humans, concerns for the effects of TSEs on human welfare and for the loss of millions of dollars to sheep producers from this debilitating disease have prompted the National Institute for Animal Agriculture to promote a program for worldwide scrapie eradication by 2010. 17
Genotyping sheep for genes that appear to confer resistance to contracting classical scrapie has been used for several years in an attempt to eradicate scrapie through selective breeding. Eight commercial laboratories are approved by the US Department of Agriculture (USDA) Animal Plant Health Inspection Service (APHIS) to perform the official scrapie genotyping. Approved laboratories are eligible to conduct privately funded official scrapie genotype testing and compete for National Scrapie Eradication Program funded official genotyping. Their tests are reliable (most claim 99% reliability) and accurate, but relatively expensive.
Currently, there are numerous methods for detecting single nucleotide polymorphisms (SNP) in genes. 12 Many of these use real-time PCR methodologies for detecting the SNP. 6 The Taqman® 5′ Nuclease SNP genotyping assay was recently developed and is widely used for SNP detection in humans. Thus, this method might also detect the polymorphisms at codon 136, alanine (A136) or valine (V136), and codon 171, arginine (R171), glutamine (Q171), histidine (H171), or lysine (K171), that affect susceptibility to scrapie in sheep. In the absence of V136, the R171 allele appears to confer resistance to classical scrapie, with the AA136 RR171 genotype the most resistant to scrapie and QR171 only rarely infected. V136 increases susceptibility to contracting scrapie and also appears to affect survival time; infected sheep that are AV136 live longer than those that are VV136. 2,8,9 Additionally, in the presence of the V136 allele, sheep that also carry the allele encoding R171 are less likely to be infected and when infected, they survive longer than those homozygous for Q171 or having H171 or K171 with Q171. Thus, the objective of the present study was to develop a Taqman SNP genotyping assay that would reliably test for A/V136 and Q/R171 of the sheep prion gene.
The Taqman 5′ Nuclease SNP genotyping assay relies on the cleavage of sequence-specific fluorescently labeled probes during PCR amplification. The probes are chemically modified at their 3′ end to make them nonextendable and to have a nonfluorescent quencher. Each sequence-specific probe, representing alternate alleles of the SNP, has a different fluorescent label and both are combined in a reaction with 1 set of primers. During PCR amplification, cleavage of the bound probes, and their subsequent separation from the nonfluorescent quencher, results in generation of fluorescence that is based on having one or both alleles present in the DNA sample. 6 Ideally, probes would only bind tightly enough to their exact sequence to be cleaved when displaced during the PCR amplification. Thus, careful design of probes should prevent cross-reactions with a mismatched probe and generation of a nonspecific signal.
To design the appropriate sequence-specific probes and primers, 14 or more complete cDNA or protein sequences of the sheep prion gene were selected from the National Center for Biotechnology Information (NCBI) and European Molecular Biology Laboratory (EMBL) databases and compared for homology. Variations in codon 136 (amino acids corresponding to nucleic acids 406–408 of the prion mRNA) and codon 171 (amino acids corresponding to nucleic acids 511–513 of the prion mRNA) were noted and codon bias for the amino acids corresponding to A, V, Q, R, H, and K in the sheep prion gene were determined. For codon 136, GCC coded for A and GUC coded for V; for codon 171, CAG coded for Q, CGG coded for R, CAU coded for H, and AAG coded for K. The nucleic acid sequence from base pair (bp) 351 through 570 of the prion cDNA of GenBank Accession Number AJ000736 was selected as the template for preparation of SNP probe and primer sets for codons 136 and 171. 3 The probes and primers were designed using Primer Express®. a The NCBI Basic Local Alignment Search Tool (BLAST) (National Center for Biotechnology Information) for codon 136 and 171 amplicons confirmed their identity within the sheep prion gene.
After a series of experiments to optimize PCR temperatures (finally using 62°C for the amplification reaction instead of the default value of 60°C), the probes and primers designed for codon 171 using Primer Express performed successfully, but not those for codon 136. Thus, the cDNA template sequence from bp 351 through 509 was submitted to Applied Biosystems for their Assays-by-DesignSM Service for successful optimization of probes and primers. Fluorescent probes and unlabeled primers for codon 171 that were designed using Primer Express were purchased from Applied Biosystems and provided in separate aliquots. For codon 136, the fluorescent probes and unlabeled primers were included in a 40 × master mix. The sequences of probes and primers were disclosed with the order, but their exact concentration in the 40X master mix was not.
Proper DNA isolation is an important factor in a successful SNP determination. However, the DNA concentration and the A260/A280 ratio of the DNA could vary widely and still give a reliable test. It is very important to avoid contamination of the DNA sample with PCR inhibitors. Blood samples were collected using K3 EDTA as the anticoagulant, because heparin inhibits the PCR-reaction. 10 Commercially available kits were used for DNA isolation from 40–200 μl of whole blood using a column-based DNA isolation technique b or from muscle by homogenizing the tissue in a commercial DNA isolating reagent. c Clotted blood samples were homogenized briefly with a polytron and then processed successfully. DNA samples that did not run well in the PCR reaction (most often isolated from a very small amount of blood) were ethanol precipitated, washed, and concentrated before a second PCR run.
For the SNP assay, the PCR protocols and solutions from Applied Biosystems were used, but the reaction was scaled down to a 12.5-μl total reaction volume per sample per well of the 96-well PCR plate. For codon 171, this volume contained 2.5 μl of the DNA sample (diluted in nuclease-free H2O) combined with a 10 μl-aliquot containing Taqman Universal PCR Mastermix, a forward and reverse primers (900 nM each, final dilution), and the 2 fluorescent probes (200 nM each, final dilution). For codon 136, the 40 × mastermix was diluted in nuclease-free H2O to 1 × in the10-μl aliquot containing Taqman and combined with 2.5 ml of the DNA sample. The PCR reaction was conducted exactly as for gene expression assays and “read” by comparing the relative exponential increase in fluorescence specific to each probe in the sample tested. Controls of known genotype d were included on each 96-well plate and compared to unknown samples. For both codons, a typical PCR run on the ABI 7000 was 2 minutes at 50°C, 10 minutes at 95°C, and then 40 cycles of 95°C for 15 seconds and 62°C for 1 minute.
The cDNA sequences used as probes and primers for each codon in the SNP real-time PCR reaction, as well as their Primer Express software-estimated melting temperature (Tm), GC content, length, and the fluorescent labels on the probes are shown in Table 1. For the Taqman SNP assay (allelic discrimination), the primers ideally should have a Tm of 58–60°C and the probes a Tm of 65–67°C, preferably only having a 1 °C difference in Tm between the 2 probes. Amplicon size should be between 50–150 bases. Primer Express has most of the criteria for primer and probe development such as those related to primer Tm requirements, primer-dimer formation, GC content, GC clamp, and so on, built into the selection protocol. However, there are numerous selections of probe/primer combinations offered by the Primer Express program for each SNP determination and these require user selection for the best performance. Thus, if the first choice of probes and primers is not performing well, the Assays-by-Design Service of Applied Biosystems may be needed, using the company's algorithm for probe and primer selections.
For the initial confirmation of the validity of these probes, the Taqman SNP profile for several North Dakota State University (NDSU) sheep of known homozygous and heterozygous genotypes was determined. Then, 20 samples from sheep that had been commercially genotyped were run without prior knowledge of their genotype and all were correctly identified at both codons. Finally, 50 more SNP determinations for each codon were compared to commercial testing; only 1 sample had an error at 1 codon (136), giving an error rate for 70 samples of less than 1%.
Nucleotide sequences of the Taqman probes and primers used in the SNP genotyping assay for codon 136 and codon 171 of the sheep prion gene.

Probes are labeled with a fluorescent reporter dye at the 5′ end, either 6FAM™ or VIC™, and a quencher at the 3′ end; MGBNFQ = minor groove binding nonfluorescent quencher.
The patterns of results from the real-time SNP assays for the known homozygous and heterozygous alleles for codon 136 are shown on the left in Fig. 1 (A-C) and for codon 171 are shown on the right in Fig. 1 (D-F). These patterns were used thereafter as the standard for identifying homozygous or heterozygous alleles. Ideally, the fluorescence resulting from amplification and cleavage of probes for both alleles would be nearly equal if heterozygous (Fig. 1; B, E) or not seen, if homozygous for the other allele. This was true only for the homozygous RR genotype, in which case there was no fluorescence from the binding and cleavage of the Q allele probe (Fig. 1; F). For the other genotypes, the homozygous identification was based on whether fluorescence from cleavage of the probe for 1 allele was dominant over any fluorescence resulting from cleavage of the probe for the other allele (Fig. 1A, 1C, and 1D). In these cases, it appears that a small amount of the mismatched probe must bind to the sequence of the other allele and be cleaved, generating a low level of fluorescence. However, the DNA samples, used as controls, had been sequenced or tested by other methods for accuracy. If the patterns of the samples were not similar to those of the known genotype controls, or if the fluorescent amplification was inadequate for both alleles, the DNA was concentrated or re-isolated before repeating the assay.
After the initial tests to validate the SNP assays, uninfected ewes and lambs from the NDSU Experimental Research flocks were selected for genotyping. In this study, 928 out of 935 samples from blood (n = 818) and muscle (n = 117) were tested successfully for both codons. The results of the genotyping of the 928 sheep are shown in Table 2. Only 7 samples ({LT} = <1% of total samples) could not be determined. These samples may have had extremely low DNA concentrations or were samples containing unknown inhibitors that did not perform in the PCR reaction. If a second sample could have been taken, it is likely that the problem(s) would have been resolved.
Of the 928 sheep tested, 86% (n = 798) contained R171 (Table 2). Of these, the most abundant genotypes were AA136 QR171 (42.7%), and AA136 RR171 (37.5%). Least abundant genotypes were AV136 QQ171 (3.0%), AV136 QR171 (5.8%), and AA136 QQ171 (11.0%). There was no VV136 QQ171 genotype in this group of animals, which is not surprising because very few sheep (less than 2%) of this genotype exist in the U.S. 4,15–17 In comparison, the 2002–2003 national genotype distribution for 1,784 ovine cerebellum samples analyzed by genotype at codons 136 and 171 was as follows: AA136 QR171 (41.2%), AA136 RR171 (15.1%), AV136 QQ171 (5.4%), AV136 QR171 (3.6%), AA136 QQ171 (33.4%), and VV136 QQ171 (0.5%) with 0.8% designated as H171, K171, or untested samples. 17 Thus, our genotyping results demonstrate increased numbers of the AA136 RR171 genotype and decreased numbers of the AA136 QQ171 genotype in our flocks, which was a goal of our scrapie eradication project at NDSU.
The Taqman SNP technology is versatile enough to design probe and primer sets for detection of new SNP as additional novel protective alleles are identified. Although homozygosity for the allele encoding A136 Q171 is the major determinant 16–18 of susceptibility to the predominant strain of classical scrapie in the United States, the A136 R171 genotype is not protective in AV136 QR171 sheep. 5 Likewise, atypical scrapie strains such as Nor98 are reported in sheep homozygous or heterozygous for R171, particularly in sheep heterozygous for an allele encoding a SNP at 141 (leucine/phenylalanine) or 154 (arginine/histidine). 14 The SNP at codon 154 is in a potential nucleation site 11,13 and is also associated with susceptibility or incubation time in some studies of classical scrapie. 7 Moreover, a recent study revealed a rare change to leucine at codon 154 in 2 breeds of Spanish sheep that may affect susceptibility to scrapie as well. 1 Thus, the scrapie eradication program in the United States may eventually include identification of several different SNP within prion alleles.
In summary, the Taqman 5′ Nuclease genotyping assay for detecting SNPs in the sheep prion gene is an efficient, reliable, and accurate method of genotyping sheep for the genes that control susceptibility to scrapie or contribute to the incubation times for progression of the disease. As a screening procedure, this SNP assay has high throughput and good potential for automation, particularly by using the Applied Biosystems 7900HT robotic real-time PCR machine along with an automated DNA isolation platform, such as the ABI Prism™ 6100 Nucleic Acid PrepStation. This SNP assay is also cost-effective; the cost of the assay is primarily based on the price of DNA isolation kits, Taqman probes and the PCR mastermix; but reducing the reaction volumes from 50 μl to 12.5 μl decreases the cost considerably without compromising the results. Our estimated cost of the assay, based only on disposable supplies and not on labor and equipment, is between $2.00-$3.00/sample and approximately half of the cost is based on the price of the DNA isolation kit. The system is flexible and will allow testing for established and novel mutations associated with scrapie susceptibility.
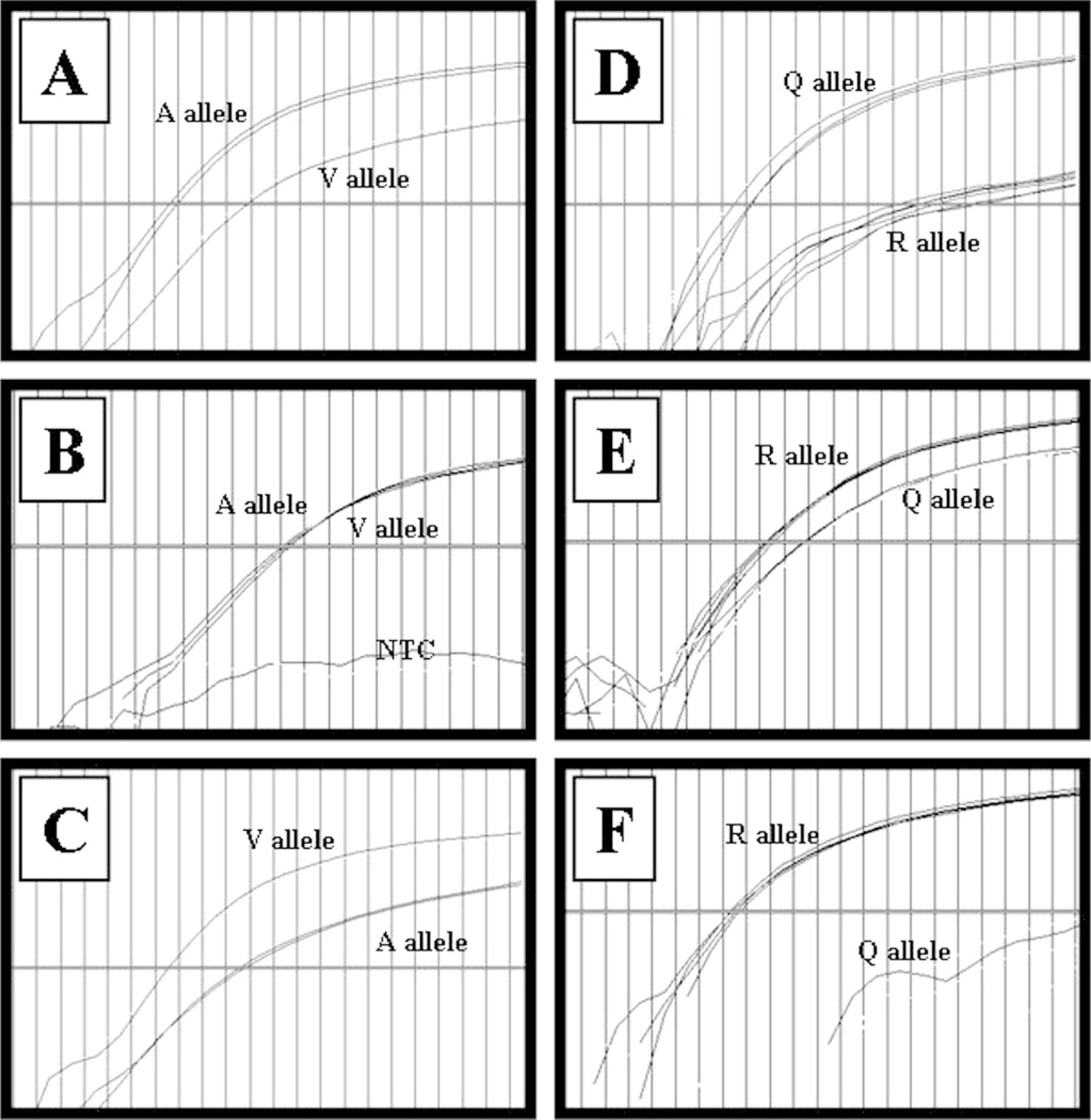
Patterns of results from the real-time PCR SNP assays for known homozygous and heterozygous alleles of the sheep prion gene at codon 136 (A-C) and at codon 171 (D-F). For codon 136,
Distribution of genotypes in sheep tested for SNPs at codon 136 and 171 of the prion gene.

Footnotes
a.
Primer Express,® Taqman Universal PCR Mastermix Catalog #4324020. Applied Biosystems, Foster City, CA.
b.
GenElute blood genomic DNA kit, Sigma-Aldrich, Bellefonte, PA.
c.
DNAzol, Molecular Research Center, Inc., Cincinnati, OH.
d.
GeneCheck, Fort Collins, CO.
