Abstract
Toxoplasma gondii is a zoonotic protozoan pathogen that infects many endothermic vertebrates, including humans; the domestic cat and other felids serve as the definitive host. Macropodids are considered highly susceptible to toxoplasmosis. Here, we describe the clinical, pathologic, and immunohistochemical findings of an outbreak of systemic toxoplasmosis in a mob of 11 red kangaroos (Macropus rufus), with high morbidity (73%) and mortality (100%) rates. Affected animals had either severe and rapidly deteriorating clinical conditions or sudden death, which was correlated with widespread necrotizing lesions in multiple organs and intralesional T. gondii organisms identified via MIC3-specific immunohistochemistry and confirmed by REP529-specific rtPCR. Quantification of parasites demonstrated the highest parasite density in pulmonary parenchyma compared with other tissues. Our study highlights the continued importance of this severe condition in Australian marsupials.
Introduction
Toxoplasma gondii is a zoonotic protozoan pathogen in the phylum Apicomplexa. T. gondii is a coccidian parasite with a worldwide distribution, an indirect life cycle, and is capable of infecting nearly all endothermic vertebrates. Domestic cats and other members of the family Felidae are definitive hosts; non-felids serve as intermediate hosts.11,12,35 There are 3 infectious stages of the life cycle, including the rapidly dividing and motile tachyzoites, the slow-dividing bradyzoites that form tissue cysts, and the sporozoites within sporulated (infective) oocysts.5,8 The sexual reproduction phase occurs within the intestinal tract of the definitive host, and oocysts released in the feces contaminate the environment, including food and water sources. 13 Therefore, domestic and wild cats play a central role in the dissemination of T. gondii, particularly to herbivorous animals. The prepatent period depends on the infectious stage ingested by the felids: 3–10 d after ingesting tissue cysts and ≥ 19 d after ingesting oocysts or tachyzoites. Infected felids can shed the organism for 7–21 d; oocysts sporulate in the environment and become infectious in 1–3 wk. 14 Following ingestion by the intermediate host of either sporulated oocysts containing sporozoites or infective tissue cysts containing bradyzoites, sporozoites or bradyzoites convert into rapidly dividing and highly motile tachyzoites that disseminate throughout the intermediate host and infect various cell types in different tissues.
The development of a host immune response and intracellular killing mechanisms by the infected cells finally leads to either elimination of tachyzoites or differentiation into the slowly replicating bradyzoites, which form life-long cysts within affected tissues.5,8 Although cysts can develop in any tissue type, they have preferential site specificity for the central nervous system, eye, heart, and skeletal muscle. Bradyzoites are the infective form for definitive and intermediate hosts. At any time, bradyzoites can be released from cysts, transform back into tachyzoites, and cause recrudescence of infection; this is commonly seen in immunocompromised patients.5,8
Toxoplasmosis is a disease of major public health concern, with T. gondii infecting over one-third of the global human population. 11 Seroprevalence among animals is also high.12,14,22 Although infections in healthy human or animal intermediate hosts are usually asymptomatic or only cause mild clinical signs (malaise, fever, and lymphadenomegaly), severe, systemic, and often fatal disease may develop. The immune status of the host and/or other factors, such as pregnancy, can impact the host’s ability to effectively control the rapidly replicating tachyzoites. Lesions are diverse and can include myocarditis, myositis, ocular toxoplasmosis with choroiditis or retinochoroiditis, and encephalitis.12,14
Australian marsupials are among the most susceptible intermediate hosts for disease caused by T. gondii, and this infection is considered an important cause of decline in wild and captive marsupial populations.9,23,32,34 This increased susceptibility is thought to be the result of the historic absence of definitive hosts in Australia, precluding the development of resistance to T. gondii by Australian mammals. 27 Although several pathologic and serologic studies have been reported in different marsupial species, there are no detailed immunohistochemical studies.1,3,4,6,15,16,28,31–34 We describe here the clinical, pathologic, and immunohistochemical findings of an outbreak of acute, systemic toxoplasmosis in captive red kangaroos (Macropus rufus) from eastern Louisiana.
Materials and methods
Animals included in our study and clinical history
On 13 January 2020, a red kangaroo was received at the Louisiana Animal Disease Diagnostic Laboratory (LADDL), Louisiana State University (LSU; Baton Rouge, LA), for gross examination after being found dead on the premises (case 1). By the time of submission, 2 additional animals were reported dead on the same premises. Subsequently within that week, 2 additional red kangaroos from this premises were submitted to the LSU Veterinary Teaching Hospital (LSU-VTH).
The first animal was a 5-y-old, 33-kg male with a 24-h onset of dyspnea and weakness (case 2). Examination revealed that the kangaroo was 5% dehydrated, dyspneic, recumbent, had a delayed capillary refill time (> 3 s), and had no pupillary light response in either eye. Survey radiographs revealed a bronchointerstitial pattern in the lungs. Bloodwork revealed hemoconcentration (packed cell volume: 0.55 L/L; reference interval [RI]: 0.46–0.51 L/L), hyponatremia (135 mmol/L; RI: 146–152 mmol/L), hyperproteinemia (87 g/L; RI: 60–68 g/L), hyperkalemia (8.3 mmol/L; RI: 4.6–5.6 mmol/L), and elevated alanine aminotransferase (49 U/L; RI: 30–41 U/L), creatine kinase (> 2,036 U/L; RI: < 1,776 U/L), and total bilirubin (80.4 µmol/L; RI: < 1.7 µmol/L).25,39 The kangaroo was hospitalized overnight and provided oxygen (40%, oxygen cage), intravenous (IV) fluids (lactated ringer solution, 72 mL/kg/d), clindamycin (11 mg/kg, q12h, IV), enrofloxacin (5 mg/kg, q12h, IV), carprofen (4 mg/kg, q12h, IV), and famotidine (0.6 mg/kg, q24h, IV). The animal declined overnight and was euthanized (pentobarbital, 177 mg/kg IV) and submitted for postmortem examination.
The second kangaroo was a 2-y-old, 16.5-kg male with acute onset of lethargy, diarrhea, and abnormally friendly behavior (case 3). Given that this animal was alert and responsive, and hospitalization would have been difficult, no additional blood work or ancillary testing was performed. Based on the history of the facility and the preliminary results from the examination performed at the LADDL on case 1, toxoplasmosis was suspected and this animal, and all remaining animals (n = 6), were treated on-site by the caretakers with clindamycin (18 mg/kg, q12h, PO) and ponazuril (18 mg/kg, q24h, PO for 10 treatments) for a period of 14 and 10 d, respectively.
In April 2020, an additional kangaroo (3.5-y-old, intact female) was found dead on the premises (case 4) and submitted for postmortem examination at the LADDL (Table 1). In this case, death occurred ~ 3 mo following the institution of treatment.
Kangaroo cases submitted to the Louisiana Animal Disease Diagnostic Laboratory and included in our study.

Postmortem examination and sample collection
Postmortem examinations were performed on 3 animals submitted to the LADDL (Table 1). Samples from lungs, liver, spleen, kidneys, heart, and brain (cerebrum, cerebellum, brainstem) were collected aseptically and stored at −80°C for ancillary testing. Samples from all organs were also fixed in 10% neutral-buffered formalin for 24 to 48 h.
Histopathology
Sections of formalin-fixed, paraffin-embedded (FFPE) tissues (lung, liver, adrenal glands, spleen, kidneys, heart, brain, pituitary gland, mesenteric lymph nodes, diaphragm, pancreas, gastrointestinal tract, eyes, and female reproductive organs [corresponding to only one case]) were stained with hematoxylin and eosin (H&E) prior to histologic evaluation.
Bacteriology
Samples from the lungs were subjected to routine bacteriologic testing by inoculating commercial blood agar, chocolate agar, and MacConkey agar plates (Remel). Inoculated blood and chocolate agar plates were incubated at 37°C with 5% CO2 for the first 24 h, and without CO2 for the next 48 h. Inoculated MacConkey agar plates were incubated at 37°C for up to 72 h. Bacterial colonies were subsequently screened using a MALDI-TOF MS (Biotyper Compass; Bruker) for identification.
Immunohistochemistry
Antibody screening
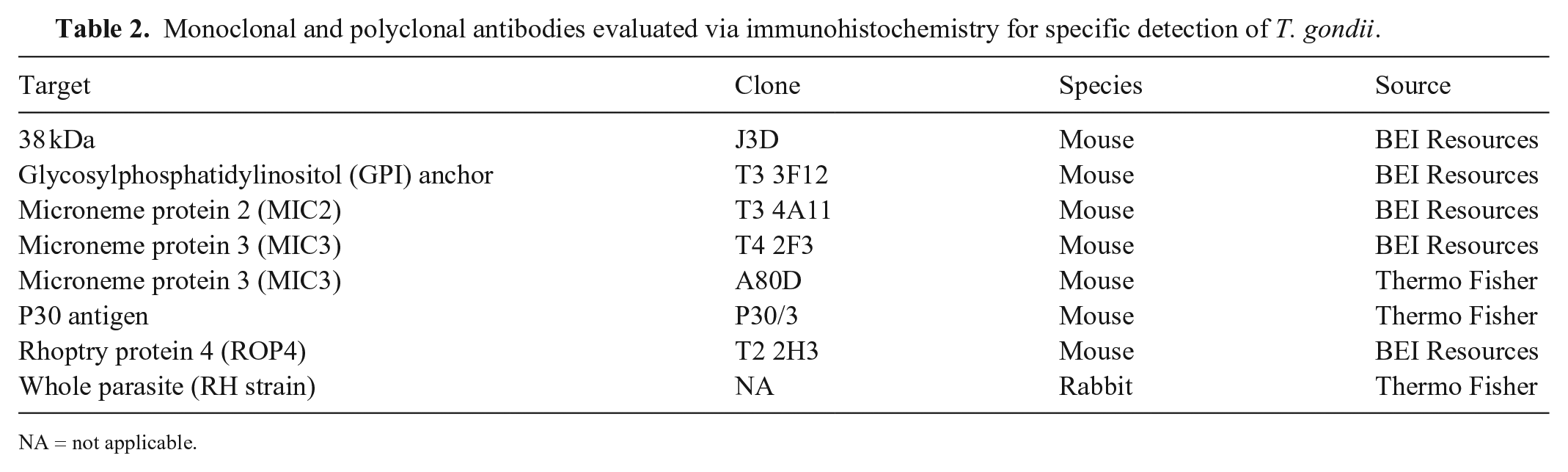
We tested 8 monoclonal and polyclonal antibodies directed to different proteins of T. gondii (Table 2) via immunohistochemistry (IHC), and those with adequate performance were titrated to the end-point dilution. A reference tissue (heart) containing parasitophorous vacuoles with T. gondii zoites and confirmed by REP529-specific real-time PCR (rtPCR; derived from case 2) was utilized for T. gondii–specific antibody screening. Briefly, 4-μm sections of FFPE tissues were mounted on positively charged slides (Superfrost Plus; VWR) and subjected to IHC (automated BOND-MAX, Polymer refine red detection kit; Leica) as described previously. 7 Following automated deparaffinization, heat-induced epitope retrieval (HIER) was performed using either a ready-to-use citrate-based (pH 6.0; Leica) or an EDTA-based (pH 9.0; Leica) solution at 100°C for 20 min. Sections were then incubated with the primary antibody for 30 min at room temperature, followed by a rabbit anti-mouse IgG (30 min) and a polymer-labeled goat anti-rabbit IgG coupled with alkaline phosphatase (30 min). Fast Red was used as the chromogen (15 min), and counterstaining was performed with hematoxylin. Slides were mounted with a permanent mounting medium (Micromount; Leica).
Monoclonal and polyclonal antibodies evaluated via immunohistochemistry for specific detection of T. gondii.
NA = not applicable.
To assess antibody specificity of those antibodies that were determined to be adequate for IHC (2 of 8 antibodies evaluated), bovine turbinate cells infected with either T. gondii strain RH, Neospora caninum strain Liverpool, or Sarcocystis neurona strain SN3 were used. Infected cells were fixed in 10% neutral-buffered formalin, placed in specimen processing gel (HistoGel; Thermo Fisher) following the manufacturer’s recommendation, embedded in paraffin following standard procedures, and immunostained with the respective anti–T. gondii antibodies as described above.
Immunostaining of tissues derived from infected kangaroos
Sections from the lung, liver, heart, spleen, adrenal gland, kidney, brainstem, cerebellum, cerebrum, pituitary gland, choroid/retina, gastrointestinal tract, pancreas, mesenteric lymph node, ovary, uterus, and mammary gland (n = 46 total) were immunostained using a monoclonal antibody (mAb) specific to the microneme protein MIC3 of T. gondii (T4 2F3, BEI Resources, American Type Culture Collection) based on the results of antibody screening. Tissue sections were immunostained with anti-MIC3 (clone T4 2F3, diluted 1:50) following HIER using the ready-to-use citrate-based buffer as indicated above. The number of parasitophorous vacuoles containing aggregates of zoites and tachyzoites was quantified in 5 random, 40× fields equivalent to 0.238 mm2/field or 1.19 mm2 total area. T. gondii–infected cell pellets and a reference tissue (heart) containing parasitophorous vacuoles with T. gondii zoites and confirmed by REP529-specific rtPCR (derived from case 2) were used as a positive control; tissues incubated without the primary antibody were used as negative controls.
Nucleic acid extraction from tissue specimens
Ten percent tissue homogenates (lung, liver, heart, and brain from each animal) were prepared in 1× phosphate-buffered saline (pH 7.4; TissueLyser II; Qiagen). Tissue homogenates were centrifuged at 1,400 × g for 15 min at 4°C, and 50 μL of the supernatant was subjected to nucleic acid extraction (MagMAX CORE nucleic acid purification kit; Thermo Fisher) following the manufacturer’s instructions. Extracted nucleic acids were stored at −80°C until processed.
TaqMan rtPCR
A primer and probe set targeting T. gondii REP52918,26 and N. caninum Nc52 were used. The reaction was set up (Path-ID qPCR master mix; Thermo Fisher) following the manufacturer’s recommendations. Briefly, the 25-μL reaction contained 12.5 μL of 2× qPCR master mix, 120 nmol TaqMan fluorogenic probe, 400 nmol of each primer, and 5 μL of template DNA. Amplification was carried out (ABI 7500 fast real-time PCR system; Applied Biosystems). The program included 10 min at 95°C (PCR initial activation step), followed by 40 cycles at 95°C for 15 s (denaturation), and 60°C for 1 min (combined annealing/extension).
Statistical analysis
Pearson analysis was performed to determine the correlation between the number of parasites identified histologically and via IHC using statistical analysis software (JMP15; SAS Institute). The agreement (kappa statistic) between the 2 methods was also estimated using the GraphPad online tool (https://www.graphpad.com/quickcalcs/kappa1/).
Results
History and clinical findings
A mob of 11 kangaroos (1.5–5.5 y old) was housed at the premises before the disease outbreak began. The animals were maintained in an enclosed pen, and their diet consisted of a commercial pelleted diet (Kangaroo/Wallaby diet; Mazuri) and pasture in their enclosure. Free-choice water was provided in large bowls. Domestic cats were not intentionally housed on the property but were seen on the premises. Eight of 11 (73%) animals were affected during the outbreak, all of which died, and 3 (cases 1, 2, 4) were submitted to the LADDL for postmortem examination (Table 1).
Postmortem and histologic findings
All kangaroos were in fair body condition at the time of postmortem examination. Macroscopically, the lungs were bilaterally mottled to diffusely dark-red to purple, variably firm, and failed to collapse, with some noticeable rib impressions in all cases (Fig. 1). On cut surface, the affected pulmonary parenchyma was deeply dark-red. The lumen of the trachea also contained variable amounts of white to red-tinged froth. The epicardial surface of the heart had short, pale-tan to white streaks in all cases. In 2 of the 3 cases examined, the liver had a slightly accentuated lobular pattern. The inflammatory histologic changes identified in the lungs, liver, heart, brain, and eyes were similar in all cases, with variation in lesion severity and distribution (Table 3).
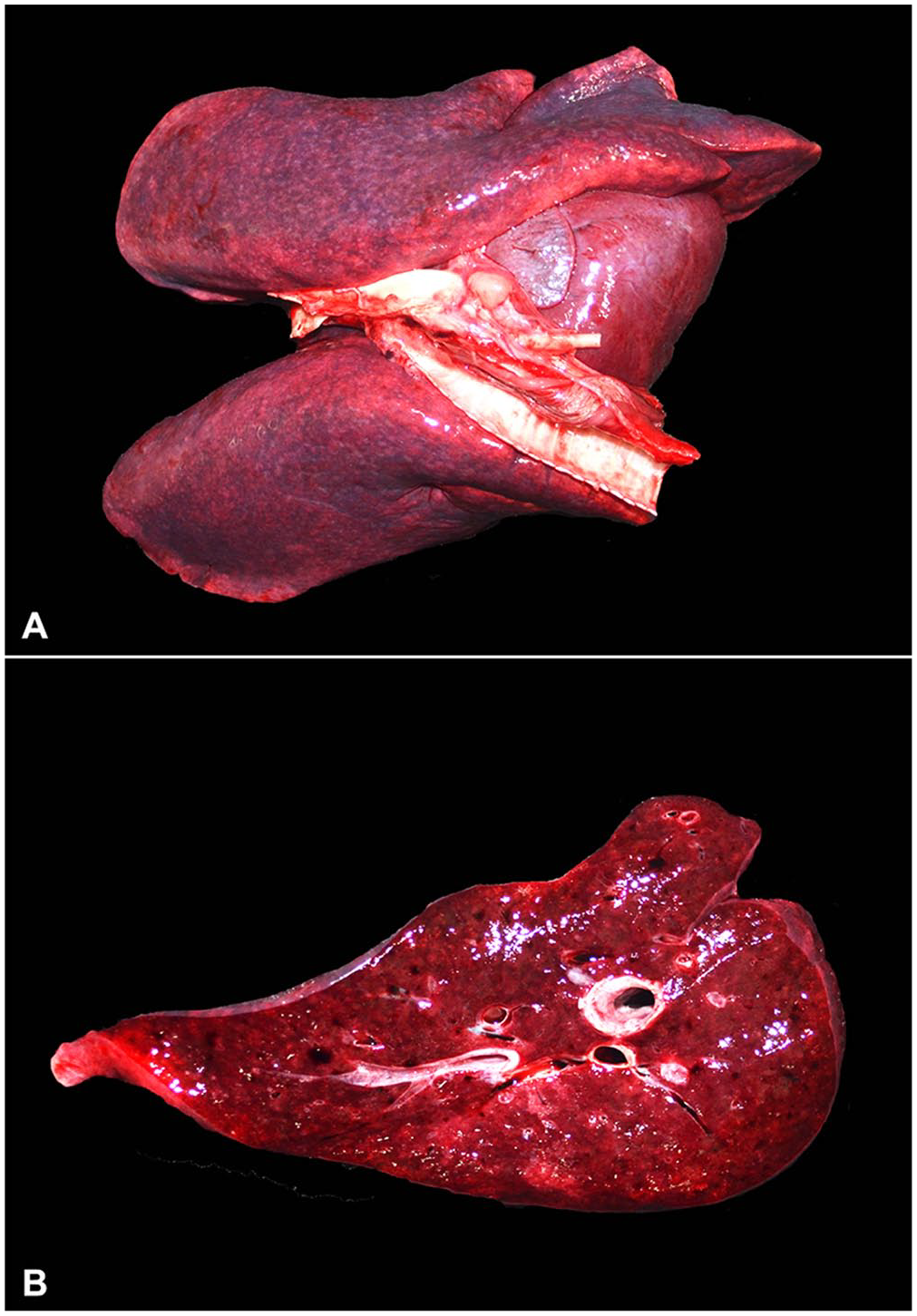
Acute toxoplasmosis in red kangaroos.
Histologic findings associated with toxoplasmosis in 3 red kangaroos.

D = diffuse; F = focal; H = histiocytic; He = hemorrhagic; L = lymphocytic; M = multifocal; MC = multifocal-to-coalescing; N = necrotizing; Neu = neutrophilic; P = plasmacytic.
Number of animals in which specific histologic changes were identified/total animals that were examined histologically.
Only evaluated in the one female kangaroo received.
Lung
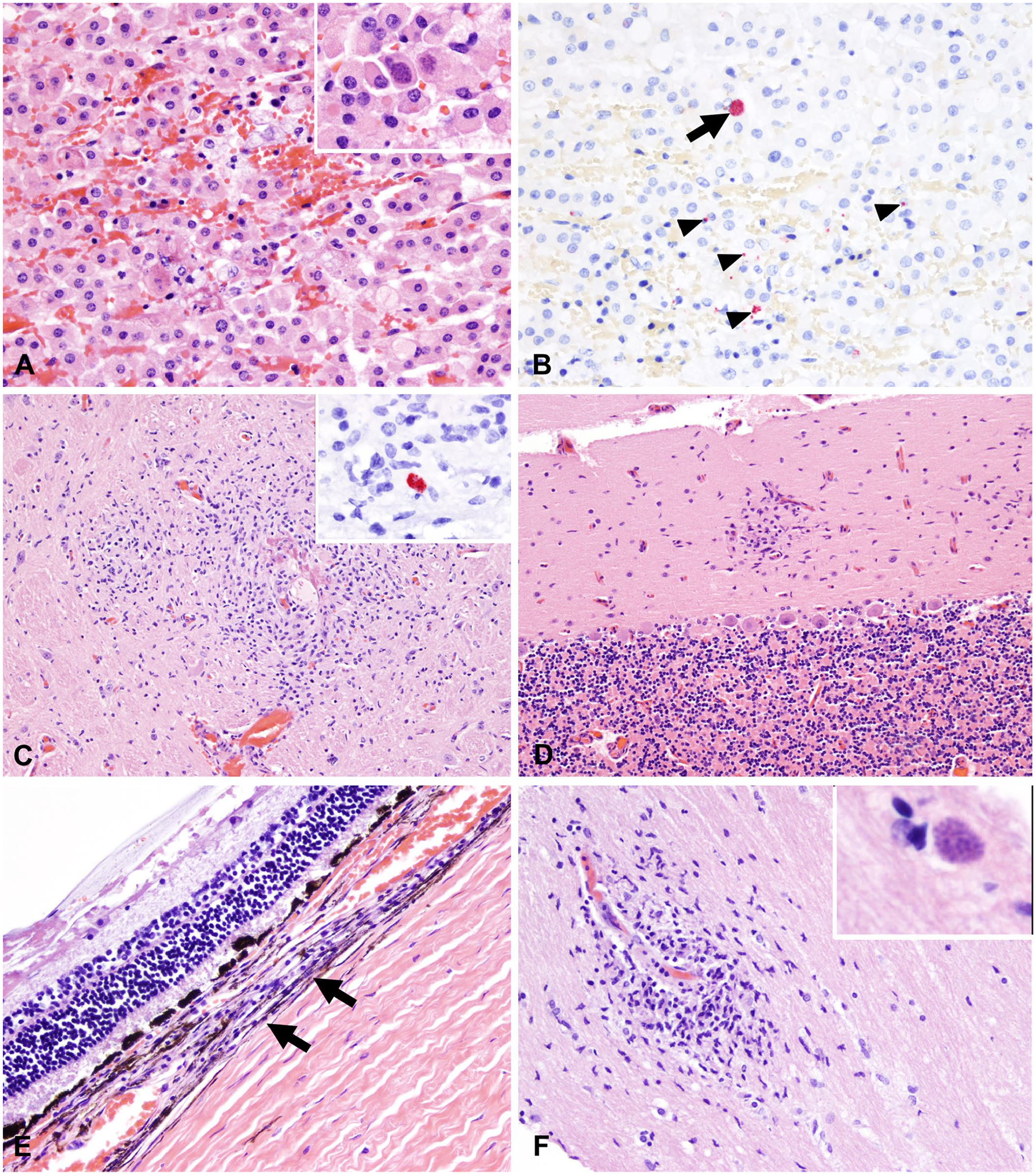
The histologic changes in the pulmonary parenchyma were equivalent in all affected animals and consisted of severe and diffuse lymphoplasmacytic and histiocytic interstitial pneumonia with prominent peribronchial, peribronchiolar, and perivascular cuffing (Fig. 2A). Alveolar spaces were flooded with alveolar macrophages, edema fluid, and, less commonly, fibrin and hemorrhage (Fig. 2A, inset). Parasitophorous vacuoles containing aggregates of zoites measured ~ 15 × 5 μm and were only evident histologically in one case (case 4); these parasitized cells were located near affected blood vessels and small airways (Fig. 2B).
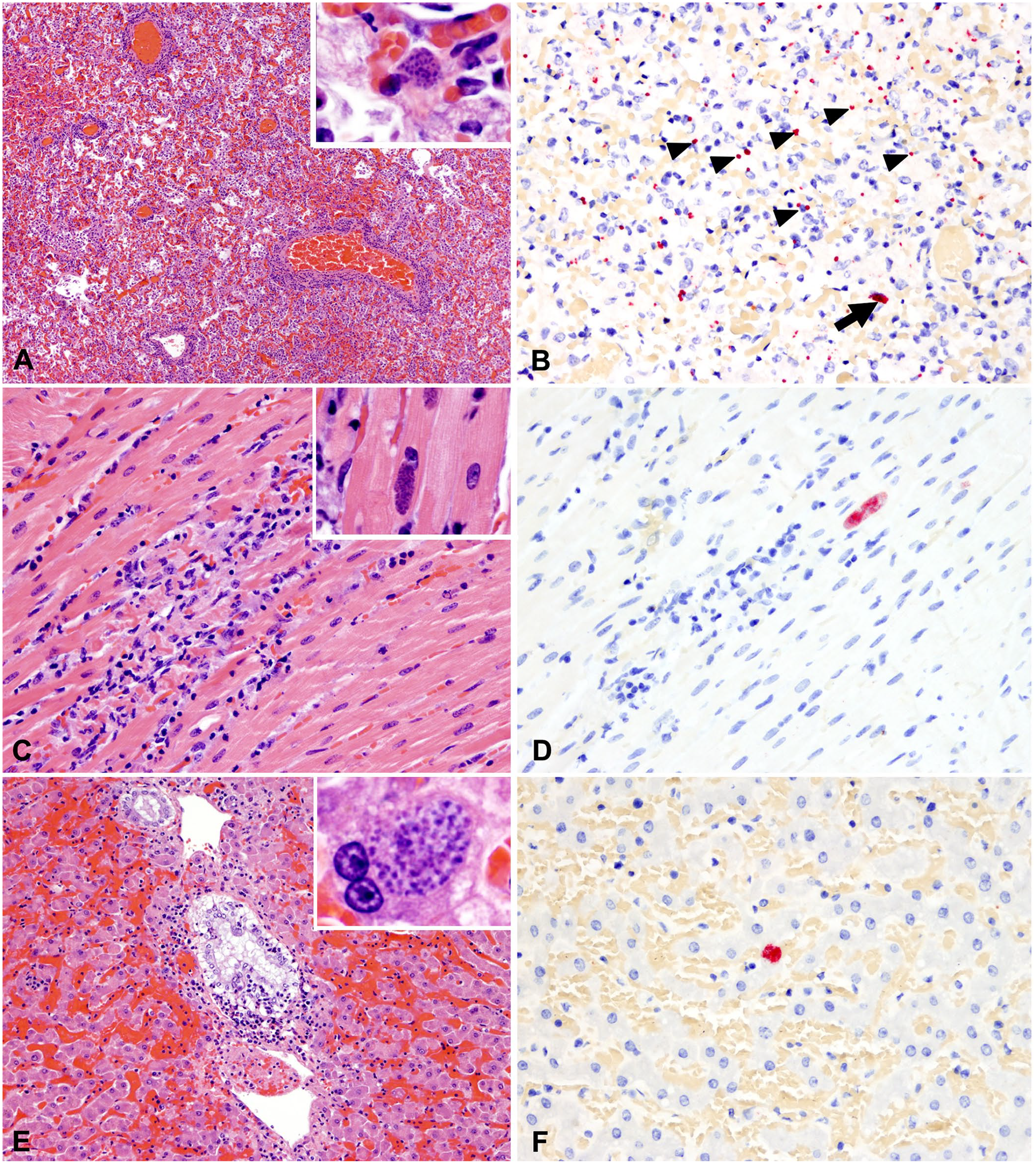
Histologic and IHC findings in tissues with the highest Toxoplasma gondii burden.
Heart
The white streaks observed grossly in the epicardium corresponded with foci of necrotizing myocarditis of variable severity (Table 3) with mixed inflammatory cells (lymphocytes, plasma cells, histiocytes), proliferating fibroblasts, and rare-to-frequent intra-sarcoplasmic parasitophorous vacuoles containing zoites (Fig. 2C, 2D; Table 4). Interestingly, these clusters of intracellular zoites were more frequently observed in cases of mild rather than severe myocarditis.
Immunohistochemical and real-time PCR findings in 3 red kangaroos.
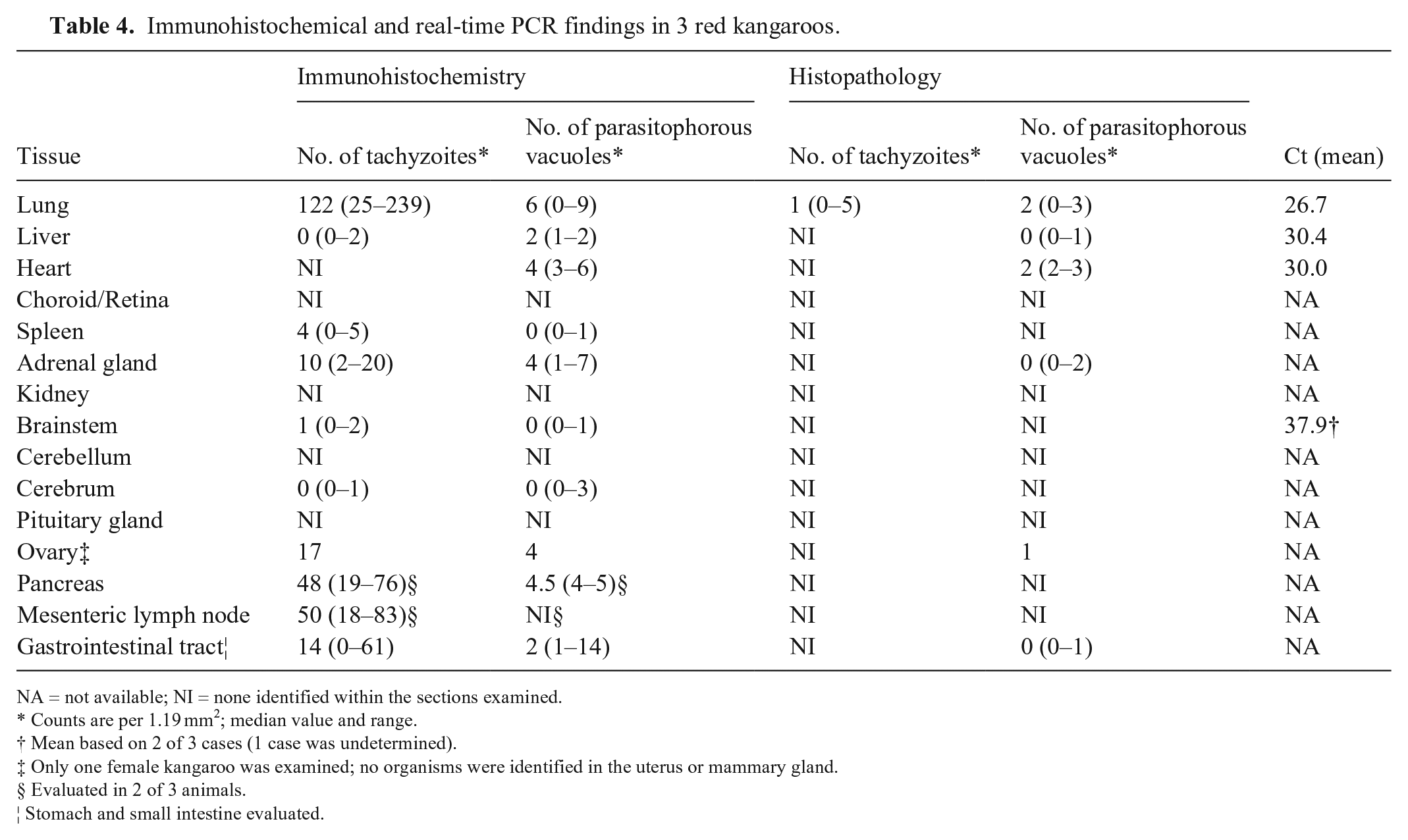
NA = not available; NI = none identified within the sections examined.
Counts are per 1.19 mm2; median value and range.
Mean based on 2 of 3 cases (1 case was undetermined).
Only one female kangaroo was examined; no organisms were identified in the uterus or mammary gland.
Evaluated in 2 of 3 animals.
Stomach and small intestine evaluated.
Diaphragm
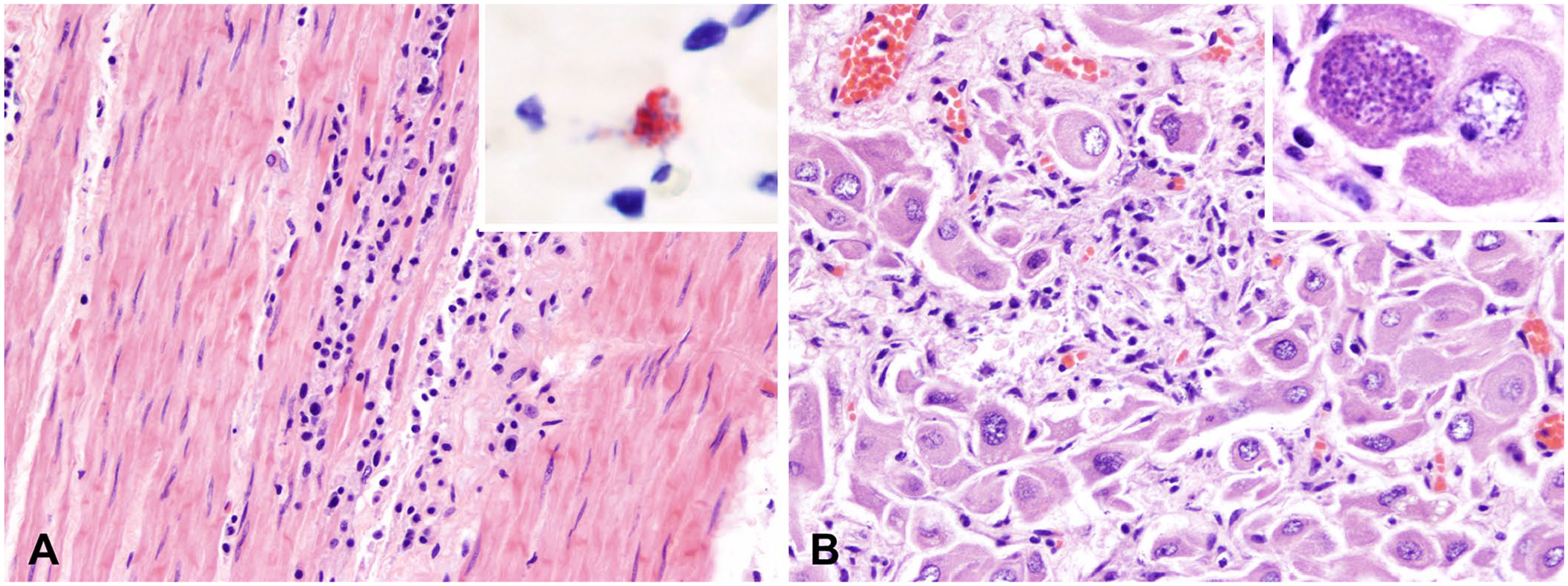
The histologic lesions within the skeletal muscle were similar to those affecting the myocardium and occurred in all animals, with sporadic intracellular aggregates of zoites.
Liver
Histologic alterations included random necrotizing hepatitis or cholangiohepatitis. Portal spaces were frequently expanded by infiltrating lymphocytes, histiocytes, and, in one of the cases, a few eosinophils (Fig. 2E). In 2 of the 3 cases (cases 1, 2), bile ducts were frequently delimited by karyorrhectic debris, and the bile duct epithelium was infiltrated by transmigrating lymphocytes and/or neutrophils (Fig. 2E). Parasitophorous vacuoles containing zoites within hepatocytes were rarely evident in one of the cases (case 4; Fig. 2F).
Adrenal glands
The adrenal cortex was characterized by either acute hemorrhage or small foci of necrosis with a few lymphocytes and histiocytes and rare intralesional aggregates of intracellular zoites (Fig. 3A, 3B).
Histologic and IHC findings in other tissues.
Brain
Microscopic findings in the brain of all cases examined included random necrotic foci and reactive gliosis to glial scarring multifocally affecting the brainstem, cerebellum, and cerebrum (Fig. 3C, 3D). In one of the cases, an aggregate of reactive glial cells was noted within the neurohypophysis. No protozoal organisms were evident histologically.
Eyes
The eyes were another commonly affected organ. In all the animals, microscopic changes were characterized by multifocal lymphocytic, plasmacytic, and histiocytic choroiditis (Fig. 3E). Optic neuritis was recognized in case 2 (the kangaroo was reportedly blind). This case was characterized by clusters of reactive glial cells and lymphocytes, with intralesional aggregates of intracellular zoites and axonal vacuolation (Fig. 3F).
Gastrointestinal tract
The stomach and intestines had mild infiltration of the lamina propria with lymphocytes, plasma cells, and fewer eosinophils in all cases; in case 2, the tunica muscularis of the stomach and small intestine was multifocally disrupted and composed of fragmented smooth muscle cells, karyorrhectic debris, and infiltrating lymphocytes, plasma cells, and histiocytes (Fig. 4A).
Histologic and IHC findings in other tissues (continued).
Female reproductive tissues
Female reproductive tissues from case 4, a female kangaroo with a recent history of parturition, were examined microscopically after a mucus plug was noticed within the vaginal lumen during postmortem examination. Interestingly, the ovarian parenchyma was comprised mostly of a single corpus luteum, which was multifocally replaced by small amounts of karyorrhectic debris and a few infiltrating lymphocytes and neutrophils, with rare luteal cells containing parasitophorous vacuoles filled with zoites (Fig. 4B).
Ancillary testing
T. gondii REP529 DNA was successfully amplified from the vast majority of the tissues evaluated. Mean cycle threshold (Ct) values ± SD were 26.7 ± 4.2, 30.4 ± 4.8, and 30.0 ± 4.39 for the lung, liver, and heart, respectively (Table 4). Late Ct values (37.1 and 38.7) were obtained from the brain of 2 of 3 cases; no amplification of T. gondii DNA was obtained from the brain of case 4. The N. caninum Nc5 genomic marker was not detected in any case, and no bacteria were cultured from lung specimens in any of the cases.
Antibody screening
We evaluated 8 anti–T. gondii antibodies by IHC (Table 2). Among these, 3 antibodies (namely monoclonal anti-microneme protein MIC3 clones T4 2F3 and A80D, and a polyclonal antibody (pAb) generated to the RH strain of T. gondii [whole cell]) demonstrated immunoreactivity in FFPE tissues. Monoclonal anti-microneme protein MIC3 clone A80D provided a very weak signal; immunostaining was, therefore, not considered adequate. Monoclonal anti-microneme protein MIC3 clone T4 2F3 and the pAb generated to the RH strain of T. gondii provided adequate immunostaining, and specificity testing was performed using bovine turbinate cell pellets infected with T. gondii, N. caninum, and S. neurona. Only anti-MIC3 clone T4 2F3 was specific for T. gondii, with no cross-reactivity to the closely related apicomplexan protozoan organisms N. caninum and S. neurona (Fig. 5). Although the pAb to the RH strain of T. gondii did not cross-react with S. neurona, it showed cross-reactivity to both T. gondii and N. caninum.
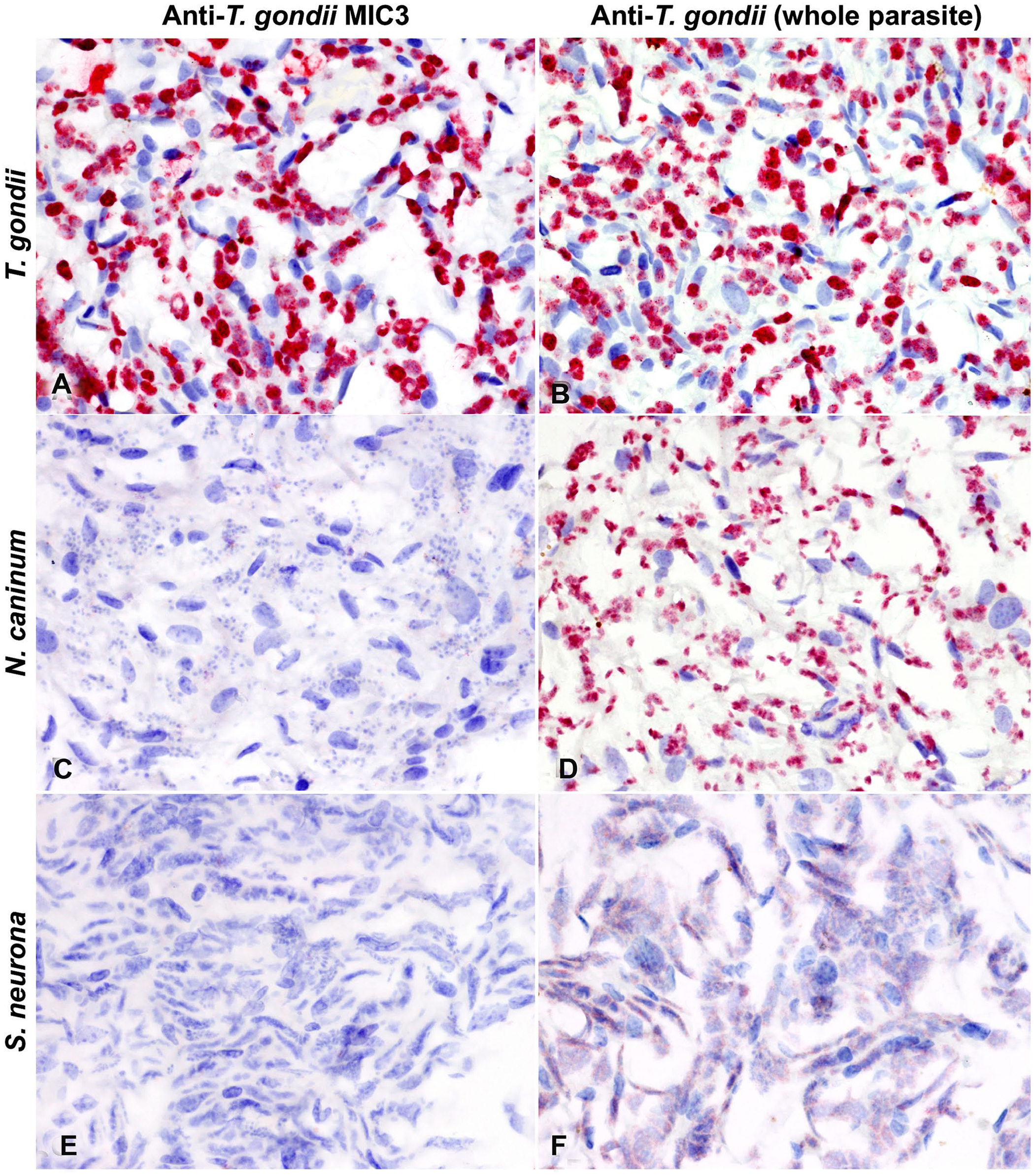
Specificity of 2 anti–Toxoplasma gondii antibodies (anti-MIC3 T4 2F3 monoclonal antibody [
IHC findings in tissues from affected kangaroos
Based on the screening results, anti-MIC3 clone T4 2F3 was used for immunostaining of kangaroo tissues. Sections from the lung, liver, heart, spleen, adrenal gland, kidney, brainstem, cerebellum, cerebrum, pituitary gland, choroid/retina, gastrointestinal tract, pancreas, mesenteric lymph node, ovary, uterus, and mammary gland were evaluated by IHC using the anti–T. gondii microneme protein MIC3 T4 2F3 mAb. This anti-MIC3 mAb identified both parasitophorous vacuoles containing zoites as well as tachyzoites within tissue sections. Among the tissues examined, the lungs had the highest number of organisms, mostly as intracellular (presumptively in intracytoplasmic parasitophorous vacuoles within histiocytes and pneumocytes) and free tachyzoites (Fig. 2, Table 4), with lower numbers of parasitophorous vacuoles containing zoites, followed by the heart (median of 4 parasitophorous vacuoles per 1.19 mm2) and liver (median of 2 parasitophorous vacuoles per 1.19 mm2). Free tachyzoites were also frequent in other non-pulmonary tissues (adrenal gland, pancreas, gastrointestinal tract, mesenteric lymph node) with sporadic parasitophorous vacuoles containing zoites prevailing elsewhere (Table 4, Fig. 3).
As expected, a larger number of parasites were identified by IHC compared to regular histologic evaluation, which mainly identified larger parasitophorous vacuoles containing zoites and sporadically free tachyzoites. Although there was a significant positive correlation (r = 0.715; 95% CI: 0.529–0.836; p < 0.0001) between the number of parasites identified by IHC and histologically (n = 46 total tissues), the agreement between these was low (kappa = 0.188; 95% CI: 0–0.390), demonstrating that parasites are more readily identifiable via IHC than by histopathology on H&E-stained tissue sections.
Discussion
Macropodids are animal species known to be highly susceptible to toxoplasmosis, which can have devastating effects on marsupial populations. 32 Although previous studies have evaluated the gross and microscopic lesions in several Australian marsupials, reports on IHC findings are scarce and limited to the use of antibodies that have been poorly characterized, with no known cross-reactivity to other apicomplexan protozoa.6,10,17,29 Gross and histologic findings in our outbreak of acute toxoplasmosis in captive red kangaroos in Louisiana were similar to previous reports (Table 5), and were characterized by diffuse interstitial pneumonia, myocarditis, myositis, encephalitis, hepatitis, and occasionally adrenalitis. Interestingly, all animals evaluated here had multifocal choroiditis, with one case of optic neuritis with intralesional parasitophorous vacuoles containing zoites and another with a small glial nodule within the neurohypophysis. The only female kangaroo examined (case 4) also had similar changes in the ovary. Unaffected organs in these animals included the spleen and kidneys.
Comparison of pathologic findings associated with Toxoplasma gondii infection in 3 red kangaroos.

The primary challenge associated with antibody-based assays of apicomplexan parasites, such as IHC and serologic assays, is the extensive cross-reactivity, particularly between T. gondii and N. caninum, that makes confirmation of the diagnosis difficult.20,37,40 In a previous study, which used polyclonal T. gondii antiserum, tachyzoites stained specifically, but tissue cysts were inconsistently and unevenly stained. 6 As part of our study, we evaluated a panel of monoclonal and polyclonal antibodies to T. gondii to identify suitable and highly specific antibodies for IHC, which could allow the specific detection of T. gondii over other closely related apicomplexan protozoa in FFPE tissues. To our knowledge, the specificity of these antibodies has not been reported previously. We identified a mAb specific to the MIC3 microneme protein of T. gondii for use in IHC that does not cross-react with N. caninum or S. neurona. This antibody demonstrated reactivity to both parasitophorous vacuoles containing zoites and tachyzoites located in the lungs, liver, and myocardium. Parasitophorous vacuoles were distributed more widely than tachyzoites, which were only identified in the lung, spleen, adrenal gland, and brainstem. Among the latter tissues, tachyzoites were overly abundant in lung sections from all animals, but rare in other tissues. We hypothesize that this difference could be associated with the stage of the disease, efficiency of the host immune response in different tissues, and differences in cellular tropism.
We also adapted a TaqMan rtPCR assay from human to animal tissues for the specific identification of T. gondii. 18 Even though the small number of animals and tissues analyzed precludes establishment of a precise correlation between T. gondii genomic DNA copies and the number of parasites (tachyzoites and parasitophorous vacuoles containing zoites) quantified in tissue sections, Ct values may be in line with the theoretically expected correlation with parasite burden. In our study, although there was a significant positive correlation between detection of the parasites histologically and via IHC, the detection rate of parasites was, as expected, higher with IHC than on H&E-stained sections, which accounts for the low agreement between these 2 methodologies. Thus, our study demonstrates that IHC is a useful tool for the detection of T. gondii when their identification on H&E-stained sections may be elusive. Although the use of PCR has revolutionized infectious disease testing, it is important to highlight that IHC has proven to be a useful tool for the pathologist to diagnose toxoplasmosis in several animal species as well as for screening purposes (e.g., at the slaughterhouse).21,24,36,38 IHC is also available for use in humans and can be performed easily on different body fluids for the diagnosis of acute toxoplasmosis. 30 To our knowledge, a large panel of antibodies to T. gondii used to identify suitable and specific antibodies for IHC has not been reported previously, nor has the use of monoclonal anti-MIC3 T4 2F3 for IHC procedures been reported. 19
In our outbreak, as in many others described in the literature, the presence of domestic cats within the facility was the most likely epidemiologic link. Contamination of the environment with oocysts, including water and food, was the most likely source of infection for this group of kangaroos. It is possible that older animals transferred from other facilities could have been infected in the past and developed recrudescence of disease; however, the high attack rate during this outbreak makes this an unlikely possibility, and the active infections in young animals born on-site could only have occurred at the facility based on the herbivorous nature of this species and the epidemiology of T. gondii. Therefore, our outbreak highlights the importance of controlling the access of cats to facilities in which Australian marsupials are housed.
Footnotes
Acknowledgements
We thank the members of the Histology and Immunohistochemistry sections at the Louisiana Animal Disease Diagnostic Laboratory (LADDL), School of Veterinary Medicine, Louisiana State University for their assistance. We also thank Ms. Michelle Yeargan at the University of Kentucky for her help in generating cell pellet controls. The following reagent was obtained through BEI Resources, National Institute of Allergy and Infectious Diseases (NIAID), National Institutes of Health (NIH): Monoclonal Anti-Toxoplasma gondii Micronemal Protein 3, Clone T4 2F3 (produced in vitro), NR-50261.
Declaration of conflicting interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
This study was partially supported by Louisiana State University, School of Veterinary Medicine start-up fund (PG 002165) to Dr. Udeni B. R. Balasuriya.
