Abstract
Porcine brucellosis, which is caused by Brucella suis biovar (bv) 2, is a re-emerging disease that causes reproductive problems in pigs in Europe. The pathogenesis and lesions of B. suis intrauterine infection are poorly characterized; characterization could facilitate the diagnosis and investigation of porcine brucellosis. We collected samples of placentas and fetuses for histologic and microbiologic studies during an outbreak of abortions on a pig-breeding farm in Spain. Brucella was cultured from the vaginal swabs obtained from sows that had aborted, some placentas, and fetal tissues (spleen, liver, lung, gastric content); molecular testing confirmed B. suis bv 2 infection. Histologically, there was necrotizing and hemorrhagic placentitis; suppurative hepatitis; lymphoid depletion and sinusoidal histiocytosis in the spleen, lymph nodes, and thymus; and bronchointerstitial pneumonia. Hemorrhages were observed in the umbilical cord, heart, kidneys, and brain. We detected Brucella by immunohistochemistry (IHC) in all of the placentas and fetal organs studied, specifically in the trophoblasts of the chorionic epithelium, in the cytoplasm of macrophages in the chorionic stroma, and extracellularly in necrotic debris. Furthermore, we assessed the lymphocyte population in the placentas through the use of IHC (anti-CD3, anti-Pax5 antibodies), revealing that the lymphocytic response was composed of T cells but not B cells.
Brucella is a pathogen that is widespread throughout the world, with enormous relevance for public health owing to its zoonotic potential and recognized economic impact on animal production. Brucellosis in domestic swine is caused by B. suis biovars (bv) 1–3 and, to a lesser extent, by B. melitensis and B. abortus.36,47 The host range, geographic distribution, and pathogenicity of B. suis differ according to the biovars: B. suis bv 1 and 3, which are zoonotic and endemically present in the Americas, Asia, Oceania, and occasionally in Europe, affect several animal species, particularly the Suidae family, but also humans, cattle, horses, and dogs.2,8,9,26,32,33 B. suis bv 2 is, in contrast, localized to Europe and has limited zoonotic potential, although it threatens European pig production and can infect cattle.12,36 Wild boar (Sus scrofa) and the European hare (Lepus capensis) are reservoirs of B. suis bv 2 and appear to be relevant in transmission to extensively farmed pigs. 47 B. suis bv 4 infects reindeer (Rangifer tarandus), the arctic fox (Vulpes lagopus), the arctic wolf (Canis lupus arctos), cattle (without causing disease), and humans.15,47 B. suis bv 5 infects rodents. 39
Sources of infection by Brucella are horizontal through uterine secretions, fetal membranes, aborted fetuses, and milk, or vertical through the placenta.1,32,39 Brucella infects the reproductive systems of both males and females, leading to infertility and abortions in many domestic and wildlife species. 48 B. suis infects the testis, epididymis, prostate, vesicular glands, and bulbourethral glands in boars.1,34,35,37,45,47 Testicular and epididymal lesions in males have been defined as necrotizing, suppurative, lymphocytic, granulomatous, or pyogranulomatous, depending on the study.1,34,36,39,45,47 Suppurative or granulomatous seminal vesiculitis with fibrosis and lymphocytic prostatitis have been reported. 45 Nonreproductive gross lesions in adults, regardless of their sex, include lymphadenitis, hepatitis, splenitis, nephritis, cystitis, pleural and mesenteric adhesions, encephalitis, arthritis, and osteomyelitis.12,34,35,37,39,45
Sows that become infected outside the gestational period will not abort in future pregnancies, but can suffer from infertility, and uterine or mammary gland abscesses.1,18,37,39,47 Histologically, suppurative endometritis is observed. 45 Infection of primiparous pregnant sows by B. suis leads to late-term abortions. Brucella tropism in the placenta causes multifocal-to-coalescent necrosuppurative placentitis and placental edema and hyperemia. 36 Of the 23 feral sows that were positive for Brucella spp. in another study, 2 had placental necrosis. 45
The pathogenesis and histologic lesions of B. suis intrauterine infection of fetuses are poorly characterized. Although the pathogenesis is not completely understood, B. suis reaches the fetus through chorionic vessels and induces fetal malnutrition and hypoxia. 1 Porcine brucellosis leads to fetal resorption, and aborted, stillborn, premature, or weak piglets.36,37,39 Fetuses born from B. suis–infected gilts have hemoperitoneum. 1 Most piglets infected in utero recover from the infection before 6 mo of age, with the risk of permanent infection being negligible. 37
Histologic lesions in the placenta and fetuses have been described for some Brucella spp. in infected ruminants. 36 In contrast, studies in swine focus on gross changes induced by B. suis bv 1 or 3, which may be adequate for the diagnosis of porcine brucellosis, but knowledge of histologic lesions of placental and fetal tissues is lacking. We retrieved no histologic or immunohistochemical reports of placental and fetal lesions in naturally occurring B. suis bv 2 abortions in domestic swine in a search of Google, PubMed, CAB Direct, Web of Science, and Scopus. Our objective was therefore to describe the histologic, immunohistochemical, and microbiologic findings in the placentas and fetuses of a series of naturally occurring abortions caused by B. suis bv 2 in domestic swine.
Materials and methods
Sample collection
During an outbreak of late-term abortions and stillbirths on a pig farm in Spain, autopsies of placentas from 4 sows and 2 fetuses or stillbirths from each of 4 aborted litters were performed in the field. Vaginal swabs from sows that had aborted, along with aseptically sampled placenta, spleen, liver, lung, and gastric content, were collected in Amies transport medium (Deltalab) for bacterial culture. Placentas and fetal tissues were collected in 10% neutral-buffered formalin.
Microbiology
The procedures for Brucella detection were performed following the Manual of Diagnostic Tests and Vaccines for Terrestrial Animals. 47 Briefly, vaginal swabs and tissue samples were cultured in parallel in Thayer–Martin modified medium (Becton Dickinson) and CITA medium, 10 and incubated at 37°C in a 10% CO2 atmosphere. Plates were checked every 2 d for 14 d.
Morphologically compatible colonies were isolated and extracted using a commercial extraction kit (MagMAX CORE nucleic acid purification kit; Applied Biosystems) and an automated extraction robot (KingFisher Flex; Thermo Fisher). The DNA extracted was quantified using a spectrophotometer (NanoDrop 2000; Thermo Fisher) and stored at −40°C until analyzed.
Brucella spp. DNA detection was performed using a qPCR protocol described previously. 3 A commercial multiplex PCR (INgene Bruce-ladder Suis; Ingenasa) was then used for the identification of B. suis bv 1–5 according to the manufacturer’s instructions. The PCR assay was conducted in a thermocycler (Bio-Rad).
The DNA/RNA from vaginal swabs was directly extracted using the DNA/RNA commercial extraction kit and automated extraction robot detailed above. Specific PCR assays for swine abortive agents (porcine reproductive and respiratory syndrome virus [Betaarterivirus suid], porcine herpesvirus 1 [Suid alphaherpesvirus 1], porcine herpesvirus 2 [Suid betaherpesvirus 2], porcine circovirus 2, porcine parvovirus 1 [Ungulate protoparvovirus 1], Leptospira interrogans, Chlamydia spp., and Toxoplasma gondii) 40 yielded negative results.
Histologic and immunohistochemical study
Placenta and fetal tissue samples were fixed in 10% neutral-buffered formalin for 24 h and processed routinely; sections were stained with H&E. For immunohistochemistry (IHC), paraffin sections were placed on charged slides, deparaffinized in xylene, and rehydrated in an ethanol series. After heat-induced epitope retrieval using a pressure cooker with citrate buffer pH 6 (Panreac Química), the samples were incubated in a hydrogen peroxide solution in methanol (Panreac Química) to quench endogenous peroxidase. The samples were then incubated, first in horse serum and then in primary antibodies: rabbit polyclonal anti-Brucella (1:200; Bioss Antibodies) in the case of the placentas and fetal samples; rabbit polyclonal anti-human CD3 (clone UCHT1, 1:200; Dako-Agilent) and mouse monoclonal anti-human Pax5 (1:25; Dako-Agilent) antibodies for placentas. Commercial reagents were used for the secondary antibody (ImmPRESS-VR horse anti-rabbit IgG polymer kit; Vector) and chromogen (ImmPACT NovaRED peroxidase substrate; Vector). Sections of tissue with culture results that were positive for Brucella were used as positive controls for Brucella; spleen from an unrelated pig was used as a positive control for the CD3 and Pax5 antibodies. With regard to the negative controls, the primary antibody was replaced with a commercial universal negative control reagent. Finally, the samples were counterstained with hematoxylin and coverslipped.
Results
Microbiology
Brucella was cultured from the vaginal swabs obtained from all 4 sows, 2 of the 4 placenta samples, and all fetal tissues (spleen, liver, lung, gastric content) from 8 of 8 fetuses. B. suis bv 2 was confirmed in all of the isolates (Table 1).
Results of histologic, immunohistochemical, bacteriologic, and molecular studies of porcine placentas and fetuses naturally infected by Brucella suis biovar 2.
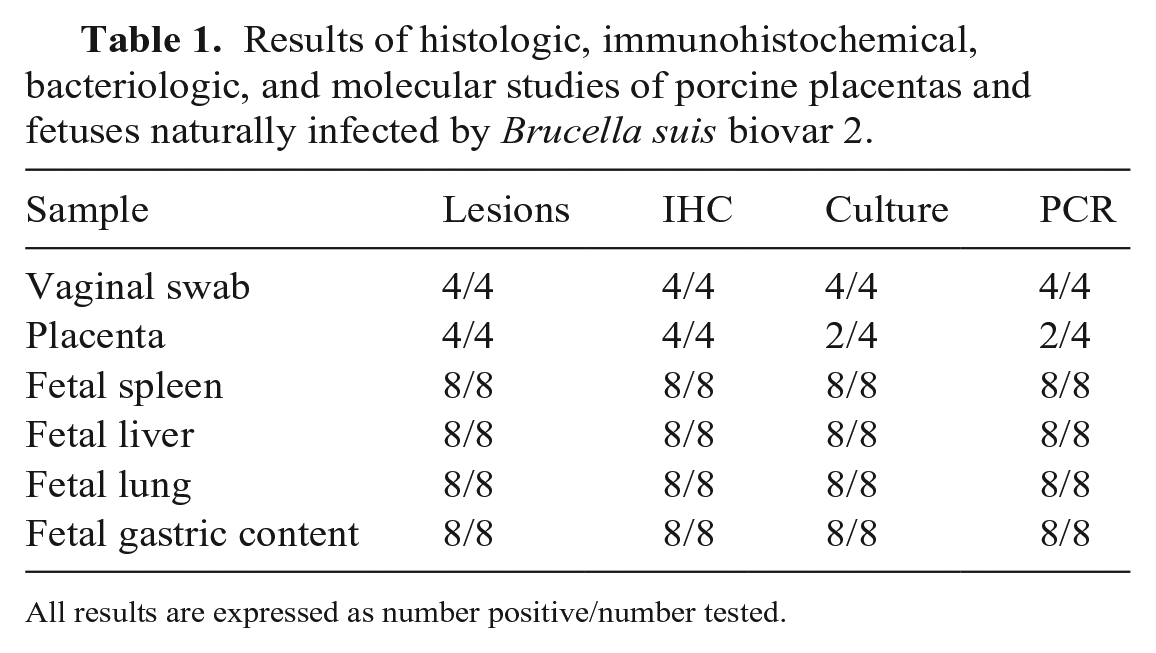
All results are expressed as number positive/number tested.
Histopathology
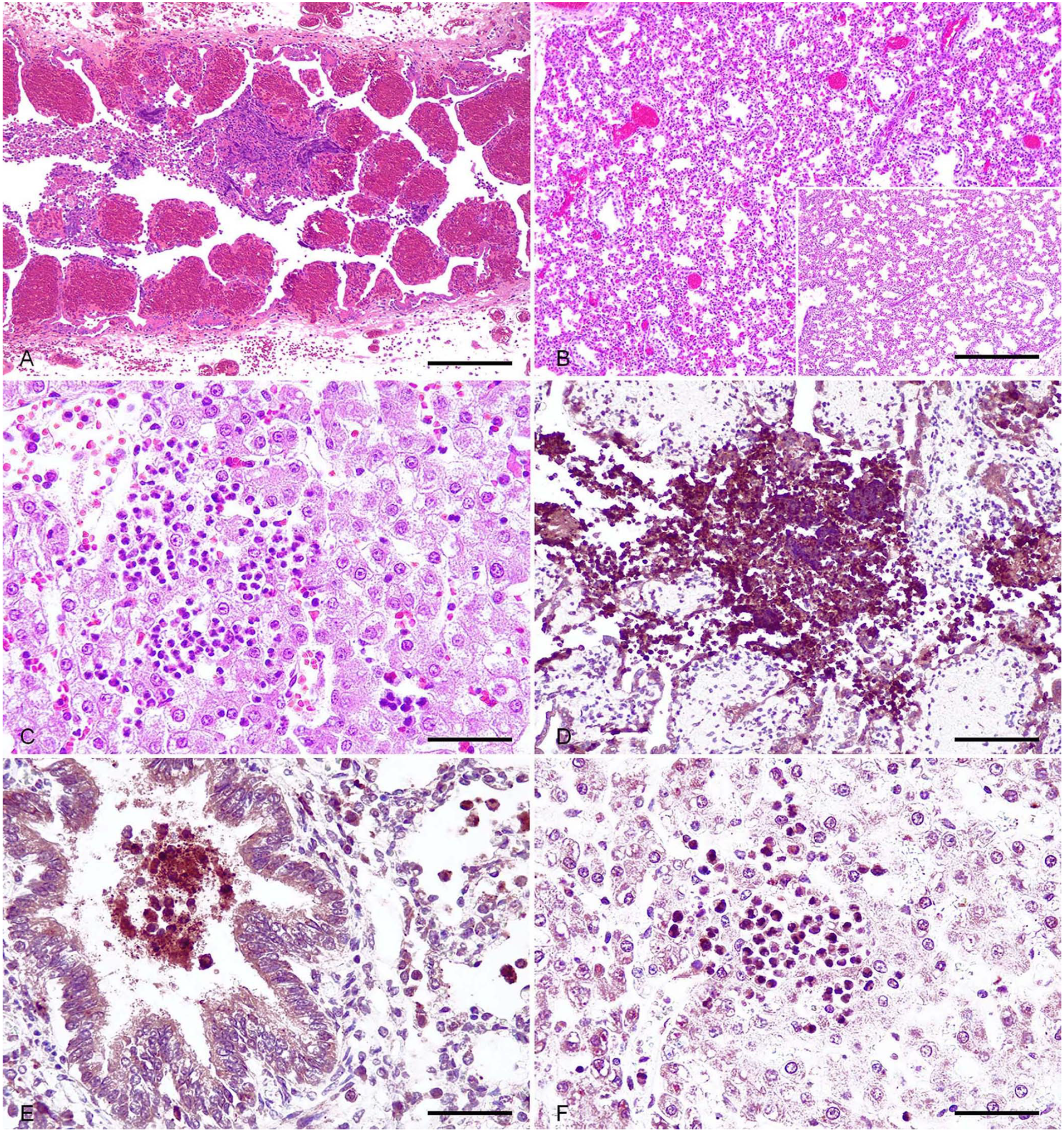
In all cases (including those with no bacterial growth), histologic evaluation of the placentas revealed severe, acute, focally extensive, necrohemorrhagic placentitis. The chorionic stroma of the placental villi was expanded by hemorrhage (Fig. 1A), and occasional macrophages, neutrophils, and lymphocytes. Trophoblasts of the chorionic epithelium were necrotic and had desquamated occasionally into the lumen. The stroma of the villi not displaying hemorrhage and necrosis was markedly expanded by edema. The umbilical cords had moderate-to-severe perivascular hemorrhage in all cases.
Histopathologic and immunohistochemical findings in the placentas and fetuses of domestic swine naturally infected with Brucella suis biovar 2.
The lungs of half of the fetuses from the litters of sows 2 and 3 had mild-to-moderate, diffuse-or-focally extensive lymphocytic bronchointerstitial pneumonia characterized by thickening of alveolar septa, especially in the peribronchial and peribronchiolar areas (Fig. 1B). Bronchi and bronchioles contained abundant necrotic debris, and there were viable and degenerate neutrophils in all of the fetuses, as well as desquamative alveolitis. Amniotic epithelial cells and meconium were occasionally observed in terminal bronchioles and alveoli.
The livers of all the fetuses were marked by moderate acute multifocal suppurative periportal and random hepatitis, with occasional hepatocellular necrosis (Fig. 1C). Groups of lymphoid cell precursors were observed occasionally in periportal areas.
Fetal spleens were unremarkable. Lymphoid depletion was marked in the cortex of the thymus and lymph nodes. The splenic red pulp, medullary sinuses of the lymph nodes, and thymic medulla additionally contained increased numbers of macrophages and, to a lesser extent, neutrophils, with the latter occasionally infiltrating Hassall corpuscles in the thymus. The heart, kidney, and brain had mild multifocal interstitial hemorrhage.
Immunohistochemistry
In the placenta, Brucella IHC revealed an intense granular positive immunoreaction that was diffuse in the epithelium of the chorionic villi and the cytoplasm of macrophages surrounding necrotic areas in all placentas, including those with an absence of bacterial growth (Fig. 1D). IHC targeting CD3 revealed a few T cells infiltrating chorionic villi (Suppl. Fig. 1A). The Pax5 IHC yielded negative results (Suppl. Fig.1B).
Positive granular cytoplasmic immunoreaction to Brucella was observed in the macrophages surrounding umbilical vessels, in alveolar macrophages, and in necrotic debris in the lung airways (Fig. 1E), as well as in the cytoplasm of neutrophils infiltrating periportal areas in the liver and Kupffer cells (Fig. 1F).
In the spleen, intense granular cytoplasmic immunoreaction was observed in macrophages surrounding penicillar arterioles and in the red pulp. In the thymus, immunoreaction was observed in the medulla, specifically in the cytoplasm of macrophages.
In the heart, kidney, and brain, Brucella immunoreaction was intracytoplasmic in scattered macrophages in the heart, renal glomeruli and tubules, and the choroid plexuses in all of the fetuses. The intestinal epithelium was also immunoreactive to Brucella.
Discussion
Brucellosis is a notifiable zoonotic disease that has a great impact on ruminants, causing serious economic losses, especially in developing countries. The hallmark lesion of brucellosis is placentitis and fetal infection, leading to abortions and stillbirths. However, the literature contains limited data regarding the histologic characterization of placental and fetal lesions caused by B. suis, and its systemic distribution. The collection of these samples is invaluable for diagnosis, but histologic analysis is sometimes hampered by availability or autolysis.4,32
Brucella placental pathology likely differs among species because of variation in placentation. 17 Four types of placentas are recognized in domestic species based on gross morphology: diffuse, multicotyledonary, zonary, and discoid. According to their histologic structure, placentas are subclassified into epitheliochorial, endotheliochorial, or hemochorial. 21 The placentas of swine, horses, dolphins, and llamas, among other wildlife species (camels, hippopotami, lemurs, pangolins, peccaries, rhinoceroses, whales), are diffuse epitheliochorial. 6 Epitheliochorial placentas are composed histologically of the chorionic villi that come into contact with endometrium, and the chorionic plate is composed of fetal mesenchymal stroma.
In the porcine placentas that we studied, B. suis bv 2 induced necrohemorrhagic placentitis characterized by villus expansion by hemorrhage and edema, and by necrosis of villus epithelial trophoblasts. The inflammatory infiltrate was scarce and composed principally of macrophages, neutrophils, and T lymphocytes in the stroma of the villi and, to a lesser extent, in the chorionic plate. Similar lesions have been observed in abortive infections by B. ceti or B. pinnipedialis in free-ranging dolphin species (Stenella coeruleoalba, Tursiops truncatus, Cephalorhynchus hectori).4,16,19,27,31 The experimental infection of a llama with B. abortus led to lymphohistiocytic placentitis with trophoblast necrosis, as well as edema, necrosis, and mineralization of the underlying stroma. 17
We detected Brucella immunoreactivity in the epithelium of the villi, in the necrotic debris, and in macrophages present in the chorionic stroma of the porcine placentas studied. Positive immunoreactivity has been reported similarly in the chorionic villus epithelium, chorionic plate, inflammatory cells, and luminal exudate of placentas from dolphins infected with Brucella.4,19,27,31 Immunohistochemical studies carried out on a llama experimentally infected with B. abortus showed cytoplasmic immunoreactivity in trophoblasts. 17
We identified T cells (CD3+) infiltrating the stroma of the chorionic villi by IHC and immunophenotyping of lymphocytes in swine placentas that tested positive for B. suis bv 2. Gamma-delta T-cell receptor–positive cells have been recognized as a resident population in the intercotyledonary placental tissue of sheep but have not been reported in swine. 6 Moreover, CD4+ CD8+ T cells are present in equine endometrial cups. 6 The T-cell infiltration that we observed may be part of the physiologic lymphoid population in swine placenta or may be part of the placental immune response to B. suis. Therefore, further studies focused on the characterization of the physiologic lymphocyte population in the porcine placenta, and on the cellular immune response to Brucella infection in swine, are warranted.
Brucella organisms gain access to the fetus through the placenta and umbilical cord, 27 and may also be present in the amniotic fluid. Here, we identified suppurative hepatitis in the periportal areas of porcine fetuses infected with B. suis bv 2. Periportal hepatitis has been reported in canine fetuses infected with B. canis, 22 in ovine fetuses infected with B. melitensis, 24 and in bovine fetuses following abortion as the result of B. abortus or B. abortus strain RB51 infections.13,38,48 Experimental B. melitensis infection in pregnant dams leads to severe hepatic necrosis in fetuses. 28 Experimental infections may cause severe lesions compared with natural infections, given that we discovered only discrete foci of hepatocellular necrosis. Here, immunoreaction to Brucella yielded positive results in scattered cells in the umbilical cord, as well as in the areas of inflammation and in Kupffer cells in the liver. Positive immunoreactivity has been reported similarly in the umbilical cord and liver of stillborn and neonatal dogs infected with B. canis, 11 in the liver of a bovine fetus infected with B. abortus, 38 and in ovine, caprine, and mouse fetuses infected with B. melitensis,24,28,46 mainly in macrophages located in periportal areas and in Kupffer cells.
Brucella placentitis and placenta insufficiency lead to anoxia and fetal distress, causing the aspiration of amniotic fluid containing bacteria, and hence pneumonia in many species. 24 We identified bronchointerstitial pneumonia in 2 porcine fetuses. Bronchointerstitial pneumonia with occasional fibrinous pleuritis caused by B. abortus infection has also been identified in bovine fetuses.5,14,25,48 B. abortus experimental intrauterine infection in sheep led to suppurative bronchopneumonia and fibrinous pleuritis in fetuses. 20 B. melitensis caused bronchopneumonia or interstitial pneumonia in naturally infected ovine fetuses 24 and experimentally infected caprine fetuses. 28 B. abortus RB51 vaccination has resulted in fetal suppurative bronchopneumonia in aborted bovine fetuses, 13 and suppurative interstitial pneumonia and periportal hepatitis in swine fetuses. 44 B. abortus S19 vaccine did not cause lesions in porcine fetuses. 49 The difference in the virulence and lesions caused by B. abortus RB51 and S19 strains in swine may be a consequence of different lipopolysaccharide (LPS) structures (smooth LPS in S19, rough LPS in RB51).44,49 Dolphin and whale fetuses infected with Brucella develop interstitial pneumonia, amniotic squames, and macrophages in the airways, as we observed in swine fetuses.4,29 We additionally observed meconium in the airways in some cases, which could be attributed to fetal anoxia that led to the aspiration of amniotic fluid. 25 This phenomenon has been reported in the Brucella-infected fetuses of cattle, bison, and dolphins, and is evidence of fetal distress.7,42
We saw the immunohistochemical detection of Brucella in porcine fetuses infected with B. suis bv 2 as intracellular immunoreactivity in bronchiolar epithelium and alveolar macrophages, and extracellular immunoreactivity in the luminal exudate. Brucella detection utilizing IHC in cattle, ovine, caprine, bison, canine, and dolphin fetuses similarly revealed a cytoplasmic reaction in alveolar macrophages and occasionally in neutrophils, and was extracellular in the luminal debris and meconium.4,11,28,38,41
B. abortus and B. melitensis infection in bison and caprine fetuses, respectively, cause splenic necrosis.28,41 We observed less severe lesions in porcine fetuses, characterized by a lack of development of white pulp components. This contrasts with the white pulp hyperplasia reported in the B. melitensis infection of goat fetuses 28 and bovine abortions caused by B. abortus or B. abortus RB51,13,32,48 and could be explained by the greater pathogenicity of these Brucella species, especially B. melitensis. We discovered increased numbers of macrophages in the red pulp in the porcine fetal spleens in concordance with the lesions observed in caprine fetuses infected with B. melitensis. 28 As observed for B. melitensis–infected goat fetuses, 28 we detected immunoreactivity in porcine fetal spleen in the cytoplasm of macrophages.
We noted lymphoid depletion in the thymus of aborted porcine fetuses, and, to a lesser extent, in lymph nodes. Our observations contrast with the lymphoid hyperplasia observed in bovine fetuses infected with B. abortus 14 but coincide with the thymic lymphoid depletion reported in bovine fetuses that were aborted as the result of B. abortus and B. abortus RB51.13,14,32,48 In agreement with our findings, sinus histiocytosis in lymph nodes has been documented in ovine fetuses after B. abortus experimental intrauterine infection, 20 and in bovine abortions resulting from B. abortus RB51. 13 Positive immunoreaction has been reported in macrophages of the red pulp in the spleen of ovine fetuses infected with B. melitensis, 24 as we observed in porcine fetuses infected with B. suis bv 2.
We observed no renal lesions, aside from multifocal hemorrhages, in porcine fetuses infected with B. suis bv 2, although suppurative or lymphocytic interstitial nephritis has been reported in bovine fetuses infected with B. abortus or the B. abortus RB51 vaccine, respectively,13,38,48 as well as in safety and efficacy vaccination studies using a natural rough mutant of B. suis (strain 353-1) in 6-wk-old pigs.34,43 Experimental B. melitensis infection in caprine fetuses can cause necrosis of glomeruli and renal hemorrhages, and Brucella immunoreaction has been observed in glomeruli, tubules, and interstitium of both ovine and caprine fetuses.24,28 We observed interstitial hemorrhages in aborted porcine fetuses, but not necrosis. This may be explained by a higher dose of Brucella inoculated in experimental compared to natural infections, or possibly by the higher pathogenicity of B. melitensis compared with B. suis. We observed positive immunoreaction in glomeruli and tubules of swine infected by B. suis bv 2, as reported in canine fetuses infected by B. canis. 11
One of the most common routes of entry for Brucella is via the alimentary tract. 48 This route occurs more frequently in neonates and adult animals through milk ingestion or contact with secretions, respectively. However, fetuses can also ingest amniotic fluid containing Brucella. 11 Indeed, we detected B. suis bv 2 by culture in the gastric content in all of the porcine fetuses studied here, thus supporting this hypothesis. Furthermore, the Brucella IHC of the intestine revealed positive immunoreactivity in some enterocytes. The same findings have been reported in stillborn and neonatal dogs naturally infected with B. canis, suggesting that the oral route may make a significant contribution to in-utero infections caused by B. canis and B. suis. 11 Brucella is known to be internalized by the M cells of the gut-associated lymphoid tissue (GALT) in cattle and mice.11,48 We were unable to demonstrate Brucella in GALT, given that GALT had not yet developed in the fetal intestinal samples studied.
B. abortus rarely causes diffuse lymphoplasmacytic and granulomatous leptomeningitis in bovine fetuses. 23 We observed no lesions in the CNS in aborted porcine fetuses, despite finding positive immunoreactivity in the choroid plexus, as reported for B. canis in canine fetuses. 11
The detection of Brucella in aborted samples requires bacterial isolation, which can be time-consuming, hazardous, and not very sensitive in decomposing tissues. 24 The immunohistochemical detection of Brucella in formalin-fixed, paraffin-embedded tissues is not used routinely but may contribute to the diagnosis of, and research into, Brucella abortions in domestic and wildlife species.23,24,25,30,32,38 The global distribution of Brucella and the presence of regions in which veterinary diagnostic laboratories are poorly equipped emphasizes the need to use detection techniques that are complementary to bacteriology.24,25,48 The main advantages of IHC over culture are its rapidity and specificity. 38 With regard to bovine brucellosis caused by B. abortus, IHC had a sensitivity of 82% and a specificity of 94%. 38 We demonstrated here the usefulness and accuracy of IHC in the detection of Brucella in placentas and fetal tissues resulting from porcine abortions caused by B. suis bv 2 given that all tissues had positive immunoreactivity for Brucella despite the bacteria not being recovered from some placentas. The placenta was found to have lower levels of recovery compared with fetal tissues in cattle infected with B. suis bv 1. 35 Brucella culture yielded positive results for 2 of our 4 porcine placentas, but positive Brucella immunoreactivity was detected in all 4 cases. Negative results by culture and positive results for IHC have similarly been reported in fetuses with lesions compatible with B. abortus born from a cow with a positive serologic diagnosis. 38 The concordance between results for bacterial culture and IHC in tissue samples requires a larger sample size to evaluate the complementary use of these techniques for Brucella detection. The usefulness of gross and histologic findings along with IHC should be considered during the diagnosis of B. suis bv 2 abortions.
Supplemental Material
sj-pdf-1-vdi-10.1177_10406387231163867 – Supplemental material for Histopathologic and immunohistochemical findings in the placentas and fetuses of domestic swine naturally infected with Brucella suis biovar 2
Supplemental material, sj-pdf-1-vdi-10.1177_10406387231163867 for Histopathologic and immunohistochemical findings in the placentas and fetuses of domestic swine naturally infected with Brucella suis biovar 2 by Agustín Rebollada-Merino, Teresa García-Seco, Marta Pérez-Sancho, Lucas Domínguez and Antonio Rodríguez-Bertos in Journal of Veterinary Diagnostic Investigation
Footnotes
Acknowledgements
We thank Gabriela Torre (VISAVET Health Surveillance Centre) for her technical support.
Declaration of conflicting interests
The authors declared that there are no potential conflicts of interest with respect to the research, authorship, and/or publication of this paper.
Funding
Agustín Rebollada-Merino is the recipient of a Spanish Government–funded PhD contract for Research Staff Training (FPI) granted by the Spanish Ministry of Science and Innovation and the Spanish Ministry of Universities (RTI2018-098658-B-C22; PRE2019-087439).
Supplemental material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
