Abstract
Real-time reverse transcription polymerase chain reaction (RRT-PCR) is commonly used for the rapid detection, as well as to determine the subtype, of avian influenza viruses (AIVs). There are 16 known serologically distinct hemagglutinin (HA) subtypes of AIV described. Currently, determination of the subtypes of AIVs by RRT-PCR tests has been limited to the H5 and H7 subtypes. In this study, RRT-PCR assays were developed in simplex formats for rapid detection of AIV subtypes H6, H9, and H11. The primers and probes for RRT-PCR were designed from nucleotide sequences of the HA genes, which were either downloaded from GenBank (for H6 and H9) or sequenced for this study. The specificity and sensitivity of the RRT-PCR assays were determined based on the detection of the virus from a proficiency panel consisting of 15 different HA subtypes of AIVs and from serial dilutions of target viral RNA. The subtype-specific RRT-PCR assays were used to detect the virus in cloacal and oropharyngeal swabs of experimental chickens inoculated with H6, H9, and H11 AIVs, and the test results were compared with validated RRT-PCR assays based on the amplification of AI matrix (MA) gene. A high correlation of the matrix test and the specific H6, H9, and H11 by the RRT-PCR assays was observed; kappa coefficients for the agreement of test results in cloacal and oropharyngeal swabs combined were 0.927, 0.962, and 0.981, respectively.
Keywords
Intoduction
Avian influenza viruses (AIVs) are negative-sense RNA viruses belonging to the family
Low pathogenic avian influenza (LPAI) viruses, although causing little or no disease in specific pathogen-free chickens, in the field can cause serious disease and high mortality in conjunction with other infectious agents or poor environmental conditions. 5 There have been several reports on outbreaks in poultry with LPAI viruses other than the subtypes H5 and H7, the most notabe of which are the subtypes H6N2 and H9N2. 2,20,38 Many of the LPAI virus subtypes, including H6 and H9, can be routinely detected in wild birds, live bird markets, and poultry raised with exposure to wild birds. 21,33,38 Lowly pathogenic avian influenza virus H9N2 infections have also been reported in humans and pigs. 27,34,35 Evidence for gene transfer (reassortment) between LPAI and HPAI viruses has been suggested for the origin of the HPAI virus A/Hong Kong/156/97 (H5N1) that infected humans in Hong Kong in 1997. 10,27
The most commonly used protocols for determination of the subtype of AIVs are hemagglutination inhibition (HI) and immunofluorescence assays. These methods are generally reliable but time consuming and require large serologic panels to identify all HA and NA subtypes. Molecular methods, on the other hand, can be rapid, sensitive, and specific. Examples of molecular methods are reverse transcription polymerase chain reaction (RT-PCR), 23,36,49 real-time RT-PCR (RRT-PCR), 13,40,45 nucleic acid sequence-based amplification (NASBA), 12 RT-PCR enzyme-linked immunoabsorbent assay (RT-PCR-ELISA), 9 restriction fragment length polymorphism (RFLP) assay, 8 and combined PCR-heterodu-plex mobility assay (HMA). 15 The most commonly used molecular methods for rapid subtype detection of AIVs are RT-PCR and RRT-PCR. The use of RT-PCR for rapid detection of AIVs based on the amplification of the matrix, nucleoprotein, and non-structural genes and for subtyping of AIVs based on the amplification of the HA gene have been re-ported, 15,23,36,49 but the test results often need to be confirmed by additional tests, such as nucleotide sequencing of the PCR-amplified products. Subtyping of ArVs by RRT-PCR is sensitive and rapid and typically does not need further verification. In this study, RRT-PCR assays were developed for subtyping H6, H9, and H11 viruses based on the amplification of the HA gene. The sensitivity and subtype specificity of RRT-PCR assays were compared to the previously validated ATV matrix RRT-PCR from reference and experimental samples, including oropharyngeal and cloacal swabs from chickens inoculated with AIVs of the subtypes H6, H9, and H11. The results show that all 3 RRT-PCR tests have high specificity and sensitivity in the subtyping of the viruses tested.
Materials and methods
Viruses
All viruses used in this study were propagated in 9- to 10-day-old embryonating chicken eggs and harvested from amnioallantoic fluids (AAF) of inoculated eggs after 2 to 3 days of incubation at 37°C. The 50% egg infectious dose (EID50) titer of the virus in allantoic fluid was determined by serial dilutions in eggs and calculated by the method of Reed and Muench. 37
RNA extraction
RNA was extracted from AAF stocks or cloacal and oropharyngeal swabs with an RNeasy mini kit a or Trizol LS b with modified National Veterinary Services Laboratory (NVSL) standard operating procedures as previously described. 13 The RNA was eluted with 50 μl of RNase-free water and 8 μl of RNA used as template in a 25-μl master reaction mixture for RRT-PCR and a 50-μl master reaction mixture for RT-PCR.
Sequencing of the HA gene from†isolates
The nucleotide sequence of the full-length HA gene was sequenced from 9 North American (NA) and 1 Eurasian (EA) isolates of the AIV subtype H11. The full-length HA gene from each isolate was amplified by RT-PCR from the RNA extracted from the virus using the forward primer Hgga+ and the reverse primer H-T7. These primers were designed to match the complementary sequences at the 5′ (for Hgga+) and 3′ (for H-T7) ends of the target amplicon (HA gene). The RT-PCR was carried out with an Qiagen OneStep RT-PCR kit
a
in 50 μl of reaction mixture with the following components: kit-supplied 5x buffer 10 μl (final concentration lx), kit-supplied enzyme mixture 2 μl, the forward primer Hgga+ 0.5 μl (10 pmol), the reverse primer H-T7 0.5 μl (10 pmol), kit-supplied 10 mM dNTP mix 2 μl, RNase inhibitor RNasin,
c
0.5 μl (6.5 units), template RNA 8 μl, and RNase-free water to adjust the volume to 50 μl. The RT-PCR amplification was carried out on a GeneAmp PCR System 2700 thermocycler
d
under the following thermocycling conditions: 1 cycle of RT at 50°C for 30 min followed by heat activation of the hot start
RT-PCR and RRT-PCR
All RT-PCR assays were carried out with an Qiagen OneStep RT-PCR kit
a
in 50 μl of master reaction mixture as described previously.
13,40
The amplification of the MA gene by RRT-PCR (MA-RRT-PCR) was carried out in a multiplex format using dried down reagents (AI matrix beads) that also contained an internal positive control (IPC) to detect if PCR inhibitors are in the sample.
13
The reactions were carried out in a multiplex format using the same AI matrix forward and reverse primers but with 2 different probes: a FAM-labeled probe for detection of the virus in the FAM channel and a Cal-Fhior 610-labeled probe for detection of the IPC in the Texas Red channel.
13
The amplification of the HA gene by RRT-PCR (HA-RRT-PCR) (this report) was carried out in a simplex format according to the protocols described below. All HA-RRT-PCR assays were performed in a 25-μl master reaction mixture containing the following components: kit-supplied 5x buffer 5 μl (final concentration lx), kitsupplied enzyme mix 1 μl, RNase inhibitor0 0.5 μl (6.5 units), 25 mM MgCl2
c
1.25 μl (final concentration 3.75 mM), 10 mM dNTP mix (kit supplied), 0.8 μl (final concentration 320 μM each for dATP, dCTP, dGTP, and dTTP), RNA template 8 μl and required amounts of primers (forward and reverse), probe, and RNase-free water (described below) in a final volume of 25 μl. The final concentrations of the primers and probes in the master mixtures for HA-RT-PCR (50 μl final volume) and HARRT-PCR (25 μl final volume) varied depending on the subtype of the virus. For HA-RRT-PCR the following amounts of the primers and probes were used per reaction: 30 pmol of H6+1461N (forward primer), 10 pmol of H6–1706 (reverse primer), and 3 pmol of H6+1620 (probe) for the H6 subtype; 20 pmol of H9+1612 (forward primer), 30 pmol of H9–1706 (reverse primer), and 9 pmol of H9+1644 (probe) for the H9 subtype; and 10 pmol of H11+150 (forward primer), 10 pmol of H11–300 (reverse primer), and 4.5 pmol of H11+230 (probe) for the H11 subtype. For HA-RT-PCR the concentrations of the buffer, dNTPs, and enzymes in the reaction mixture were were the same as used for the amplification of the full-length HA gene from H11 as described above, while the concentrations of the primers were the same as used for HA-RRT-PCR. The thermocycling conditions for HA-RT-PCR were the same as used for amplification of the full-length HA gene from H11. The thermocy cling conditions for HA-RRT-PCR test had a 30-min reverse transcription step at 50°C, followed by a 15-min heat activation of the
Bird experiments
Four-week-old specific pathogen-free White Plymouth rock chickens from the Southeast Poultry Research Laboratory flocks were used in all bird experiments. Fifteen birds were used in a group for each experiment, and each bird was inoculated orally with 0.2 ml of a viral suspension (in AAF) with EID50 virus titer for each isolate as follows: 1060 for A/CK/NY/14677–13/98 (H6N2); 106.5 for A/CK/CA/K0301417Ct/03 (H6N2); and 107.0 for A/CK/NJ/12220/97 (H9N2), A/Turkey/TX/81 (H9N2), A/Chicken/NJ/15906–9/ 98 (H11N1), and A/GF/NY/2641O-17/95(H11N9). Oropha-ryngeal and cloacal swabs were collected from each bird on day 2 and day 3 postinoculation (PI) and suspended in 1.5 ml of brain heart infusion broth containing a mixture of antibiotics (per ml) including penicillin G 10,000 IU, gentamicin 100 μg, and amphotericin B 20 μg. Five hundred microliters of the brain heart infusion suspension was used for RNA extraction with RNeasy mini kit a as described above, and 8 μl of the extracted RNA was used per 25 μl of master reaction mix for RRT-PCR.
Serum samples and HI test
Serum samples were collected from all infected birds on day 28 PI before the birds were euthanized with sodium pentobarbital (100 mg/kg body weight, injected intravenously). Serum samples were also collected from some birds on day 7 and day 14 PI. Hemagglutination inhibition assays on serum were performed by standard methods as previously described. 6 The HI test was performed on 96-well plates 8 with 4 hemagglutinating units (HAU) of viral antigen and 0.5% chicken erythrocytes. 6 The HI endpoint titers were determined as the reciprocal of the highest serum dilution that completely inhibited the hemagglutination activity.
Nucleotide sequence accession numbers
The nucleotide sequences reported in this study have been deposited in GenBank under the following accession numbers: DQ424858 (A/chicken/NJ/15906–9/96), DQ435821 (A/chicken/NJ/4236–18/93), DQ435284 (A/guinea fowl/NJ/8848–17/98), DQ435282 (A/chicken/NJ/14172–14/94), DQ435283 (A/duck/Memphis/546/74), DQ435285 (A/guinea fowl/NY/26410–17/95), DQ424859 (A/green-winged Teal/LA/9GW/87), DQ424860 (A/mallard/MN/346246/00), DQ424861 (A/ruddy turnstone/NJ/650678/02), and EF200063 (A/shoveler/Netherlands/18/99).
Results
Cloning, sequencing, and nucleotide sequence analysis of the HA genes from different isolates of the AIV subtype H11
At the beginning of this project, little sequence information was available for the H11 HA gene, and sequence from additional isolates was needed for the development of specific primers and probes that could identify most H11 isolates. During manuscript preparation, additional sequences on the H11 HA gene became available that were included as part of the phylogenetic analysis. The full-length HA genes were cloned and sequenced from 10 AIV isolates of the H11 subtype as described in the Materials and Methods section. Analysis of the nucleotide sequences of the HA genes revealed higher sequence similarity (90.3–99.6% identical residues) between the 9 NA isolates. The HA gene from the single EA isolate (A/shoveler/Netherland-18/99) had lower sequence similarity (79.2–80.7% identity) with the NA isolates. A phylogenetic tree was constructed based on the nucleotide sequences of the HA genes from 31 H11 isolates including 21 sequences from the GenBank and 10 sequences from this study. The results (Fig. 1) show 2 distinct lineages, an NA lineage consisting of 22 isolates and an EA lineage consisting of 9 isolates with all the isolates clustered in the expected geographic lineages. There have been 89% to 99% identities at the sequence level between the isolates from the same geographic region (NA or EA) and 79% to 82% identities between the isolates from different geographic regions (NA and EA). When compared with HA sequences from the isolates of the 16 AI subtypes (H1-H16), all the H11 isolates were grouped in a single cluster well separated from other subtypes, with the closest subtypes being H13 (60.5–61.6% identity) and H16 (59.3–60.2% identity).
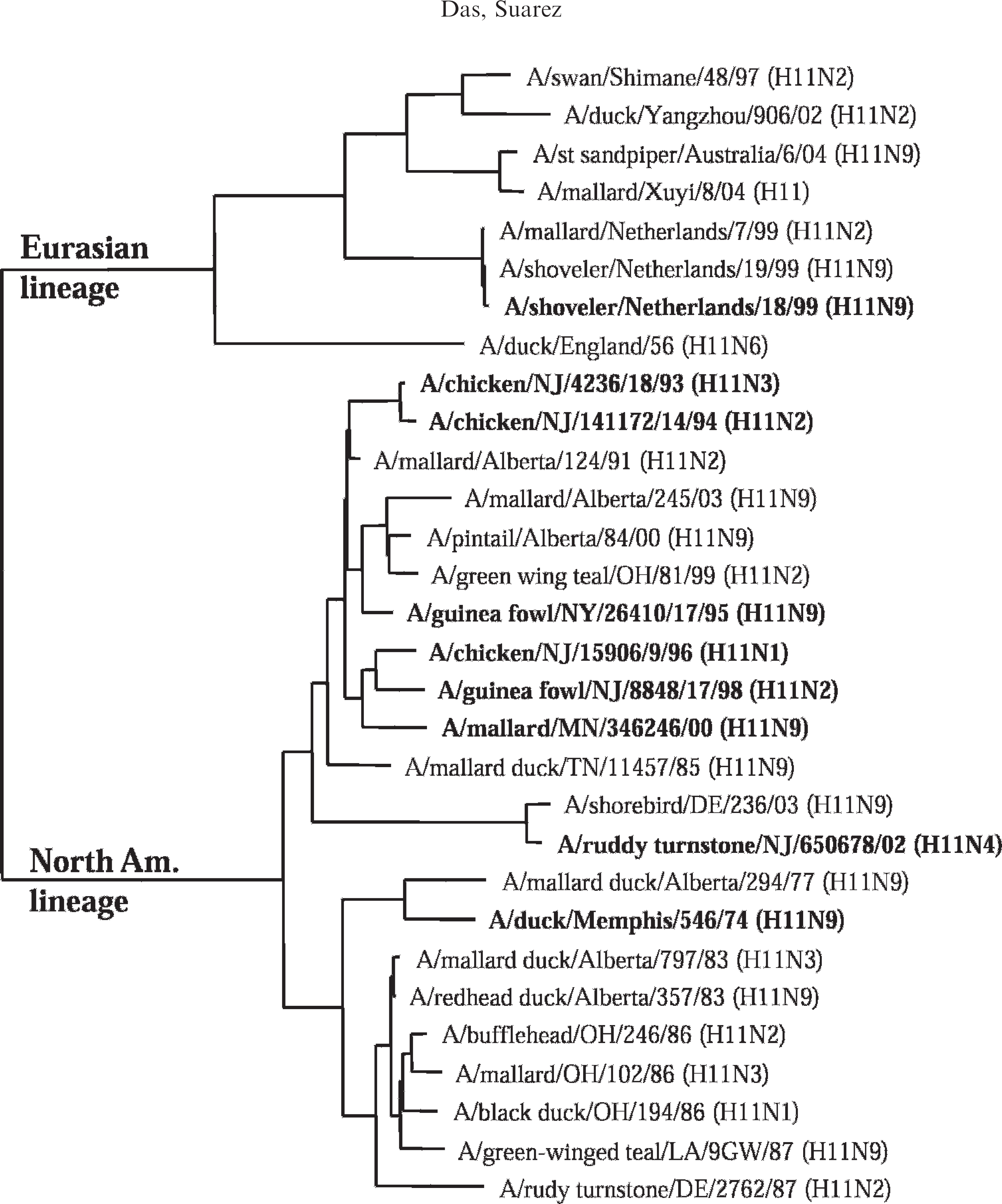
Phylogenetic tree of the different isolates of the avian influenza virus H11 subtype from North American and Eurasian origins based on the nucleotide sequences of their hemagglutinin genes. The sequences were aligned by Clustal W method using the MegAlign program. e The tree was constructed with phylogenetic analysis using parsimony neighbor joining midpoint rooting. The sequences used in the alignment were derived from either the influenza sequence database or in-house sequencing (in bold face) as reported in this study. States were abbreviated by their 2-letter postal code.
Development of RT-PCR and RRT-PCR protocols for rapid detection of the H6, H9, and H11 subtypes of AIVs
The RT-PCR and RRT-PCR protocols specific for detection of the viral RNA of the AIV H6, H9, and H11 subtypes were developed based on the amplification of the HA gene. The primers and probes were designed after a multiple sequence alignment of the available nucleotide sequences of the HA gene of the corresponding subtypes derived from GenBank or from in-house sequencing. Nucleotide sequences of all the primers and probes used in the RT-PCR and RRT-PCR are listed in table 1. Most of the primers and probes contained ambiguous bases at one or multiple sites in order to facilitate their binding to the template of multiple isolates of the given AI subtype.
Sequences of oligonucleotide primers and probes used for sequencing and real-time reverse transcription polymerase chain reaction (RRT-PCR) in this study.

The RNA from different isolates of the AIV H6, H9, and H11 subtypes were used as templates to determine the subtype specificity of the RRT-PCR protocols developed in this study. The results (Tables 2–4) show specificity in the detection of the subtype of each isolate irrespective of their geographic origins (NA or EA). The Cts for the detection of the viral RNA by RRT-PCR were below 30 for all isolates except A/Turkey/TX/89 (H9). Additionally a panel of AIVs representing 15 HA subtypes was subjected to subtype-specific HA-RRT-PCR protocols developed in this study. The results (Table 5) show specific amplification of the RNA from the AIV H6, H9, and H11 subtypes only with no cross-reactivity with RNA from the other subtypes. The subtype specificity of the HA-RRT-PCR assays was further confirmed by RT-PCR with the same HA primers that were used for HA-RRT-PCR. Only the viral RNA from the AIV H6, H9, and H11 subtypes were amplified with the primers corresponding to the subtype of the virus, and there was no amplification of the viral RNA from other subtypes (not shown).
Performance evaluation of the real-time reverse transcription polymerase chain reaction protocol for amplification of hemagglutinin gene from different isolates of the avian influenza virus subtype H6.∗
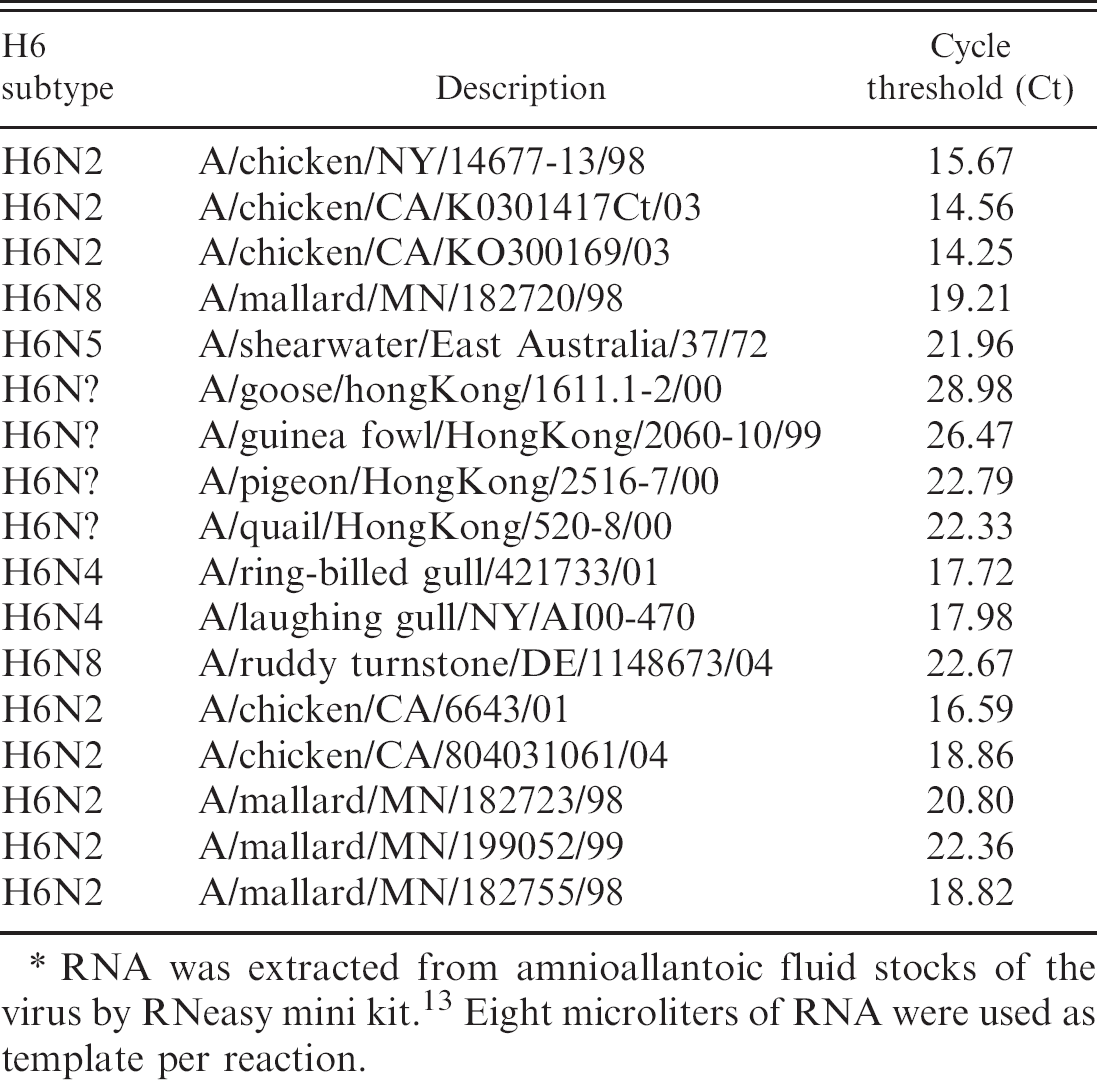
RNA was extracted from amnioallantoic fluid stocks of the virus by RNeasy mini kit. 13 Eight microliters of RNA were used as template per reaction.
Performance evaluation of the real-time reverse transcription polymerase chain reaction protocol for amplification of hemagglutinin gene from different isolates of the avian influenza virus subtype H9.∗
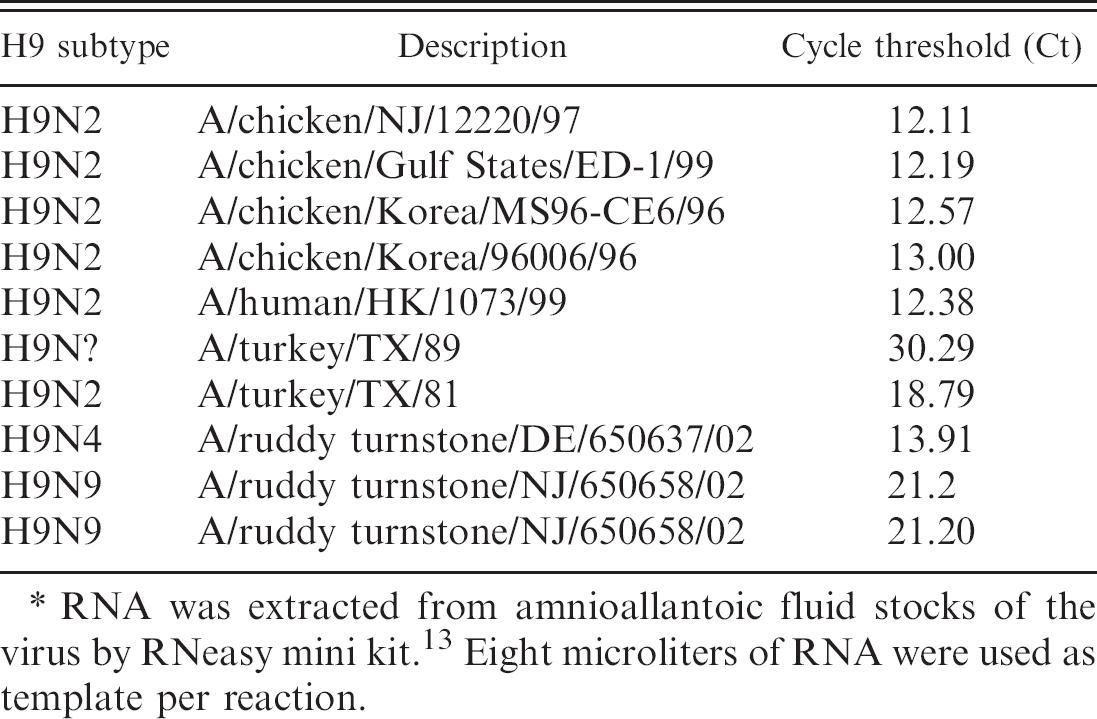
RNA was extracted from amnioallantoic fluid stocks of the virus by RNeasy mini kit. 13 Eight microliters of RNA were used as template per reaction.
Analytical sensitivity of detection of the RNA from AIV H6, H9, and H11 subtypes by RT-PCR and RRT-PCR
The analytical sensitivity of detection of the viral RNA by subtype-specific RT-PCR and RRT-PCR based on the amplification of the HA gene was carried out with serially diluted viral RNA as template (100–10−8 dilution), and the results were compared with standardized RT-PCR and RRT-PCR based on the amplification of the viral MA gene according to the conditions described in the Materials and Methods section. The results show similar sensitivity of detection of the virus by RT-PCR based on the amplification of HA or MA gene (Fig. 2). On the other hand, detection of the virus by RRT-PCR was found to be more sensitive via amplification of the matrix gene (MA-RRT-PCR) than the amplification of HA gene (HA-RRT-PCR) for the AIV H6 and H11 subtypes, while both protocols exhibited similar sensitivity for the detection of the AIV H9 subtype (Table 6). The Ct standard curves calculated from HA-RRT-PCR of the serial dilutions of the viral RNA (Table 6) revealed high correlation coefficients (
Performance evaluation of the real-time reverse transcription polymerase chain reaction protocol for amplification of hemagglutinin gene from different isolates of the avian influenza virus (AIV) subtype H11.∗
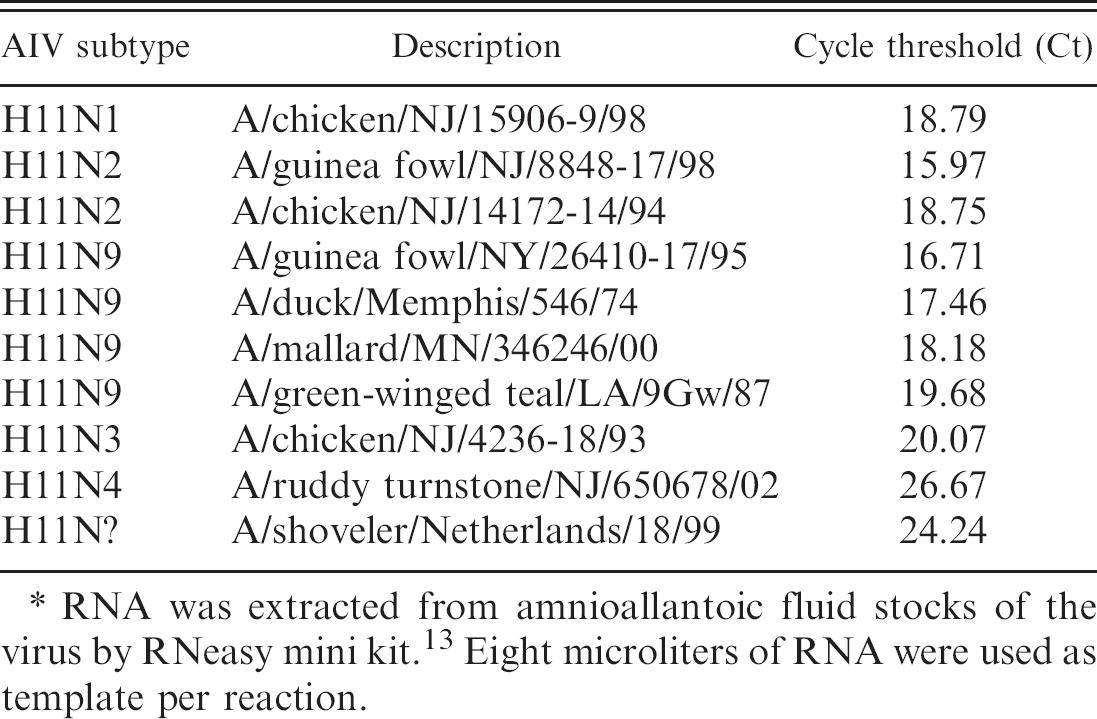
RNA was extracted from amnioallantoic fluid stocks of the virus by RNeasy mini kit. 13 Eight microliters of RNA were used as template per reaction.
Subtype specificity of the amplification of hemagglutinin (HA) gene by real-time reverse transcription polymerase chain reaction from a 15 avian influenza virus (AlV)-subtype (H1–H15) proficiency panel.∗
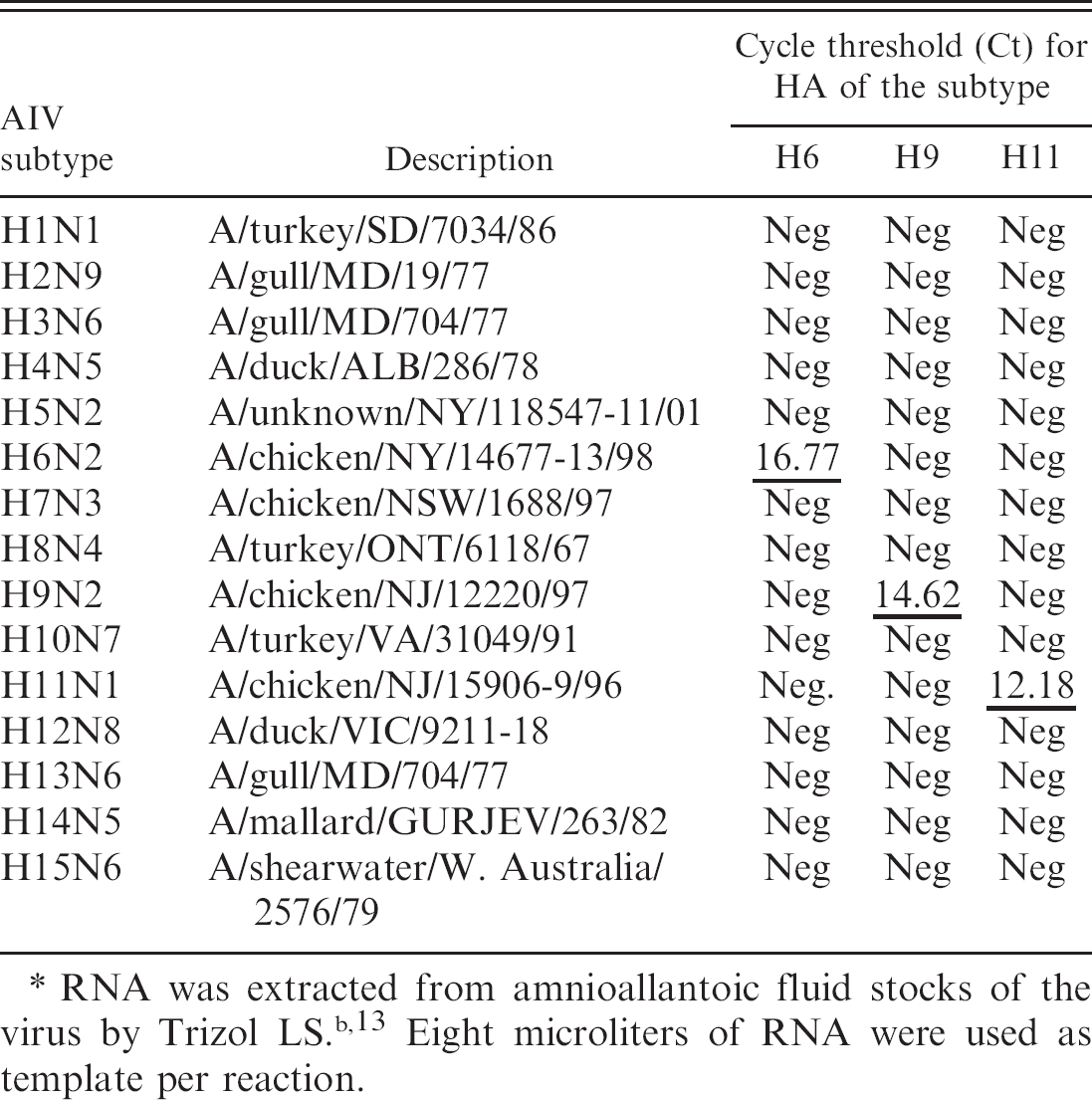
RNA was extracted from amnioallantoic fluid stocks of the virus by Trizol LS. b , 13 Eight microliters of RNA were used as template per reaction.
Detection of the virus in cloacal and oropharyngeal swabs from bird experiments with chickens inoculated with AIVs of the subtypes H6, H9, and H11
The pathogenicity of the LPAI viruses in chickens was determined by detecting the viruses in cloacal and oropharyngeal swabs of the inoculated birds by RRT-PCR as described in the Materials and Methods section. The results (Table 7) show relatively higher occurrence of the virus in oropharyngeal swabs than the cloacal swabs. The virus was not detectable in cloacal swabs of birds inoculated with A/chicken/CA/K0301417Ct/03 (H6N2) and in cloacal swabs sampled on day 2 PI from birds inoculated with A/guinea fowl/NY/26410–17/95 (H11N9) and in all but 1 cloacal swab on day 2 PI of birds inoculated with A/chicken/NJ/12220/97 (H9N2) (Table 7). There was between 91% and 98% correlation of test results between the HA-RRT-PCR and MA-RRT-PCR assays. A total of 120 samples (cloacal and oropharyngeal swabs combined) collected from 2 days PI and 3 days PI were analyzed from 2 isolates of each subtype by RRT-PCR. The numbers of samples that tested positive were 42/120 by HA-RRT-PCR and 46/120 by MA-RRT-PCR for H6; 40/120 by HA-RRT-PCR and 41/120 by MA-RRT-PCR for H9; and 47/120 by HA-RRT-PCR and 48/120 by MA-RRT-PCR for H11. The MA-RRT-PCR disagreed with the HARRT-PCR for 5, 2, and 1 on positive samples for H6, H9, and H11, respectively, and the number of samples tested positive by HA-RRT-PCR but not by MA-RRT-PCR was 1, 0, and 0 for H6, H9, and H11, respectively. The correlation of test results between MA-RRT-PCR and HA-RRT-PCR was verified by kappa statistics, h and the kappa coefficients were calculated to be 0.968, 0.790, and 0.964, for cloacal swabs and 0.898, 0.88, and 1.0, for oropharyngeal swabs for the AIV subtypes H6, H9, and H11, respectively. The overall agreements of test results between MA-RRT-PCR and HA-RRT-PCR for cloacal and oropharyngeal swabs combined (120 samples) were calculated to be 0.927, 0.962, and 0.961, for H6, H9, and H11, respectively, indicating close agreement of test results between the 2 protocols, which supported the accuracy of the subtype-specific HA-RRT-PCR for H6, H9, and H11 reported in this study.
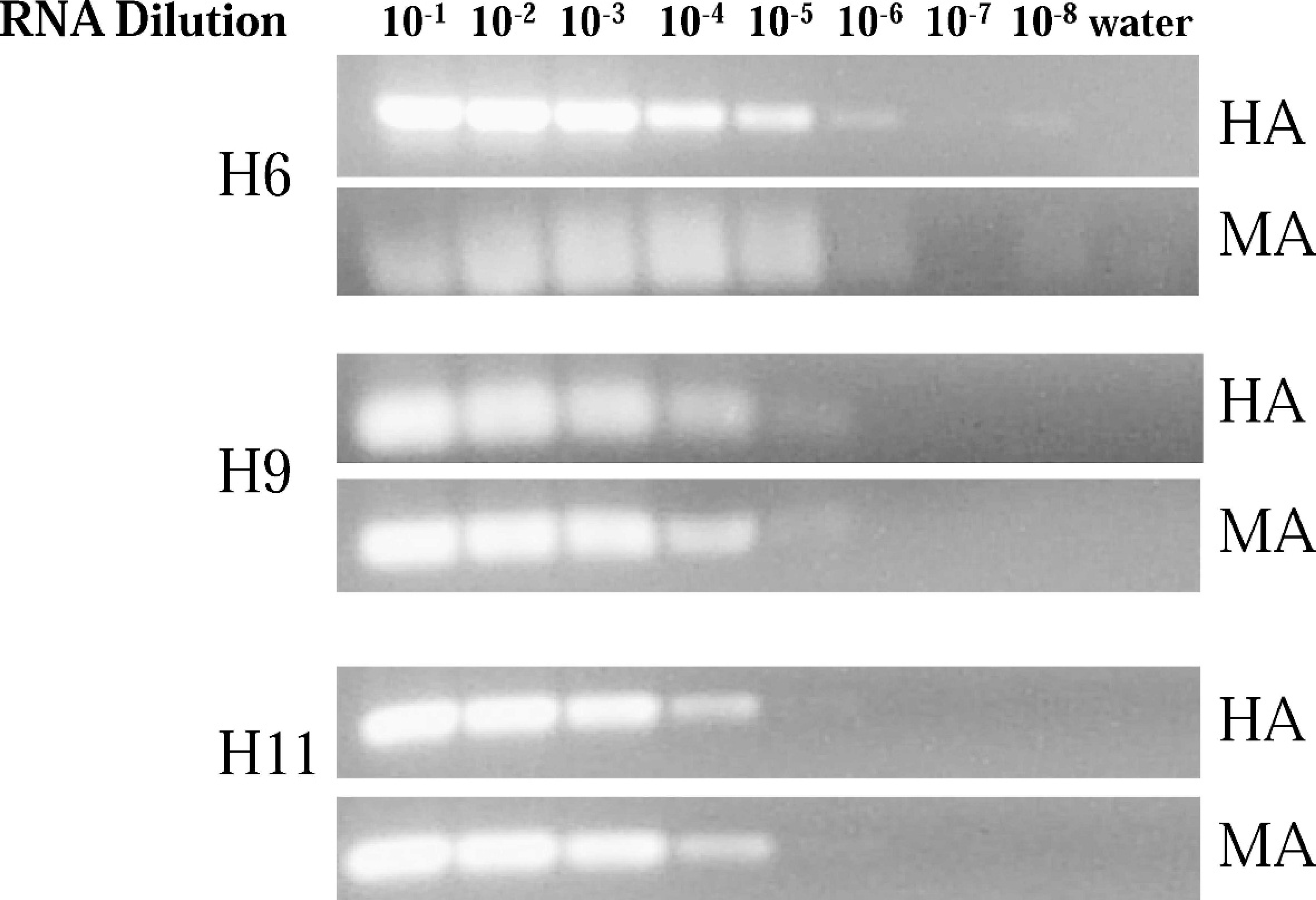
Reverse transcriptase polymerase chain reaction (RT-PCR) amplification of the hemagglutinin (HA) and matrix (MA) genes from serially diluted viral RNA corresponding to the avian influenza virus H6, H9, and H11 subtypes. Amplification of the MA gene was carried out using the primers as described previously, 40 and the amplification of HA gene was carried out with subtype-specific primers as described in the Materials and Methods section. The viral RNA used as a template for RT-PCR was from A/shearwater/East Australia/37/72 (H6N5), A/chicken/NJ/12220/97 (H9N2), and A/Ruddy Turnstone/NJ/650678/02 (H11N4) for the AIV H6, H9, and H11 subtypes, respectively. The PCR products were separated by electrophoresis on 1% agarose gels containing 0.5 μg/ml of ethidium bromide. The sizes of the PCR products amplified by RT-PCR were 100 bp for the MA gene (for all subtypes), and 245 bp, 94 bp, and 150 bp for the HA gene from AIV H6, H9, and H11 subtypes, respectively.
The antibody response to viral infection was monitored by HI assays. The sera were collected on days 7, 14, and 28 PI from birds inoculated with A/chicken/CA/K0301417Ct/03 (H6N2) and A/chicken/NJ/12220/97 (H9N2) and on day 28 PI from all inoculated birds. The average HI titers were much higher in birds inoculated with A/chicken/CA/K0301417Ct/03 (H6N2) than in birds inoculated with A/chicken/NY/14677–13/98 (H6N2) (Table 8). Differences in antibody response were also evident in birds inoculated with A/chicken/NJ/15906–9/98 (H11N9) (high HI titers) and A/guinea fowl/NY/26410–17/95 (H11N9) (low HI titers) (Table 8). Higher antibody titers against the inoculated virus correlated with increased virus shedding in cloacal and oropharyngeal swabs of the inoculated birds (Tables 7, 8).
Discussion
Rapid detection and characterization of AIVs in poultry is an important step for the control and eradication of AIV outbreaks. The subtype-specific tests were developed as a companion test to the previously validated matrix RRT-PCR test. Currently, the matrix test is recommended as one of the primary screening tests for the detection of any type A influenza virus. Further determination of the HA subtype is important to characterize the virus. Currently, HA subtyping is performed by serologic methods that require the isolation of the virus. The use of molecular diagnostic tests provides a rapid and sensitive method of determining the HA subtype either from a viral isolate or directly from the original clinical sample. The availability of HA subtype can provide a rapid confirmation of the MA test results and provide important epidemiologic information about an outbreak virus. In this study, subtype-specific RRT-PCR assays were developed for the rapid confirmation of the H6, H9, and H11 subtypes of the AIVs based on the amplification of the HA gene. The H6 and H9 subtypes of viruses are responsible for persistent outbreaks in the United States and other countries. 2,20,38 The AIVs of the subtype H11 are less common in poultry but were included as part of our long-term goal to develop RRT-PCR assays for the rapid detection of all 16 subtypes of AIVs.
End-point detection of the viral RNA of the avian influenza virus subtypes H6, H9, and H11 from serial dilution of the templates determined by real-time reverse transcription polymerase chain reaction (RRT-PCR) based on amplifications of the matrix (MA) and hemagglutinin (HA) genes.∗
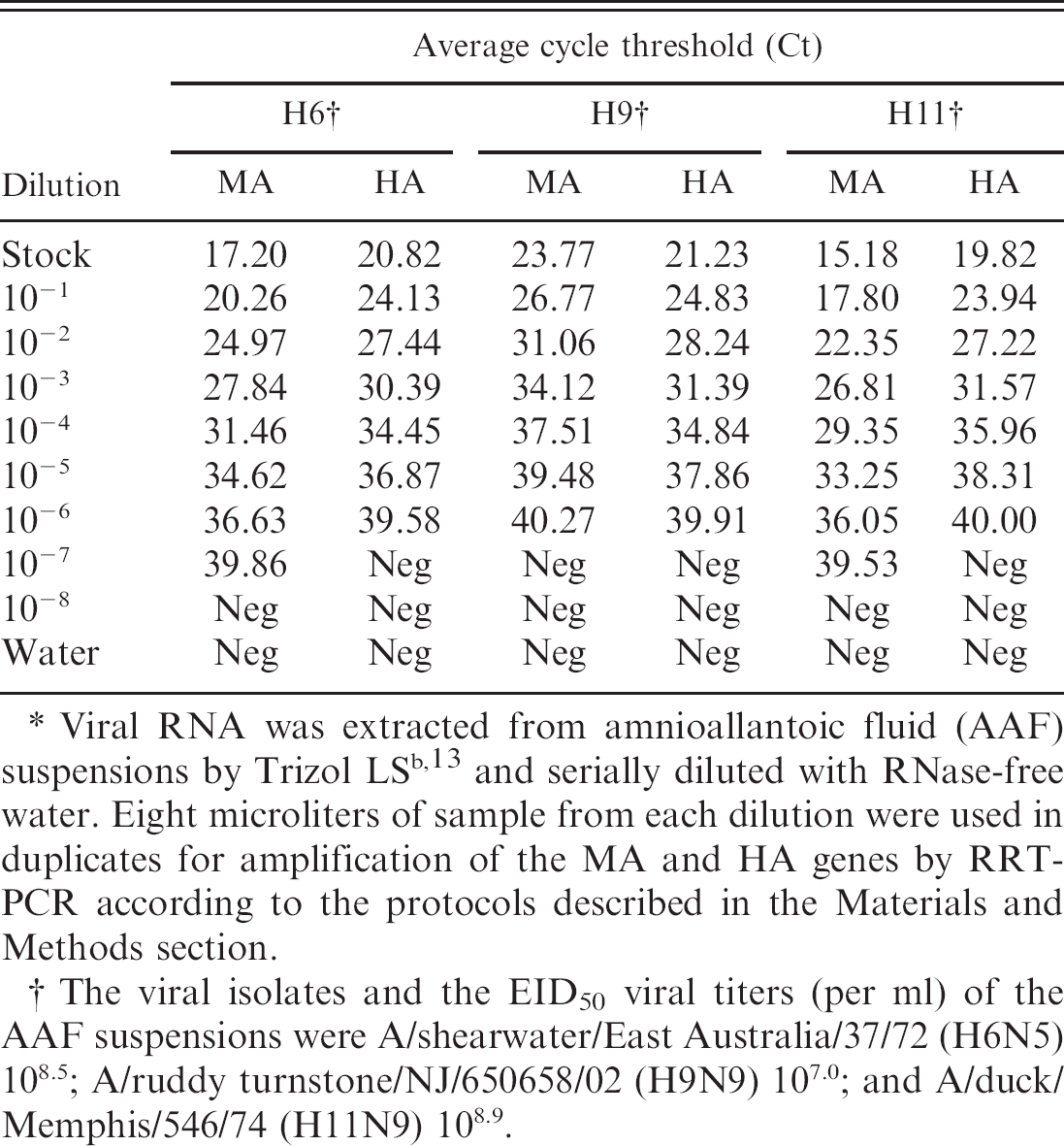
Viral RNA was extracted from amnioallantoic fluid (AAF) suspensions by Trizol LS b , 13 and serially diluted with RNase-free water. Eight microliters of sample from each dilution were used in duplicates for amplification of the MA and HA genes by RRT-PCR according to the protocols described in the Materials and Methods section.
The viral isolates and the EID50 viral titers (per ml) of the AAF suspensions were A/shearwater/East Australia/37/72 (H6N5) 108.5; A/ruddy turnstone/NJ/650658/02 (H9N9) 107.0; and A/duck/Memphis/546/74 (H11N9) 108.9.
Multiple sequence alignment of the HA sequences of the H11 isolates from different geographic origins indicate 2 distinct lineages, NA and EA, with approximately 18% to 21% differences at the sequence level between them. Similar differences between the HAs at the nucleotide sequence level were also found for the AI isolates of the H6 and H9 subtypes from NA and EA origins (not shown). Based on the differences in nucleotide and amino acid sequences of the HAs, the viral isolates of other AIV subtypes, including H5, H7, H4, and H2, were also classified into NA and EA lineages. 4,14,22,29 Due to the large variation in the nucleotide sequence of the HA genes between the isolates, no completely conserved sites were available for the primer and probe design that match 100% for both NA and EA isolates for any given subtype. Degenerate oligonucleotide primers were used to increase the specificity of binding of the primers and probe to the target amplicon (HA) of the virus from different geographical origins. Although considerable care was taken to select primers and probes that could detect all viruses within a subtype, a potential for false negatives because of sequence variability is always a possibility. The results (Tables 2–5) show 100% detection of all the isolates tested of the subtypes H6, H9, and H11, but the panel was biased to NA isolates because of their availability. Ongoing review of test sensitivity and specificity before and during an outbreak to identify potential problems remains imperative for any molecular diagnostic test, especially for RNA viruses, which have high mutation rates.
Rapid diagnosis of the avian influenza virus (AIV) of the subtypes H6, H9, and H11 by real-time reverse transcription polymerase chain reaction (RRT-PCR) in cloacal and oropharyngeal (oroph) swabs of experimental chickens inoculated with the virus.
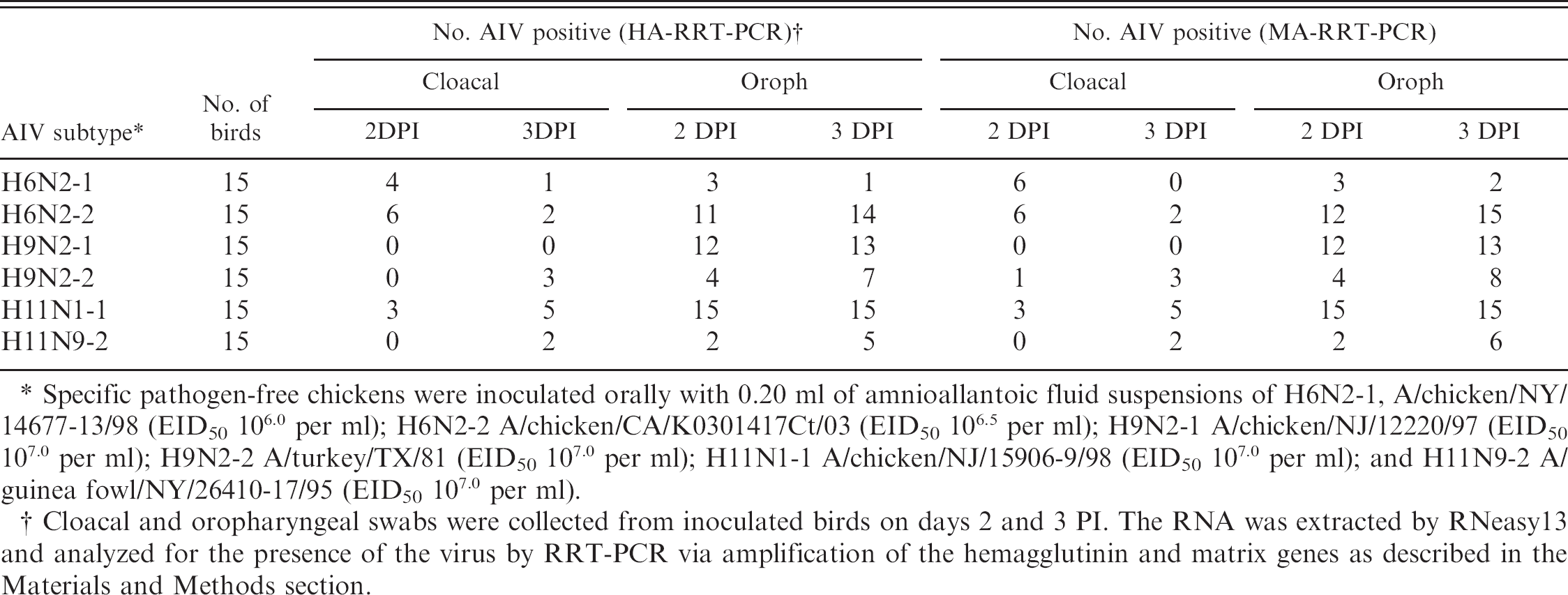
Specific pathogen-free chickens were inoculated orally with 0.20 ml of amnioallantoic fluid suspensions of H6N2–1, A/chicken/NY/14677–13/98 (EID50 106.0 per ml); H6N2–2 A/chicken/CA/K0301417Ct/03 (EID50 106.5 per ml); H9N2–1 A/chicken/NJ/12220/97 (EID50 107.0 per ml); H9N2–2 A/turkey/TX/81 (EID50 107.0 per ml); H11N1–1 A/chicken/NJ/15906–9/98 (EID50 107.0 per ml); and H11N9–2 A/guinea fow1/NY/26410–17/95 (EID50 107.0 per ml).
Cloacal and oropharyngeal swabs were collected from inoculated birds on days 2 and 3 PI. The RNA was extracted by RNeasy13 and analyzed for the presence of the virus by RRT-PCR via amplification of the hemagglutinin and matrix genes as described in the Materials and Methods section.
Hemagglutination inhibition (HI) titers corresponding to viral antigens in blood sera collected from inoculated chickens at different day postinoculation (DPI).∗
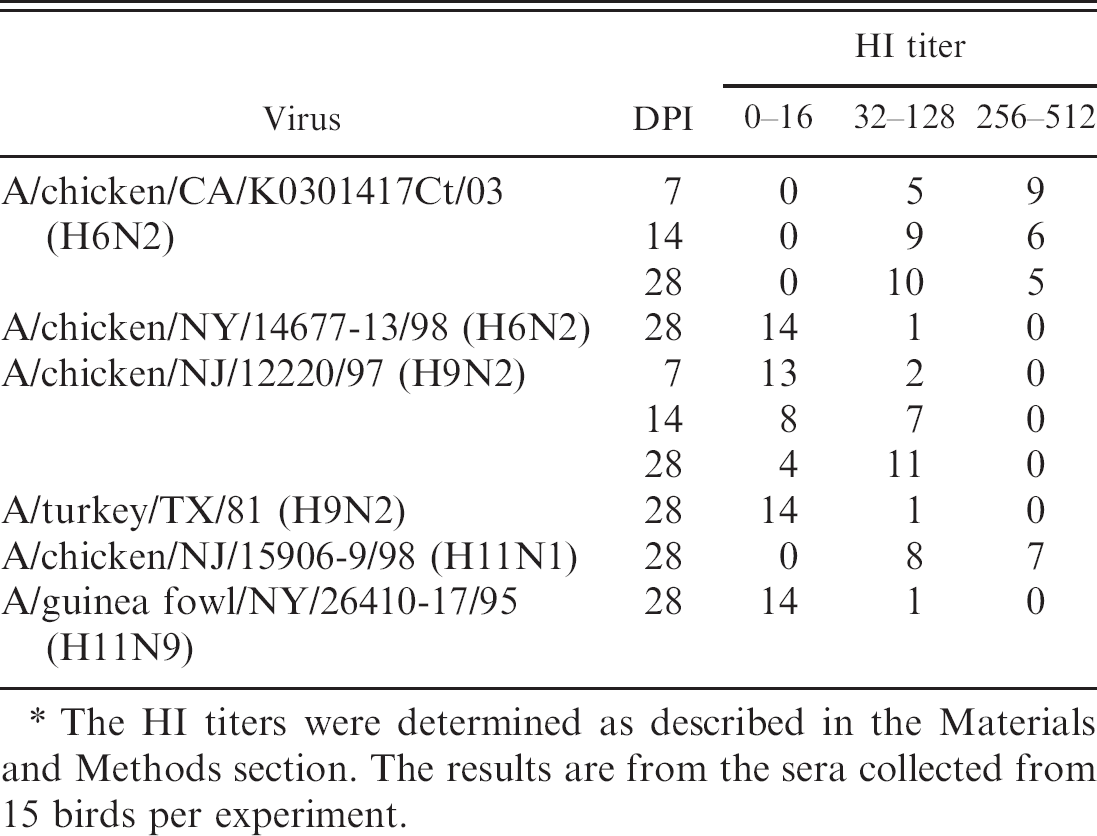
The HI titers were determined as described in the Materials and Methods section. The results are from the sera collected from 15 birds per experiment.
The subtype specificity and sensitivity of the RRT-PCR assays were evaluated by detecting the virus in experimental samples (oropharyngeal and cloacal swabs) from chickens inoculated with the virus. The minimum detection levels of the virus titer (EID50 per ml) of various isolates of the AIV H6, H9, and H11 subtypes were determined to be between 101 and 102.9 by HA-RRT-PCR and between 101 and 101.9 by MARRT-PCR, indicating relatively higher sensitivity of detection of virus by MA-RRT-PCR than HA-RRT-PCR. A similar difference (10-fold) in the detection limit of the virus between MA-RRT-PCR and HA-RRT-PCR was also reported for the AIV H5 and H7 subtypes (40). The sensitivity of detection of the virus from different subtypes was found to be similar by RT-PCR based on the amplification of the MA or HA gene (Fig. 2). Close agreement of test results between MA-RRT-PCR and HA-RRT-PCR for detection of the virus in experimental samples (cloacal and oropharyngeal swabs) (Table 7) indicates the accuracy of the HA-RRT-PCR assays developed in this study. The detection of the virus by HA-RRT-PCR was found to be less sensitive than MA-RRT-PCR for AIV subtypes H6 and H11 but similar for the AIV subtype H9 (Table 6). Due to higher conservation of the nucleotide sequences of the matrix genes (94.8%) compared with the HA genes (83.1%) of the AIVs from different origins, 42 there was limited flexibility for primer and probe design for HA-RRT-PCR than the MA-RRT-PCR. Efforts to improve the sensitivity of the primer and probe design for HAs of the subtypes H6 and H11 were difficult due to this limitation.
In bird experiments, the viruses were detected more in oropharyngeal swabs than in cloacal swabs, which are common for LPAIV infections in chickens, including the subtypes H6 and H9, as reported by others. 3,7,19,46 Poor infectivity of the NA isolates of H9N2 viruses in chickens has also been reported. 19 Poor replication of the viruses in experimental chickens was further supported by low HI titers of the inoculated chickens.
The HA-RRT-PCR assays developed in this study were designed to determine the subtypes of the AIVs, and they are not recommended for primary screening to detect AIV in test samples. Primary screening should still be performed by standard AI matrix tests followed by virus isolation.
Acknowledgement
The authors thank Suzanne DeBlois for technical support and help with the bird experiments.
