Abstract
Use of meat juice and muscle swabs at slaughterhouses may provide an easy-to-collect sample for African swine fever (ASF) surveillance. Meat juice has been experimentally shown to be a reliable sample for the detection of ASF virus (ASFV). We compared the detection of ASFV nucleic acid from diaphragm meat juice, diaphragm muscle swab, spleen, and spleen swabs from pigs with signs of ASFV infection at slaughterhouses around Kampala, Uganda. Pigs with ≥2 clinical or pathology signs at the time of slaughter had a spleen sample, spleen swab, diaphragm muscle sample, and diaphragm muscle swab collected. Meat juice was collected from muscle samples through a freeze–thaw cycle. Each sample was tested individually, and 72 spleen, meat juice, and muscle swab sample pools of 4 negative and 1 positive sample were tested, as well. Standard operating procedures from the USDA–Foreign Animal Disease Diagnostic Laboratory for viral DNA extraction and real-time PCR (rtPCR) were used. Of the 493 pigs evaluated, we classified as positive 357 (72.4%) diaphragm meat juice samples, 218 (44.2%) diaphragm muscle swabs, 247 (50.1%) spleen samples, and 241 (48.9%) spleen swabs. All spleen sample pools were positive (72 of 72; 100%), as were 71 of 72 (98.6%) meat juice pools and 67 of 72 (93.1%) muscle swab pools. Meat juice samples provided a reliable sample type for the detection by rtPCR of ASFV in pigs with natural infections.
African swine fever virus (ASFV; Asfarviridae, Asfivirus) is a large, enveloped DNA virus that is the only member of its family,2,46 although a new virus that affects abalone may be related. 27 Based on sequencing of the p72 segment of the ASFV genome, 24 genotypes are recognized. 35 ASFV causes a hemorrhagic disease that can have very high morbidity and mortality in domestic swine, wild boar, and feral swine; warthogs, bush pigs, and giant forest hogs typically exhibit subclinical infection.29,47 As a result, the World Organisation for Animal Health (WOAH) 48 has classified African swine fever (ASF) as a notifiable disease, meaning that its detection in a country can impact trade. ASFV was first described in 1921 in Kenya, 29 and has been reported in many parts of the world in the last 5 y, including Africa, Asia, Europe, and Hispaniola in the Caribbean (https://wahis.woah.org/#/dashboards/country-or-disease-dashboard, accessed 2022 Aug 08).
Surveillance and laboratory testing are the cornerstones of disease control, and their use should align with the needs for outbreak investigations, disease recognition, and proof of disease freedom. A widely accepted vaccine for ASFV is not available, to date. Biosecurity plays a key role in ASF control programs, given that ASFV is transmitted directly through pig-to-pig contact 20 and can survive in excretions for a long period 11 thus allowing contaminated fomite transmission. In Uganda, community-led biosecurity programs have been shown to be effective. 36 One considerable area of risk for ASFV entry into a country is from pork products, 9 and that risk is only increasing as the virus continues to spread globally. 23 In addition, there is a need for testing of carcasses to understand the distribution of the virus in wildlife when wild swine species become infected. 48 Finally, compartmentalization has created an option to use biosecurity compartments to prove disease freedom and allow for continuity of business when a country is positive for ASFV infection. A compartment must include all downstream entities based on the product that the compartment makes. Therefore, if pork is the final product from a compartment, slaughterhouses must be included. 39 It is likely that slaughterhouses will conduct testing to prove disease freedom to ensure trust in the compartment.
It is well-documented that pooling is a cost-effective way to test more animals with fewer samples to detect viruses, but pooling is impacted by test sensitivity, sample dilution, and herd prevalence.30,31,38 Sample pooling can be a reliable detection method, but, when prevalence is low, pooling reduces detection sensitivity. In a study using a low-virulence porcine reproductive and respiratory syndrome virus (PRRSV; Betaarterivirus suid 1, 2), sensitivity was decreased by 6% and 8% when blood and blood swabs were pooled, respectively. 42 It has been shown that, in low-prevalence endemic settings in which lower viral titers are exhibited in animals, the larger the pool, the lower the sensitivity. 30 Therefore, pooling should be evaluated carefully when considering new sample types for any pathogen.
Ongoing evaluation of sample types is critical for preparing for an outbreak. The WOAH recommends the use of whole blood, serum, spleen, lymph nodes, bone marrow, lung, tonsil, and kidney for detection. 47 The Department of Animal Health of the Ugandan Ministry of Agriculture, Animal Industry, and Fisheries (MAAIF) uses the WOAH guidance for approved samples (Assistant Commissioner, Animal Disease Control, MAAIF, pers. comm., 2022 Oct 17). In the United States, samples currently approved for testing by the U.S. Department of Agriculture–Foreign Animal Disease Diagnostic Laboratory (USDA-FADDL) include spleen, blood, tonsil, and lymph nodes8,44; blood swabs, blood cards, and spleen swabs can be used after ASFV is confirmed in the United States. 33 Although the use of swabs reduces workload, such samples are challenging for swine producers given that they require venipuncture of a large number of animals, or opening a carcass of a potentially infected animal, which could lead to environmental contamination. Additional sample types are being evaluated in various laboratories, with work highlighting the potential usefulness of oral fluids, 18 inguinal lymph nodes, 19 and ear notch samples. 12 Surveillance and monitoring systems will benefit from having more sample options available and characterized. Sample types that are simple to collect in terms of training and workload will allow the collection of better-quality samples, which will enhance surveillance systems. Having a wide variety of well-characterized sample types will allow for greater participation regardless of the site of collection, which could be the field, barn, market, or slaughterhouse.
Meat juice and muscle swabs have not been assessed widely for detection of ASFV. One study used a novel PCR probe, which when used on meat juice was as reliable as spleen samples in detection. 28 In another study, meat juice from the diaphragm and other muscles was used for the reliable detection of moderately and highly virulent ASFV and antibodies against ASFV in pigs infected under experimental conditions. 37 Meat juice was also shown to be a reliable sample for the detection of classical swine fever virus (CSFV; Pestivirus C),24,26 a differential for ASFV. 47 Outbreak testing can require a great number of resources. Sample pooling is a resource management strategy to reduce testing and costs 30 that could be used during an epidemic. Overall, the scientific literature has limited information on meat juice and muscle swabs as individual or pooled samples, which would be simple for those with limited training to collect postmortem from feral swine and wild boar carcasses, from meat products at import points, on farms, and in slaughterhouses.
We compared ASFV detection in diaphragmatic meat juice and muscle swab samples to spleen and spleen swab samples. The diaphragm was a low-value muscle chosen as a source of meat juice. Spleen and spleen swab samples are approved sample types in the United States,33,44,45 and spleen is a sample recommended by the WOAH. 47 All 4 sample types are readily available postmortem, and thus useful for sampling on-farm carcasses, recently deceased wildlife, and carcasses in slaughterhouses. In particular, our focus was on pigs that would be naturally infected. We also evaluated the impact of sample pooling.
Materials and methods
Live animals were only observed and were not handled physically as part of our project. Animals were assessed and sampled immediately after being slaughtered. The Pipestone Institutional Animal Care and Use Committee reviewed and approved our study (protocol 2021-4).
We used 501 pigs that were presented at slaughterhouses around Kampala, Uganda with clinical signs consistent with ASFV infection with the aim of detecting acute ASFV infections in ≥139 pigs, with ≥90% diagnostic sensitivity, 95% confidence, and 5% error for each sample type (https://epitools.ausvet.com.au/oneproportion). The spleen sample was the gold standard sample type used to identify infected animals given that it is a sample type approved by the WOAH 47 and the USDA swine fever surveillance program. 45 Detection in spleen also has been shown to have very high sensitivity that is comparable to studies using blood. 40 Blood is a known PCR inhibitor,41,10 and may require different procedures for accurate detection. 10 Uganda previously reported that ASFV genotypes IX3,15,16,32 and X 15 circulated in the country, and that acute disease with high mortality16,32 and subclinical 41 disease was present. With targeted surveillance of sick animals, we expected to capture animals experiencing acute disease.
Trained veterinarians visually assessed animals antemortem and recently slaughtered carcasses for evidence of diarrhea, skin discoloration or ecchymoses, enlarged and/or hemorrhagic lymph nodes, enlarged and/or hemorrhagic spleens, as well as petechiae on organs (kidney, intestines). Pigs with ≥2 of these signs were included in the study. The sampling date, location, lesions, and clinical signs were recorded. A 4 × 4-cm piece of the diaphragm muscle was taken from the thickest part of the muscle body and placed in a tissue collection bag (Lasec). A muscle swab was then taken of the entire diaphragm and its cut surface by vigorously rubbing the surfaces with a polyester swab on a plastic shaft (Wuxi NEST Biotechnology). The swab was placed in 2–3 mL of sample storage medium (Wuxi NEST Biotechnology) in a sample transport tube (Wuxi NEST Biotechnology), swirled, the liquid pressed out of the swab, and the swab discarded. A 3 × 3-cm piece of spleen was collected as well and placed in a tissue collection bag (Lasec); the spleen swab was collected in the same manner as the muscle swab using the same materials. Samples were transported in a cooler with ice packs to the Makerere University Central Diagnostic Laboratory (Kampala, Uganda) at the end of each sampling day and stored for 2–9 mo in a −20°C freezer until they were processed and extracted.
In preparation for extraction, diaphragmatic muscle samples were thawed, and meat juice collected. If <0.5 mL was collected after the first freeze–thaw, the sample went through a second freeze–thaw cycle. If ≥0.5 mL of meat juice was still not available, 0.5–2 mL of PBS (Fisher) was added, and the freeze–thaw cycle repeated a final time. The amount of PBS added was at the discretion of the laboratory technician and dependent upon the amount of muscle, tendon, and fat visible on the sample; samples with a smaller amount of muscle tissue and more tendon and fat had more PBS added. The spleen tissue was also processed further before extraction;1 g of the spleen sample was homogenized (Stomacher 80 Biomaster; Seward), combined with 9 mL of Dulbecco modified Eagle medium (Life Technologies), and then centrifuged at 1,000 × g for 10 min; the supernatant was collected and used for viral DNA extraction.
Extractions were performed on each sample from each pig (DNeasy tissue and blood kit; Qiagen). The USDA-FADDL SOPs were used for these extractions, 13 which followed the manufacturer’s instructions. Real-time PCR (rtPCR) assay was used as described previously, 50 and the USDA-FADDL SOP 14 was again used. The master mix (TaqMan fast virus 1-step master mix), forward primer (5′-CCTCggCgAgCgCTTTATCAC-3′), reverse primer (5′-ggAAACTCATTCACCAAATCCTT-3′), and probe (6FAM-CgATgCAAgCTTTAT-MGB/NFQ), all from Thermo Fisher, were used in the rtPCR procedure. The rtPCR assay was run on a QuantStudio 5 thermocycler (Thermo Fisher). The internal positive control and assay (VetMax Xeno DNA IPC, VetMax Xeno IPC LIZ assay; Thermo Fisher) were used for each sample following USDA-FADDL SOPs, and the internal PCR control (IPC) was also used in the extraction control for pooled samples. Limited supplies prevented use of the VetMax Xeno assay for each of the pooled samples.
We created 72 pooled samples for each of spleen, muscle swab, and meat juice samples using 4 known-negative samples (IPC positive and ASFV target undetermined) and 1 known-positive sample (IPC positive and ASFV target positive). This allowed detection with >95% diagnostic sensitivity, 95% confidence, and 5% power (https://epitools.ausvet.com.au/oneproportion). ASFV-positive targets were those with a Ct >10 and <40. The samples with the lowest Ct values for each sample type were used as the positive sample in the pool during the sample pooling process. In this way, the ideal positive samples were used for each sample type. The extraction and rtPCR assay were performed in the same manner as for the individual samples.
Clinical and pathology sign (CPS) scores were created. Each of the 5 CPSs assessed was given 1 point, and scores were tallied for each pig. Percentage of positive samples detected was determined, and 95% CIs were calculated using the Wilson or Agresti–Coull method based on sample sizes. 6 Multiple pairwise comparisons were done with a McNemar chi-squared test for matched data with a Bonferroni adjustment (α = 0.0083); a Cochran Q test was used for comparing proportions for different sample types from the same pigs. Agreement was assessed using 2 kappa statistics: Cohen kappa and the Brennan–Prediger inter-rater agreement. The latter adjusted the data for bias and prevalence.7,21 Agreement was described. 25 The normality of Ct values was evaluated using the Shapiro–Wilk test, and median and range were used to summarize. The Kruskal–Wallis test was used to compare Ct values between sample types. Scatter plots and lines of best fit were used to visualize relationships between Ct values from different sample types using scatter plots, and correlation coefficients were calculated. Excel v.16.63.1 (Microsoft) was used for data entry and compilation; Stata v.16.1 IC (StataCorp) was used for descriptive and analytical statistics.
Results
There were 8 pigs (1 [0.2%] with a CPS score = 0; 7 [1.4%] with a CPS score = 1) that did not have CPS scores ≥2 and were dropped from further analysis (Table 1). The remainder of the 493 pigs had CPS scores of 2 (153; 31.0%), 3 (196; 39.8%), and 4 (138; 28.0%). Only 6 pigs (1.2%) had the highest possible score of 5. Of the 493 pigs with adequate CPS scores, only 7 (1.4%) had signs of diarrhea. The most common CPSs were cutaneous ecchymoses (464; 94.1%) and enlarged and/or hemorrhagic spleens (467; 94.7%). Of the remaining CPSs, 349 (70.8%) pigs had enlarged and/or hemorrhagic lymph nodes, and 189 (38.3%) had petechiae on internal organs. Each pig had meat juice, muscle swab, spleen, and spleen swab samples collected. Of the meat juice samples, 114 (23.1%) required a second freeze–thaw, and 73 (14.8%) required additional PBS and a third freeze–thaw; 32 samples had 0.5 mL added, 38 with 1 mL, 1 with 1.5 mL, and 2 with 2 mL.
Summary statistics of clinical and pathology signs and sample types from pigs sampled in Kampala, Uganda slaughterhouses for African swine fever virus real-time PCR testing, 2021–2022.

Meat juice had the highest positivity (357 of 493; 72.4%) with a 95% CI (68.3%, 76.2%) that did not overlap with the CIs for the positivity of the other sample types (Table 2). The statistical significance of meat juice compared to the other sample types was confirmed in the pairwise comparisons. None of the other samples differed from one another statistically; spleen had the next highest positivity (247 of 493; 50.1%), then spleen swab (241 of 493; 48.9%), and finally muscle swab (218 or 493; 44.2%). The overall agreement between the samples was 66.6%; spleen and spleen swab had the highest agreement overall (70.4%) and meat juice and spleen had the lowest agreement (61.1%). The Cohen and Brennan–Prediger kappa statistics were almost identical when comparing sample types. Most samples had fair agreement (range: 0.2–0.4), and spleen and spleen swab had moderate agreement (0.41; range: 0.4–0.6). 25 When comparing positivity rates for sample types against the CPS score, the same pattern emerged. Meat juice samples did not have overlapping of percentage positivity 95% CIs with any other sample type for CPS scores of 2–4. The percentage positivity 95% CIs of all of the other sample types did overlap for the same CPS scores. The percentage positivity 95% CIs for all sample types overlapped for CPS scores of 5; however, there were only 6 samples.
Percent positivity and agreement comparisons between sample types from pigs with clinical and pathology signs of African swine fever virus infection, Kampala, Uganda, 2021–2022.
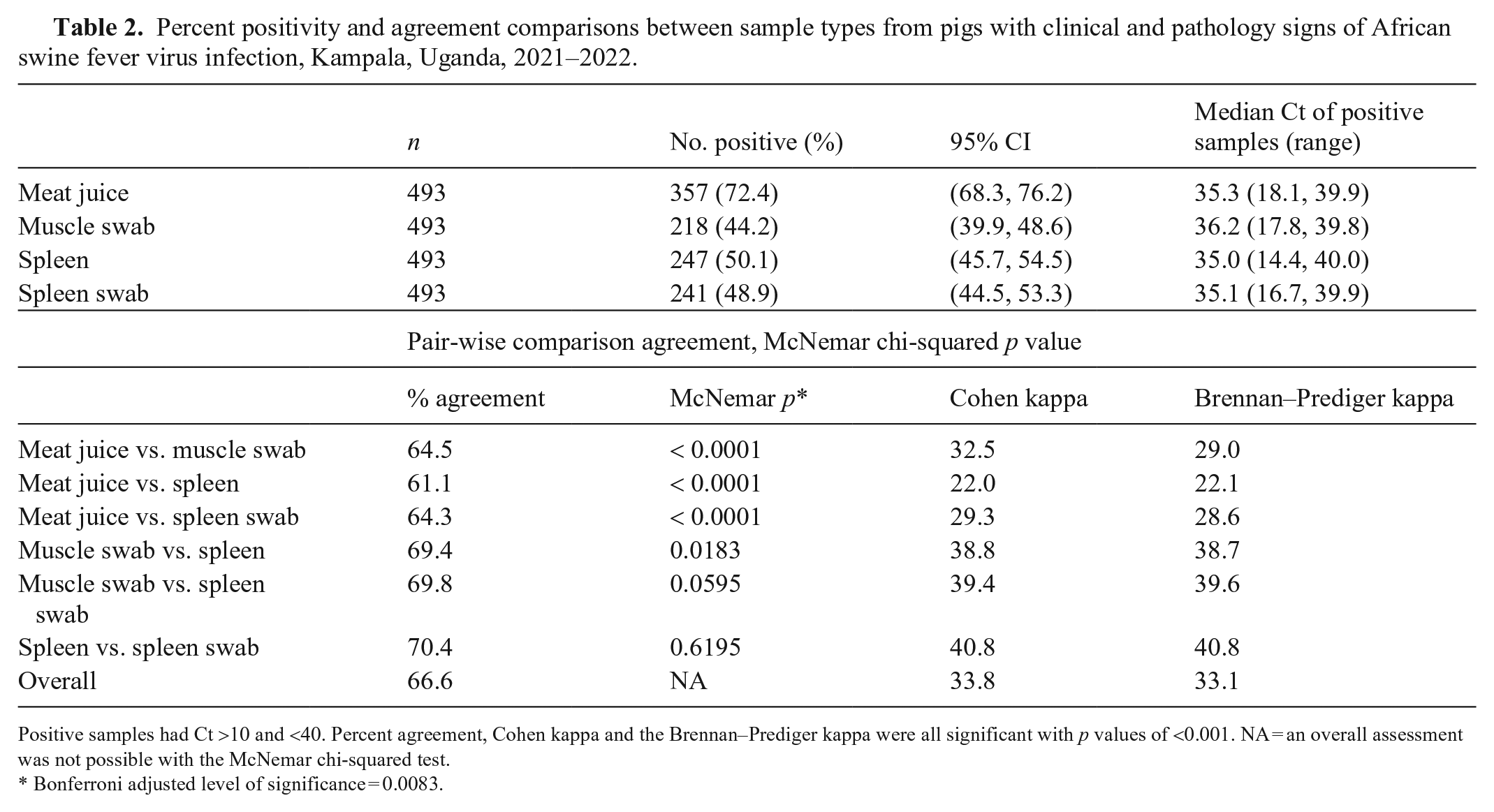
Positive samples had Ct >10 and <40. Percent agreement, Cohen kappa and the Brennan–Prediger kappa were all significant with p values of <0.001. NA = an overall assessment was not possible with the McNemar chi-squared test.
Bonferroni adjusted level of significance = 0.0083.
Ct values were compared between different sample types from the same pig where both samples were positive (Figs. 1–6). Spleen samples had the lowest Ct values, which correlated most highly with meat juice sample Ct values (r = 0.904), followed by spleen swab (r = 0.873), and finally muscle swab samples (r = 0.777). The latter had the poorest correlation across all comparisons, with the muscle swab having Ct values of 25–30, whereas the spleen swab had Ct values of 15–20. Meat juice and muscle swabs also had a high correlation of 0.822. The meat juice samples had lower Ct values (20–25) compared to the muscle swabs (Ct 25–30).
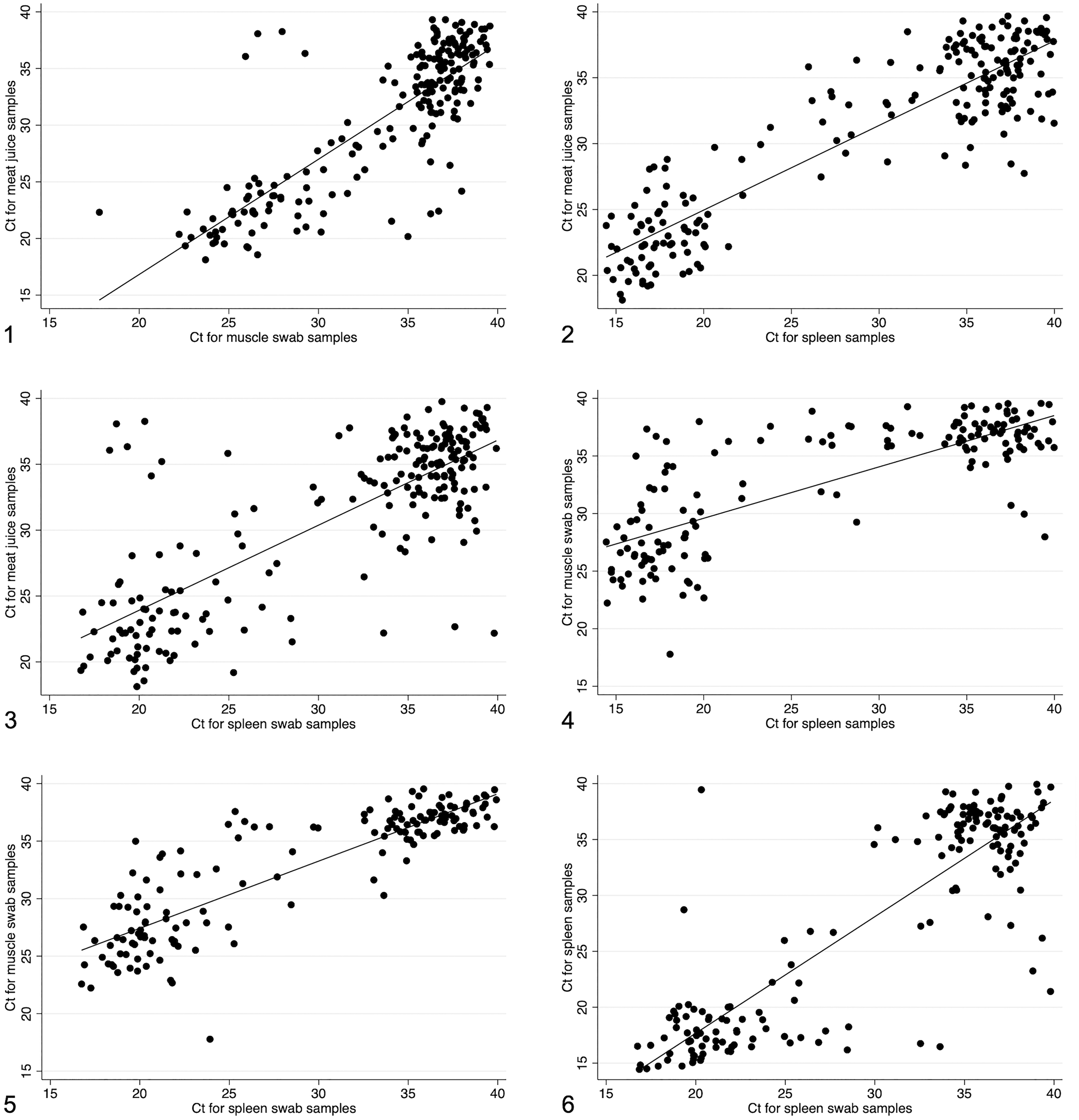
Scatterplots of Ct values from samples tested using a real-time PCR assay for the detection of African swine fever virus (ASFV). Pigs were sampled at slaughterhouses around Kampala, Uganda and had clinical and pathology signs of ASF. r = correlation coefficient. Lines = lines of best fit.
Spleen sample pools had the highest positivity rate (72 of 72; 100%), followed by meat juice (71 of 72; 98.6%), and finally muscle swabs (67 of 72, 93.1%; Table 3). The Ct values of the individual positive samples used to make the pools were statistically different, with spleen samples having the lowest median Ct of 17.9, followed by meat juice (23.2) and muscle swabs (26.7). The meat juice pool that was negative used a positive sample that had a Ct of 25.9 when tested individually. For muscle swabs, the 5 pools that were negative by rtPCR testing used positive samples with a median Ct of 28.9 (range: 25.9, 33.3) to make the pools. A similar trend was found with the pooled sample Ct values; spleen samples had the lowest pooled sample Ct value (21.9), and muscle swabs had the highest (31.0).
Summary of percent positivity and Ct values for pooled meat juice, muscle swab, and spleen samples collected from slaughterhouse pigs with clinical and pathology signs of African swine fever, Kampala, Uganda, 2021–2022.
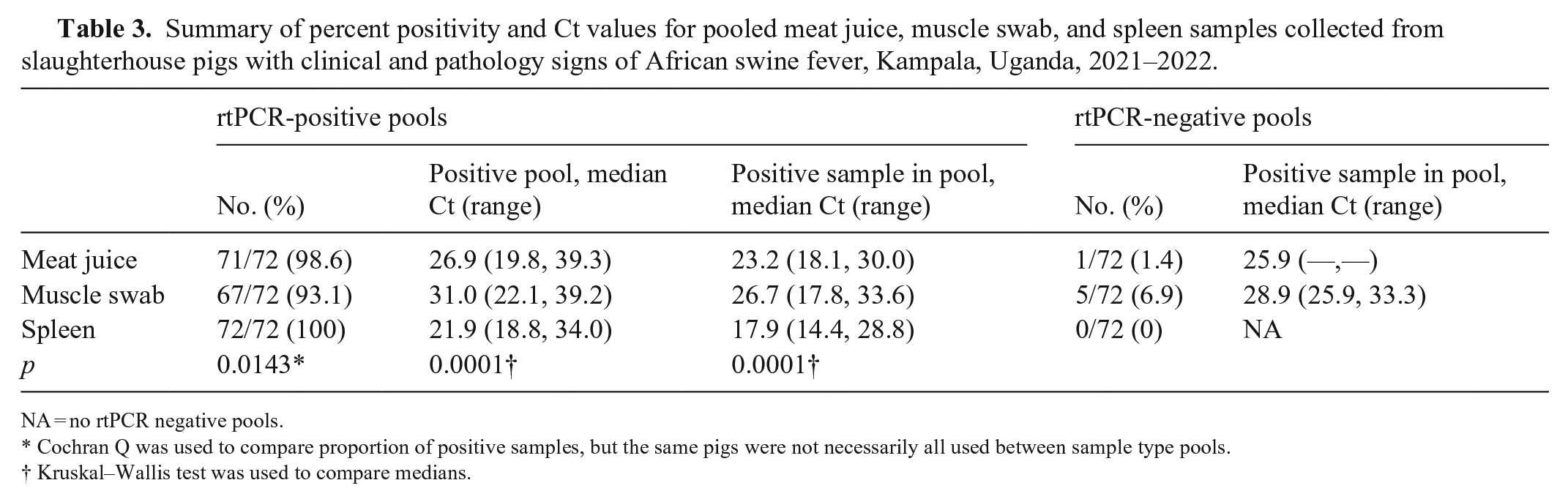
NA = no rtPCR negative pools.
Cochran Q was used to compare proportion of positive samples, but the same pigs were not necessarily all used between sample type pools.
Kruskal–Wallis test was used to compare medians.
Discussion
We found splenic hemorrhage and enlargement as well as skin discoloration to be the most common clinical signs of ASFV infection in the pigs sampled during our study. Enlarged and hemorrhagic spleens are nonspecific signs of ASF that can occur in any severe viral infection in swine.43,47 Cutaneous ecchymotic hemorrhage is also a clinical sign of ASF, but is also seen in erysipelas, salmonellosis, porcine dermatitis and nephropathy syndrome, and classical swine fever.43,47 The prevalence of these differential diagnoses is not well studied in Uganda, therefore laboratory testing is necessary to rule out ASFV infection.
Meat juice resulted in the highest positivity rate among all sample types, and 95% CIs of the positivity rates of meat juice did not overlap with spleen, spleen swab, or muscle swab. It is unclear why meat juice had the greatest positivity rate, but there are 2 major considerations. The first is that meat juice may be a more reliable sample type, and the second is that meat juice may have a higher number of false-positives.
A study found that use of meat juice reliably detected moderately virulent and highly virulent forms of ASFV as reliably as in whole blood of experimentally infected animals, although blood had a lower Ct value. 37 Work has also shown that CSFV can be detected using meat juice in experimentally infected animals, but at lower sensitivity than serum.24,26 Foot-and-mouth disease virus serotypes A and SAT2 have been detected as reliably in meat juice as in serum and for a longer duration. 49 All previous reports on viral detection have originated from experimental settings. Meat juice may be a more reliable sample for ASFV genotypes IX1,4,32 and X, 34 known to be circulating in Uganda.4,32 This would need further study and could be assessed through a thorough validation study.
It may be that there were more false-positive results with the meat juice samples. Cross-contamination of samples is always a possibility, but it is not likely in our study. If contamination occurred at the slaughterhouse, we would have expected all samples to be contaminated equally. Fresh gloves and instruments were used for each sample collected, and individuals were trained to collect samples properly. Each collector had an assistant to ensure proper collection and reduced contamination, as well. All testing was done at the same time in the laboratory; contamination issues would have been similar between all sample types. Furthermore, negative extraction and template controls were analyzed alongside the samples, and there was no indication of contamination on any of the plates run; the Ct values for meat juice were lower than spleen, further suggesting that the issue was not contamination. The biological plausibility of false-positive results is doubtful. Hemoglobin and myoglobin are known inhibitors of PCR given their iron content, which inhibits DNA synthesis.5,41 Use of such samples may require use of a different polymerase to address inhibition.5,41 If inhibition was the issue, then false-negatives, not false-positives, would be expected. Further work using a large-scale negative cohort and a full analytic and diagnostic validation will be needed to confirm the reliability of meat juice as a sample type against blood as well as against other tissue sample types. Nonetheless, our study suggests that meat juice may be a reliable sample type for slaughterhouses, pork products, and potentially for mortality on farms.
The results from the pooled samples in our study suggested that the pool is highly impacted by the viral load of the positive sample. Given that spleen samples had the highest viral load, they were the most reliable. Because internal controls could not be used in each pooled sample to confirm extraction in our study, it will be important to ensure that extractions are successful in future work. Further, it will be important to evaluate ASFV variants of different virulence levels to understand what differences in sensitivity may occur; attenuated viruses have exhibited different patterns of detection in different sample types.17,22,37
A challenge with meat juice samples is their utility for high-throughput laboratories. Samples must go through a freeze–thaw cycle before collecting meat juice. If meat juice is readily collected after the first freeze–thaw, no further processing of the sample is needed. In our study, some samples required a second freeze–thaw and then the addition of PBS, which may have been the result of animal dehydration or collection of tendons rather than muscle. This issue may be corrected by animal welfare requirements in different countries, but if an ongoing challenge, these extra steps delay testing and add to the workflow. Nonetheless, meat juice samples are noninvasive, simple to collect, and can be collected by unskilled workers at the farm or slaughterhouse, and thus may fill a needed gap for on-farm, wildlife, and slaughterhouse sampling.
Footnotes
Acknowledgements
We thank the U.S. Department of Agriculture–Foreign Animal Disease Diagnostic Laboratory members who provided training on rtPCR techniques and extraction, as well as quality lab management practices, including Steven Pauszek, Kate Schumann, Heather Petrowski, Diane Holder, and Hannah White.
Declaration of conflicting interests
The authors declare no potential conflicts of interest with respect to research, authorship, and/or publication of this article.
Funding
Funding was provided by the National Pork Board (NPB 20-150) for sample collection and laboratory work. Our project benefited from the training and equipment provided by an ongoing project sponsored by the Department of the Defense, Defense Threat Reduction Agency (grant HDTRA1-20-1-0007). The content of the information provided here does not necessarily reflect the position or the policy of the U.S. federal government, and no official endorsement should be inferred.
