Abstract
Among 172 hearts from clinically normal adult sheep aged 1.5 to 7 years evaluated for the presence of cartilage and/or bone in the right atrial myocardium, 3.49% (6/172) had palpable evidence of osteocartilaginous foci. An additional 8% prevalence was estimated based on radiographs of hearts that contained ≤1 mm foci of nonpalpable, radiographically dense bone. Microscopically, the nodules in the atria were characterized by mature lamellar bone enclosing adipose tissue, with occasional new bone formation by endochondral ossification. No degenerative changes were evident in the affected atrial myocardium, suggesting that these lesions were clinically insignificant background changes.
Introduction
Sheep are increasingly being used as animal models for preclinical safety evaluation of bioprosthetic heart valves and devices necessitating thorough documentation of the background changes in this species. In most mammalian species, the atrial and ventricular musculature is inserted into the cardiac skeleton composed of fibrous rings (annuli fibrosi) and the fibrous trigone (trigona fibrosa). Coarse interwoven collagen fibers and some elastic fibers surrounding the atrioventricular, aortic, and pulmonary arterial openings constitute the fibrous rings. The fibrous trigone consists of areas of dense connective tissue between the aorta and atrioventricular openings, which in some mammalian species may contain fibrocartilage, hyaline cartilage, or bone (os cordis). The os cordis is a regular constituent of the ruminant heart, 13,16 the right and left counterparts of which are related to the trigone between the atrioventricular ostia and the aortic ostium, respectively. In one study, the presence of os cordis has been described at the base of the atrial septum and the anterior portion of the left atrioventricular ring in all 25 sheep hearts examined. 8 Occurrence of cardiac bone or cartilage has also been described in the Syrian hamster, 5 otter, 6 dog, 14 and several other species of animals. 17 However, there are few reports in the literature about the prevalence of bone or cartilage in the atrial myocardium of sheep, not associated with the os cordis. 1,18 A study was conducted on the prevalence and structural features of metaplastic bone and/or cartilage within the right atrial myocardium of clinically healthy adult sheep, an important animal model for cardiovascular research and preclinical safety evaluation of heart valve prostheses and devices developed for use in humans.
Materials and methods
Hearts were harvested at necropsy from 172 (4 males and 168 females) clinically healthy, commercially raised, adult sheep (aged 1.5–7 years) from a variety of breeds and diverse backgrounds, most of which were submitted to the Animal Disease Diagnostic Laboratory at Purdue University as part of the national surveillance program for scrapie in sheep and goats. Sampled population included Suffolk (40), Black face (11), Dorset (2), Shropshire (1), and crossbred sheep (118). Hearts were weighed, examined in the routine manner (gross examination), and preserved in 10% neutral buffered formalin. The right atria were examined for the presence of bone and/or cartilage by palpation. Atria in which bone/cartilage was identified by palpation were separated and radiographs made before 5-mm-thick sections were made perpendicular to the atrioventricular plane for macroscopic evaluation. In addition, right atria removed from 50 randomly chosen hearts in which bony tissue was not palpable in the atrial myocardium were examined for the presence of bone/cartilage within the myocardium by radiographs. A beryllium window x-ray machine a was used to make radiographs using (Kodak Portal Pack) nonscreen film. Exposure factors were 150 milliamperage seconds and 20 kilovoltage potential.
Atrial specimens in which bone was palpable were decalcified in 10% buffered aqueous solution of formic acid, dehydrated, and routinely embedded in paraffin. Sections (5 μm) were cut and stained with the following procedures: hematoxylin and eosin, Van Geison stain (for connective tissue), and Alcian blue/PAS (pH 4.0; to demonstrate acidic glycosaminoglycans).
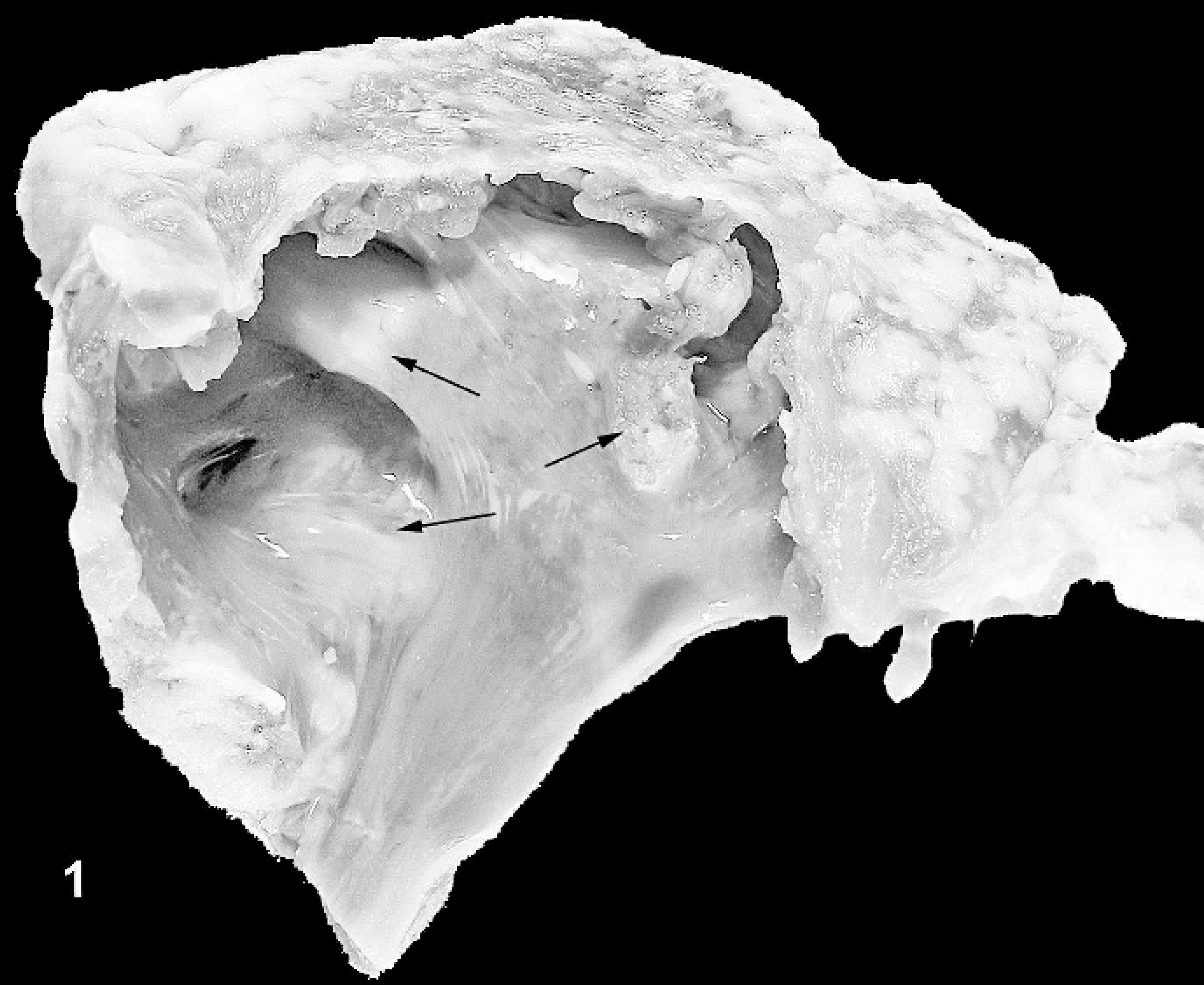
Gross photograph of the endocardial surface of right atrium, with palpable foci of ossification. Ossified areas appear as white, slightly raised firm nodules as indicated by the arrows.
Results
Gross anatomy and radiographic evaluation
The right atrial wall of 6 hearts (among 172 examined by palpation) contained round to irregularly shaped, occasionally confluent firm nodular foci measuring 0.5 cm to 2.0 cm in maximal dimension. On the endocardial surface, these nodules projected minimally into the lumen and were white and gritty on the cut surface (Fig. 1).
Among the 56 atria (including 6 that contained palpable bone and an additional 50 in which ossification was not palpable) subjected to radiological evaluation, 10 contained foci of ossification/mineralization. The ossified areas that were palpated in 6 hearts were confirmed by radiographs, and 4 additional atria were detected among 50 atria that did not have palpable evidence of metaplastic change. In the 4 atria in which foci of ossification/mineralization were not palpable, the foci measured ≤1 mm in greatest dimension. In the 6 atria that contained palpable bone or cartilage, a linear articulated pattern of ossification was observed on radiographs (Figs. 2, 3). A reticulated (spongiform) ossification pattern was also observed within linear articulated foci (Fig. 4).
Based on the gross observations, the prevalence of atrial osteocartilaginous metaplasia was estimated to be 3.49% (6/172). In the 50 samples in which bone or cartilage was not grossly evident in the atrial myocardium but identifiable in radiographs, the prevalence was 8% (4/50). Assuming an estimated 8% prevalence of nonpalpable centers of ossification in the rest of the atria that were not radiographed, an additional 9.3 hearts could be expected to have tiny foci of ossification. Therefore, an overall prevalence of grossly palpable and/or radiographically identifiable foci of atrial ossification is estimated to be 11.2%. The breed distribution of sheep hearts in which bone was grossly evident were as follows; 2 Blackface, 1 Suffolk, and 3 crossbred sheep (all females aged between 2 and 6 years). Hearts that were subjected to radiographic evaluation were collected from 1.5- to 7-year-old Suffolk (31), crossbred (17), and Dorset (2) ewes.
Microscopy
The atrial free wall contained anastomosing trabeculae of mature lamellar bone with roughly parallel walls, measuring 1 to 2 mm or less in thickness (Fig. 5). These interconnected trabeculae were frequently bordered by cartilage or alcianophilic connective tissue that blended into the bony trabeculae. Less frequently attenuated osteoblasts and/or collagenous connective tissue fringed the bony trabeculae. Trabecular bone incorporated numerous osteocyte-bearing lacunae and often enclosed spaces of variable dimension that contained mature adipocytes. In some sections, new bone formation by endochondral ossification was evident (Fig. 6). Cardiomyocytes adjacent to the metaplastic tissue were sometimes minimally compressed.
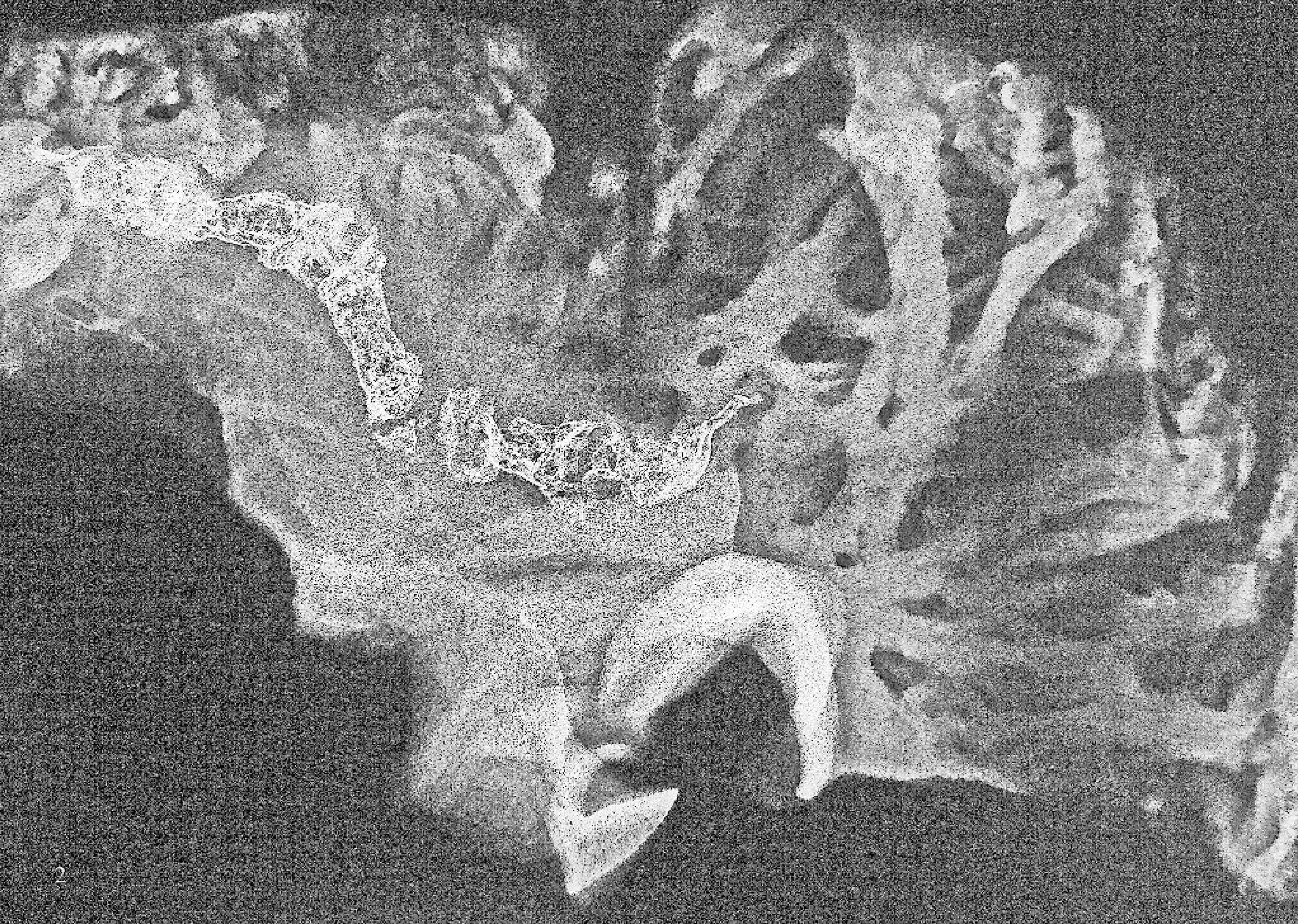
Radiograph of the right atrial myocardium containing extensive foci of bone with linear articulated pattern.
In the atrial myocardium of 1 sheep, a few neutrophils, occasional macrophages (sometimes containing phagocytosed erythrocytes), and extravasated erythrocytes were present, unassociated with the metaplastic tissue. Per-acute interstitial hemorrhages were present in some sections. This was probably due to the immediate postmortem harvesting of heart and handling of tissues.
Discussion
Many investigators consider sheep to be the most appropriate animal model for heart valve research. Accordingly, delineating the background changes in the cardiovascular system, especially changes hitherto incompletely characterized, behooves prompt consideration. The bone and cartilage in the right atrial myocardium of sheep included in this study were not associated with the os cordis. Bone incorporated into the atrial myocardium was mature and not associated with inflammation or degeneration of surrounding myocardium. Therefore, these changes were interpreted as clinically insignificant background lesions in healthy adult sheep. This interpretation is supported by Anderson, 1 who also reported osteocartilaginous metaplasia in the right atrium of 9 of 53 clinically healthy adult male Suffolk sheep. However, sheep from a variety of breeds and both sexes originating from diverse backgrounds were included in the current study. Furthermore, radiographs were used to better characterize the morphology of the metaplastic tissue and also to demonstrate the prevalence of osteocartilaginous foci when it could not be identified on macroscopic evaluation.
In Syrian hamsters, cartilage has been identified within the cardiac skeleton but not the atrial free wall. This metaplastic change occurred in areas where mechanical stress was particularly intense. 19 Localized strong mechanical stimulation even within physiological limits has been proposed to be a key factor leading to cartilage formation in the vertebrate heart, 11,17,19 with the localization being limited to areas of excessive mechanical stress such as the trigona fibrosa. Bhagawan et al. 3 described the presence of cartilage in the septa at the origin of the aorta in 4% of sheep hearts examined. Hyaline cartilage and its ossification were considered a metaplastic change by these authors, who also proposed that this alteration most likely resulted from added myocardial stress due to underlying pathologic conditions such as Johne disease (15 of 40 sheep that had osteocartilaginous metaplasia in the heart were concurrently diagnosed with Johne disease). On the contrary, the sheep included in the current study were clinically healthy and did not have overt evidence of any underlying disease processes.
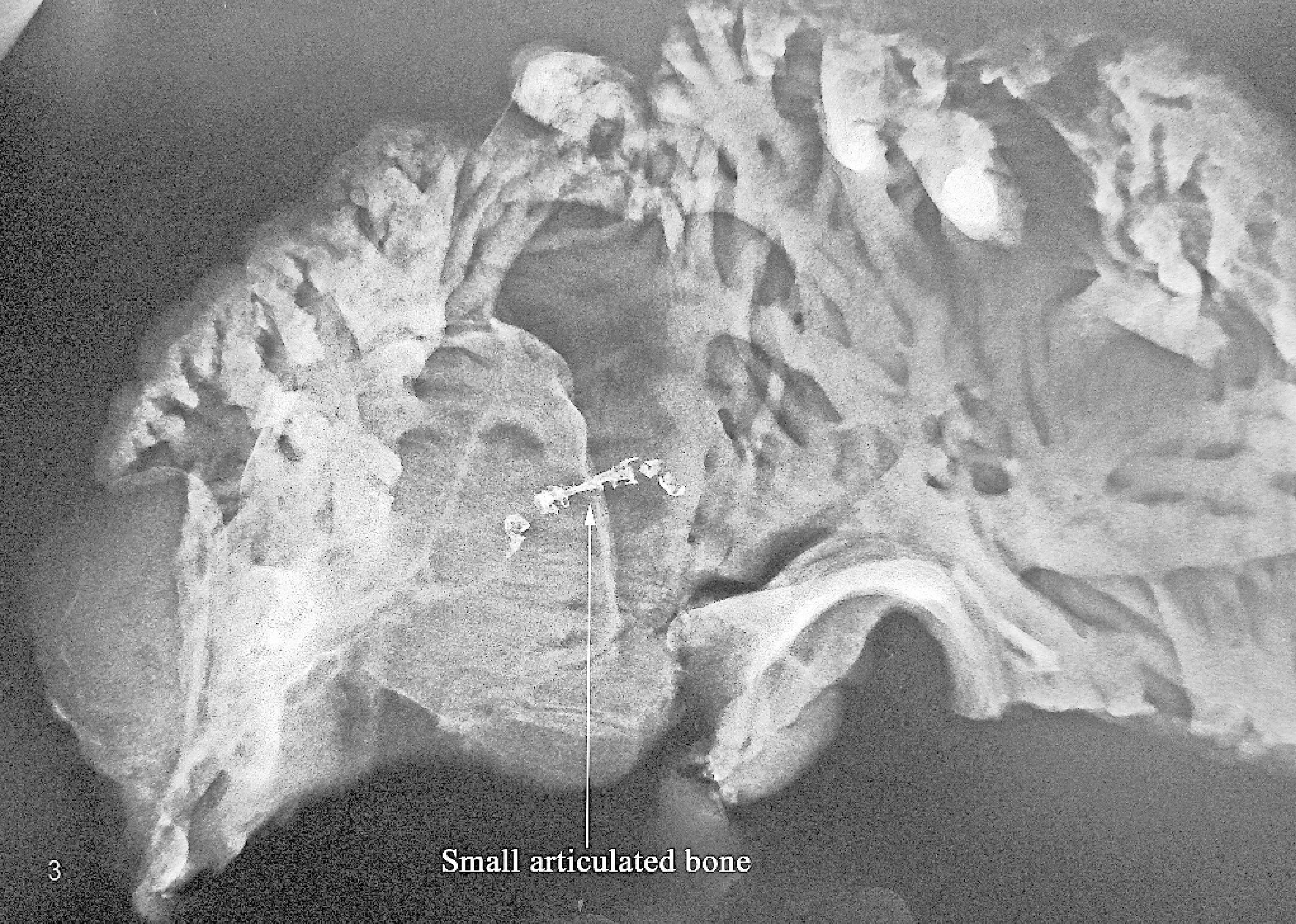
Small linear focus of ossification in the right atrial myocardium of a sheep (arrow; white area toward the top right of the picture is an artifact).
Osteocartilaginous metaplasia of experimentally induced infarcts that correlated well with the extend of infarction within the papillary muscles and/or subendocardial myocardium has been described in spontaneously hypertensive rats and Wistar-Kyoto rats. 2 Nevertheless, in the present study, no evidence of myocardial ischemia/infarction was apparent, and the atrial myocardium adjacent to the metaplastic tissue was microscopically unremarkable.
The neural crest refers to a cellular population contained within the leading edge of neural folds that after neural tube closure migrates to various tissues in the developing vertebrate embryo, generating a diverse range of differentiated cell phenotypes. 7,9,10,15 The neural crest cells play a critical role in the maturation of avian and mammalian heart by contributing smooth muscle cells for the development of aortic arch arteries and participating in the formation of the outflow tracts. 4,12 The neural crest cells provide substantial cellular contribution to the zebrafish myocardium. 20 Cartilage occurring in the cardiac semilunar valves of birds is thought to originate from neural crest cells. 21 The cardiac areas reached by normal or aberrant migration of neural crest cells are still uncertain and could potentially include the atrial myocardium. It is tempting to hypothesize that a population of aberrantly migrating neural crest cells that becomes lodged in the atrial myocardium differentiates into hyaline cartilage and subsequently to bone, at least in part due to the mechanical stimulation resulting from continuous movement of the heart during life. The consistent localization of the lesions also supports the possible presence of dormant undifferentiated tissue in the atrial wall, with subsequent differentiation and proliferation.
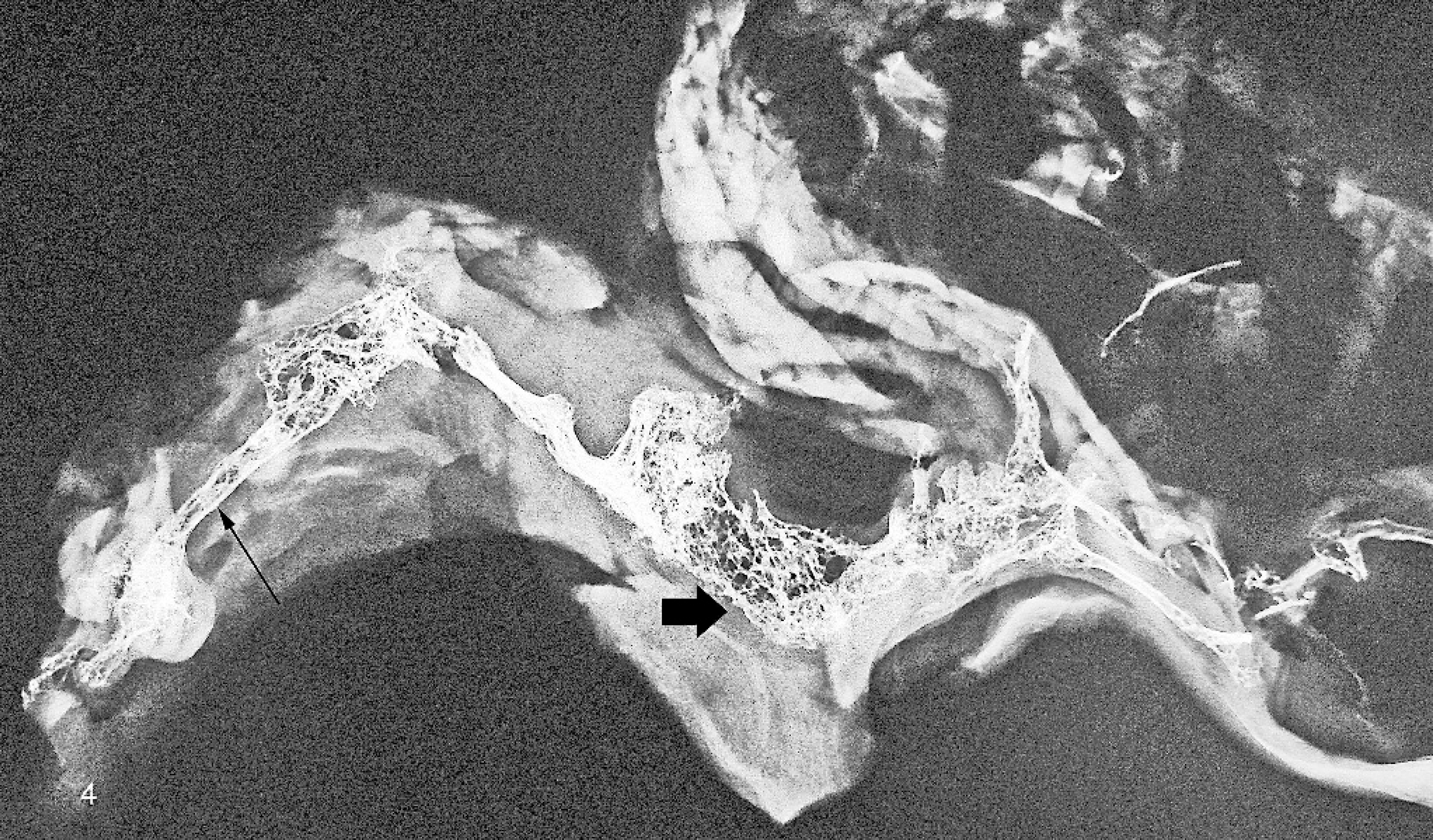
Linear reticulated (spongiform) pattern of ossification (short arrow) admixed with foci of tubular bone formation toward the left-hand side of the radiograph (long arrow) in the right atrial myocardium of a sheep.
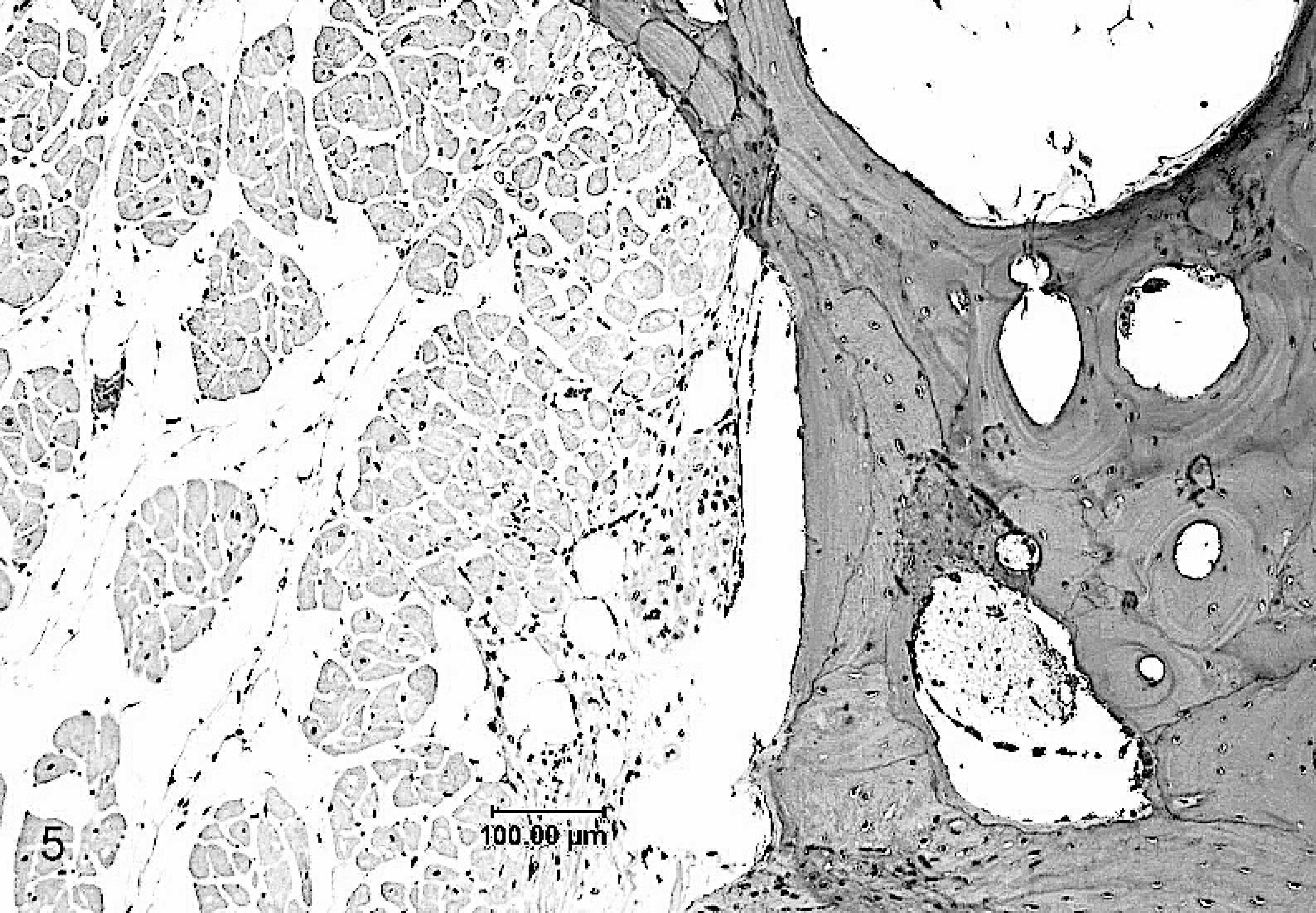
Mature lamellar bone forming interconnected trabeculae adjacent to microscopically unremarkable atrial myocardium.
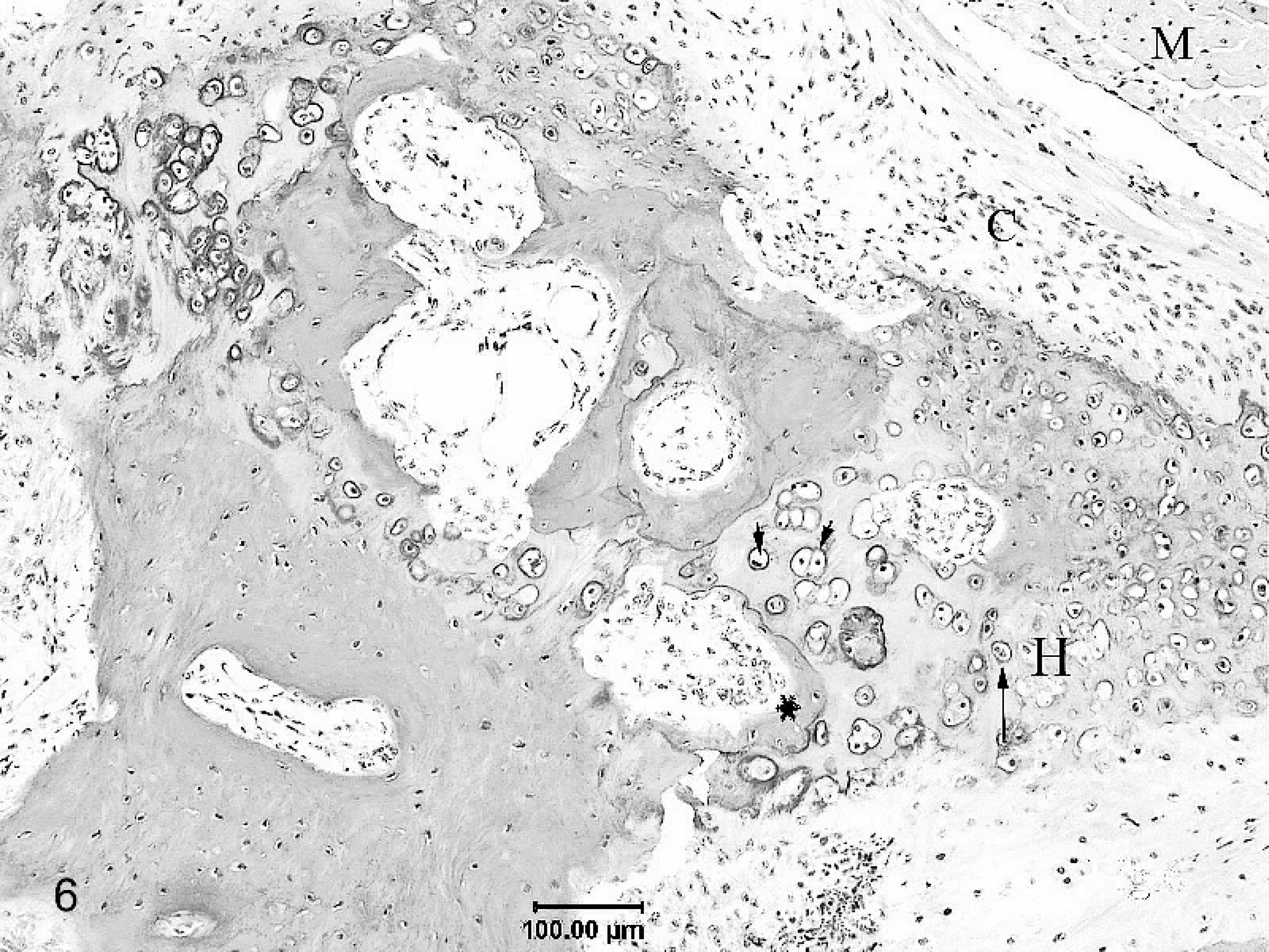
Formation of new bone by endochondral ossification in the right atrial myocardium of a sheep. The intervening connective tissue (C) present between the atrial myocardium (M) and the cartilaginous foci can be seen blending into the cartilage. Chondrocytes show haphazard columnization and a zone of hypertrophy (H). Hypertrophied chondrocytes (long arrow) undergoing degeneration and necrosis with pyknotic nuclei (arrow head/short arrows) can be observed preceding the zone of osteoid deposition (∗).
Although the prevalence of the osteocartilaginous metaplasia was relatively more frequent in Suffolk or crossbred sheep (mostly Suffolk-cross), this alteration was not restricted to this breed (palpable bone/cartilage was present in the right atrial myocardium of 2 female Blackface ewes among 6 in which this alteration was identified by palpation). Moreover, most of the sheep included in this study were Suffolk or crossbred animals. Similarly, sex predilection was also was not ascertained because of the limited number of male sheep included in the study. Nevertheless, an earlier report on the similar condition was described exclusively in adult male Suffolk sheep. 1
In the present study, gross abnormalities were observed neither in the atrioventricular and outflow valves nor in the myocardium. The functional and/or pathologic significance of the metaplastic alteration in the right atrial myocardium is still incompletely understood. Because the sheep included in this study were clinically healthy, the atrial ossification described here probably had no untoward effect on cardiac function and was interpreted as an incidental finding.
Acknowledgements
The authors wish to thank Dr. Langohr for critical review of the manuscript and Ms. Sharon Albregts and Mr. Bob White for help with collection and examination of specimens.
Footnotes
a.
General Electric LC-90-Model 11CE5, GE Biosciences, Piscataway, NJ.
