Abstract
Fumonisin B1 is a mycotoxin that causes lethal pulmonary edema in swine. Sphinganine, sphingosine, and the sphinganine to sphingosine ratio are important biomarkers for fumonisin B1 exposure. Currently, tissues selected for sphinganine and sphingosine analyses are frozen at −80°C until analyses take place. However, for diagnostics and some research projects, formalin is used more routinely as a preservative for long-term storage of tissues. To determine whether formalin-fixed tissues could be used for sphinganine and sphingosine analyses, sphinganine and sphingosine concentrations were quantified in both frozen and formalin-fixed lung, liver, kidney, and heart from fumonisin B1-treated and control pigs. Tissues were evaluated 3 months after freezing and 3, 6, and 12 months after formalin fixation. Sphinganine, sphingosine, and the sphinganine to sphingosine ratio of both frozen and formalin-fixed lung and liver from fumonisin B1-treated pigs were elevated. Formalin-fixed tissues had lower sphinganine and sphingosine concentrations but higher sphinganine to sphingosine ratios than the corresponding frozen tissues. Storage in formalin for up to 12 months did not affect the results. Sphingosine analysis could not be performed in formalin-fixed heart and kidney because of noninterpretable chromatograms. Therefore, formalin-fixed lung and liver can be used to determine fumonisin B1-induced sphinganine and sphingosine alterations in swine, with the sphinganine to sphingosine ratio being the most useful.
Fumonisin B1, a mycotoxin produced predominantly by the fungus Fusarium verticillioides, frequently contaminates corn. Ingestion of toxic doses of fumonisin B1 causes fatal pulmonary edema in pigs, leukoencephalomalacia in horses, and hepatotoxicity in all species studied. 5,6,19 In 1989, the corn crop in the midwestern and southeastern parts of the United States was heavily contaminated with fumonisins, and thousands of pigs died. Based on these outbreaks of porcine pulmonary edema and subsequent experimental studies, guidance levels for fumonisins were set by the US Food and Drug Administration, with the maximum level for total fumonisins in swine feed set at 20 ppm. Outbreaks of porcine pulmonary edema have not been reported until recently from subsequent corn crops presumably because of these guidance levels and the overall lower levels of fumonisins. A study by the US Department of Agriculture (USDA) found that only 7% of the 1995 Midwest preharvest corn contained greater than 5 ppm of fumonisin B1. However, an outbreak of porcine pulmonary edema was documented in Illinois by the authors0' laboratory from the 2006 corn crop. Submitted corn samples contained 23 ppm fumonisin B1. Because pulmonary edema is not specific for fumonisin toxicity and because feed is rapidly turned over in large operations and often not available for mycotoxin analysis from a specific time period, an alternative diagnostic tool for current and retrospective analysis is desirable.
Because of conformational similarity, fumonisin B1 is a dominant inhibitor of ceramide synthase that synthesizes complex sphingolipids from sphinganine and sphingosine. 18 Increased sphinganine and sphingosine concentrations have been observed in cells lines, animals, plants, and fungi following fumonisin treatment. 4,7,9 The use of sphinganine, sphingosine, and the sphinganine to sphingosine ratio as biomarkers for fumonisin B1 exposure has been validated in urine, serum, tissues, and cultured cells. 1,2,8,15,17 In pigs, serum and tissue sphinganine and sphingosine concentrations and the sphinganine to sphingosine ratio are elevated within 48 to 72 hours, and lethal pulmonary edema develops within 4 to 7 days after initial exposure to fumonisin B1 at ≥20 mg/kg body weight/day orally or ≥1 mg/kg body weight/day intravenously. 8,12,13,14 In addition to pulmonary edema, hepatocellular apoptosis has been documented. 6 Chronic hepatic injury has been induced with fumonisin exposures that do not cause pulmonary edema. 6
In experimental studies, sphingolipid analysis has been performed on fresh tissues. Fresh tissues, if not analyzed immediately, can be stored at −80°C but not −20°C because freezer burn (lipid oxidation) can occur after 3 months. 11 For diagnostic purposes and research projects, formalin is a more routine preservative for multipurpose, low-cost, long-term storage of animal tissues. Thus, in many cases, frozen tissues are not available for retrospective studies. The purpose of this study was to determine whether 1) formalin fixation interferes with sphinganine and sphingosine analysis by high-performance liquid chromatography (HPLC), 2) the concentrations of sphinganine and sphingosine in formalin-fixed tissues are similar to those from frozen tissues, and 3) the length of time in formalin fixative affects HPLC results.
In the present study, tissues were obtained from twelve 13- to 17-kg male castrated cross-bred pigs a and used to characterize the clearance of sphinganine and sphingosine from blood and urine following exposure to fumonisin B1. Group A (n = 4) and B pigs (n = 4) received 1 mg of purified fumonisin B1 b per kg of body weight intravenously at 0, 24, and 48 hours and were killed at 72 hours and 144 hours, respectively; group C pigs (n = 4) received equivalent volumes of sterile phosphate-buffered saline intravenously at 0, 24, and 48 hours and were killed at 144 hours. Two pigs given fumonisin B1 developed respiratory distress, fever, and anorexia at 48 to 72 hours. Both had gross and histological evidence of pulmonary edema. Group C pigs were unaffected. Lung, liver, kidney, and heart were obtained from each pig at necropsy and preserved at − 80°C for 3 months or in 10% buffered formalin c at room temperature for 3, 6, or 12 months prior to sphinganine and sphingosine analyses. Approximately 1 g of either frozen or formalin-fixed tissue was homogenized with 9 volumes of cold 0.05 M K2HPO4 d by Polytron e on ice at a moderate speed (4/10) with 20 up-and-down strokes. The homogenates were further diluted with 0.05 M K2HPO4 to achieve a final tissue to buffer ratio of 1: 9 for fixed heart; 1: 24 for frozen heart and fixed lung, liver, and kidney; and 1: 99 for frozen lung, liver, and kidney. Tissue to buffer ratios for different tissues were determined in a pilot study for optimized signals on HPLC chromatograms since the concentrations of sphinganine and sphingosine vary from organ to organ. In addition, 100 μl of formalin solution was sampled from fixed tissue containers of group A and C pigs for sphinganine and sphingosine analyses.
The method for quantitation of sphinganine and sphingosine in biological specimens by HPLC was described by Merrill 7,11 and subsequently revised by Yoo 10,20 to allow rapid extraction of free sphinganine and sphingosine from cultured cells. Yoo's method 10,20 was more economical and sensitive and consisted of simplified steps with improved recoveries that allowed rapid, simultaneous processing of a larger number of samples. Although the method worked for rodent kidney, its use in rodent liver and tissues from other species was not recommended because the chromatograms were often plagued by mystery peaks. 10,20 Prior to the study reported here, the authors reevaluated the methods described by Yoo 10,20 and developed a modified method for sphinganine and sphingosine analysis that has the advantages of Yoo's method 10,20 but also can be used for a variety of specimens, including body fluids (serum and urine from pigs and calves), cells in culture (HepG2 hepatocytes, PK15 renal cells, H9C2[2−1] cardiomyocytes and A10 aortic smooth muscle cells), and tissues (liver, kidney, lung, and heart from pigs and calves). Critical modifications from the method described by Yoo 10,20 included a higher amount of C20-sphinganine added per sample (50 instead of 20 pmol), a lower concentration of potassium phosphate buffer in the HPLC mobile phase (8% instead of 10%), a lower HPLC flow rate (1 ml/minute instead of 2 ml/minute), and a lower fluorescence excitation wavelength (230 nm instead of 340 nm).
Briefly, 100 μl of tissue homogenate, with C20-sphinganine f as an internal standard, was base hydrolyzed by 500 μl of 0.125 M methanolic c KOH d in chloroform c (4:1) for 1 hour at 37°C and extracted with 450 μl of chloroform, 450 μl of alkaline water, and 90 μl of 2 N NH4OH. d Base hydrolysis cleaves acylglycerolipids and hydrolyses lysosphingolipids to release free sphinganine and sphingosine but does not free sphinganine and sphingosine from complex sphingolipids. 7 The chloroform phase was transferred to a new tube, rinsed with 900 μl of alkaline water, and evaporated under an N2 stream (Reacti-Vap III). g The dry residue was resuspended in 100 μl of mobile phase buffer (90% methanol and 10% 5 mM KHPO4 pH 7.3) with 2 μl o-phthaldialdehyde (OPA) d reagent (25 mg OPA, 5 μl 2-mercaptoethanol, d and 500 μl absolute ethanol c in 50 ml of 3% of boric acid, d adjusted pH to 10.5 with KOH). Chloroform/methanol extraction following base treatment effectively removes phospholipids (phosphatidylethanolamine and phosphatidylserine) that are reactive to the OPA reagent. 7 Fluorescence was measured at an emission wavelength of 430 nm and an excitation wavelength of 230 nm by a fluorescence detector (HP1046A) h after separation through a Nova-Pak C18 4 μ column (WAT086342 Radial-Pak cartridge) i on the Hewlett Packard 1100 series HPLC system. h The mobile phase was 92% methanol and 8% 5 mM KHPO4, pH 7.3, at a flow rate of 1 ml/minute at 20°C. Resulting profiles were evaluated using the HP ChemStation for LC software. h The amounts of sphingosine, sphinganine, and C20-sphinganine in the final sample were determined by comparison with a concomitantly run standard curve (average R 2 = 0.997) composed of known amounts of sphingosine, f sphinganine, f and C20-sphinganine (2.5, 5, 10, 15, and 20 pmol). This set of standard solutions also allowed verification of retention times, column performance, and stability of OPA reagent. Since the extraction efficiency of C20-sphinganine is the same as sphinganine and sphingosine, 7 final sphinganine and sphingosine concentrations were normalized to the recovery of the C20-sphinganine internal standard. Retention times for sphingosine, sphinganine, and C20-sphinganine were at approximately 11, 14, and 22 minutes, respectively. The findings for selected samples for which this method was used were verified by liquid chromatography mass spectrometry analysis performed at the Toxicology and Mycotoxin Research Unit, USDA-ARS.
All analyses were run in triplicate. All values are presented as means ± standard errors. Statistical analysis was conducted by analysis of variance using the general linear model procedure of SAS/STAT 8.0. j Differences with P < 0.05 were considered significant. Correlation coefficients between the 2 data sets were determined by the CORREL function of Microsoft Office Excel 2003. k
Results indicated that the modified method for analysis of sphinganine and sphingosine by HPLC worked for swine lung and liver preserved in formalin. As shown on their HPLC chromatograms (Fig. 1), there were optimal signals and clear separation of sphinganine and sphingosine from other unknown peaks. After 3 months of formalin fixation for the heart and 6 months for kidney, overlapping peaks on the chromatogram interfered with quantification of sphingosine (Fig. 1). The presence of formalin fixative decreased the extraction efficiency of the C20-sphinganine internal standard and resulted in a lower average recovery (40%) and a higher detection limit (0.35 pmol/mg) for formalin-fixed tissues. For frozen tissues, the average recovery of the C20-sphinganine internal standard was 70%, and the detection limit was 0.20 pmol/mg.
Formalin fixation decreased tissue sphinganine and sphingosine concentrations (Figs. 2, 3). After fixation, decreases in sphinganine and sphingosine, averaging 66% and 76%, respectively, were present in comparison to their corresponding frozen specimens. Because the decreases in sphingosine concentrations were greater than those of sphinganine, each formalin-fixed specimen had a higher sphinganine to sphingosine ratio (Figs. 2, 3). The correlation coefficients of sphinganine, sphingosine, and the sphinganine to sphingosine ratios between fresh frozen and formalin-fixed tissues were greater than 0.95. Only trace amounts of sphinganine (accounting for 0.36% of the lost sphinganine) were detected in the formalin solution from fumonisin B1-treated pig tissues, while sphingosine was not detected (detection limit = 0.35 pmol/mg). There were no differences in the concentration of sphinganine and sphingosine and the sphinganine to sphingosine ratio of livers or lungs after 3, 6, and 12 months of formalin fixation (Figs. 2, 3). Both frozen (lung, liver, kidney, and heart) and formalin-fixed (lung and liver) tissues from fumonisin B1-treated pigs (groups A and B) had elevated concentrations of sphinganine and sphingosine as well as elevated sphinganine to sphingosine ratios as compared to control pigs (Figs. 2, 3).
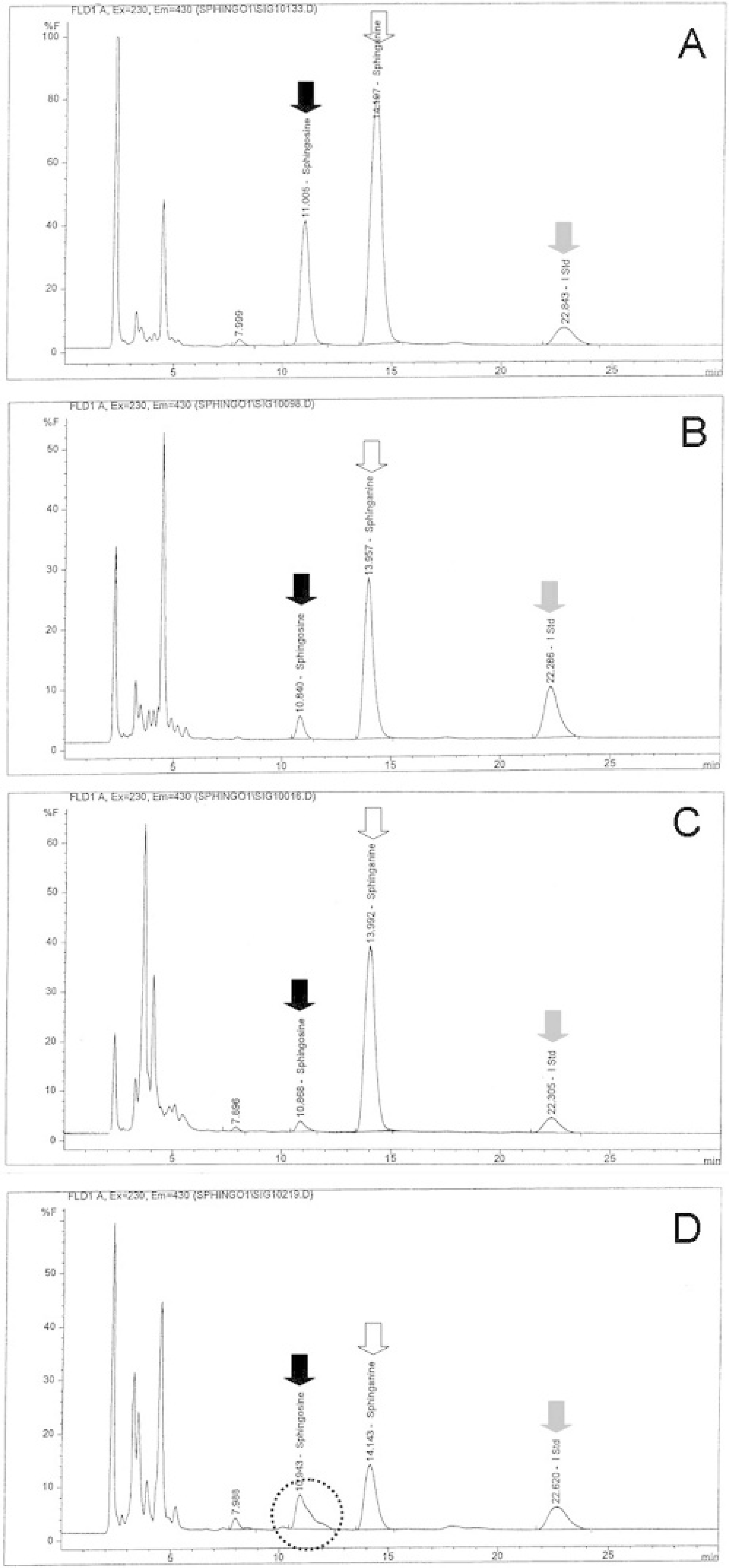
Representative sphingosine (solid arrow), sphinganine (open arrow), and C20-sphinganine internal standard (I Std; gray arrow) HPLC chromatograms of lung
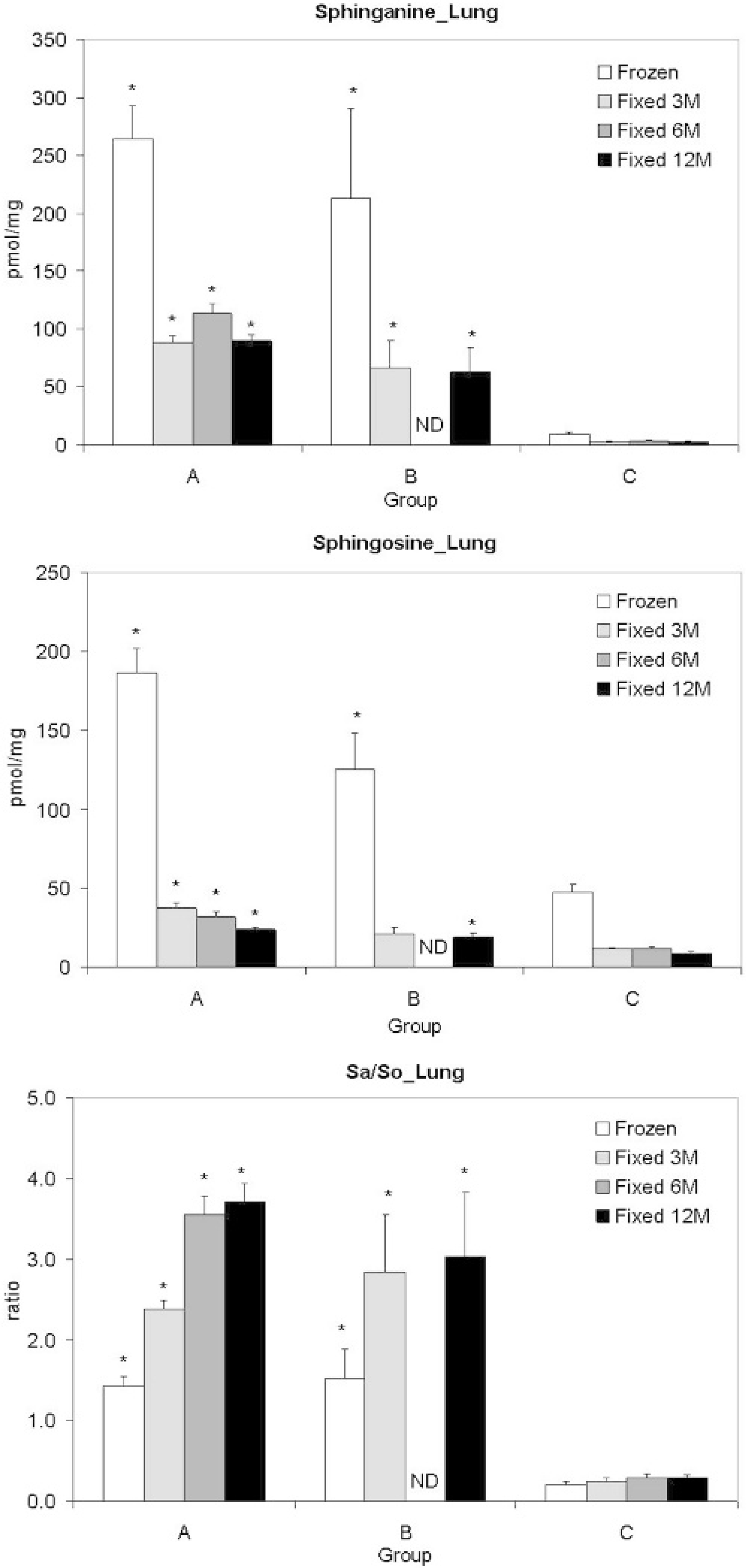
Tissue sphinganine
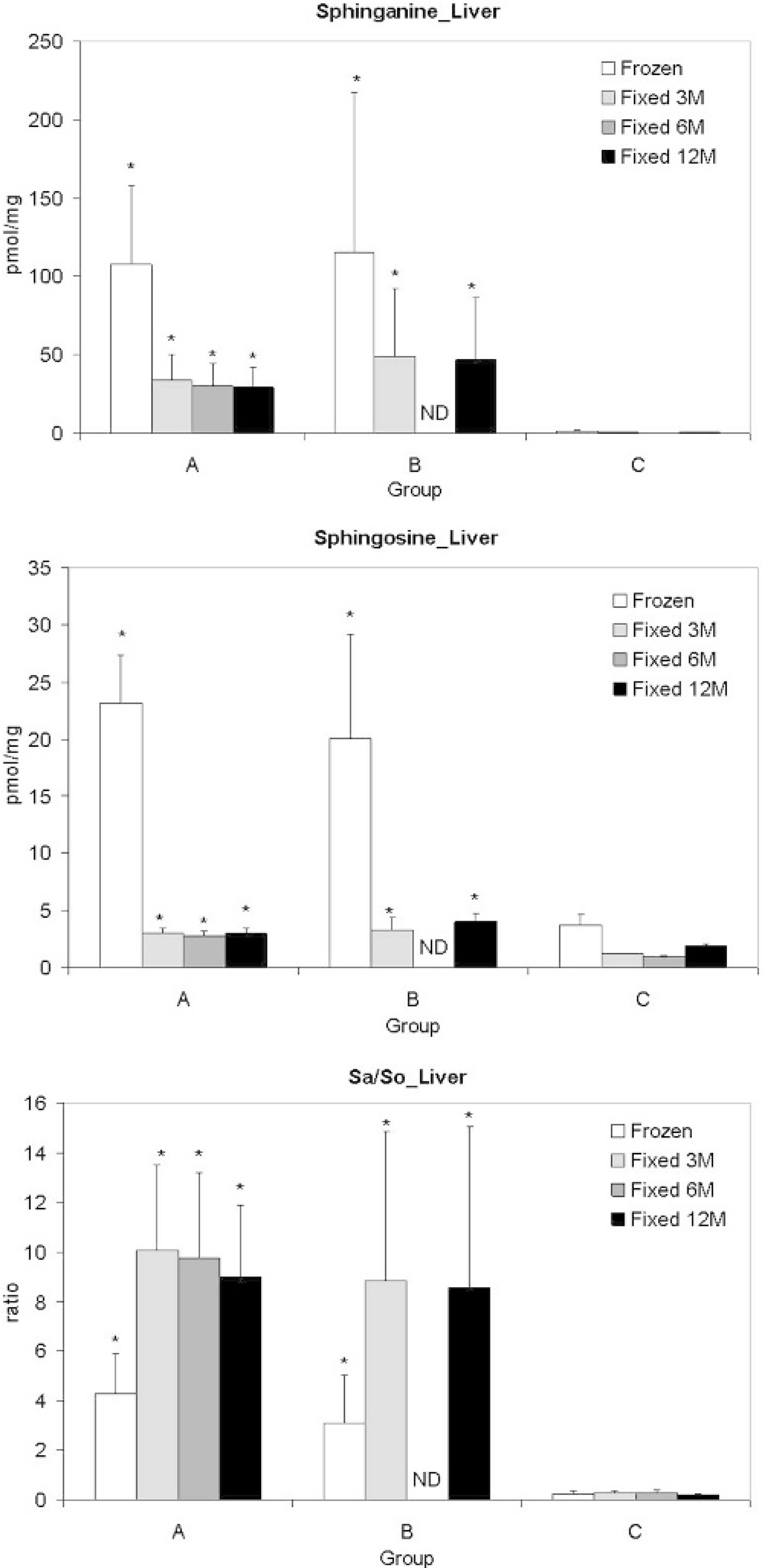
Tissue sphinganine
Preliminary reference ranges for sphinganine and sphingosine concentrations and the sphinganine to sphingosine ratio (Sa/So) in swine tissues and serum.*
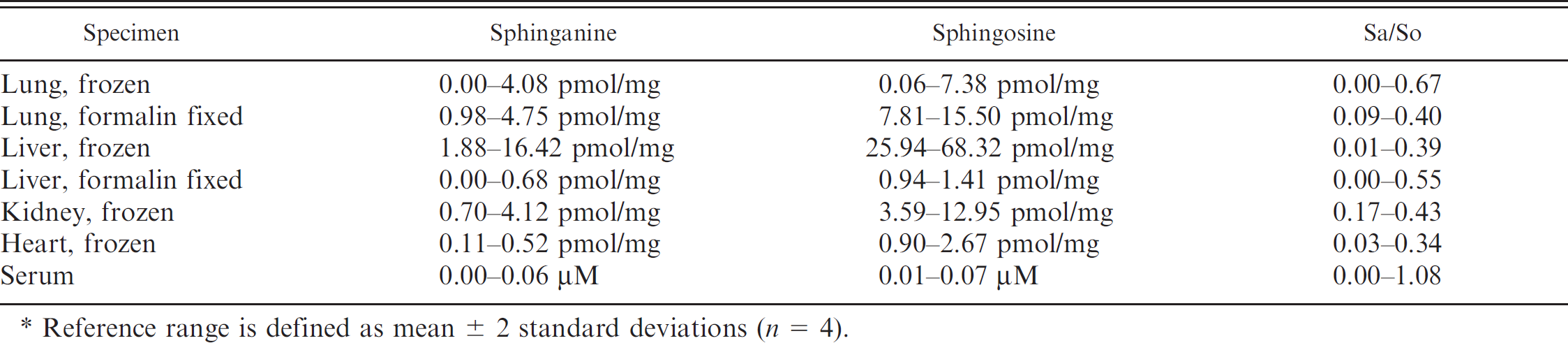
Reference range is defined as mean ± 2 standard deviations (n = 4).
This study demonstrated that formalin-fixed liver and lung can be used to determine fumonisin B1-induced alterations in sphinganine and sphingosine concentration in swine, with the calculated sphinganine to sphingosine ratio being the most useful. Although formalin fixation decreased tissue sphinganine and sphingosine concentrations and increased the sphinganine to sphingosine ratio, storage in formalin for up to 12 months did not further affect the concentrations of sphinganine and sphingosine or the sphinganine to sphingosine ratio in tissues, indicating their usefulness as biomarkers for fumonisin B1 exposure. This information should allow retrospective determination of sphinganine and sphingosine in formalin-fixed tissues in cases in which fumonisin B1 exposure is considered a potential cause of toxicity in spontaneous disease. Also, it would allow tissues to be fixed in formalin for storage in experimental cases.
Based on data from the control pigs, preliminary reference ranges for sphinganine and sphingosine concentrations and the sphinganine to sphingosine ratio in swine tissues and serum are presented in Table 1. Data from frozen tissue presented here are consistent with those published in the literature. Fumonisin B1 exposure in pigs results in increased tissue sphinganine concentration, sphinganine to sphingosine ratio, and, to a lesser extent, sphingosine concentration. 8,12,13,14 Sphinganine concentrations in tissue (lung, liver, kidney, and heart) of control pigs are always lower than sphingosine, with lung having the highest sphingosine concentration. 10 Sphinganine and sphingosine alterations in pigs with naturally occurring fumonisin B1 toxicosis are still poorly documented in the literature. In the recent field outbreak of acute pulmonary edema and death associated with fumonisin B1 consumption investigated by the authors' laboratory, the average serum sphinganine concentration was 1.67 μM, the average sphingosine concentration was 5.30 μM, and the average sphinganine to sphingosine ratio was 3.22 (unpublished data).
Quantification of sphinganine and sphingosine in formalin-fixed heart and kidney was not successful because overlapping peaks on the chromatogram interfered with quantification of sphingosine. The cause of the interference is not known. Prolonged formalin fixation was shown to reduce the total amount and the composition of gangliosides in the brain of rats: the acidity of buffered formalin was suspected to be responsible for the decreases. 16 Because only a small amount of sphinganine was detected in formalin from the fixed tissues in this study and because both sphingosine and sphinganine seem resistant to acid treatment, 11 the lower extraction efficiency and the decrease in concentration of sphinganine and sphingosine in formalin-fixed tissues is probably a result of formaldehyde-induced cross-linking of sphinganine and sphingosine to other molecules, 3 which is not reversible by the base hydrolysis and extraction procedures used in this study.
In conclusion, formalin-fixed liver and lung can be used to determine fumonisin B1-induced sphinganine and sphingosine alterations in swine, with the sphinganine to sphingosine ratio being most useful.
acknowledgements. Supported, in part, by the International Life Sciences Institute, the Cooperative Regional Project NC-129 (mycotoxins in cereal grains), and the University of Illinois at Urbana-Champaign, Urbana, Illinois.
Footnotes
a.
Veterinary Research Farm, University of Illinois at Urbana-Champaign, Urbana, IL.
b.
Center for Food Safety and Applied Nutrition, Food and Drug Administration, Laurel, MA.
c.
Fisher Scientific International, Fair Lawn, NJ.
d.
Brinkmann Instruments Inc., Switzerland.
e.
Sigma-Aldrich Co., St. Louis, MO.
f.
Matreya Inc., Pleasant Gap, PA.
g.
Pierce Biotechnology, Rockford, IL.
h.
Hewlett-Packard Development Co., Palo Alto, CA.
i.
Waters Corp., Milford, MA.
j.
SAS Institute, Cary, NC.
k.
Microsoft Corp., Redmond, WA.
