Abstract
African swine fever (ASF) causes fatal disease in pigs and is an escalating threat to the global swine industry. ASF has re-emerged from Africa as a transcontinental epidemic spreading through the Caucasus into Europe, Russia, China, numerous Asian countries, and the Caribbean. ASF virus (ASFV) is a U.S. select agent requiring handling in high-containment biosafety level 3 (BSL-3) laboratories for pathogen work. Formalin-fixation eliminates infectivity and preserves the genome, providing noninfectious specimens for BSL-2 work. Recovery of DNA from formalin-fixed, paraffin-embedded tissue (FFPET) is challenging and cumbersome. A reliable and easy-to-perform method for DNA recovery from FFPET would facilitate surveillance. To meet this objective, we developed a high-throughput protocol for the recovery of ASFV DNA from FFPET. Deparaffinization, tissue lysis, and reversal of cross-linking were performed in a single tube, followed by DNA purification via automated magnetic bead extraction. Quantitative PCR (qPCR) detection was used to determine the copy number of the B646L gene that encodes for the ASFV p72 protein in tissues (5 pigs, 4 tissues) from pigs with lesions consistent with acute ASF. Copy numbers obtained from FFPET were within one log of copy numbers obtained from fresh tissue, thus enabling ASF qPCR surveillance from formalin-inactivated and preserved tissues at BSL-2 at diagnostic sensitivity similar to fresh tissues tested at BSL-3.
African swine fever virus (ASFV; Asfarviridae, Asfivirus), a large, double-stranded DNA virus, is the only known mammalian arthropod-borne DNA virus, transmitted by soft ticks (Ornithodoros spp.), with reservoirs in wild members of the Suidae family.1,4,9,17 ASFV infection in domestic pigs results in African swine fever (ASF), a contagious, hemorrhagic disease with nearly 100% mortality with virulent strains.13–15 Clinical signs include fever, cutaneous hyperemia and cyanosis, incoordination, abortion, and sudden death. Subacute, subclinical, and chronic forms of ASF are described, most commonly with less-virulent ASFV strains whereby subclinical pigs can shed virus for extended times. 3 The virus predominantly infects monocytes–macrophages resulting in massive lymphocyte destruction most likely mediated via a proinflammatory cytokine storm. 14 Acute ASF lesions include hemorrhage, and necrosis and/or inflammation in various tissues, including lymph nodes, spleen, lung, liver, kidney, and skin.13–15
ASF, first described in Kenya in 1910–1921, is endemic in sub-Saharan Africa, causing sporadic outbreaks.2,12 In the mid-20th century, a genotype I ASFV caused the first transcontinental epidemic of ASF when it spread to various countries in Europe, Asia, and the Americas before becoming eradicated from outside Africa in the late 1990s. ASF genotype 1 only remained endemic on the island of Sardinia, Italy. 2 In 2007, a genotype II ASFV was identified in the Republic of Georgia and spread through the Caucasus and Russia, and into Eastern Europe and China.2,14 Since its spread into China, ASFV has been detected in numerous countries in eastern Asia and the Americas, with a recent incursion into the Caribbean island of Hispaniola (Dominican Republic, Haiti; https://www.oie.int/app/uploads/2022/01/asf-situation-report-3.pdf).
ASFV poses a high risk to global pork production given its: 1) high mortality, 2) efficient transmission, 3) extended survival in the environment, animal feed, and meat products, 4) a global presence of susceptible wild pigs and competent arthropod vectors that establish reservoirs and facilitate transmission, and 5) the absence of an efficacious treatment or vaccine. ASFV poses a considerable risk as a potential bioterrorism agent and is a U.S. select agent, requiring specialized biosafety level 3 (BSL-3) laboratories and specialized training and certification for personnel to perform research and diagnostic work, further complicating efforts to mitigate its impact on global agriculture.8,13,19 Formalin-fixation is an effective method to inactivate ASFV in tissues while maintaining access to nucleic acids and proteins.6,11 Laboratory the use of formalin-fixed, paraffin-embedded tissue (FFPET) is a sound strategy for the safe transport of ASFV-suspect or ASFV-positive samples for active and passive surveillance, laboratory testing, epidemiologic studies, and biomedical research. However, inherent complications with FFPET can impact their utility as a molecular detection specimen including: 1) paraffin wax inhibition of extraction and PCR, 2) chemical modification and crosslinking between nucleic acids and proteins, and 3) nucleic acid fragmentation and rapid degradation as a result of tissue manipulation.7,18 One study demonstrated that ASFV can be detected by gel-based PCR following phenol:chloroform:isoamyl alcohol purification methods using formalin-fixed tissues; however, ASFV detection and recovery from FFPET was unsuccessful using commercial purification kits. 10 Here, we describe a simple and reliable protocol for the purification, detection, and quantification of ASFV DNA from FFPET from acutely infected pigs using a single-tube deparaffinization protocol, followed by magnetic-bead total nucleic acid extraction and qPCR detection. This protocol for FFPET could be applied to diagnostic and surveillance efforts to monitor and curtail the global spread of this pathogen and lessen global impacts.
We included 5 tissues (i.e., heart, liver, spleen, tonsil, prescapular lymph node) from 5 pigs, 5–7 d post-inoculation (dpi) with ASFV, in our study. Pigs were housed at BSL-3 Ag level (Biosecurity Research Institute at Kansas State University [KSU], Manhattan, KS, USA) and infected with 360 HAD50 (50% hemadsorbing dose) of genotype II ASFV strain Armenia 2007 (Arm07) intramuscularly. Two pigs (11, 14) died from peracute ASF (5 dpi); the remaining pigs were euthanized because of severe disease at 5 dpi (12, 13) and 9 dpi (15). Tissues collected at autopsy were formalin-fixed for a minimum of 10 d in 10% neutral-buffered formalin at a 1:20 tissue:formalin ratio, then processed and paraffin-embedded using the Kansas State Veterinary Diagnostic Laboratory (KSVDL) standard BSL-2 procedures. Given that these studies were performed at the BSL-3 Ag containment facility at KSU, the validated protocol for formalin fixation requires a minimum of 10 d for tissues kept in formalin prior to release to the lower BSL-2 containment. Tissue sets from 2 noninfected pigs served as negative controls. Paired fresh-frozen tissues (FFTs) were collected, frozen at −80°C, and stored in the BSL-3 laboratory until processed. For molecular testing of FFPET, five, 5-µm thick, microtome sections from blocks were processed as a single sample (see details below). H&E slides for histologic evaluation were prepared following KSVDL standard procedures. Postmortem images were captured (G12 camera; Canon) as were microscopic images (BX46 light microscope, DP27 camera, CellSens standard v.1.16; Olympus). Gross and microscopic images were further color calibrated (EZImage Prep function, ChromaCal v.2.5.1; Datacolor). Images and panels were finished and assembled (Photoshop and InDesign; Adobe).
Gross and histologic pathology findings in pigs were consistent with peracute-to-acute ASF (Fig. 1; Suppl. Fig. 1). Gross lesions included splenomegaly; severe, diffuse, pulmonary edema and congestion; and moderate-to-severe lymphadenopathy with hemorrhage and edema of lymph nodes (Fig. 1). Representative histologic lesions included mild-to-marked lymphocytolysis, fibrinonecrotic vasculitis, hemorrhage and edema within the tonsils, lymphadenitis, and splenitis (Suppl. Fig. 1A–C). Liver lesions were variable and ranged from minimal-to-mild to severe, nonsuppurative, necrotizing, periportal hepatitis (Suppl. Fig. 1D).
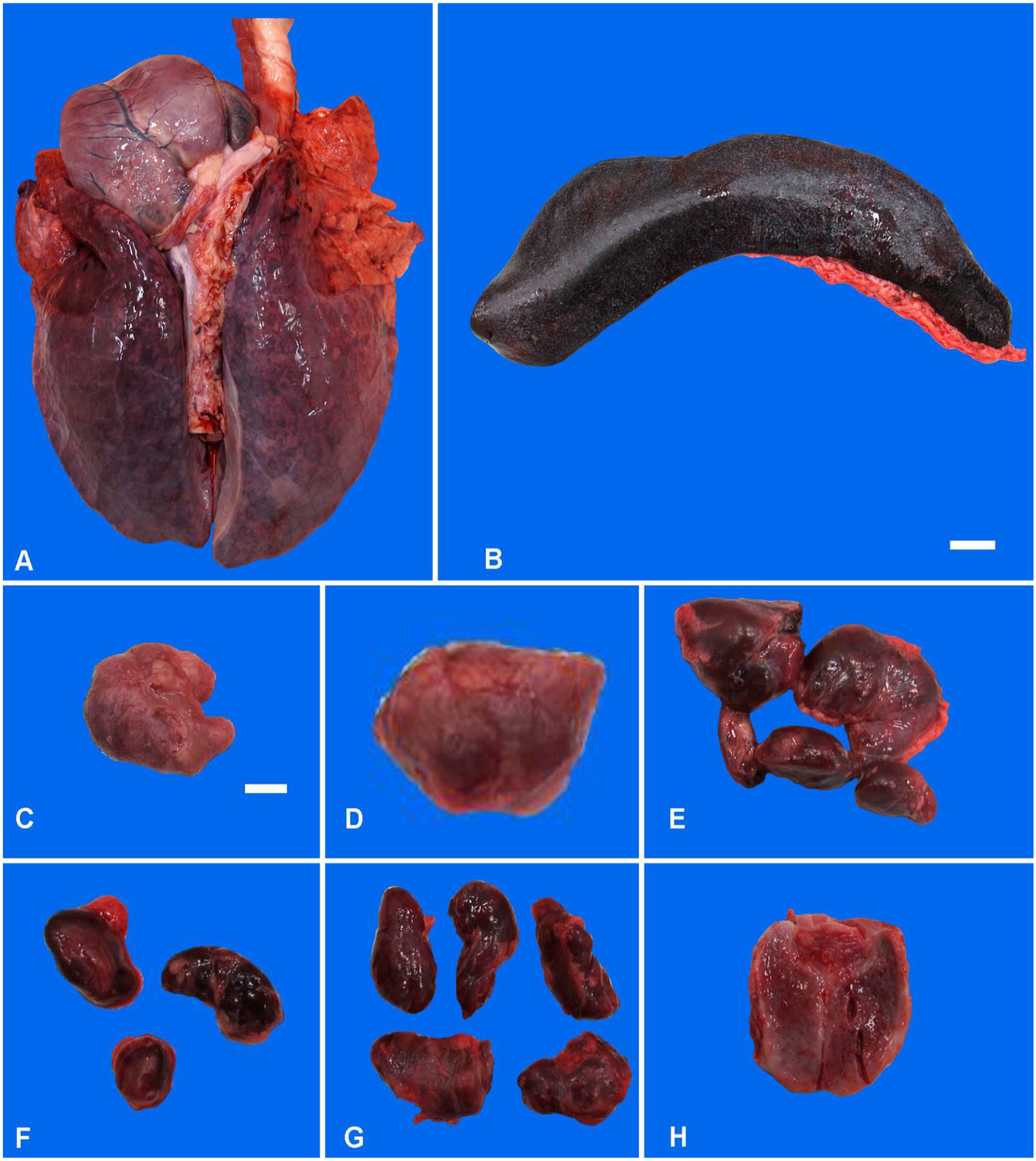
Gross lesions in pigs infected experimentally with African swine fever virus strain Armenia 2007.
FFPET processing was performed in a BSL-2 laboratory. Paraffin sections in 2-mL safe-lock microcentrifuge tubes (Qiagen) containing 320 µL of deparaffinization solution (Qiagen) were incubated at 56°C for 3 min; 250 µL of ATL buffer (Qiagen) was added. Tubes were centrifuged (9.0 × g) for 1 min for phase separation: paraffin (upper phase) and tissue/ATL buffer (lower phase). Following the addition of 40 µL (20 ng/µL) of proteinase K (Qiagen), tubes were incubated at 56°C for 1 h, then heat-treated for reversal of formalin-induced DNA crosslinking (90°C for 1 h). The tissue lysate below the paraffin gel was transferred to a tube containing 280 µL of AL buffer (Qiagen) and vortexed. Nucleic acids from FFPET were purified using automated magnetic-bead extraction (TacoMini; GeneReach) or on a workstation (BioSprint96; Qiagen) using the DNA/RNA extraction kit (GeneReach).
Fresh tissues (previously frozen) were processed in an ASFV BSL-3 laboratory, by trained and certified personnel. For FFT, 200 mg of tissue was minced and homogenized in Dulbecco modified Eagle medium (20% weight/volume [w/v]; Corning; LT tissue lyser, Qiagen); 200 µL of the clarified homogenate was added to 300 µL of ATL buffer containing 40 µL (20 ng/µL) of proteinase K and digested (56°C for 2 h). The clarified tissue lysate (200 µL) was mixed with an equal volume of AL buffer. DNA from FFT was purified using automated magnetic-bead extraction (KingFisherDuo Prime device, Thermo Fisher; DNeasy blood & tissue kit, Qiagen).
DNA extractions were performed according to manufacturers’ instructions with modifications. Briefly, 100 µL of FFPET or FFT lysate was added to 500 µL of lysis buffer (GeneReach) or AL buffer and 50 µL of magnetic beads (provided in the extraction kits). Before extraction, 200 µL of molecular-grade isopropanol (Thermo Fisher) was added to the lysis well. DNA bound to beads was washed 2 times with 750 µL of wash buffer A (GeneReach) or AW1 buffer (Qiagen), once with 750 µL of wash buffer B (GeneReach) or AW2 buffer (Qiagen), followed by a final wash with 750 µL of 100% molecular-grade ethanol (Thermo Fisher). Following a 5-min drying period, DNA from FFPE and FFT was eluted in 100 µL (FFPET) or 70 µL (FFT) of elution buffer, respectively. Negative and positive controls included were molecular-grade water and a phage-based ASFV extraction positive control (gift from FADDL, USDA-APHIS), respectively.
We used primers and probes from a published qPCR ASFV p72 gene detection assay 20 for the detection of the ASFV p72 encoding gene. PCR primers (Integrated DNA Technology) and FAM probe (Thermo Fisher) at 2 µM (2 µL) and 1 µM (0.1 µL), respectively, were used in a 20-µL qPCR reaction 16 (PerfeCTa FastMix II; Quanta Biosciences). PCR was performed (CFX96 Touch real-time PCR detection system; Bio-Rad) in triplicate using 2.5 µL of DNA. Thermocycling conditions were 95°C for 5 min, 45 cycles of 95°C for 10 s, and 60°C for 1 min. Negative and positive PCR controls were included in each qPCR run and consisted of molecular-grade water (Thermo Fisher) and the quantified phage-based ASFV-positive amplification control (USDA-APHIS-FADDL) and/or the positive control (ASFV p72 plasmid).
For the quantification of ASFV copy number (CN), serial dilutions (10×) of the positive control (ASFV p72 plasmid) were used to generate an 8-point standard curve (109–101 copies) using 16 qPCR well replicates performed as 2 scientific replicates. The ASFV p72 CN/reaction was mathematically determined using the mean cycle threshold (Ct) value and the slope and intercept of the DNA standard curve. The qPCR CN represents quantification of ~25% of the total tissue lysate from the FFPET (5 paraffin sections) and 5% of the FFT (20% w/v homogenate). For normalization of ASFV p72 CN from the 2 distinct specimens, 1 of which cannot be quantitated by tissue weight (FFPET), an endogenous reference gene (RG) assay was used for qPCR CN normalization. This qPCR was run in duplicate, and the RG CN was determined by the reference standard curve methodology as described above. The ASFV p72 CN was normalized to 1,000 copies of the RG to allow for direct comparison of the quantitative data sets (Table 1).
Quantitative PCR copy number determination for African swine fever virus p72 gene* recovered from fresh-frozen tissue (FFT) and formalin-fixed, paraffin-embedded tissue (FFPET).
PS LN = prescapular lymph node.
Copy number is reported per 1,000 copies of porcine reference gene.
Using this rapid single-tube processing method for FFPET, followed by automated magnetic-bead extraction, all 25 FFPET samples from ASFV-infected pigs tested positive for the ASFV p72 gene (Table 1). In most cases, the CNs detected were equivalent to those detected using FFT (within a log), demonstrating <10% variability of DNA recovery between FFPET and FFT, when comparing CN that is reported normalized to RG equivalents. We acknowledge that this is not a traditional way of reporting PCR CN or Ct data for veterinary diagnostic work 5 ; however, it does allow for the direct comparison of the PCR data sets when one sample preparation method is based on paraffin sections cut from embedded tissue whereas the other is based on a 20% w/v tissue homogenate. The estimated weights of tissue used for extraction were 20 mg of fresh tissue and 1–2 mg of tissue from paraffin sections (Suppl. Table 1). For all FFPET samples, mean Cts were <35, allowing for detectable ASFV DNA in all tissue types tested (Suppl. Table 1). The highest concentration of ASFV DNA was recovered from the spleen, followed by the tonsil, lymph node, and liver. The lowest ASFV CN was detected in the heart (Table 1). qPCR results were consistent with gross and histologic findings, with high viral DNA recovery in pigs with severe acute ASF lesions (Fig. 1; Suppl. Fig. 1). The liver of pig 13 had the highest ASFV CN of the group, and this finding is supported by the severity of the inflammation and necrosis detected histologically in the liver (Suppl. Fig. 1D). Our results are consistent with a study that used fresh tissues from experimentally infected pigs and demonstrated high CNs of ASFV DNA that was correlated with severe, acute ASF lesions. 20 ASFV DNA was not detected in FFT or FFPET tested from various tissues (lung, liver, spleen, lymph node) from noninfected controls (data not shown).
Formalin-fixation inactivates infectious agents by crosslinking RNA, DNA, and proteins. Formalin-fixation provides a simple and routine method of tissue preservation for histopathology, and for immunohistochemical or in situ detection methodologies. It also enables safe ambient temperature transport of samples suspected or confirmed to contain high-consequence pathogens. Because the pathogen is inactivated, processing and molecular-based detection methods can be safely performed in BSL-2 laboratories. Our objective was to develop a user-friendly, high-throughput, DNA purification protocol for sensitive detection of infectious agents using FFPET. The promising results of our pilot study, warrant further evaluation of the use of FFPET as specimens for PCR detection of ASFV, especially in countries with limited resources and/or in areas without, or geographically distant from BSL-3 laboratories. All reagents that we used are available commercially, and our protocol is applicable for routine laboratory testing, surveillance, and retrospective studies to further advance ASFV biomedical research and aid global efforts to mitigate this pathogen.
Supplemental Material
sj-pdf-1-vdi-10.1177_10406387231158534 – Supplemental material for Development of a sensitive, high-throughput extraction protocol for qPCR detection of African swine fever virus in formalin-fixed, paraffin-embedded tissues
Supplemental material, sj-pdf-1-vdi-10.1177_10406387231158534 for Development of a sensitive, high-throughput extraction protocol for qPCR detection of African swine fever virus in formalin-fixed, paraffin-embedded tissues by Kinga Urbaniak, David A. Meekins, A. Sally Davis, Juergen A. Richt and Jessie D. Trujillo in Journal of Veterinary Diagnostic Investigation
Footnotes
Acknowledgements
We thank Dr. Michael T. McIntosh from the U.S. Department of Agriculture, Animal and Plant Health Inspection Service, Foreign Animal Disease Diagnostic Laboratory (FADDL), Plum Island, New York, for providing the phage-based positive control for qPCR for ASFV; Lauren Pearson and Jodee Webster from the Kansas State Veterinary Diagnostic Histology laboratory for providing paraffin scrolls; William Wilson (USDA ARS), Sun Young Sunwoo, Chester McDowell, and Tammy Koopman (KSU) for their assistance with the autopsy; certified medical illustrator, Mal Rook Hoover, KSU-CVM, for technical assistance with gross and histologic photographic panels; and the faculty and staff at the Biosecurity Research Institute at KSU for their support with BSL-3 animal work.
Declaration of conflicting interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article. The JAR laboratory received support from Tonix Pharmaceuticals, Genus, Xing Technologies, and Zoetis, outside of the reported work. Juergen A. Richt is an inventor, and patents have been issued on the use of antivirals and vaccines for the treatment and prevention of virus infections, owned by Kansas State University, KS, USA.
Funding
Our work was funded through grants from the National Bio and Agro-Defense Facility (NBAF) Transition Fund from the State of Kansas (Juergen A. Richt), the Department of Homeland Security for the Center of Excellence for Emerging and Zoonotic Diseases (CEEZAD), grant DHS 2010-ST-061-AG0001, and the AMP Core of the Center of Emerging and Zoonotic Infectious Diseases (CEZID) from the National Institute of General Medical Sciences (NIGMS) under award P20GM130448 (J.A. Richt).
Supplemental material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
