Abstract
Necropsy of 4 neonatal calves with a peracute syndrome of abdominal distention, diarrhea, dehydration, shock, and death revealed abomasal tympany, marked edema, hemorrhage, and emphysema of ruminal and abomasal walls and histopathologic lesions characteristic of forestomach acidosis. The presence of acid damage and the production of substantial quantities of gas strongly suggested that the pathogenesis of the syndrome involved exuberant fermentation of intragastric substrate. This supposition led to attempts to experimentally induce the syndrome by dosing neonatal calves with an alimentary tract flora, followed by milk replacer supplemented with excessive fermentable carbohydrate (D-glucose and cornstarch). Two of the 5 calves thus treated developed a syndrome very similar to that observed in naturally affected calves. Literature suggests involvement of several Clostridial species, Sarcina spp., and possibly other microbes or a combination thereof to be involved in very similar syndromes. Therefore it is suggested that this complex of lesions and clinical signs is not dependent on a single etiologic agent. Rather, it is proposed that the natural occurrence of disease requires a quantity of highly fermentable substrate (starch, glucose, lactose, etc.) and a bacterial flora that is capable of rapidly fermenting that particular substrate with a resultant production of gas and acid.
There are a number of reports of acutely fatal syndromes generally in young (0-10 weeks) calves, 5,13,15,16 lambs, 2,4,6,10,12,18 and goat kids 3 characterized clinically by acute onset, rapid course, abdominal distention, pain, shock, and death. At necropsy there are variously fore-stomach and abomasal gaseous distention, hemorrhage, inflammation, mucosal necrosis, and often mural emphysema of the stomachs. These syndromes have been reported as abomasal tympanya 11,13,17 abomasal bloat, 3,10,16,18 braxy-like disease, 15 and abomasitis. 4,5 They are etiologically ascribed to or associated with a number of bacterial pathogens (Clostridium septicum, 4,5,15 Clostridium fallax, 18 Clostridium sordellii, 8,18 Clostridium perfringens, 11,13,15,16 Campylobacter spp. 11 and Sarcina spp 3,18 ) and associated with certain dietary regimens, feeding systems, 2,6,12 and nutritional deficiencies. 9 It is unclear whether these reports concern a single or multiple closely related syndromes that appear to have a similar pathogenesis. Among cases reported, all are characterized by excessive gas production and gaseous distention within the stomachs.
Four naturally affected calves that shared a number of features in common with animals of the cited reports were examined. They were 2–3 days old and included 1 of dairy and 3 of beef breed. The first, a Holstein, was an embryo transfer product whose birth was induced and delivery assisted. The calf was treated for partial failure of passive antibody transfer and bacterial septicemia. Necropsy revealed acute hemorrhagic and emphysematous rumenitis together with abomasitis and multifocal hepatic necrosis and interstitial pneumonia. The second calf, a purebred Angus delivered by caesarian section on the farm, developed diarrhea and was treated with antibiotic by the owner. The calf became recumbent at 2 days of age and died during transport for veterinary attention. Necropsy revealed erosion and mural emphysema of ruminal, reticular, and omasal mucosa and abomasal edema. Coincidental to alimentary tract lesions there were multiple renal cysts, cardiomegaly, multiloculated myocardial hematocysts, a 2.5-cm-diameter patent foramen ovale, and a patent ductus arteriosus. The third calf was of mixed beef breeding, had not been observed to stand after birth, was tube-fed whole milk (amount unknown), then presented for veterinary attention because of abdominal distention and recumbency. Based on physical and neurologic examinations, the animal was euthanized a and necropsied. There were abomasal and ruminal hemorrhages, edema, erosion, and emphysema. Further, there were medullary and cervical spinal dysraphism and bronchopneumonia. The fourth calf, a 3-day-old Brangus, presented with anorexia, depression, and abdominal distention. Approximately 4 liters of whole milk had been tube-fed in a single feeding. Feces had not passed since birth. Intestinal obstruction was suspected. The calf died, and necropsy 3 hours after death revealed a large amount of curdled milk in the abomasum and abomasal emphysema. There were segmental aplasia of the colon, pulmonary edema, and interstitial pneumonia.
Salient gross lesions that occurred in each animal were abomasal distention with gas or ingesta, serosal and mucosal hemorrhage, marked edema, and emphysema of the abomasal wall. These lesions were usually accompanied by mucosal erosion and mural emphysema of the rumen, reticulum, and omasum (Fig. 1). Histopathologic findings in forestomachs of calves 1, 2, and 3 included hydropic change, vesiculation, pustule formation, and erosion of forestomach epithelium, lesions that are characteristic of ruminal acidosis 1 (Fig. 2). Numerous gaseous bullae were noted in the lamina propria and submucosa of the forestomachs of calves 1, 2, and 3. The abomasums of each of the 3 variously contained focal areas of mucosal degeneration, hemorrhage (often transmural), and marked submucosal edema, occasionally with a sparse, well dispersed, mixed inflammatory cell infiltrate. Gaseous bullae were present in the abomasal wall of calf 1 and 4.
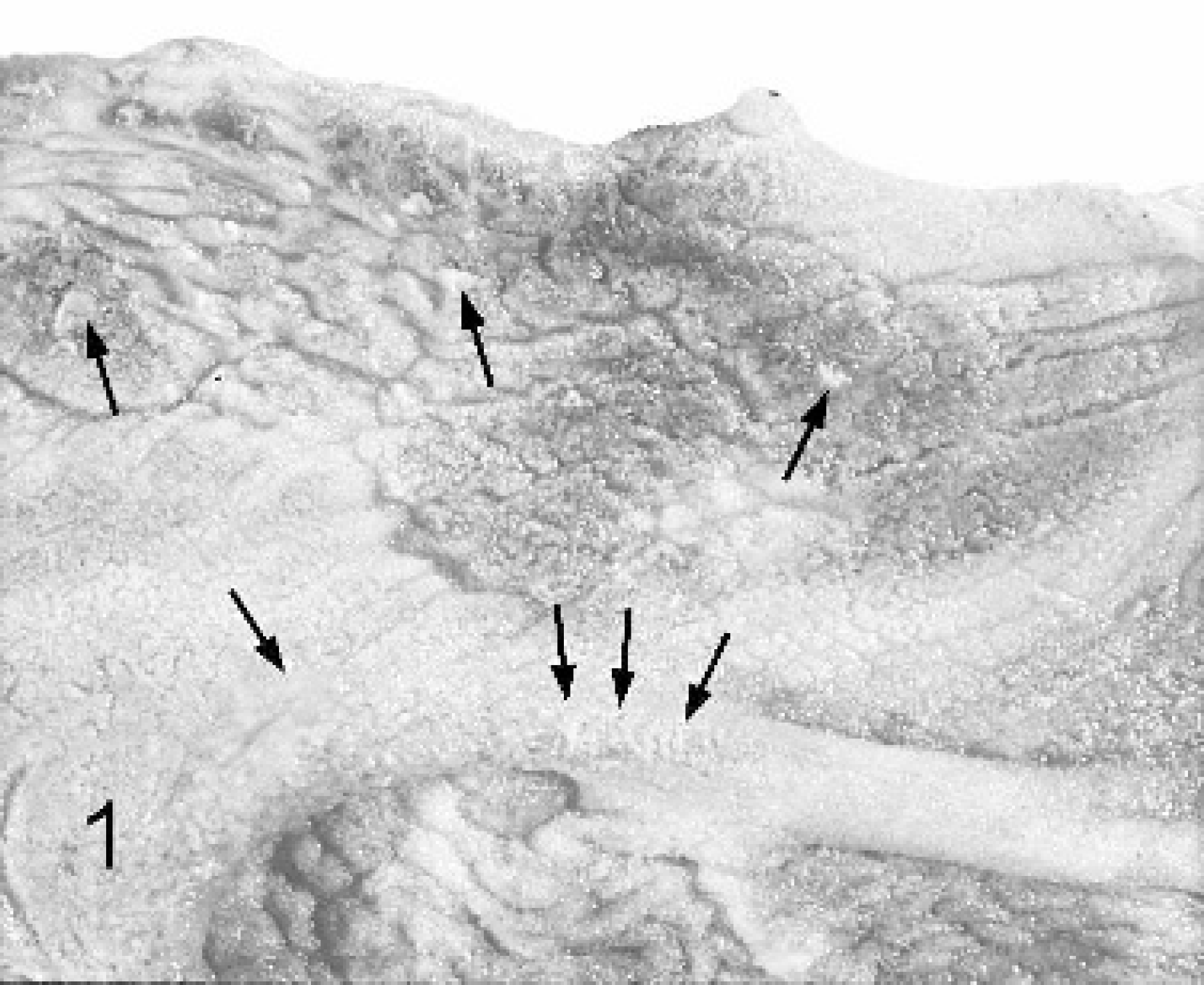
Rumen mucosa of calf 2 with areas of elevated, discolored mucosa and pale adherent exudate (arrows). Mucosal elevation is due primarily to mural emphysema.
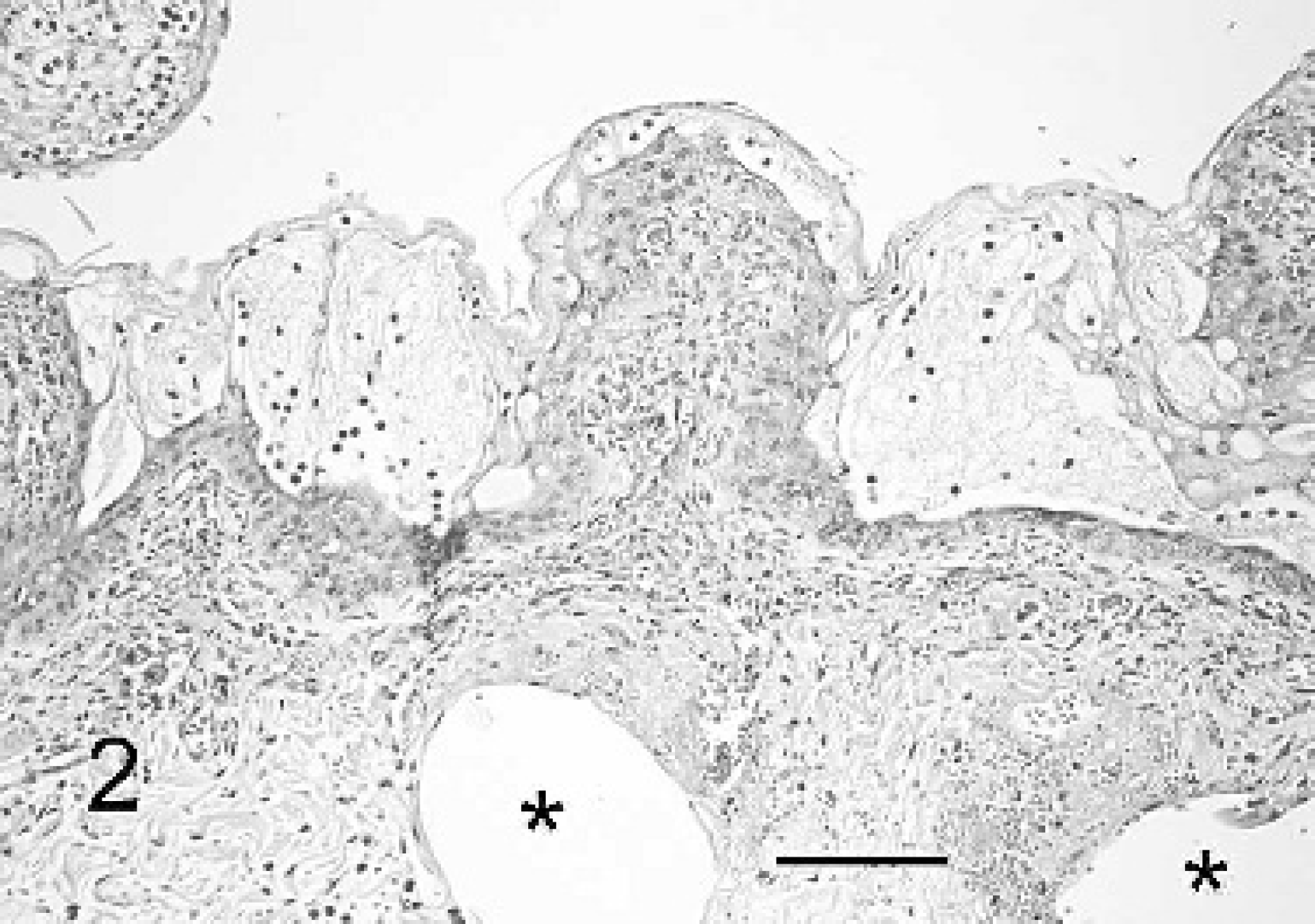
Rumen wall of calf 3 with epithelial vesiculation and early pustule formation. The lamina propria is congested, edematous, hemorrhagic, and mildly infiltrated with neutrophils. Note the clear, rounded gaseous spaces in the submucosa (*). HE. Bar = 100 μm.
Microbiologic assay included aerobic and anaerobic culture of stomachs and morphologic examination of forestomach impression smears (Gram stain) and of histologic sections of each animal (hematoxylin and eosin [HE], Gram, Giemsa, Steiner stains). Cultural results of either or both abomasum and rumen yielded a Streptococcus spp. in 4 of 4 animals; Escherichia coli in 3 of 4; and Staphylococcus aureus, Bacillus sp. Enterococcus sp, Enterobacter spp. in 1 of 4. A Clostridium spp. was cultured only from calf 4; smears and sections of the stomach of calf 4 contained a profuse population predominantly of large gram-positive rods suggesting that Clostridium spp. may have had an etiologic role in this animal. Morphologic examination of smears and histologic sections of calves 1, 2, and 3 revealed a variety of gram-positive and gram-negative rods, many gram-positive cocci (chains and diplococci), relatively few palisading rods, and moderate numbers of large gram-positive rods (Clostridium-like). Packeted organisms (Sarcina spp.) were occasionally observed, but always in small numbers.
There is limited evidence of access to excessive quantities of fermentable substrate in these 4 animals. Calf 4 was known to have been tube fed approximately 4 liters of colostrum when 12 hours of age. The dietary history of calf 3 is obscure. Calf 1 and 2 appear to have received normal quantities of colostrum and milk. Calf 1 received 62-g dextrose intravenously during the course of 6 hours. There was no record of dextrose administration to calves 2, 3, and 4.
The naturally occurring syndrome reported here includes not only forestomach and abomasal gas production and emphysema, but also evidence of concurrent acid production in quantity sufficient to induce histologic lesions characteristic of forestomach acidosis. Thus, the pathogenesis of this complex of signs and lesions appears dependent on exuberant fermentation resulting in large quantities of both gas and acid. On that basis reproduction of the syndrome was attempted by experimentally administering a readily fermentable substrate to neonatal calves that were inoculated with a microbial flora contained in the rumen liquor of a mature cow.
Five healthy colostrum-fed Holstein dairy calves (4 males, 1 female) ranging from 34 kg to 45.5 kg and originating from one farm were subjected to experimental treatment at ages 24–60 hours. All calves (XP-1 through XP-5) were born normally except for the female that required veterinary assistance for delivery (twin with a male). The study was conducted on the farm for a 1-year period. Each calf in the study was housed in a barn stall exposed to ambient temperature. Data gathered before and during experimental treatment included physical examination (general attitude, appetite, presence of sucking reflex, hydration status, mucous membrane color, capillary refill time, rectal temperature, pulse, respiratory rate, and gut sounds), packed cell volume, total plasma protein, and blood glucose. Urine glucose was monitored b when urine was obtainable. To provide these neonatal calves an immediate gastrointestinal flora, they were initially dosed by esophageal intubation with 200-ml rumen fluid obtained from a healthy, fistulated cow fed a strict roughage (hay) diet. Dosing with ruminal fluid was followed by a rinse of 200-ml 5% glucose solution. Approximately 4 hours later, substrate administration either by nipple bottle or by esophageal feeder commenced. The 5 calves, XP-1-XP5, each received a mixture consisting of 250-g dry milk replacer, 70-g corn starch, c 180-g glucose, and warm tap water to a final volume of 2 liters. Calves received 4 to 11 doses of substrate at 8–12-hour intervals for a period of 22130 hours. After natural death or euthanasia, necropsy was performed, and microbiologic examinations regularly included culture of rumen and/or abomasal content or wall; examination of Gram-stained smears of mucosal surfaces of rumen and abomasum; and HE, Gram, Steiner, and Periodic Acid Schiff-stained histologic sections of fore-stomachs and abomasums.
Clinical signs were generally mild and inconsistently included diarrhea, mild abdominal distention with fluid and gas, splashiness on abdominal succussion, and mild depression. Hyperglycemia (189-516 mg/dl) accompanied by glucosuria (1,000-2,000 mg/dl) consistently developed. Signs in severely affected animals (XP-1 and XP-4), in addition to the above, were perceptible dehydration, colic, prominent abdominal distention, diarrhea, and recumbence. Calves XP-1 and XP-2 were clinically ill 20–24 hours after initial dosing. Calf XP-1 was critically ill 30 hours and 34 hours postdosing and died shortly thereafter. Calf XP-4 was euthanized and necropsied 3–4 hours after the onset of clinical illness. Calves XP-2, XP-3, and XP-5 were subjected to euthanasia and necropsy 96, 144, and 96 hours postdosing, respectively.
Gross lesions in calves XP-1, XP4, and XP-5 included hemorrhage, edema, and necrosis of abomasal and ruminal mucosae. Emphysematous bullae were present in the stomach walls of calves XP-1 and XP-4. The pH of rumen content of calves XP-1, XP-3, XP-4, and XP-5 ranged from 4.3 to 4.6: abomasal pH ranged from 2.4 to 4.3. Histopathologic lesions in the affected calves corresponded to gross lesions and included hemorrhage, submucosal edema, emphysema, inflammation, and erosion of the mucosa. Hydropic degeneration, vesiculation, and pustule formation were present in forestomach epithelium of calves XP-1, XP-4, and XP-5.
Alpha Streptococci, other Streptococcal spp., and E. coli were rather consistently obtained. Anaerobic cultures failed to yield positive results. Morphologic types of organisms recognizable in smears and in histologic sections included both gram-positive and gram-negative rods and cocci and a few large diplococci. Yeast-like organisms were occasionally noted. There were numerous segmented hyphae compatible with Candida spp. in calf XP-5. Gram-positive rods compatible with Clostridium spp. were numerous in preparations from calves XP-1, XP-3, and XP-4.
This syndrome clinically characterized by acute onset and course, abdominal distention, pain, shock, and death and at necropsy by abomasal tympany, forestomach and abomasal edema, hemorrhage, mucosal necrosis, and often mural emphysema occurs sporadically, predominantly in suckling calves and other young ruminants. 17 The apparent basis for the development of the signs and lesions is the production of gas and, as has been revealed in the present study, the concurrent production of acid quantities sufficient to induce damage to forestomach mucosa. The production of acid and gas in stomachs strongly implies that the underlying pathogenetic basis of this complex of syndromes is excess fermentation of readily fermentable contents. Such a process is further implied by the frequent occurrence of abomasal tympany/bloat in young ruminants nourished by ad lib feeding systems (potential overfeeding), 3 by consumption of large quantities of diet at a feeding, by nursing high milk production dams, by consumption of high starch milk replacers, 6 by prevention of disease achieved by adding 0.05% formalin to the feed mixture, 7 and by feeding cooler rather than warmer milk replacer. 12 An anecdotal report indicates a relationship between stomach distention and mural emphysema of stomachs and urinary bladder in calves and the existence of hyperglycemia following the oral or parenteral administration of glucose solutions (King JM, Cornell University, College of Veterinary Medicine, personal communication 2006). Additionally, the experimental evidence described in this article help to substantiate the premise that this complex is due to exuberant fermentation of carbohydrate. Some reports associate the syndromes with various Clostridial sp., 4,8,11,13,15,16 and there are reports of a similar syndrome in calves, sheep, and goats associated with Sarcina spp. 3,18 C. perfringens type A experimentally instilled intraruminally to neonatal calves induced an acute abdominal syndrome with abomasal and ruminal tympany and inflammation. 14 In neither spontaneous nor experimentally induced cases was a specific microbial etiology able to be identified with confidence. There was evidence for a role for Clostridium spp. in one of the experimental cases, but in others evidence was limited to modest numbers of morphologically Clostridium-compatible organisms in smears and histologic sections of forestomachs and abomasal mucosa.
This would suggest that the source of fermentative enzymes (microbial) for induction of the syndromes is not microbial species specific. Rather it is suggested that the requirements for disease are: 1) there be a dietary or therapeutic provision of excessive quantities of a highly fermentable substrate and (2) a fermentative enzyme appropriate to the available fermentable substrate be present in a concentration capable of sustaining rapid fermentation. Many Clostridium spp., Sarcina spp., Lactobacillus spp., and other microbes have the potential to provide those enzymes. Disease prevention, then, would more appropriately be by dietary management than by prophylactic or therapeutic medication.
Additional studies are needed to clarify interrelationships between the clinicopathologic and etiologic factors in this complex of similar syndromes. Nevertheless, the observation of 4 naturally occurring cases and success in experimentally reproducing a similar disease characterized by stomach tympany, mural emphysema, and forestomach acidosis strongly supports exuberant fermentation as a major pathogenetic mechanism in the development of these closely related syndromes.
Footnotes
a.
Sleepaway, Fort Dodge Animal Health, Fort Dodge, IA.
b.
Diascreen 10, Hypoguard, Minneapolis, MN.
c.
Argo, ACH Food Companies, Inc., Memphis, TN.
