Abstract
To detect bovine antibody directed to smooth lipopolysaccharide (LPS), cell lysate (LYS), O-polysaccharide (OPS), and LPS-deprived chromatographic fractions (ChF) of Brucella abortus, 2 bi-antigenic diagnostic models based on the enzymatic rapid immunofiltration assay (ERIFA), ERIFALPS/LYS and ERIFAOPS/ChF, were developed. Their diagnostic performance was compared with complement fixation test (CFT), Rose Bengal test (RBT), indirect in-house and commercial enzyme-linked immunosorbent assays (iELISA and com-ELISA, respectively), based on the smooth LPS antigen, by using a total of 420 cattle sera collected from aborted-unvaccinated, aborted-unvaccinated and culture-positive, healthy-unvaccinated, and healthy-vaccinated cattle. The results demonstrated excellent agreement and no statistical difference between iELISAs and LPS-, LYS-, OPS-based ERIFA models. However, diagnostic performance of CFT, RBT, and ChF-based ERIFA was less significant than that of LPS-, LYS-, and OPS-based ERIFA models, and iELISAs. The results demonstrated a successful adaptation of the multi-antigenic ERIFA model to anti–B. abortus antibody in bovine sera and suggest that the ERIFA model can be considered as an “individual rapid ELISA” due to its similarity with ELISA, individual applicability, and rapidity in determining reactor animals within 5 minutes. In conclusion, the potential of multi-antigenic applications can make the rapid ERIFA model not only an alternative screening method but also a confirmatory test for bovine brucellosis diagnosis.
Introduction
Brucellosis, characterized by abortions and reproductive failures causing heavy economic losses in animals and undulant fever with severe complications in human beings, may be diagnosed by bacterial isolation, serologic methods, and molecular-based assays such as polymerase chain reaction (PCR). 3,5,22 Although conventional and IS711 real-time PCR assays were found to be highly sensitive methods for the detection of Brucella spp. in comparison with bacteriologic isolation from tissues and blood samples, 16,17 no advantages of these methods over standard serologic and bacteriologic methods have been reported. 26 Moreover, the nucleic acid–based methods need adequate infrastructure and professional expertise, which are not yet present in many countries.
Rose Bengal test (RBT), buffered antigen plate agglutination, complement fixation test (CFT), indirect in-house and competitive enzyme-linked immunosorbent assays (iELISA and cELISA, respectively), immunoblotting, and fluorescence polarization assay (FPA) are widely used methods to detect serum antibodies for bovine brucellosis diagnosis. 8,18,23,24,32,33 Although CFT has been considered as the reference test by the World Organization for Animal Health (OIE) 33 for the detection of reactor animals via titration of the Brucella antibody, this reference technique can only be applied in specialized laboratories. In addition, CFT needs to be standardized by using standard reagents to circumvent important interassay variability, and it is also time consuming and cumbersome for testing multiple samples. Currently, ELISA methods under indirect and competitive formats are largely used to screen for the presence of Brucella-specific antibody in both blood and milk. 21–23,29,32
Nevertheless, brucellosis surveillance with laboratory-based diagnostic techniques such as CFT, FPA, and ELISA is very limited in the majority of the developing countries and rural areas. For that reason, the development of new diagnostic tools with high specificity and sensitivity is still needed for control of the disease. In this regard, the lateral flow assay (LFA) has been recently adapted to brucellosis screening in different livestock species, and its usefulness as a rapid test has been demonstrated. 1 Alternatively, the rapid immunofiltration assay (RIFA) is an advantageous technique for developing new diagnostic tools because of its simplicity, rapidity, high sensitivity and specificity, and adaptability to the diagnosis of different infectious disease agents. 6,9,25,27,34 The present study describes the development of 2 bi-antigenic rapid and sensitive assays for anti-Brucella antibody screening, based on the enzymatic format of RIFA (ERIFA).
Materials and methods
Field and reference serum samples and antibodies
The field sera used in the present study were categorized into 4 groups designated as I–IV. Group I was composed of sera from 109 unvaccinated and aborted cattle obtained from 6 different localities in the Kars district (Turkey), where high brucellosis seroprevalence and abortion rate, as well as the isolation of Brucella abortus biotypes 1 and 3, have been reported. 12,13,29 The sera, which were evaluated by CFT, RBT, a iELISA, and a commercial ELISA (com-ELISA), b were considered as confirmed B. abortus–positive sera. Group II consisted of sera from 18 additional aborted cattle, from the same herds as group I samples and from which B. abortus biotype 3 strains were isolated. Group III sera were collected from 59 healthy animals, without a history of brucellosis, from 3 different locations in which the animals had been injected with the B. abortus S19 strain a vaccine (3–12 × 1010 colony forming units, contained in 4 ml of vaccine diluent, subcutaneously applied). Three serum samples from each animal of group III were collected at day 0 (before vaccination, n = 59) and at days 30 (n = 59) and 120 (n = 55) after vaccination. Group IV was composed of sera from 120 healthy unvaccinated cattle from areas of the Black Sea region (Turkey) without a history of brucellosis. Brucella strong positive (SP) and weak positive (WP) reference sera c and fetal bovine serum (FBS) d were also included in all experiments as additional positive and negative controls, respectively. Mouse monoclonal anti-Brucella LPS antibody (LPS-mAb, immunoglobulin G [IgG]2a, clone 4B5A) e and monospecific anti-A and M antigenic determinants of Brucella LPS sera a were also used as reference antibodies.
Bacteriologic procedures
Culture and identification of Brucella spp. was performed as previously described, 3 from aborted fetuses, vaginal swabs, and milk and/or blood samples of aborted and pregnant animals. Samples were plated onto Farrell medium f and Brucella, f blood, f and serum dextrose agar f and incubated at 37°C under aerobic conditions in the presence of 5–10% CO2 for 5–7 days. The isolates with typical S colonies of Brucella spp. were screened for Tbilisi phage sensitivity, oxidase, f and urease activity f with Gram staining. In addition, species and biovar identification were performed according to CO2 requirement, production of H2S, growth in the presence of basic fuchsin and thionin, and slide agglutination test with monospecific anti-A and anti-M antigenic determinants of Brucella LPS sera. a Brucella abortus S19 vaccine strain a was cultured under aerobic conditions in brain heart broth g for 3 days at 37°C.
Brucella antigens
The following 4 different antigens were used: 1) smooth lipopolysaccharide (sLPS) from B. abortus 2308 strain, h 2) O polysaccharide (OPS) from B. abortus S1119-3 strain, i 3) cell lysate (LYS), and 4) chromatographic fractions of cell lysate (ChF), prepared from B. abortus S19 vaccine strain. a Briefly, freshly cultivated and formaldehyde-inactivated bacteria were heated at 95°C for 15 min in lysis buffer (125 mM Tris-Cl, 150 mM NaCl, 2% sodium dodecyl sulfate [SDS], pH 6.8). The supernatant collected by centrifugation at 25,000 × g for 30 min was dialyzed for 24 hr against deionized water and used as LYS antigen. To prepare the ChF antigen, the separation of LYS antigen was carried out by gel filtration chromatography on an S100 column j with a fast protein liquid chromatography system j at the flow rate of 0.4 ml/min. One milliliter of fractions was collected and evaluated for their immunoreactivity with reference sera and for LPS content with LPS monoclonal antibody. e Sodium dodecyl sulfate–polyacrylamide gel electrophoresis (SDS-PAGE) analysis of immunoreactive and LPS-deprived fractions showed that it essentially constituted of small molecules of 11–30 kDa. The ChF fractions were used as the LPS-deprived Brucella antigen.
Immunologic test procedures
A com-ELISA b kit was used for the detection of Brucella LPS antibodies following the manufacturer's instructions. Samples were tested in duplicate, and any sample yielding an optical density (OD) >α was considered as positive; the value of α was calculated using the formula α = 0.6 × OD value of positive control, as prescribed by the manufacturer. The iELISA was performed as previously described. 29 Briefly, 100 μl of sera diluted 1:200 were added into microwells coated with 100 μl of LPS antigen h (5 μg/ ml) incubated overnight at 4°C, then blocked with 100 μl of 1% cold water fish gelatin k in phosphate buffered saline and Tween 20 (PBST/FG) and incubated for 1 hr at 37°C. Alkaline phosphatase (AP) conjugated rabbit anti-bovine IgG k was added and incubated for 1 hr at 37°C. As substrate, 100 μl of p-nitrophenyl phosphate k was added and incubated at 37°C. The OD was measured at 405 nm in an ELISA reader. l The positive reference controls, WP and SP sera, and the negative control, FBS, at 1:200 dilutions were included in each plate. All sera were tested in duplicate. A total of 145 well-known sera (18 samples from aborted, unvaccinated, and B. abortus biotype 3 culture-positive animals of group II, 48 samples from unvaccinated and non-brucellosis history herds [before vaccination at day 0, group III], and a total of 79 B. abortus S19–vaccinated cattle samples [45 from 1-month postvaccination and 34 from 4-months postvaccination cattle of group III]) were used to determine optimal cutoff value and area under the curve (AUC) of the iELISA. The samples, pre-evaluated with RBT, CFT, and com-ELISA, were considered as confirmed positive sera and were included in receiver operating characteristics (ROC) analysis. In this respect, the sera from 18 naturally infected animals of group II and 79 sera from postvaccinated cattle of group III were used as “seropositive samples,” while 48 sera from group III cattle before vaccination were treated as “seronegative samples.” Optimal cutoff value and AUC of the iELISA were determined as 0.600% and 99.7%, respectively. Its sensitivity and specificity were calculated as 96.9% and 100%, respectively.
The RBT was applied according to a method described elsewhere. 3 Briefly, 1 drop (25 μl) of the serum was mixed with an equal volume of RBT-stained B. abortus S99 cells a on a white porcelain plate. The plate was slowly agitated for 4 min, and the reaction was scored positive if distinct agglutination occurred. The CFT was performed according to the procedure prescribed by OIE, 33 results were considered as positive when the titer was equivalent to 20 international complement fixation test units (ICFTU)/ml or more.
Enzymatic rapid immunofiltration assay
The ERIFA model developed for brucellosis diagnosis was essentially the same described earlier for diagnosing Mycoplasma gallisepticum infection in chickens. 6 The model was prepared with a large porous nitro-cellulose (NC) membrane m included in an individual plastic cassette. The NC membrane was placed onto a high-capacity absorbent pad m that was fixed into the testing units for capturing all reagents used during the assay. Five hundred nanoliters of 2 of the 4 Brucella antigens were separately coated onto the NC membrane of the same test unit to constitute 2 test formats (ERIFALPS/LYS or ERIFAOPS/ChF). Each test unit contained an additional internal control dot of 0.2 μl of purified bovine IgG e (1 mg/ml). The NC membrane was blocked with 100 μl of 1% PBST/FG, which was also used as wash solution and reagent diluent. Serum samples (50 μl, 1:50 dilution) were added and flowed through the membrane and then washed with 50 μl of PBST/FG. No incubation of each reagent was needed because their flowing time (approximately 15–20 sec) through the NC membrane enables to replace all incubation steps. Fifty microliters of AP-conjugated rabbit anti-bovine IgG e were added. The membrane was washed 4 times with PBST/FG, and then 50 μl of 5-bromo-4-chloro-3-indolyl-phosphate and nitroblue tetrazolium (BCIP/NBT) membrane substrate e were added. Color development was stopped after 1 min, and the results were evaluated by their intensity and visualization time. The cassettes presenting the test results were photographed.
Statistical analysis
The optimal positive cutoff value and the AUC for accuracy of iELISA were determined by ROC analysis. The degree of agreement of the serologic test results was assessed by determining the kappa value (κ). The diagnostic efficacy of ERIFA in terms of sensitivity and specificity was estimated by using the 2-sided contingency table. The statistical significance of differences between the tests was examined with the McNemar test to calculate exact probability P value. A P value less than 0.05 was considered as a significant difference between the tests. Win Episcope version 2.0, n Open Epi version 2.3, o and MedCalc p software programs were used for statistical evaluations. Diagnostic accuracy of the tests was calculated as described in the notes to Tables 1 and 2.
Results
As identical results were obtained with separately coated LPS and LYS antigens in one of the bi-antigenic ERIFA format in all tests, these results were presented in the text and tables as ERIFALPS/LYS. Concerning the bi-antigenic ERIFAOPS/ChF model where OPS and ChF were also separately coated in the same test unit, the results were separately presented as ERIFAOPS and ERIFAChF because of the differences observed between their results. The bi-antigenic ERIFALPS/LYS and ERIFAOPS/ChF prototypes were first evaluated with LPS-MAb, reference sera (WP, SP, and FBS), and 20 group I sera from aborted cattle. Among these 20 sera, 10 samples were seropositive and 10 were seronegative by RBT, CFT, iELISA, and com-ELISA for brucellosis. The 10 brucellosis-negative samples had been determined to be Leptospira antibody–positive using the microscopic agglutination test and ELISA. 13
As seen in representative results of ERIFA in Figure 1A, only the ChF antigen was not recognized by LPS-MAb and confirmed negative sera (N1–N4) did not react with any of the antigens (Fig. 1B). In contrast, the reference (WP and SP) and confirmed positive (P1–P4) sera from group I cattle reacted with all antigens including ChF (Fig. 1B). Similarly, 10 serologically brucellosis-positive samples were positive and 10 others were negative by ERIFA with all antigens. Following these results, all 109 samples from group I were tested with RBT, com-ELISA kit, iELISA, and ERIFA models in comparison with CFT as reference test.
The results presented in Table 1 showed moderate agreement of RBT (κ = 0.624) and ChF-based ERIFA (κ = 0.778), and a good agreement of ERIFAOPS (κ = 0.890) in relation to CFT, while almost perfect agreement was observed between CFT and com-ELISA, iELISA, and LPS/LYS-based ERIFA (κ > 0.900). Nine out of 30 CFT-WP sera (20 ICFTU/ml) from group I were negative with ERIFAChF. Among these, only 1 sample in ERIFALPS/LYS and 3 samples in ERIFAOPS were negative. Seventeen out of 87 CFT-positive sera were negative with RBT. The pairwise comparison showed no significant differences between CFT and ERIFALPS/LYS, ERIFAOPS, iELISA, or com-ELISA. However, the difference between CFT and RBT and between CFT and ERIFAChF was significant (P < 0.05).
Based on these results, diagnostic performance of the ERIFA models was evaluated in comparison with different diagnostic tests by using a large number of bovine sera from groups II (culture-positive aborted cattle), III (healthy vaccinated cattle), and IV (healthy unvaccinated cattle; Table 3). Eighteen cattle of group II from which B. abortus biovar 3 had been isolated were seropositive with all diagnostic tests, except for 1 group-II sample that was negative with ERIFAChF. All 120 samples from healthy animals of group IV and 59 samples from prevaccinated cattle (group III) were seronegative by all test methods. Among 59 sera from cattle 30 days postvaccination (group III), only 1 sample was detected as nonreactive with RBT, com-ELISA, and ERIFAOPS. In the same group, while all sera were reactive in ERIFALPS/LYS, nonreactivity was observed in CFT and ERIFAChF for 7 and 9 samples, respectively. An increased number of discordant results between diagnostic tests were observed with the sera collected at day 120 postvaccination, showing a significant decrease in Brucella antibody levels. Among 55 samples from group III (day 120), 50, 48, 47, 46, 40, 39, and 38 sera were found positive with iELISA, ERIFALPS/LYS, ERIFAOPS, com-ELISA, ERIFAChF, RBT, and CFT, respectively (Table 3).
Preliminary performance evaluation (sensitivity, specificity, and diagnostic accuracy) and statistical analysis of different diagnostic tests in comparison with the complement fixation test (CFT) using the sera of aborted unvaccinated cattle (group I) obtained from brucellosis endemic areas.
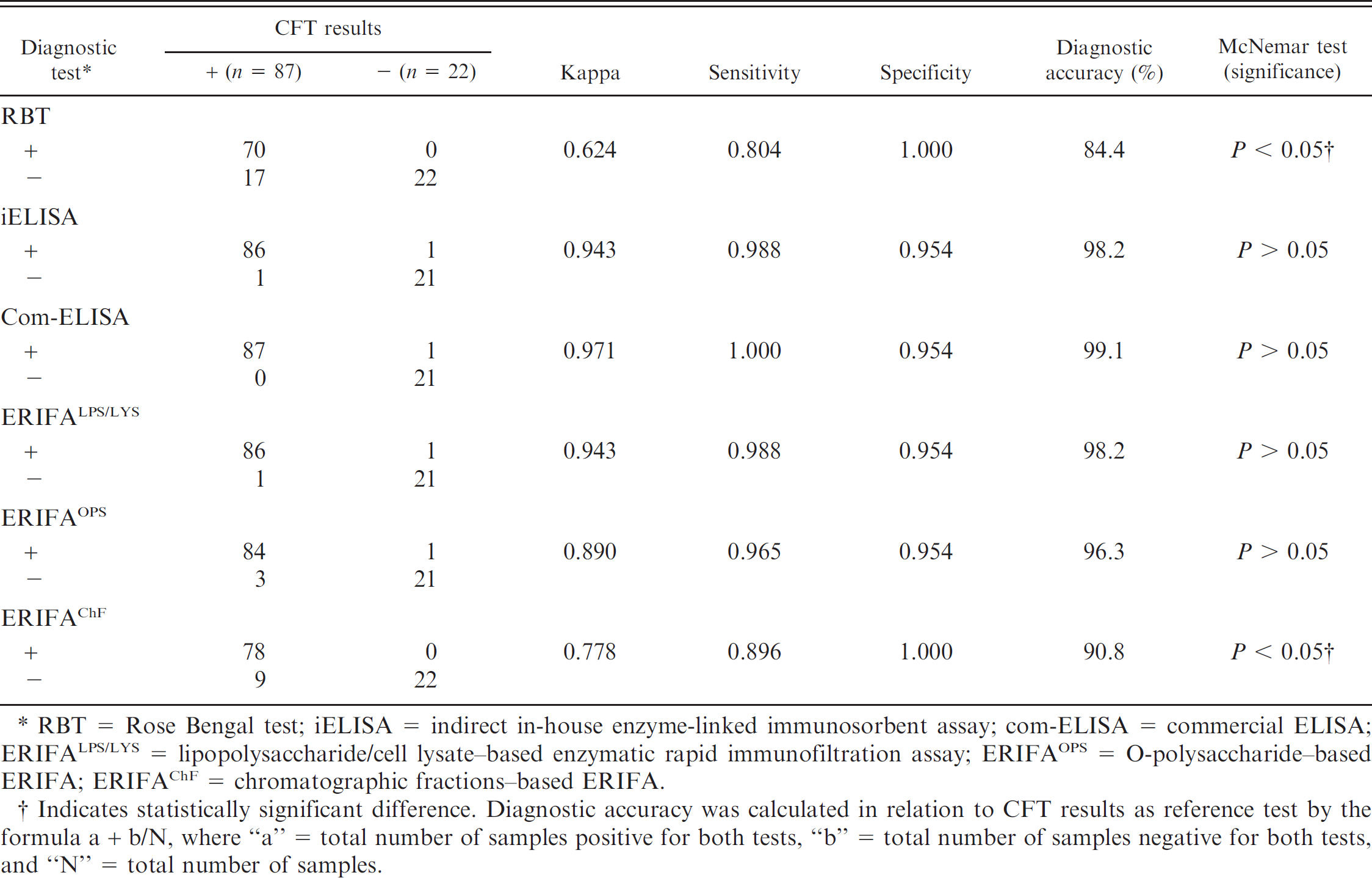
RBT = Rose Bengal test; iELISA = indirect in-house enzyme-linked immunosorbent assay; com-ELISA = commercial ELISA; ERIFALPS/LYS = lipopolysaccharide/cell lysate–based enzymatic rapid immunofiltration assay; ERIFAOPS = O-polysaccharide–based ERIFA; ERIFAChF = chromatographic fractions–based ERIFA.
Indicates statistically significant difference. Diagnostic accuracy was calculated in relation to CFT results as reference test by the formula a + b/N, where “a” = total number of samples positive for both tests, “b” = total number of samples negative for both tests, and “N” = total number of samples.
Representative results of the ERIFA methods presented in Figure 2 showed no reaction of the serum samples from prevaccinated cattle of group III (day 0) against 4 antigens, while the internal control worked correctly. In contrast, antibody response against all antigens was clearly detected with the sera of the same animals at day 30 postvaccination except that of sample 2 against ChF antigen. The decrease and/or no detection of antibody response with the sera collected from the same cattle at day 120 postvaccination was especially observed with OPS and ChF (Fig. 2, day 120). Two representative sera from group-II cattle (naturally infected) showed strong reactivity to LPS, LYS, and OPS antigens and moderate reactivity against ChF.
Based on the overall results, a comparative analysis between LPS/LYS-based ERIFA as a highly sensitive rapid assay and the other diagnostic tests is presented in Table 2. The highest agreement and diagnostic accuracy of the ERIFALPS/LYS were observed with iELISA and ERIFAOPS (κ = 0.981 and 99.0%), followed by com-ELISA (κ = 0.962 and 98.1%), CFT (κ = 0.900 and 95.0%), ERIFAChF (κ = 0.876 and 93.8%), and RBT (κ = 0.872 and 93.6%). The sensitivity of ERIFALPS/LYS in relation to iELISA, ERIFAOPS, com-ELISA, CFT, ERIFAChF, and RBT was found to be 99.5%, 98.1%, 97.6%, 91.0%, 87.7%, and 87.3%, respectively. Relative specificity was determined as 100% with respect to RBT, ERIFAOPS, and ERIFAChF, and in decreasing order with CFT (99.0%) and with iELISA and com-ELISA (98.6%). The pairwise comparison based on the overall results showed no significant differences (P > 0.05) between ERIFALPS/LYS and iELISA, ERIFAOPS, and com-ELISA. However, ERIFALPS/LYS was found to be significantly different from ERIFAChF, CFT, and RBT (P < 0.05).
Comparative performance analysis of lipopolysaccharide/cell lysate–based enzymatic rapid immunofiltration assay (ERIFALPS/LYS) model with different diagnostic tests based on the overall results of the serum samples from groups I–IV cattle. *
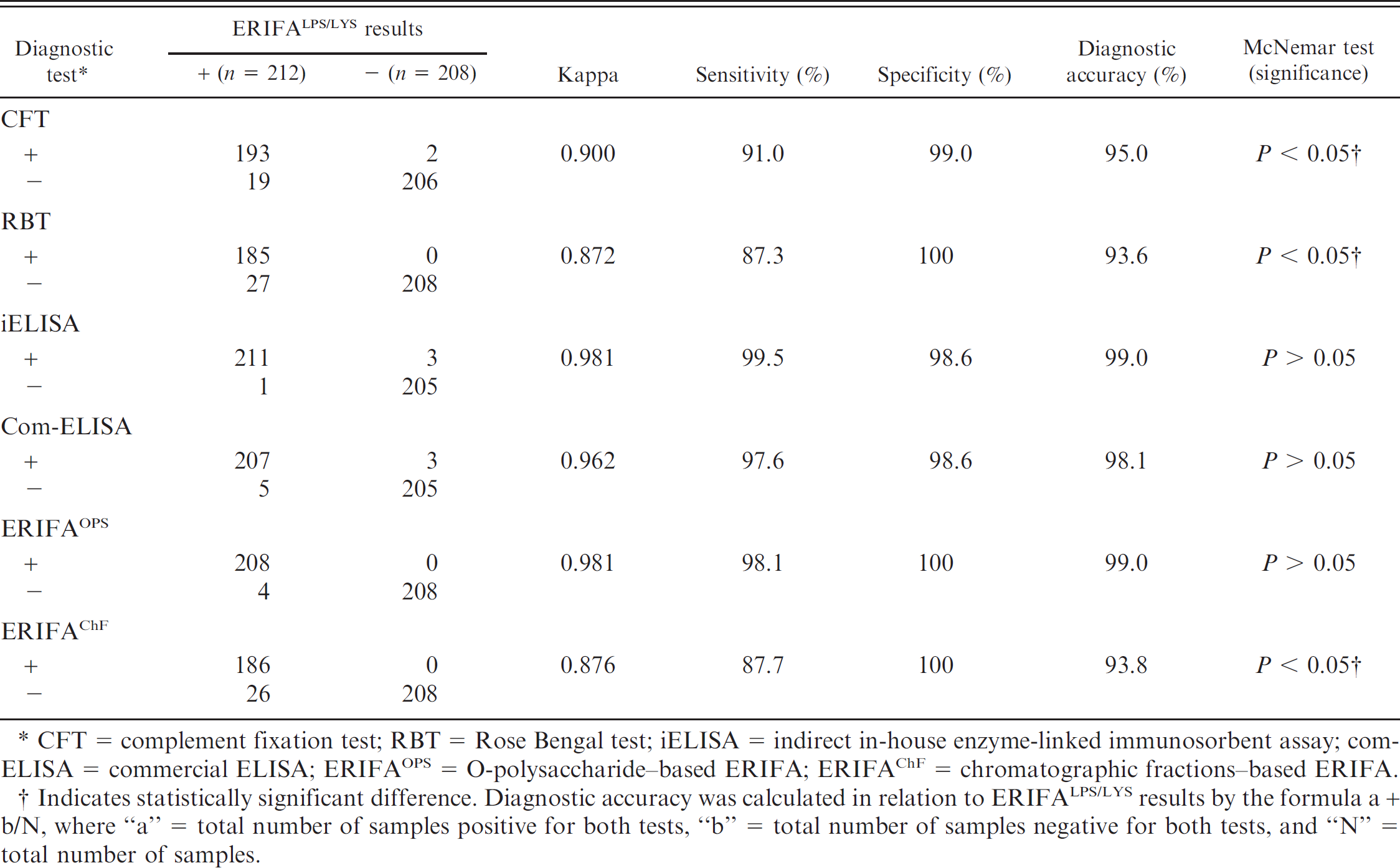
CFT = complement fixation test; RBT = Rose Bengal test; iELISA = indirect in-house enzyme-linked immunosorbent assay; com-ELISA = commercial ELISA; ERIFAOPS = O-polysaccharide–based ERIFA; ERIFAChF = chromatographic fractions–based ERIFA.
Indicates statistically significant difference. Diagnostic accuracy was calculated in relation to ERIFALPS/LYS results by the formula a + b/N, where “a” = total number of samples positive for both tests, “b” = total number of samples negative for both tests, and “N” = total number of samples.
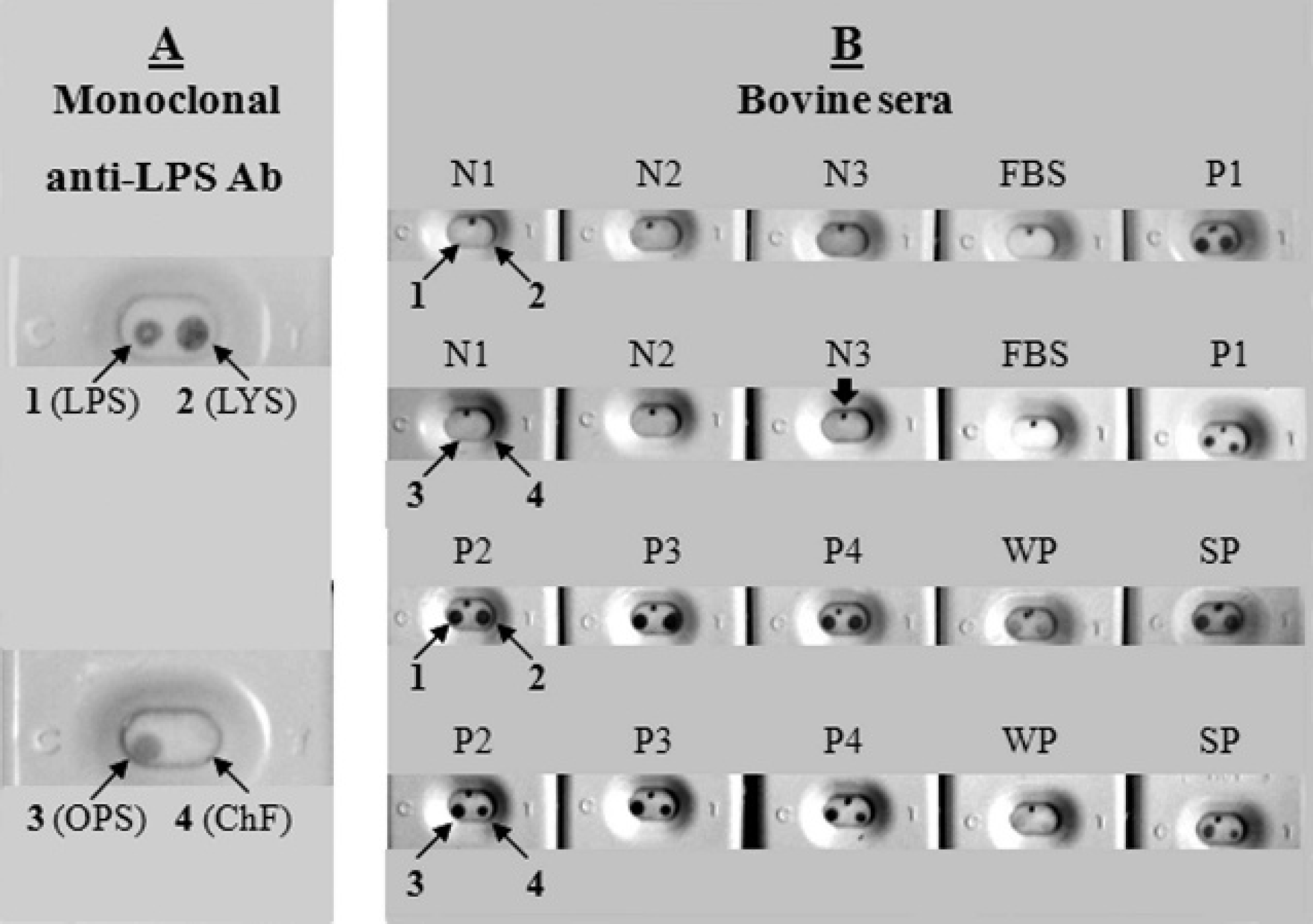
Representative results of 2 bi-antigenic enzymatic rapid immunofiltration assay models by using monoclonal anti-lipopolysaccharide antibody, reference weak and strong positive sera,
c
negative fetal bovine serum,
d
and well-defined field sera from group I aborted unvaccinated cattle. A, 4 Brucella abortus antigens tested with monoclonal anti-lipopolysaccharide antibody (LPS-mAb) are presented in 2 large cassettes.
Discussion
Being easily applicable for diagnostic purposes and adaptable to the field and limited laboratory conditions, RIFA is a simple technique widely used for the development of sensitive diagnostic tools in human and veterinary medicine. 6,9,25,27,34 Recently, the development of a non-enzymatic immunochromatographic test (lateral flow assay) for brucellosis diagnosis has been described. 1 The present study describes the development of 2 bi-antigenic models based on ERIFALPS/LYS and ERIFAOPS/ChF as alternative rapid tests for detecting Brucella IgG antibodies. Comparative analysis of the overall results (Table 2) demonstrated very high agreement and no significant statistical differences between iELISAs and ERIFA models based on LPS, LYS, and OPS antigens. However, diagnostic performance of LPS-, LYS-, and OPS-based ERIFA models was significantly higher than that of CFT, RBT, and ChF-based ERIFA.
Detection capability of different diagnostic tests for Brucella antibody positivity of the sera collected from culture-positive aborted (group II), healthy-vaccinated (group III), and healthy-unvaccinated (group IV) cattle. *
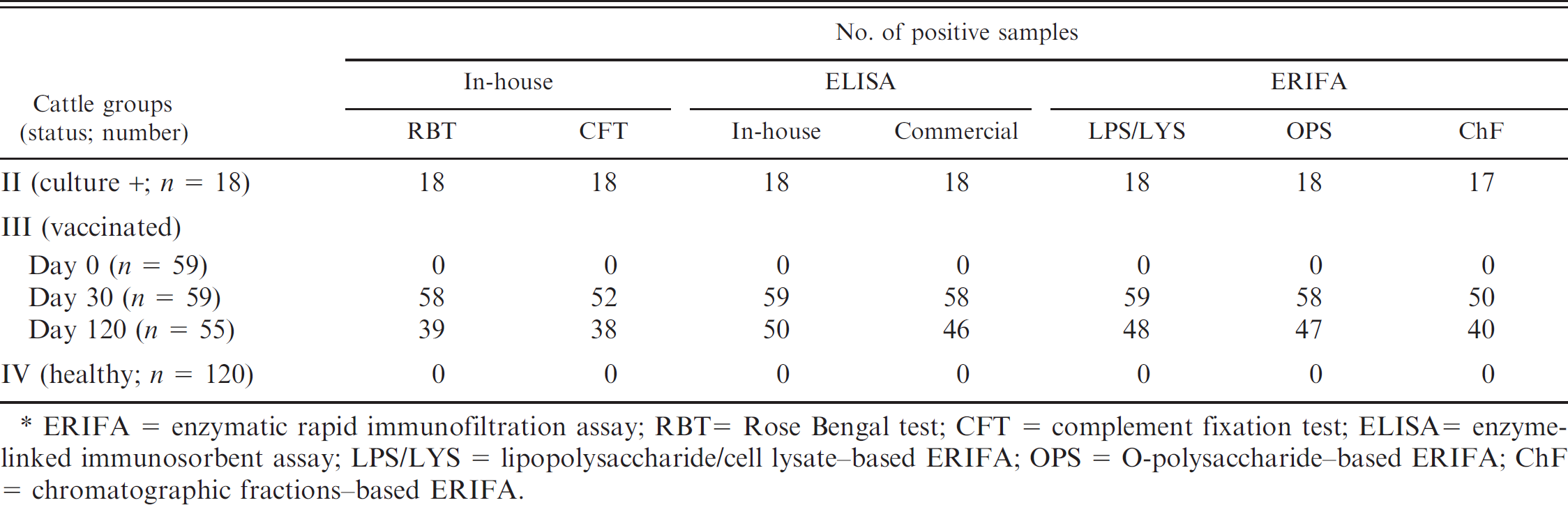
ERIFA = enzymatic rapid immunofiltration assay; RBT = Rose Bengal test; CFT = complement fixation test; ELISA = enzyme-linked immunosorbent assay; LPS/LYS = lipopolysaccharide/cell lysate–based ERIFA; OPS = O-polysaccharide–based ERIFA; ChF = chromatographic fractions–based ERIFA.
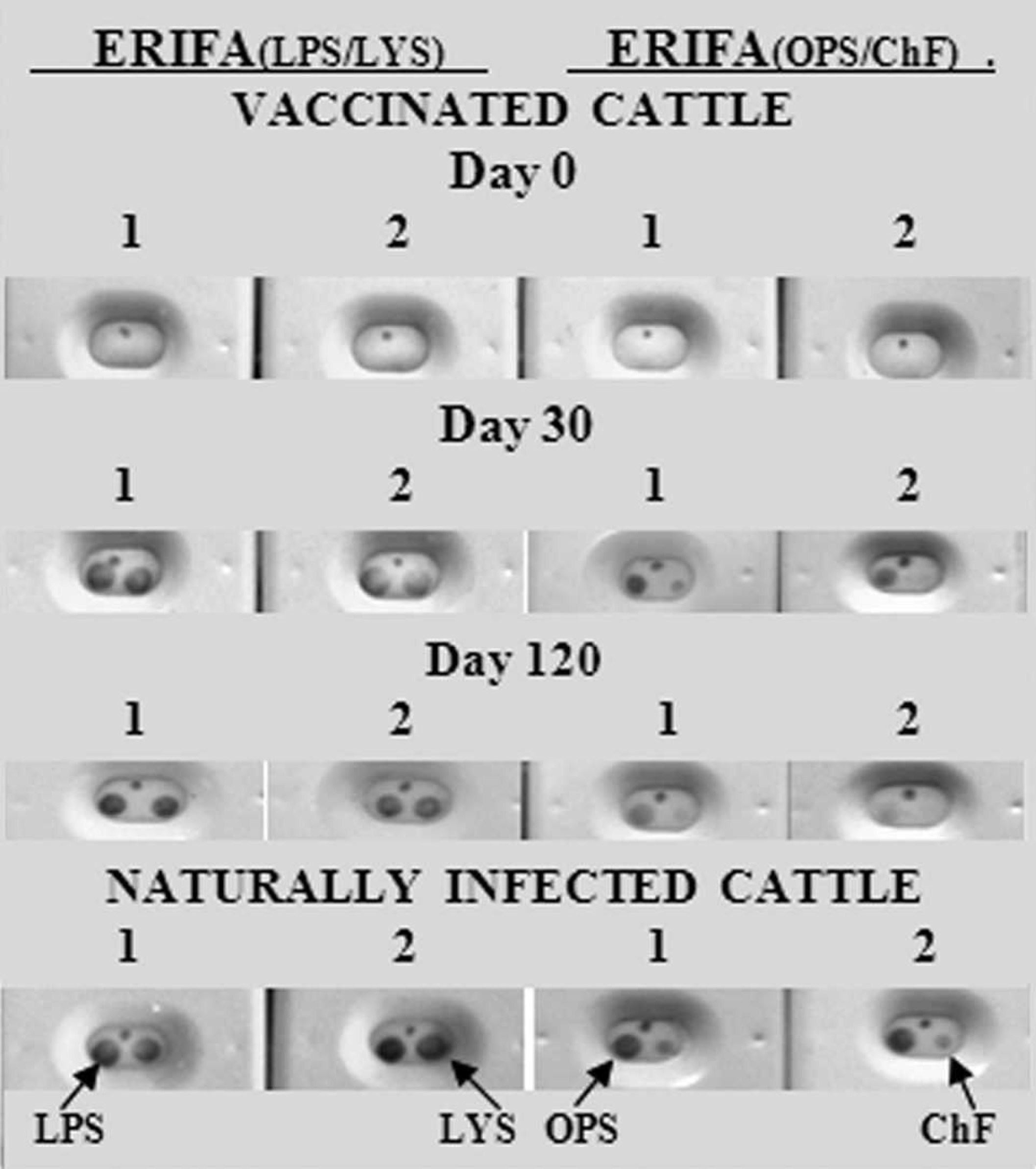
Representative results of lipopolysaccharide/cell lysate (LPS/LYS) and O-polysaccharide/chromatographic fractions (OPS/ChF)-based bi-antigenic enzymatic rapid immunofiltration assay (ERIFA) models by using healthy vaccinated (group III) and naturally infected, culture-positive, aborted (group II) cattle sera. Numbers 1 and 2 indicate sera collected from the same cattle at days 0 (before vaccination, group III) and days 30 and 120 postvaccination, or from naturally infected, culture-positive, aborted cattle (group II). Eight test cassettes on the left side correspond to ERIFALPS/LYS, and the cassettes on the right side correspond to ERIFAOPS/ChF. The arrows indicate location of the antigens.
Although false positivity due to the similarity between LPS of Brucella and Yersinia enterocolitica O:9 strains has been reported, 18 the use of sLPS has been found to be the best in immunoassays, especially in iELISA. 2,22 In addition, better performance of sLPS-based iELISA than the classical RBT/CFT combination was demonstrated to overcome false-positive serologic cross-reactions due to Y. enterocolitica O:9 when it is applied as a unique test in low-prevalence or brucellosis-free areas. 20 Furthermore, close accordance between recent review articles 10,23 based on the accumulated data and overall results obtained in the current study supports higher performance of iELISA methods, especially sLPS-based ones, with respect to CFT and RBT. As an additional diagnostic antigen to LPS in the ERI-FALPS/LYS model, whole cell lysate (LYS antigen) could most likely increase test performance, since it contains not only immunodominant LPS antigen but also several immunogenic molecules such as outer-membrane proteins and cytoplasmic components of Brucella, against which high antibody response occurs. 4,8,14,15,18,19,28 Cost-effectiveness and relatively simple preparation of LPS/LYS antigens also present another advantage of this bi-antigenic model. Furthermore, very high similarity and no statistical difference between iELISAs and LPS-, LYS-, and OPS-based ERIFA models suggest that the ERIFA model can be considered as an “individual rapid ELISA” because of its enzymatic immunoassay–based nature, performance similarity with ELISA methods, individual applicability, and rapidity to determine the reactor animals in less than 5 minutes. Other advantages of the prototype with respect to the proposed field-based and/or adapted ELISA models 11,31 are evaluation of the results by visual inspection and simultaneous detection of the presence of antibodies directed to different natures of Brucella antigens (Figs. 1, 2). It can also be considered as a semi-quantitative test by determining the time and intensity of the color reaction compared with that of the internal control.
The LPS/LYS-based ERIFA model allowing essentially bi-antigenic screening can also be designed in multi-antigenic format by including OPS and/or ChF antigens. Since OPS-based iELISA has been demonstrated as a better primary screening method than the conventional serologic tests, 7 OPS-enriched ERIFA format can be used not only as a sensitive screening assay but also as a differential diagnostic tool between the infected and RB51 vaccinated animals since the vaccine strain is mainly devoid of OPS. 30 However, the antibody response was found at similar levels in naturally infected and B. abortus S19–vaccinated animals at day 30 postvaccination by OPS- and LPS/LYS-based ERIFA (Table 3). At day 120 postvaccination, antibody response was not detected in 14.5% and 12.7% of cattle by ERIFAOPS and ERIFALPS/LYS, respectively. In ChF-based ERIFA, only 1 out of 18 naturally infected animals (5%) was detected as nonreactive, while the percentages of negative sera from B. abortus S19–vaccinated cattle were 15% and 27% at days 30 and 120 postvaccination, respectively. In addition, antibody response of vaccinated cattle at days 30 and especially 120 postvaccination against ChF antigen was weaker than that of naturally infected animals as observed in representative results (Fig. 2). These results indicate that LPS-deprived ChF antigen could be considered as somewhat similar to the previously described LPS-free cytoplasmic fraction (CYT), which was found useful for the differential diagnosis between B. abortus 544–infected and S19–vaccinated or Yersinia-infected animals. 4 In addition, SDS-PAGE analysis of ChF antigen showed that it essentially consisted of several small molecules, which suggests that ChF antigen may also contain specific small cytoplasmic and periplasmic proteins, against which the antibody response has been demonstrated in previous studies. 4,7,8,14,15,18,19,28 Among these, p15, p17, p18, BP26, and p39 proteins alone or in combinations were previously determined as promising brucellosis screening and/or differential diagnostic antigens. 4,7,14,15,18,28
Further investigations are therefore needed to determine specific constituents of LPS-deprived ChF antigen and to evaluate its confirmatory and differential diagnostic potential. Nevertheless, previously determined Brucella-specific proteins can also be included in the ERIFA model to develop a multi-antigenic prototype capable of confirming brucellosis status and differentiating naturally infected and B. abortus S19–vaccinated cattle simultaneously. In conclusion, the results obtained in the current study showed successful adaptation of the ERIFA as an alternative multi-antigenic rapid test for bovine brucellosis diagnosis.
Acknowledgements
The authors thank I. Moriyon, K. Nielsen, L. L. Perrett, F. Ferrajoli, and S. İyisan for kindly providing B. abortus LPS, B. abortus OPS, OIE reference sera, monoclonal anti-LPS antibody (Clone 4B5A), and vaccinated cattle sera, respectively.
Footnotes
a.
Pendik Veterinary Control and Research Institute, İstanbul, Turkey.
b.
SERELISA, Synbiotics Europe, Lyon, France.
c.
Veterinary Laboratory Agency (VLA), Weybridge, United Kingdom.
d.
Biochrom AG, Berlin, Germany.
e.
Istituto Zooprofilattico Sperimentale Del'Abruzzo e del Molise “G. Caporale,” Teramo, Italy.
f.
Oxoid Ltd., Basingstoke, Hampshire, England.
g.
Merck KGaA, Darmstadt, Germany.
h.
Kindly provided by I. Moriyon, University of Navarra, Pamplona, Spain.
i.
Kindly provided by K. Nielsen, Canadian Food Inspection Agency, ADRI, Nepean, Ontario, Canada.
j.
Sephacryl S100 column and AktaPrime Plus FPLC system, Amersham Biosciences, Uppsala, Sweden.
k.
Sigma-Aldrich Inc., Saint Louis, MO.
l.
Multiskan EX, Fisher Scientific Worldwide (Shanghai) Co. Ltd., Shanghai, China.
m.
Schleicher & Schuell BioScience GmbH, Dassel, Germany.
