Abstract
Study design
Network Meta-Analysis.
Objective
To comprehensively compare the clinical efficacy and safety of anterior controllable antedisplacement and fusion (ACAF), anterior cervical corpectomy and fusion (ACCF), and laminoplasty (LP) for treating multilevel cervical ossification of the posterior longitudinal ligament (OPLL).
Methods
PubMed, Cochrane Library, Embase, and Web of Science were systematically searched (inception to April 1, 2025) for clinical studies comparing at least two of ACAF, ACCF, or LP for multilevel (≥2 segments) cervical OPLL. Data on surgical parameters, neurological function (Japanese Orthopaedic Association [JOA] score, JOA recovery rate, Visual Analog Scale [VAS]), biomechanics (cervical curvature, Cobb angle, Range of Motion [ROM], Neck Disability Index [NDI]), and complications were extracted. A frequentist network meta-analysis using a multivariate random-effects model was performed. Treatments were ranked using the surface under the cumulative ranking curve (SUCRA).
Results
Thirty-one non-randomized studies involving 2616 patients were included (ACAF: 585; ACCF: 875; LP: 1156). NMA showed ACAF and ACCF achieved significantly better postoperative JOA scores and recovery rates than LP (p < 0.05). ACAF showed the highest probability of being the most effective treatment for postoperative VAS score (vs ACCF & LP, p < 0.05) and maintenance of cervical curvature/Cobb angle (vs ACCF & LP, p < 0.05). LP had the shortest operative time (p < 0.05). Regarding safety, ACAF was associated with the lowest probability of total complications (SUCRA 99.7%), with significantly lower risks of cerebrospinal fluid (CSF) leakage versus ACCF (p < 0.05), and C5 palsy and axial pain versus LP (p < 0.05). LP had the lowest risk of dysphagia (p < 0.05). Subgroup analysis suggested ACAF’s benefits, particularly in neurological outcome, are more pronounced in patients with severe stenosis (occupying ratio ≥60%).
Conclusion
Based on current observational evidence, ACAF appears to be a promising option for multilevel cervical OPLL, particularly for patients with severe stenosis. ACCF provides effective neurological decompression but carries a higher CSF leak risk than ACAF. LP, while having shorter operative times, results in inferior neurological and biomechanical outcomes compared to anterior approaches. Surgical decisions require individualized assessment based on patient and OPLL characteristics.However, these findings should be interpreted with caution due to the predominance of non-randomized studies and potential selection bias.
Keywords
Introduction
Ossification of the posterior longitudinal ligament (OPLL) is a degenerative condition characterized by ectopic calcification of the posterior longitudinal ligament, leading to cervical spinal canal stenosis, subsequent spinal cord compression, and neurological dysfunction. It predominantly affects East Asian populations, with prevalence rates reported as high as 1.920134.3% in some regions. 1 Since its first description by Tsukimoto in 1960, surgery has become the mainstay treatment for myelopathy caused by OPLL. 2 Currently, three surgical techniques are widely employed for multilevel OPLL: anterior controllable antedisplacement and fusion (ACAF), anterior cervical corpectomy and fusion (ACCF), and posterior laminoplasty (LP). 3 ACAF, a relatively newer anterior approach, involves controlled antedisplacement of the vertebral body-disc complex containing the ossified ligament, achieving effective indirect decompression without direct resection of the ossified mass, while simultaneously reconstructing cervical stability and lordosis. 4 ACCF achieves decompression through direct resection of the ossified mass and vertebral bodies, but multilevel procedures can be technically demanding and associated with increased risks of complications like cerebrospinal fluid (CSF) leakage and spinal cord injury. 5 In contrast, LP indirectly decompresses the spinal cord by expanding the spinal canal volume posteriorly; it is a relatively mature and safe technique but may be associated with postoperative loss of cervical range of motion (ROM), lordosis, and axial neck pain. 6
Although several meta-analyses have compared these techniques, most are limited to pairwise comparisons or pool different anterior approaches together, thus failing to fully elucidate the relative merits and demerits of the specific ACAF technique.7,8 Critically, a comprehensive assessment ranking all three major surgical options within a unified framework is lacking. This absence of high-level, comparative evidence hinders optimal surgical decision-making, especially when balancing multiple, often interrelated, outcomes such as operative time, blood loss, neurological recovery (e.g., JOA score, VAS score), biomechanical properties (e.g., cervical curvature, ROM, NDI), and various complication risks (e.g., CSF leakage, C5 palsy, dysphagia). Furthermore, the complex interplay between the number of involved segments, severity of stenosis, patient baseline characteristics, and surgical choice adds another layer of challenge to clinical decision-making.
Therefore, this study aimed to utilize network meta-analysis (NMA) to synthesize direct and indirect evidence from global literature, providing a comprehensive and systematic comparison of the relative efficacy and safety of ACAF, ACCF, and LP for treating multilevel OPLL. We specifically aimed to investigate potential differences in outcomes across clinical subgroups based on stenosis severity and disease extent, thereby offering more robust evidence to guide individualized surgical strategies for patients with OPLL.
Materials and methods
This network meta-analysis was conducted and reported following the Preferred Reporting Items for Systematic Reviews and Meta-Analyses1 for Network Meta-Analyses (PRISMA-NMA) guidelines.
Eligibility criteria
Studies were included if they met the following criteria: (1) Population: Patients with a clear diagnosis of symptomatic myelopathy due to multilevel (defined as ≥2 segments) cervical OPLL requiring surgical intervention. (2) Interventions: Compared at least two of the following surgical techniques: ACAF, ACCF, or LP. (3) Study Design: Randomized controlled trials (RCTs) or non-randomized comparative studies (NRCTs, e.g., prospective or retrospective cohort studies). (4) Outcomes: Reported at least one relevant clinical outcome measure.
Exclusion criteria were: (1) Non-comparative studies (e.g., case series, case reports), reviews, letters, conference abstracts, or non-clinical studies (e.g., cadaveric, pure biomechanical studies); (2) Studies involving non-standardized single procedures or planned hybrid/combined approaches (e.g., anterior combined with posterior); (3) Studies lacking extractable outcome data where data could not be obtained from the authors; (4) Studies with data unsuitable for statistical analysis; (5) Retracted publications.
Search strategy
Comprehensive searches were performed in PubMed, Cochrane Library (CENTRAL), Embase, and Web of Science (Science Citation Index Expanded) from their inception date to April 1, 2025. The search strategy combined controlled vocabulary (e.g., MeSH terms: “Ossification of Posterior Longitudinal Ligament”, “Spinal Fusion”, “Laminoplasty”, “Osteotomy”, “Cervical Vertebrae”) and free-text terms (e.g., “OPLL”, “ACAF”, “ACCF”, “anterior controllable antedisplacement and fusion”, “anterior cervical corpectomy and fusion”, “laminoplasty”, “vertebral body sliding osteotomy”, “cervical spondylotic myelopathy”). Boolean operators “AND” and “OR” were used to combine terms. (Detailed search strategies available in Appendix/Supplementary Material). Reference lists of included studies and relevant reviews were manually screened for potentially eligible studies. Corresponding authors were contacted via email if data clarification or additional information was needed. No language restrictions were initially applied, but only studies published in English or Chinese were included. This restriction was applied due to the high prevalence of OPLL in East Asian populations (where Chinese is a major scientific language) and the research team’s linguistic capabilities, while acknowledging that this may exclude relevant literature in other languages such as Japanese or Korean.
Study selection, data extraction, and quality assessment
Two reviewers (X.CH. and D.L) independently screened titles and abstracts, followed by full-text evaluation against the eligibility criteria. Disagreements were resolved through discussion or consultation with a third reviewer (W.XZ). Data were extracted using a standardized form, including: (1) Study characteristics (first author, publication year, study design, country); (2) Patient baseline characteristics (sample size per group, age, sex, OPLL severity/type, canal occupying ratio, number of involved segments, follow-up duration); (3) Intervention details; (4) Outcome measures: surgical parameters (operative time, estimated blood loss), neurological function (preoperative and final follow-up JOA score, JOA recovery rate, VAS score for neck/arm pain, NDI score), biomechanical parameters (cervical lordosis/curvature, C2-C7 Cobb angle, ROM), and complications (CSF leakage, C5 palsy, axial pain, dysphagia, hematoma, postoperative infection, hoarseness, neurological deterioration, total complications). We defined ‘Cervical Curvature’ based on the cervical curvature index (Ishihara index) or the depth of cervical lordosis (mm), whereas ‘C2-C7 Cobb angle’ referred to the Cobb angle measured in degrees. These were treated as distinct biomechanical indicators. The Newcastle-Ottawa Scale (NOS) 9 was used to assess the quality of included NRCTs based on selection (max 4 stars), comparability (max 2 stars), and outcome assessment (max 3 stars). Studies scoring ≥7 stars were considered high quality. The single included RCT was assessed using the Cochrane Risk of Bias tool (RoB 2). The observational nature of the included studies inherently introduces risks of selection and confounding biases, a critical methodological limitation that will be expanded upon in the Discussion.
Statistical analysis
A frequentist network meta-analysis was performed using the mvmeta package within Stata 18 (StataCorp LLC, College Station, Texas, USA). Network plots were generated to visualize the geometry of available comparisons.For continuous outcomes (e.g., JOA score, operative time), the Mean Difference (MD) and 95% Confidence Interval (CI) were calculated. For dichotomous outcomes (complications), Odds Ratios (OR) and 95% CIs were calculated. We calculated the I2 statistic to quantify heterogeneity. I2 values of 25%, 50%, and 75% were considered low, moderate, and high heterogeneity, respectively. Given the anticipated heterogeneity among studies, a random-effects model was used for all analyses.Overall network consistency was assessed using a design-by-treatment interaction model, with p ≥ 0.10 indicating acceptable consistency. Local inconsistency between direct and indirect evidence for specific comparisons was evaluated using the node-splitting method (P < 0.05 indicating significant inconsistency). If significant inconsistency was detected (as in the case of Cobb angle), sensitivity analyses excluding potentially outlying studies were performed, or results were interpreted with caution.Potential publication bias was assessed by visual inspection of comparison-adjusted funnel plots test where appropriate.The Surface Under the Cumulative Ranking Curve (SUCRA) and mean ranks were calculated to estimate the probability of each treatment being the best (or worst) option for each outcome. SUCRA values range from 0% to 100%, with higher values indicating a higher probability of being ranked closer to the best. Sensitivity analyses were performed for primary outcomes (e.g., JOA score, total complications) by excluding each study individually to assess the robustness of the results. Pre-specified subgroup analyses were conducted based on canal occupying ratio (<60% vs ≥60%) and number of involved segments (<3 vs ≥3) where sufficient data were available. We assessed the certainty of evidence for primary outcomes using the GRADE (Grading of Recommendations Assessment, Development and Evaluation) approach adapted for network meta-analysis. The quality of evidence was categorized as high, moderate, low, or very low. Network meta-regression was not feasible due to insufficient reporting of potential covariates across studies.All statistical tests were two-sided, and a p-value <0.05 was considered statistically significant.
Results
Literature search and study selection
The initial search yielded 1386 potentially relevant citations (1382 from databases, 4 from reference screening). After removing 459 duplicates and 328 irrelevant records, 599 abstracts were screened. Following abstract and subsequent full-text review, 31 studies met the inclusion criteria and were included in the NMA. The study selection process is detailed in the PRISMA flow diagram (Figure 1). PRISMA flow diagram illustrating the study selection process.
Characteristics and quality of included studies
Baseline characteristics of included studies.
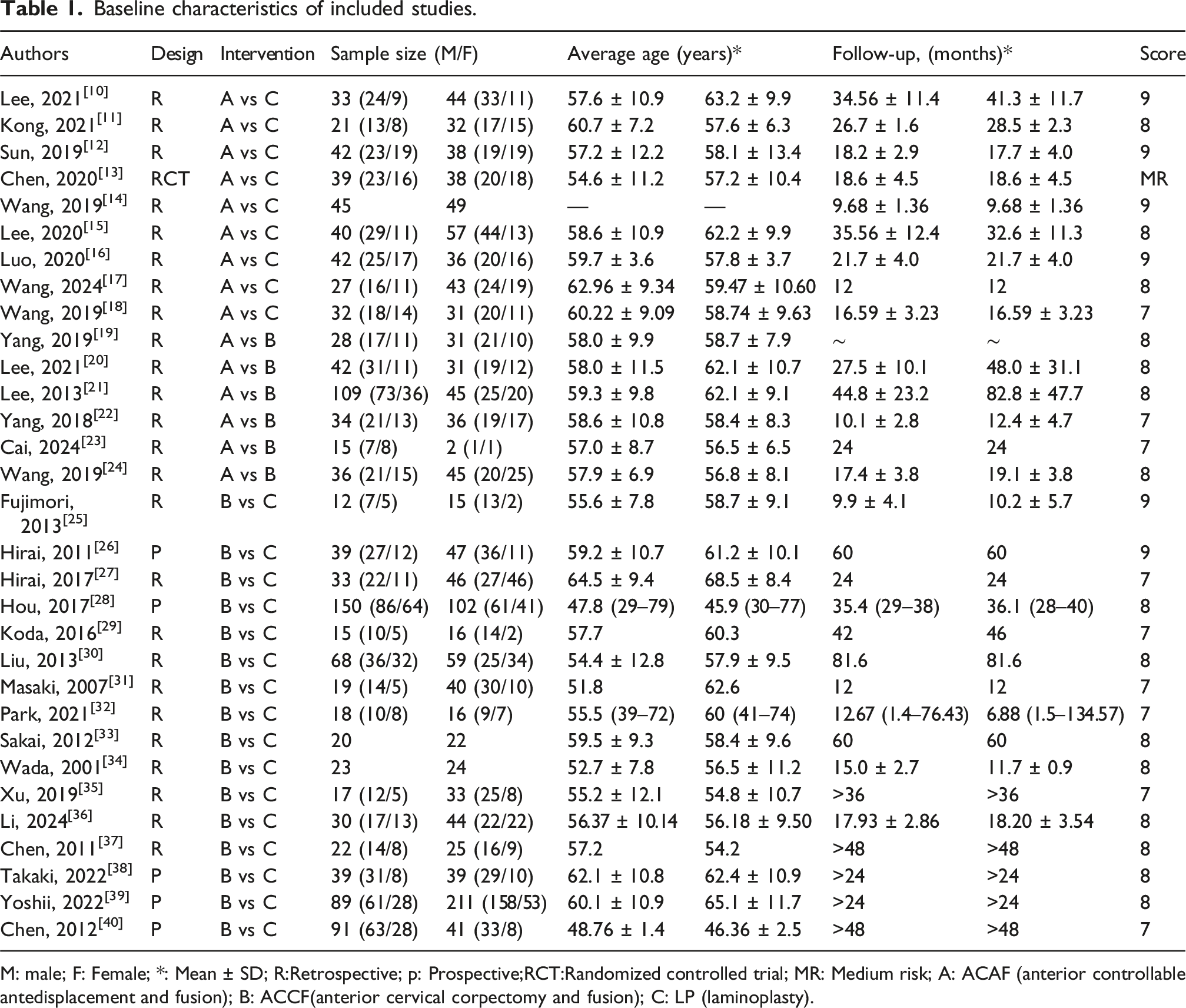
M: male; F: Female; *: Mean ± SD; R:Retrospective; p: Prospective;RCT:Randomized controlled trial; MR: Medium risk; A: ACAF (anterior controllable antedisplacement and fusion); B: ACCF(anterior cervical corpectomy and fusion); C: LP (laminoplasty).
Quality assessment of included non-randomized studies using the Newcastle-Ottawa Scale (NOS).

Network meta-analysis results
The network geometries for different outcomes are shown in Figures 2 and 3. The results of the NMA comparing ACAF, ACCF, and LP are summarized below and detailed in Table 3 and Figures 4 and 5. Global consistency tests did not reveal significant inconsistency for most outcomes (p ≥ 0.10), except for C2-C7 Cobb angle (p < 0.05). Node-splitting analyses did not show significant local inconsistency for most comparisons after sensitivity analysis for Cobb angle. Funnel plots suggested low risk of significant publication bias for most outcomes (Figures 6 and 7). Network plots for primary and secondary clinical/operative outcomes. Nodes represent interventions (ACAF, ACCF, LP), with size proportional to the number of patients. Lines represent direct comparisons, with thickness proportional to the number of studies. (a) Operative Time, (b) Estimated Blood Loss, (c) Postoperative JOA Score, (d) JOA Recovery Rate, (e) Postoperative VAS Score, (f) Postoperative ROM, (g) Postoperative Cervical Curvature, (h) Postoperative C2-C7 Cobb Angle, (i) Postoperative NDI Score. ACAF (anterior controllable antedisplacement and fusion); ACCF (anterior cervical corpectomy and fusion); LP (laminoplasty). Network plots for complications. Nodes represent interventions (ACAF, ACCF, LP), with size proportional to the number of patients. Lines represent direct comparisons, with thickness proportional to the number of studies. (a) CSF Leakage, (b) Axial Pain, (c) C5 Palsy, (d) Dysphagia, (e) Hematoma, (f) Postoperative Infection, (g) Hoarseness, (h) Neurological Deterioration, (i) Total Complications. ACAF (anterior controllable antedisplacement and fusion); ACCF (anterior cervical corpectomy and fusion); LP (laminoplasty). Final index ranking and league table. MD: Mean difference; OR: odds ratio; CI: Confidence interval; I
2
: Heterogeneity index (<25% low heterogeneity、>75%high heterogeneity); ACAF (anterior controllable antedisplacement and fusion); ACCF(anterior cervical corpectomy and fusion); LP (laminoplasty).;SUCRA(Surface under the cumulative ranking curve). Forest plots comparing ACAF, ACCF, and LP for primary and secondary clinical/operative outcomes. Results are presented as Mean Difference (MD) with 95% Confidence Intervals (CI). (a) Operative Time, (b) Estimated Blood Loss, (c) Postoperative JOA Score, (d) JOA Recovery Rate, (e) Postoperative VAS Score, (f) Postoperative ROM, (g) Postoperative Cervical Curvature, (h) Postoperative C2-C7 Cobb Angle, (i) Postoperative NDI Score. ACAF (anterior controllable antedisplacement and fusion); ACCF (anterior cervical corpectomy and fusion); LP (laminoplasty). Forest plots comparing ACAF, ACCF, and LP for complications. Results are presented as Odds Ratios (OR) with 95% Confidence Intervals (CI). (a) CSF Leakage, (b) Axial Pain, (c) C5 Palsy, (d) Dysphagia, (e) Hematoma, (f) Postoperative Infection, (g) Hoarseness, (h) Neurological Deterioration, (i) Total Complications. ACAF (anterior controllable antedisplacement and fusion); ACCF (anterior cervical corpectomy and fusion); LP (laminoplasty). Comparison-adjusted funnel plots for assessment of potential publication bias for primary and secondary clinical/operative outcomes. (a) Operative Time, (b) Estimated Blood Loss, (c) Postoperative JOA Score, (d) JOA Recovery Rate, (e) Postoperative VAS Score, (f) Postoperative ROM, (g) Postoperative Cervical Curvature, (h) Postoperative C2-C7 Cobb Angle, (i) Postoperative NDI Score. (A vs B: ACAF vs ACCF); (A vs C: ACAF vs LP); (B vs C: ACCF vs LP). ACAF (anterior controllable antedisplacement and fusion); ACCF (anterior cervical corpectomy and fusion); LP (laminoplasty). Comparison-adjusted funnel plots for assessment of potential publication bias for complications. (a) CSF Leakage, (b) Axial Pain, (c) C5 Palsy, (d) Dysphagia, (e) Hematoma, (f) Postoperative Infection, (g) Hoarseness, (h) Neurological Deterioration, (i) Total Complications. (A vs B: ACAF vs ACCF); (A vs C: ACAF vs LP); (B vs C: ACCF vs LP). ACAF (anterior controllable antedisplacement and fusion); ACCF (anterior cervical corpectomy and fusion); LP (laminoplasty).






Biomechanical outcomes
Complications
Subgroup analyses
Subgroup analysis results for postoperative JOA score.

MD: Mean difference; OR: odds ratio; CI: Confidence interval; JOA: Japanese orthopaedic Association; ACAF (anterior controllable antedisplacement and fusion); ACCF(anterior cervical corpectomy and fusion); LP (laminoplasty).
aThe subgroup analysis for total complications in patients with <3 involved levels was not performed due to an insufficient number of eligible studies to generate a valid network meta-analysis; SUCRA(Surface under the cumulative ranking curve).
JOA score
By Stenosis Severity: In patients with severe stenosis (occupying ratio ≥60%, n = 9 studies, 708 patients), ACAF yielded significantly higher JOA scores than both ACCF (MD 1.35 [0.08, 2.61]) and LP (MD 1.40 [0.59, 2.21]), ranking highest (SUCRA 98.7%). In patients with less severe stenosis (<60%, n = 9 studies, 622 patients), ACCF ranked highest (SUCRA 83.9%), significantly better than LP, but not significantly different from ACAF.
By Number of Levels: In patients with fewer involved levels (<3, n = 7 studies, 457 patients), ACCF yielded significantly higher JOA scores than ACAF (MD −0.70 [−1.11, −0.29]), ranking highest (SUCRA 96.1%). For ≥3 levels (n = 6 studies, 337 patients), ACAF was significantly better than LP (only comparison available).
Total complications
By Stenosis Severity: No statistically significant differences in total complication rates were found between the groups in either the <60% (n = 6 studies, 409 patients) or ≥60% (n = 7 studies, 636 patients) stenosis subgroups. However, SUCRA rankings consistently favored ACAF as the lowest risk approach in both subgroups (<60% SUCRA 78.4%; ≥60% SUCRA 84.9%).
By Number of Involved Levels: For the subgroup with limited OPLL (<3 involved segments, n = 2 studies, 194 patients), quantitative synthesis was not possible as only two studies provided extractable data, which was insufficient to form a connected network. In the subgroup with >3 involved levels (n = 4 studies, 290 patients), no statistically significant differences were observed in total complication rates among the three procedures (P > 0.05 for all pairwise comparisons). However, based on SUCRA rankings, LP had the highest probability of being the safest treatment (SUCRA 82.0%), followed by ACAF (SUCRA 61.4%). ACCF ranked the lowest (SUCRA 6.6%), suggesting a potentially higher risk of complications in extensive multi-level surgeries, as indicated by an Odds Ratio of 0.44 [95% CI 0.12, 1.67] compared to ACAF, although this did not reach statistical significance.
Discussion
This network meta-analysis provides the most comprehensive comparison to date of ACAF, ACCF, and LP for treating multilevel cervical OPLL, synthesizing evidence on efficacy, biomechanics, and safety. Distinct from previous pairwise meta-analyses, our NMA allows for simultaneous comparison and ranking of these three key surgical strategies. The principal finding is that ACAF showed a favorable profile, ranking highest in probability for neurological recovery, pain relief, maintenance of cervical alignment, and importantly, overall safety with the lowest total complication rate. This advantage appears particularly relevant for patients with severe stenosis (≥60% occupying ratio). However, for patients with fewer involved segments (<3), the traditional ACCF approach may offer comparable or even slightly better neurological outcomes. These findings offer crucial, evidence-based insights to refine individualized surgical decision-making for this challenging condition. It is crucial to interpret the SUCRA rankings with caution. SUCRA represents the probability of a treatment being the best among the compared options but does not quantify the magnitude of clinical difference. A high SUCRA score does not necessarily imply a clinically significant superiority, especially whe`n the underlying evidence is derived from observational studies with wide confidence intervals.
Clinical efficacy
Overall, anterior approaches (ACAF and ACCF) surpassed posterior laminoplasty (LP) in neurological recovery (JOA metrics) and pain alleviation (VAS score), aligning with established anterior decompression mechanisms. ACAF’s distinct controllable antedisplacement technique effectively mitigates cord compression and potentially better restores spinal cord morphology and alignment while preserving some cervical dynamics, which may enhance neurological outcomes.441,42 Importantly, ACAF provided significantly greater pain relief than ACCF, likely because ACAF obviates the need for extensive corpectomy, thus minimizing surgical trauma and local postoperative inflammation.4 43 The efficacy of LP, an indirect decompression method, is constrained by the degree of posterior cord migration and can be suboptimal for K-line (-) OPLL (significant canal compromise).4 44 Our subgroup analyses further elucidated these observations. For patients with a canal occupying ratio ≥60%, ACAF achieved significantly higher JOA scores than ACCF and LP, confirming its effectiveness in severe anterior compression, potentially by more effectively anteriorly displacing the ossified complex for indirect decompression. However, for OPLL involving <3 segments, ACCF led to better JOA scores than ACAF. This implies that for limited segmental disease, standard ACCF direct decompression may suffice and is technically well-established; ACAF, being more complex, may offer greater advantages in more widespread and severe pathology. These findings underscore the necessity of tailoring surgical selection to OPLL severity and extent.
Biomechanical outcomes
In terms of maintaining cervical sagittal alignment, ACAF and ACCF significantly outperformed LP, reflected in superior postoperative cervical curvature and Cobb angle measurements. By anteriorly displacing and fusing the ossified complex, ACAF aids in reconstructing cervical lordosis. Crucially, its motion-preserving design might mitigate the long-term risk of adjacent segment degeneration (ASD). 45 While ACCF also restores alignment via fusion, the requisite extensive corpectomy could negatively impact long-term stability. 46 In contrast, LP frequently leads to postoperative loss of cervical curvature and limited motion, likely attributable to factors such as posterior musculoligamentous complex injury, altered laminar anatomy, and inadequate fixation. 47 Additionally, ACAF showed a favorable trend in postoperative NDI scores over LP, reinforcing its functional recovery benefits. Despite the absence of adequate long-term data in our study to definitively evaluate ASD risk, the demonstrated biomechanical strengths of ACAF imply a potential for enhanced long-term stability.
Complication risks
One of the most significant findings of this study is that Anterior Controllable Antedisplacement and Fusion (ACAF) demonstrated the lowest overall incidence of postoperative complications. Specifically, ACAF was associated with a significantly lower risk of cerebrospinal fluid (CSF) leakage compared to Anterior Cervical Corpectomy and Fusion (ACCF), an advantage likely attributable to ACAF’s avoidance of direct resection in areas where the ossified mass may adhere to the dura mater.19,20 Concurrently, the incidence of C5 nerve root palsy following ACAF was significantly lower than that after Laminoplasty (LP), potentially because the anterior approach avoids direct manipulation or excessive posterior displacement and traction of the nerve roots often encountered in posterior procedures.48,49 ACCF carried the highest risk of CSF leakage, a finding closely related to its technical characteristic of direct ossified mass resection. While LP circumvented anterior approach-related complications such as dysphagia (lowest incidence) and hoarseness, it was associated with a relatively higher incidence of axial symptoms and C5 nerve root palsy. Notably, the incidence of dysphagia after ACAF was higher than LP but not statistically different from ACCF, possibly due to anterior exposure, tissue edema, and implant-related irritation. 50 No significant differences were observed among the three techniques regarding the risks of postoperative infection, hematoma formation, or immediate postoperative neurological deterioration, suggesting that meticulous surgical technique can mitigate these particular adverse events to some extent. The findings of our study align with trends observed in recent research. For instance, several direct comparative studies and meta-analyses have also indicated the potential of ACAF in improving neurological recovery and controlling complications.8,11,13 However, compared to other meta-analyses that primarily focused on broad comparisons between anterior and posterior approaches,3,7 the present Network Meta-Analysis (NMA), by specifically including ACAF as a distinct anterior technique and conducting three-way comparisons and rankings, provides more granular and definitive evidence. This NMA particularly highlights the advantages of ACAF over traditional ACCF in certain aspects and its comprehensive superiority over LP. It is important to note that while the overall analysis indicated ACAF had the lowest total complication risk, the subgroup analyses based on stenosis severity did not reveal statistically significant differences in total complication rates among the surgical techniques. Although SUCRA rankings consistently favored ACAF as the lowest-risk approach in both stenosis subgroups, these trending results should be interpreted with caution. Potential reasons for this lack of statistical significance include reduced sample sizes within subgroups leading to insufficient statistical power, or differing compositions of major complications across varying degrees of stenosis severity for each procedure. For example, the risk of CSF leakage with ACCF might remain consistently high regardless of stenosis severity, while the risks of axial symptoms and C5 palsy with LP might also be relatively stable. Larger, more rigorously designed studies are warranted to definitively elucidate the specific impact of stenosis severity on the complication risk profiles of these different surgical procedures. Furthermore, our subgroup analysis regarding the number of involved levels revealed an interesting divergence. For extensive OPLL (>3 segments), while no statistically significant difference was found, LP ranked highest for safety (SUCRA 82.0%), followed by ACAF (61.4%), with ACCF ranking lowest (6.6%). This suggests that for extensive multisegment disease, LP remains a reliable option with a lower risk profile compared to anterior approaches, likely due to the avoidance of complex multilevel reconstruction-related risks.
Clinical implications and recommendations
Based on this NMA, ACAF emerges as a highly promising technique for multilevel OPLL, offering an optimal balance between efficacy and safety. It should be strongly considered, particularly for patients with severe canal stenosis (≥60% occupying ratio) or extensive disease (≥3 levels), where it demonstrates superior neurological improvement and potentially the lowest complication risk. ACCF remains a viable and effective option, especially for less extensive disease (<3 levels), but surgeons must be prepared for a higher risk of CSF leakage. LP, while offering the shortest operative time, provides inferior neurological and biomechanical outcomes and carries higher risks of C5 palsy and axial pain; its use might be reserved for select cases, such as patients unfit for prolonged anterior surgery or those with contraindications to anterior approaches, provided K-line is positive. The ultimate choice requires careful individualized assessment, considering patient factors (age, comorbidities, bone quality), OPLL characteristics (severity, extent, type, K-line status, curvature), surgeon experience, and available institutional resources. The broader adoption of ACAF necessitates addressing its technical demands, learning curve, potential need for specialized navigation, and cost-effectiveness.
Limitations
This study has several limitations that warrant careful consideration. First and foremost, the evidence base relies predominantly on non-randomized comparative studies (30 out of 31). Despite the moderate-to-high quality ratings (NOS scale) of included studies, the inherent susceptibility of observational designs to selection bias and confounding by indication significantly constrains the certainty of our findings. Specifically, a critical selection bias exists regarding disease severity and alignment: surgeons may have preferentially selected anterior approaches (ACAF or ACCF) for patients with massive ossification (occupying ratio >60%) or kyphotic deformity, while reserving laminoplasty (LP) for mild-to-moderate cases with preserved lordosis (K-line positive). Consequently, the observed superiority of ACAF in this network meta-analysis might be partially driven by the inclusion of severe cases where LP is historically known to be less effective. Therefore, our findings regarding ACAF’s advantages should be interpreted primarily within the context of severe or massive OPLL and may not be generalizable to mild cases where LP remains a sufficient and safer standard. Although sensitivity analyses suggested robustness, these results cannot substitute for high-quality RCTs, and residual confounding likely persists. Second, clinical and methodological heterogeneity is inevitable across the included studies regarding patient selection, precise OPLL classification, surgical technique variations, and follow-up duration. While we employed random-effects models and subgroup analyses to mitigate this, the inability to perform meta-regression due to data limitations prevented a deeper exploration of heterogeneity sources. Third, regarding the transitivity assumption, our detailed assessment (Supplemental Table 2) revealed that while age and follow-up duration were well-balanced across comparison groups, there was a potential imbalance in disease severity. Specifically, studies directly comparing ACAF versus ACCF tended to include patients with more severe stenosis (mean proportion of occupying ratio >60%: 63.5%) compared to comparisons involving LP (35.0%–51.9%). This observation reflects confounding by indication, a common bias in observational studies where surgeons preferentially select anterior approaches (ACAF/ACCF) for massive OPLL and reserve LP for milder cases. Consequently, the indirect comparisons between ACAF and LP in our network might be influenced by this severity gradient. To mitigate this potential bias, we emphasized the subgroup analysis based on stenosis severity (>60%), which provides more precise estimates for the patient population where ACAF is most clinically indicated. Additionally, although a statistical difference in sex distribution was observed (P = 0.04), the absolute difference was small (<6%) and unlikely to significantly bias the comparative efficacy findings. Fourth, data for certain outcomes (e.g., NDI, ROM) were derived from a relatively small number of studies, potentially affecting the precision of these specific comparisons. Finally, cost-effectiveness analysis was not possible based on available data, and potential publication bias cannot be entirely excluded, although funnel plot assessments were generally reassuring.
Future directions
High-quality, multicenter RCTs with long-term follow-up are urgently needed to definitively compare ACAF, ACCF, and LP, focusing on validated patient-reported outcome measures (PROMs) and rates of ASD. Future studies should adhere to standardized reporting of OPLL characteristics (type, occupying ratio, K-line status) and detailed surgical techniques to facilitate more precise subgroup analyses and individualized treatment algorithms. Investigations into the learning curve associated with ACAF and rigorous cost-effectiveness analyses are also warranted to guide its implementation in various healthcare settings.
Conclusion
This network meta-analysis provides the most comprehensive evidence to date comparing ACAF, ACCF, and LP for multilevel cervical OPLL. ACAF showed a favorable profile, ranking highest in probability for neurological improvement, pain relief, biomechanical stability, and overall safety (lowest total complication rate). Its advantages appear particularly pronounced in patients with severe canal stenosis (≥60%). ACCF offers good neurological outcomes, especially for less extensive disease (<3 levels), but with a higher CSF leakage risk compared to ACAF. LP provides shorter operative times but inferior neurological and biomechanical results and higher risks of C5 palsy and axial pain. However, it may offer a safer profile for extensive multisegment disease (>3 levels) by avoiding complex anterior reconstruction. These findings underscore the importance of individualized surgical selection based on detailed assessment of patient and disease characteristics, while acknowledging the limitations primarily stemming from the reliance on non-randomized evidence. Future high-quality RCTs are essential to confirm these findings and evaluate long-term outcomes.
Supplemental material
Supplemental material - Comparative efficacy and safety of ACAF, ACCF, and laminoplasty for multilevel cervical OPLL: A network meta-analysis of observational studies
Supplemental material for Comparative efficacy and safety of ACAF, ACCF, and laminoplasty for multilevel cervical OPLL: A network meta-analysis of observational studies by Wei Wang, Cheng-Hao Xiang, Dan Li, Xian-Zao Wang, Xin-Hua Xu in Journal of Orthopaedic Surgery
Footnotes
Author contributions
All authors contributed to the study conception and design. Material preparation, data collection and analysis were performed by Wei Wang, Chenghao Xiang, Dan Li and Xianzao Wang. The first draft of the manuscript was written by Wei Wang and all authors commented on previous versions of the manuscript. All authors read and approved the final manuscript.
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
Declaration of conflicting interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publicationof this article.
Supplemental material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
