Abstract
Purpose
Patients withspinal degenerative diseases are often older and have multiple comorbidities. This study aims to evaluate the impact of epidural patient-controlled analgesia (PCA) on postoperative pain relief in patients undergoing lumbar spine surgeries for spinal degenerative diseases.
Methods
This retrospective case-control study included patients who underwent lumbar spine surgeries for degenerative spinal stenosis, spondylolisthesis, herniated intervertebral discs, or osteoporotic spinal fractures with spinal stenosis. The PCA group consisted of patients who received 72-h epidural PCA for postoperative pain control, while the control group received standard postoperative pain management. All patients were allowed to request intramuscular rescue analgesics for additional pain control. The primary endpoint was defined as the mean visual analogue scale (VAS) score during the effective PCA period (postoperative day [POD] 1–3). Secondary endpoints included individual daily VAS scores, rebound pain, rescue analgesic injections, morphine consumption, drainage duration, drainage volume, length of hospital stay, and complications.
Results
A total of 209 patients (mean age 73.8 years) were included, with 88 patients in the PCA group and 121 in the control group. Mean VAS score across POD 1–3 was significantly lower in the PCA group (Cohen’s d = −1.89, 95% CI = −2.22 to −1.56, p < 0.001). During hospitalization, the PCA group required significantly fewer rescue analgesic injections (Cohen’s d = −2.47, 95% CI = −2.84 to −2.11) and less total morphine consumption (Cohen’s d = −0.39, 95% CI = −0.67 to −0.11) compared to the control group. Although the PCA group experienced greater drainage volume and longer duration of drainage placement, the incidence of infection and the length of hospital stay were comparable between the two groups.
Conclusion
In this real-world cohort of elderly patients with multiple comorbidities undergoing lumbar spinal surgery, epidural PCA provided effective pain relief without an observed increase in infection rates in this study population.
Keywords
Introduction
Spinal degenerative disease is a prevalent condition in individuals over the age of 65, with a reported prevalence rate of 27.3%, which increases with advancing age. 1 Among those aged 74 and older, the prevalence rises to 38.6%. 2 These degenerative changes progressively affect all components of the vertebral unit and are a common cause of low back pain. When conservative treatments fail, surgical intervention becomes an option for many older patients.
Despite advancements in minimally invasive surgical techniques, postoperative pain management in spinal degenerative disease surgeries remains a significant clinical challenge. Extensive dissection of subcutaneous tissues, bones, and ligaments during these procedures can result in severe postoperative pain. This pain may arise from mechanical and chemical stimuli associated with surgical manipulation and inflammation of the spinal dura. Sensory innervation through the posterior rami of spinal nerves contributes to the transmission of pain signals from these structures. 3 Both direct and indirect postoperative pain can substantially impact recovery, ambulation, hospital discharge, and overall patient satisfaction. Effective pain management is essential to prevent persistent postoperative pain and facilitate timely recovery. 4
Postoperative pain management following spinal surgeries typically extends for less than 3 days. 5 Common strategies include oral or intramuscular analgesics and patient-controlled analgesia (PCA), a system allowing patients to self-administer analgesics as needed to control pain. 6 Intravenous PCA with opioids is widely used for pain control across various surgical settings.7,8 However, older patients frequently experience opioid-related adverse effects such as nausea and vomiting, which remain a considerable concern.6–9
Epidural PCA has been shown to offer advantages over intravenous PCA in several surgical contexts, including thoracic, 10 major abdominal,11–13 orthopedic, 14 pelvic, 15 and cesarean surgeries. 16 A meta-analysis demonstrated that epidural PCA provides superior postoperative pain relief and reduces the incidence of nausea, vomiting, and sedation compared to intravenous PCA. 7 Despite these benefits, there is limited research on the use of epidural PCA for surgeries related to spinal degenerative diseases. Surgeons may hesitate to use epidural PCA in spinal surgeries due to concerns about potential wound infections caused by the proximity of the epidural catheter to the surgical field.
Patients with spinal degenerative disease often belong to an older demographic and frequently have comorbid conditions, making postoperative pain management more complex. However, effective pain control is critical for promoting early mobilization and rehabilitation in this population. Early mobilization after spinal surgeries has been associated with reduced complication rates, decreased length of hospital stay, and improved functional outcomes. 17 Optimal pain management facilitates early mobilization, which is crucial for achieving these benefits.
This study aimed to evaluate the effect of epidural PCA for postoperative pain relief in patients undergoing lumbar spine surgeries for spinal degenerative diseases. The findings are expected to inform clinical decision-making and contribute to the development of effective pain management strategies tailored to this patient population.
Materials and methods
Subjects
This retrospective case-control study was approved by the Institutional Review Board of our hospital (IRB No. TCHIRB-11003008-E) and registered retrospectively on ClinicalTrials.gov (NCT06316921; March 19, 2024). Informed consent was waived due to the retrospective study design and the use of anonymized clinical data for analysis. The study adheres to the guidelines outlined in the STROCSS criteria for case-control studies. 18
The study included patients who underwent lumbar spine surgeries for degenerative spinal conditions at our hospital between January 2014 and December 2023. Eligible conditions included degenerative spinal stenosis, spondylolisthesis, herniated intervertebral disc, and osteoporotic spinal fractures with associated spinal stenosis. Patients were excluded if they had spinal infections, such as tuberculosis, epidural abscess, or vertebral body osteomyelitis (n = 15), iatrogenic cerebrospinal fluid leakage or dura tear noted during surgery (n = 4), spinal tumor or spinal metastasis (n = 8), or if they had undergone minimally invasive procedures using the bilateral paramedian approach (n = 7). Of the 243 patients meeting the initial inclusion criteria, 34 were excluded based on these criteria, leaving 209 patients for analysis.
The 209 eligible patients were categorized into two groups based on their postoperative pain management strategies: the PCA group, comprising patients who received epidural PCA, and the control group, consisting of patients who received standard postoperative pain management without epidural PCA. The decision to receive epidural PCA was made through a discussion between the orthopedic surgeon, the anesthesiologist, the patient, and the patient’s family. Patients presenting with any of the following were excluded from epidural PCA: patients with known coagulation disorders or those currently receiving anticoagulant medications; patients with preexisting neural deficits, burns, or trauma in the area of PCA placement; the presence of systemic infection, or localized infection at the site of PCA placement; increased intracranial pressure; and patients unable to understand the procedure or cooperate with self-administered pain relief. A flowchart of patient inclusion, exclusion, and group assignment was provided in Figure 1. Flow of patient selection.
Surgical procedure
Under general anesthesia, patients were positioned prone on a radiolucent spine frame. The targeted spinal level was identified and marked preoperatively using a portable radiograph, and a midline incision was designed above the spinous process. Throughout the procedure, the incision size was minimized to reduce unnecessary tissue destruction and mitigate pain induction.
Bilateral dissection of the paraspinous muscles was performed to expose the lamina, bilateral facet joints, and transverse processes. Using radiographic image guidance, fixation instrumentation was implanted prior to the laminectomy decompression. The decompression involved the complete removal of the lamina, including the pars interarticularis and the inferior facet process, at the designated spinal level. Posterior lumbar interbody fusion was performed for all patients using interbody fusion cages. 19 Posterolateral fusion on the transverse processes was also conducted, utilizing a mixture of artificial bone graft substitutes and autograft material obtained from the laminectomy. Pedicle screws were placed bilaterally and connected with rods and crosslinks to stabilize the spinal structure. To manage postoperative drainage, a wound drainage tube was placed at the laminectomy site. The surgical wound was meticulously closed in layers, and the patient was transferred to the postoperative recovery room for further management.
Postoperative pain management
In the PCA group, the epidural PCA catheter was inserted by the surgeon prior to wound closure. The catheter tip was positioned 3 cm cephalad to the upper edge of the laminectomy within the epidural space. Proper catheter placement was confirmed by aspirating with a syringe and injecting saline. If bloody aspiration was detected or if saline injection was obstructed, the catheter was repositioned. Once appropriate positioning was ensured, the catheter was securely anchored and affixed to the patient’s back to prevent displacement.
Following spinal surgery, epidural analgesia was initiated after a comprehensive assessment of sensory and motor function in both legs. The PCA regimen consisted of a solution containing 20 ml of 0.5% bupivacaine and 5 mg of morphine, diluted in 500 ml of normal saline. The PCA was administered without a loading dose, with a continuous infusion rate of 8 ml per hour. A bolus dose of 8 ml was available, with a lockout interval of 30 min. The maximum allowable dose over a 4-h period was set at 80 ml. The PCA was maintained for 72 h postoperatively and discontinued on the fourth postoperative day (POD 4), at which point the catheter was removed.
Standard postoperative pain management for the control group included oral administration of acetaminophen (500 mg, four times daily) and a muscle relaxant (chlorzoxazone 200 mg, four times daily). To mitigate the risk of adverse events in elderly postoperative patients, nonsteroidal anti-inflammatory drugs were not administered.20,21
All patients, irrespective of group, were allowed to request rescue analgesia for additional pain relief if necessary. The rescue analgesic regimen consisted of 5 mg of intramuscular morphine, with a minimum interval of 6 h between doses.
Outcome assessment and data collection
The primary endpoint of this study was defined as the mean VAS score during the effective PCA period (POD 1–3). Secondary endpoints included individual daily VAS scores, rebound pain, the number of rescue analgesic injections, total morphine consumption, duration of drainage, total drainage volume, length of hospital stay, and complications.
Pain levels were assessed using a visual analogue scale (VAS) on the first four postoperative days (POD 1–4) and on the day of discharge. The VAS ranged from 0 to 10, where 0 indicated no pain and 10 represented the worst imaginable pain. Before each VAS assessment, patients were instructed to lie supine in bed and rest without any physical activity for at least 30 min. All VAS assessments were conducted at scheduled times (e.g., 8:00 AM) and were measured a minimum of 2 h following any patient-requested intramuscular rescue analgesic dose to prevent the measurement from reflecting the peak effect of the rescue medication. Furthermore, the assessment was performed prior to any scheduled physical therapy or rehabilitation sessions.
On POD 4, pain assessment was conducted following the removal of the PCA catheter. Rebound pain was defined as an increase in VAS score of ≥4 within 24 h after PCA removal. This threshold was selected based on prior studies of neuraxial analgesia to represent a clinically significant deterioration in pain control following cessation of regional block. 22 Additionally, the number of rescue analgesic injections administered during the postoperative period was recorded. Other data were collected retrospectively from medical records, including demographic details, clinical and surgical variables, postoperative complications, and length of hospital stay.
Statistical analysis
Continuous variables were summarized as mean (standard deviation [SD]), while categorical variables were presented as counts (percentages). For comparisons of baseline characteristics, two-sample t-test was performed for continuous variables and Chi-square test was used for categorical variables. For comparisons of outcomes, standardized mean difference using Cohen’s d with 95% confidence interval (CI) was calculated for continuous variables, risk ratio (RR) with 95% CI was calculated for categorical variables.
Univariable and multivariable linear regression analyses were performed for the primary endpoint (mean VAS on POD 1–3). The model adjusted for age, sex, ASA status, hypertension, diabetes mellitus, surgical indication, number of surgical levels, sacrum fusion, operation time, and blood loss. A sensitivity analysis was conducted using propensity score matching. A propensity score was calculated for each patient using a logistic regression model incorporating the aforementioned covariates. Patients were matched to control for baseline differences, and the effect size (Cohen’s d with 95% CI) was calculated for the matched cohort.
The longitudinal pattern of postoperative pain on POD 1–4 was analyzed using a linear mixed-effects model. The fixed effects structure included the treatment group, time, and the group × time interaction term. To account for the correlation of repeated measures within the same patient, the model included a random intercept for each patient. Model fitting was performed using restricted maximum likelihood. The inclusion of a random intercept accounted for patient-specific baseline differences in pain perception. The overall effect of the intervention was evaluated by the fixed effect estimate for the PCA group and the group × time interaction P-value. Results were reported as estimated marginal means with 95% CIs derived from the linear mixed-effects model.
The clinical meaningfulness of pain relief was assessed by calculating the proportion of patients achieving the minimal clinically important difference (MCID), defined as a ≥2.0-point reduction in VAS score from the preoperative baseline. 22 The proportion achieving MCID was reported with a 95% Wilson Score CI. A two-tailed p-value of <0.05 was considered statistically significant. Statistical analyses were performed using SPSS software (Version 22; IBM Corp., Armonk, NY, USA).
Results
Baseline characteristics and operative information.

Data were presented as mean (standard deviation) or count (percentage)*.
Abbreviation: PCA, patient-controlled analgesia; ASA, American society of anesthesiologists physical status classification system; vas, visual analogue scale.
Primary endpoint
Results of primary and secondary endpoints.
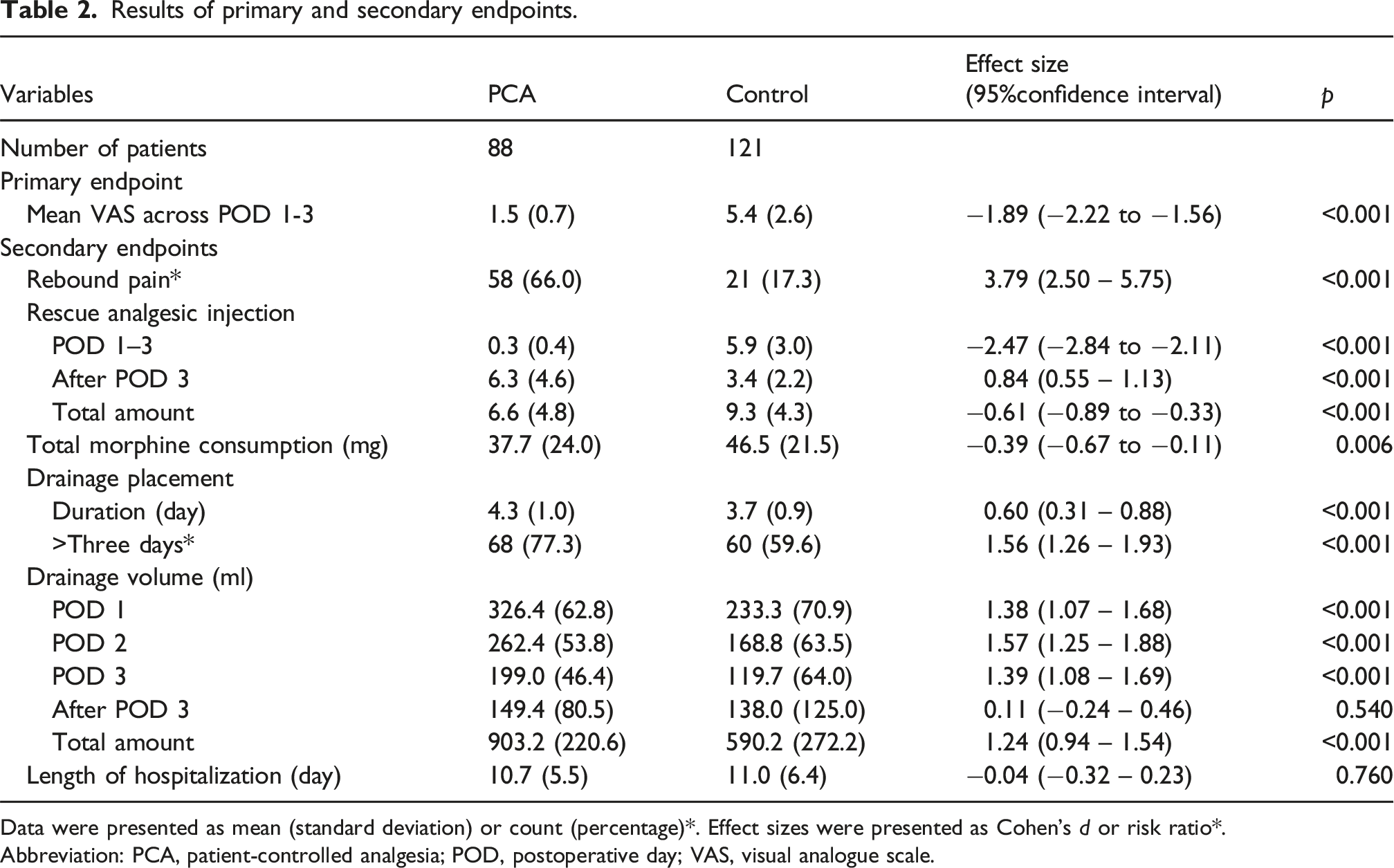
Data were presented as mean (standard deviation) or count (percentage)*. Effect sizes were presented as Cohen’s d or risk ratio*.
Abbreviation: PCA, patient-controlled analgesia; POD, postoperative day; VAS, visual analogue scale.
The multivariable linear regression showed that the association between epidural PCA and reduced postoperative pain remained significant after adjustment. The unadjusted regression coefficient (β) was −3.837 (95% CI: −4.397 to −3.278, p < 0.001), and the multivariable-adjusted coefficient (β) was −3.830 (95% CI: −4.401 to −3.259, p < 0.001), indicating that the pain relief effect was independent of baseline characteristics. In the propensity score-matched sensitivity analysis, the mean VAS score on POD 1–3 in the matched PCA group was 1.5 (SD 0.68) compared to 5.5 (SD 2.58) in the matched control group (p < 0.001). This difference represented a very large effect size (Cohen’s d = −2.11; 95% CI = −2.52 to −1.71), confirming the efficacy of epidural PCA.
Secondary endpoints
Daily VAS scores
The linear mixed-effects model demonstrated a significant fixed effect for the PCA group (coefficient β = −4.90 on POD 1, p < 0.001), confirming that the PCA intervention provided immediate and more significant pain relief compared to the control group. Furthermore, a highly significant group × time interaction was observed across POD 1–4, indicating a significantly different trajectory of pain reduction between the groups (interaction β = −1.63 on POD 3, p < 0.001; interaction β = −2.61 on POD 4, p < 0.001). As shown in Figure 2, the model-based estimated marginal mean VAS scores for the PCA group remained low (ranging from 1.76 on POD 1 to 0.38 on POD 4), while the control group showed higher scores (ranging from 6.66 on POD 1 to 3.97 on POD 4), confirming that the epidural PCA provided a more significant effect on pain relief throughout the first four postoperative days. Model-based estimated marginal mean of VAS Scores between patients who received epidural PCA and those who received standard postoperative pain care following lumbar spinal surgery. The data represented the estimated marginal mean of VAS score derived from a linear mixed-effects model, with error bars indicating the 95% confidence interval. Abbreviation: PCA, patient-controlled analgesia; POD, postoperative day.
Rebound pain
Following the removal of the PCA catheter on POD 4, the mean VAS score in the PCA group was higher than that in the control group (5.2 vs 4.6). The PCA group had a significantly higher proportion of patients experiencing rebound pain compared to the control group (66% vs 17%, p < 0.001, Cohen’s d = 3.79, 95% CI = 2.50 – 5.75) (Table 2). Despite this, comparable pain levels were observed between the two groups on the day of discharge (2.3 vs 2.3). Pain levels in the control group gradually decreased during hospitalization.
Rescue analgesic injections & morphine consumption
During hospitalization, patients in the PCA group required significantly fewer rescue analgesic injections compared to those in the control group (6.6 vs 9.3, p < 0.001, Cohen’s d = −0.61, 95% CI = −0.89 to −0.33). However, following the removal of the PCA catheter, the PCA group required a significantly greater number of rescue analgesic injections than the control group (6.3 vs 3.4, p < 0.001, Cohen’s d = 0.84, 95% CI = 0.55 to 1.13) (Figure 3). Despite this, the total morphine consumption over the entire hospitalization period was significantly lower in the PCA group than in the control group (37.7 vs 46.5 mg, p = 0.006, Cohen’s d = −0.39, 95% CI = −0.67 to −0.11) (Table 2). Number of rescue analgesic injections administered to patients with epidural PCA compared to those receiving standard postoperative pain care after lumbar spinal surgery. The data represented the mean, with error bars indicating the 95% confidence interval. *p < 0.05. Abbreviations: PCA, patient-controlled analgesia; POD, postoperative day.
Duration of drainage, drainage volume, & hospital stays
The PCA group exhibited a significantly longer duration of drainage placement (4.3 vs 3.7 days, p < 0.001, Cohen’s d = 0.60, 95% CI = 0.31 to 0.88) and a greater total drainage volume (903.2 vs 590.2 ml, p < 0.001, Cohen’s d = 1.24, 95% CI = 0.94 to 1.54) during the entire hospitalization. Following the removal of the PCA catheter on POD 4, there were no significant differences in drainage volume between the two groups (149.4 vs 138.0 ml, p = 0.540, Cohen’s d = 0.11, 95% CI = −0.24 to 0.46). The length of hospital stays was comparable between the groups (10.7 vs 11.0 days, p = 0.760, Cohen’s d = −0.04, 95% CI = −0.32 to 0.23) (Table 2).
Complications
Postoperative complications.

Data were presented as count (percentage).
Abbreviation: PCA: patient-controlled analgesia.
MCID analysis
The PCA group demonstrated significantly greater MCID achievement rates during the early postoperative period. On POD 1, 98.6% of the PCA group achieved MCID (95% CI = 92.6–99.8%), compared to only 1.6% of the control group (95% CI = 0.3–8.7%). This difference persisted through POD 2 (PCA 100% [94.9–100%] vs control 12.4% [7.8–19.2%]) and POD 3 (PCA 100% [94.9–100%] vs control 30.6% [23.1–39.3%]). The difference in MCID achievement narrowed at later time points, reflecting the tapering of the epidural PCA: PCA 1.1% (0.2–6.2%) versus control 5.0 % (2.3–10.4%) on POD 4. At discharge, the achievement rate was comparable (PCA 26.1% [18.1–36.2%] vs control 31.4% [23.8–40.1%]).
Discussion
Despite advancements in pain management, this study underscored the potential benefits of epidural PCA for postoperative pain control and recovery in elderly patients with multiple comorbidities undergoing lumbar spine surgeries. The findings demonstrated that epidural PCA effectively managed pain throughout the hospital stay, as evidenced by significantly lower VAS scores, fewer rescue analgesic injections, and less total morphine consumption in the PCA group compared to standard pain management. Although the PCA group experienced greater drainage volume and longer duration of drainage placement, the incidence of infection and the length of hospital stay were comparable between the two groups.
A key limitation of retrospective studies involving complex spinal surgery is the inherent heterogeneity of surgical procedures. Our cohort included patients with a wide spectrum of pathology, including degenerative stenosis, spondylolisthesis, and spinal fractures, undergoing both single-level decompressions and multi-level fusions. We addressed this potential confounding through rigorous statistical methods: all primary analyses were adjusted for key surgical complexity variables, including surgical indication, number of surgical levels, sacrum fusion, operation time, and blood loss. The finding that the unadjusted and adjusted effect sizes for pain relief remained virtually identical (e.g., adjusted β = −3.824 vs unadjusted β = −3.837) provided evidence that the pain control provided by the epidural PCA was independent of variations in surgical procedures.
Ensuring the correct placement of the PCA catheter tip within the epidural space is critical to prevent serious complications such as systemic local anesthetic toxicity, seizures, cardiac arrest, and even death.23,24 Previous studies have indicated that approximately 10% of epidural catheter placements may result in misplacement into an epidural vessel, with up to 10% of these misplacements going undetected. In our study of 88 patients, we successfully avoided intra-vascular placement of the PCA catheter. This was likely due to our method of catheter introduction, which was performed visually with a surgical instrument rather than blindly with the epidural needle. Additionally, we placed the catheter along the midline of the dural sac, thereby avoiding the para-median epidural venous plexus in the lateral recess, which could be inadvertently accessed by the epidural needle. Proper catheter placement is essential to minimize the risk of potential complications.
This study found that the pain relief effect of epidural PCA did not persist after its removal, leading to rebound VAS scores and an increased need for rescue analgesic injections. However, the total number of rescue analgesic injections and total morphine consumption during hospitalization was still lower in the PCA group compared to the control group, which contributed to a lower incidence of sedation. Furthermore, rebound pain in the PCA group was transient, resolving within a few days and returning to a level comparable to the control group on the day of discharge. Rebound pain following epidural PCA has been documented in previous studies, with several risk factors identified, including surgical time, sex, postoperative pain intensity, preoperative nutritional and immunologic status, and intraoperative transfusion.25–27 Preventive strategies, such as multimodal pain management and preoperative education, have been proposed to address this issue. 28 While the transient nature of rebound pain may reduce the overall benefits of epidural PCA, its known advantages in pain relief, early mobilization, and functional recovery after spinal surgery underscore its value—particularly in older patients—during the critical early postoperative period. The high incidence of rebound pain (66% in the PCA group) underscored a critical clinical consideration: the necessity of implementing a robust analgesic transition strategy. Clinically, short-term management with multimodal analgesia, including scheduled non-opioid agents and patient counseling on expected rebound discomfort, can effectively control this transient pain. As the rebound phenomenon did not extend beyond the early postoperative period, no long-term adverse effects on recovery or pain outcomes were observed.
This study found that the use of epidural PCA was associated with a longer duration of drainage placement and a higher total drainage volume compared to standard pain management. The PCA regimen may leak from the epidural space into the wound space, where it can mix with tissue fluid and blood, leading to increased drainage volume. During the first three postoperative days, the PCA group had an average drainage volume that was 100 ml greater than that of the control group. However, after the removal of the epidural PCA, there was no significant difference in drainage volume between the two groups. The substantially increased drainage volume (903.2 ml vs 590.2 ml, Cohen’s d = 1.24, 95% CI = 0.94 – 1.54) and longer duration of drain placement (4.3 vs 3.7 days, Cohen’s d = 0.60, 95% CI = 0.31 – 0.88) were clinically relevant factors. This necessitated careful wound care management and may delay the attainment of standard drain removal criteria, potentially impacting the timing of full patient mobilization in elderly patients. Nevertheless, the increased drainage in the PCA group was not associated with any additional complications or an extended length of hospitalization.
Infection is a significant concern for spine surgeons when using epidural PCA. However, the in vitro and in vivo antimicrobial effects of local anesthetics have been extensively studied.29,30 In this study, we used 0.5% bupivacaine as part of the PCA regimen. Previous research has demonstrated the inhibitory effect of bupivacaine on the growth of microorganisms commonly found on the skin, such as Staphylococcus, coagulase-negative Staphylococcus, Streptococcus, Klebsiella enterobacter, Escherichia coli, and Proteus species.31–35 This suggested a potent antimicrobial property of bupivacaine. In our study, the observed incidence of wound infection was comparable between the two groups (2.3% in the PCA group vs 1.7% in the control group), yielding a RR of 1.38 (95% CI = 0.28–6.48). Although the increased drainage volume in the PCA group was not associated with a significant rise in infection rates in this cohort, we acknowledge that the low frequency of adverse events limits the statistical power to rule out rare complications. We emphasized that while no statistically significant difference was observed, the low frequency of adverse events and the wide confidence interval indicated that the study was underpowered to exclude moderate increases in infection risk. These data suggested that the increased drainage likely reflected the physical effect of epidural infusion rather than a pathological process such as inflammation or infection. Thus, within the limits of this study, epidural PCA appeared to be a feasible method for postoperative pain management in elderly patients, though larger, adequately powered studies are needed to definitively confirm its infectious safety profile.
Although epidural PCA can be an effective pain management strategy, it also carries potential risks. In this study, two patients in the PCA group (1.7%) experienced motor block, resulting in temporary leg paralysis on the first postoperative day, whereas no motor block was observed in the control group. However, after adjusting the PCA dosage, both patients recovered normal leg function. Additionally, the PCA group experienced significantly more pruritus than the control group (29.6% vs 9.9%, RR = 2.99, 95% CI = 1.56 – 5.72). These pruritus episodes were managed with antihistamines and resolved after the removal of the PCA. While pruritus is a common adverse effect of neuraxial opioids, 36 preventive strategies and management of opioid-induced pruritus should be further explored in future research and clinical practice.
Previous studies have highlighted the benefits of epidural PCA, including shorter hospital stays, enhanced patient satisfaction, reduced morbidity, improved quality of recovery, and expedited restoration of gastrointestinal function.6,37–39 However, discrepancies between the findings of our study and those of prior reports may arise from variations in study design, surgical procedures, patient populations, and outcome assessment methods. Although epidural PCA is an established analgesic technique, its use may be particularly beneficial in elderly patients or in those undergoing extensive spinal decompression and fusion procedures, where effective early pain control and mobilization are critical. Given the potential for complications and prolonged drainage, epidural PCA should be applied selectively. For simpler cases, such as single-level lumbar disc herniation surgery, standard multimodal analgesia may provide sufficient pain relief with fewer risks. Future research should focus on comprehensive assessments of functional recovery, and explore strategies to prevent rebound pain and mitigate opioid-related complications. Additionally, alternative approaches to postoperative pain management should be investigated. For instance, fascia-based cocktail injections have shown promise; Murata et al. demonstrated the efficacy and safety of multidrug cocktail injections in managing pain after lumbar microendoscopic decompression surgery. This approach offered a potential alternative for achieving effective postoperative analgesia while potentially reducing reliance on opioids. 40
Several limitations of this study should be acknowledged. Firstly, the retrospective design inherently introduced potential selection and reporting biases that could influence the results. Specifically, the retrospective nature of data collection, particularly for adverse events and complications, carried a risk of under-reporting or inconsistent documentation. Secondly, as all patients were operated on by a single surgeon and followed up at the same institution, the generalizability of the findings to other settings may be limited. The single-surgeon model, while controlling for inter-surgeon technical variability, introduced a potential surgeon-related selection bias. Thirdly, caution must be exercised in interpreting the results due to potential confounding factors, such as variations in the indications for spinal surgeries and baseline comorbidities. Furthermore, the 10-years span of data collection implied that changes in perioperative anesthetic protocols, surgical techniques, and instrumentation over time could act as temporal confounders. Fourthly, the study did not assess postoperative functional recovery, which is a critical outcome in patients undergoing spinal surgeries. Fifthly, this study focused only on the short-term effects of PCA on postoperative pain relief, and long-term follow-up to evaluate the sustainability of pain relief and functional outcomes was not conducted. 41 Lastly, as epidural PCA was not covered by the National Health Insurance program in Taiwan, assignment to the PCA group was influenced by patient financial capacity and personal preference. This introduced a significant non-clinical selection bias, as the PCA group may inherently represent a patient population with higher socioeconomic status, which could potentially confound outcomes.
Conclusion
This case-control study, involving over 200 patients with a mean age of 74 years and multiple comorbidities, provided real-world information on the use of epidural PCA for postoperative pain control in lumbar spinal surgery for degenerative spinal diseases. Epidural PCA offered improved postoperative pain relief during the catheter placement period, which may facilitate early mobilization in this high-risk population. Notably, despite concerns about catheter proximity to the surgical field, the increased drainage volume and extended duration of drainage associated with epidural PCA were not associated with a significant increase in infection rates or prolonged hospital stay in this cohort. However, given the low incidence of postoperative infections and the sample size of this study, these safety findings regarding infection should be interpreted with caution. The use of epidural PCA should continue to be guided by careful patient selection and correct catheter placement to mitigate potential catheter-related injuries. These findings addressed a notable evidence gap, as elderly, comorbidity-rich patient populations are often excluded from randomized controlled trials. Further research is necessary to validate these findings and evaluate the long-term benefits.
Footnotes
Ethical considerations
This retrospective case-control study was ethically approved by the Institutional Review Board of our hospital (IRB No. TCHIRB-11003008-E) and retrospectively registered on ClinicalTrials.gov (NCT06316921; March 19, 2024).
Consent for publication
Informed consent was waived due to the retrospective nature of the study and the utilization of anonymized clinical data for analysis.
Author contributions
HC Chen: study design, data acquisition, data analysis, manuscript writing. JH Yu: literature research, data analysis, data interpretation, manuscript writing. MH Hsieh: data analysis, data analysis, data interpretation. SL Shih: study concept and study design, guarantor of integrity of the entire study. All authors contributed to the approval of the final version.
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
Declaration of conflicting interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Data Availability Statement
The dataset analyzed in this study is available upon reasonable request.
