Abstract
Objective
α2‑agonists and opioids have been used as intrathecal adjuvants to local anesthetics for several years, but the effect of intrathecal dexmedetomidine (Dex) or sufentanil combined with epidural ropivacaine in labor analgesia is not fully understood.
Methods
A total of 108 parturient women receiving combined spinal-epidural labor analgesia were randomly divided into three groups. Group C received l mL saline (0.9%) intrathecally, Group D received 5 µg Dex intrathecally, and Group S received 5 µg sufentanil intrathecally. All parturient women then received 0.1% epidural ropivacaine and 0.2 µg/mL sufentanil for patient-controlled epidural analgesia with standard settings. The visual analog scale score, onset time, duration of intrathecal injection, local anesthetic requirements, and side effects were recorded.
Results
The labor analgesia effects in Groups D and S were better than those in Group C. Groups D and S displayed significantly shorter onset times, longer durations of intrathecal injection, and reduced local anesthetic requirements compared with Group C. The incidence of shivering and pruritus in Group D was lower than that in Group S.
Conclusion
Intrathecal administration of 5 µg Dex could improve epidural labor analgesia effects.
This randomized controlled clinical trial was registered with the Chinese Clinical Registry Center (ChiCTR-1800014943, http://www.chictr.org.cn/).
Keywords
Introduction
The pain of childbirth is widely described to be severe and unbearable by most women, and the administration of effective labor analgesia can significantly improve intrapartum maternal–fetal well-being. 1 Combined spinal-epidural (CSE) block is commonly used for labor analgesia, with a rapid onset of analgesia and minimal side effects. 2 Currently, low-dose sufentanil is widely administered intrathecally to shorten onset times and enhance labor analgesic effects. 3 However, opioids are associated with adverse effects, such as nausea, vomiting, pruritus, urinary retention, and respiratory depression. 4 Clinicians and researchers have been investigating strategies to avoid the side effects of opioids, such as the use of α2‑adrenoreceptor agonists as an intrathecal adjuvant.5,6
Dexmedetomidine (Dex) is a new type of α2-adrenoreceptor agonist with an α2/α1 selectivity eight times higher than that of clonidine. 7 Dex has been successfully used for spinal and intravenous anesthesia in animals and humans without causing significant respiratory depression, pruritus, nausea, or vomiting.8,9 In addition to epidural administration, Dex has also been administered intrathecally as an adjuvant to local anesthetics to improve the anesthetic effect, provide stable hemodynamic conditions, and prolong postoperative analgesia.9,10 Furthermore, both intrathecal and intravenous injections of Dex alone or as an adjuvant were found to be effective for pain relief.11,12 Therefore, we hypothesized that Dex also provides an analgesic effect or enhances the analgesic effect of local anesthetics. To our knowledge, this study is the first to compare the labor analgesia effect of intrathecal Dex with sufentanil combined with epidural ropivacaine.
Materials and methods
Participant characteristics
The present randomized, double-blinded, prospective, controlled clinical trial was approved by the Ethics Committee of Shenzhen Maternity and Child Care Hospital, Southern Medical University (approval no. SZFY2018020798) and registered with the Chinese Clinical Registry Center (Registration no. ChiCTR-1800014943). The study was performed in accordance with the declaration of Helsinki (2008) and the Dutch Medical Research involving Human Subjects Act (WMO). Written informed consent was obtained from all participants.
All multipara who presented for vaginal delivery and requested analgesia in our hospital between January 2018 and February 2019 were recruited in our study. The inclusion criteria were as follows: 1) aged 18 to 45 years, 2) American Society of Anesthesiology physical status of I or II, 3) term pregnancy of a singleton fetus; 4) minimal cervical dilatation of least 3 cm, and 5) provided written informed consent. The exclusion criteria were as follows: 1) aged <18 years, 2) hypertension, endocrine diseases, or morbid obesity, 3) fetal abnormalities, 4) absolute contraindications or relative contraindications to spinal anesthesia, 5) unable to communicate, or 6) refused to take part in the trial. Patients who failed to undergo CSE block, whose epidural catheter was dislodged, or whose delivery progressed too rapidly (delivery within <120 min) were excluded from the final analysis.
Using a computer-generated random number table, 108 parturient women were randomly assigned to three groups: 1) Group C received l mL sodium (0.9%) intrathecally, 2) Group D received 5 µg Dex (Lot# 20180403; Enhua Pharmaceutical Co., Ltd., Jiangsu, China) intrathecally, and 3) Group S received 5 µg sufentanil (Lot# 1170912; Renfu Pharmaceutical Co., Ltd., Yichang, China) intrathecally. Then, all parturient women received patient-controlled epidural analgesia (PCEA, 0.1% ropivacaine + 0.2 µg/mL sufentanil). Maintenance doses of PCEA were administered using an Apon PCA pump (Jiangsu Apon Medical Technology Co., Ltd.), which was set at a rate of 7 mL/hour with a rescue bolus of 7 mL (lockout 25 min; limit 25 mL/hour). Group assignments were concealed in sealed envelopes until the anesthesia began, and the investigators and patients were unaware of the group assignments to ensure double blindness.
Study protocol
All procedures were performed by the same anesthetist group to eliminate possible effects of differences in anesthetic techniques. Vital signs, such as heart rate, blood pressure, SpO2, and respiratory rate, were monitored immediately after entering the delivery room and every 5 minutes until the end of labor. When cervical dilatation reached 3 cm, CSE block was performed at the L3/L4 intervertebral space.13,14 The epidural space was identified using the loss of resistance to saline with a 16G needle, and a 26G spinal needle was advanced through the epidural needle using the needle‐through‐needle technique. After intrathecal injection of the study agents, the spinal needle was withdrawn, and an epidural catheter was inserted into the epidural space.
Following the epidural injection of a test dose of 3 mL 1% lidocaine for 5 minutes, the Apon PCEA pump was connected to the epidural catheter. However, the epidural analgesia administration did not start until the visual analog scale (VAS) score was ≥3. The pump was set at a rate of 7 mL/hour with a rescue bolus of 7 mL (lockout 25 minutes; limit 25 mL/hour). Patients experiencing inadequate analgesia could request an additional 5 mL bolus of the medication solution via epidural administration by the nurse.
Before the analgesia began, 500 mL/hour compound sodium chloride was infused routinely. If hypotension (90/60 mmHg) occurred, the patient was placed in a left-leaning position and administered phenylephrine. If bradycardia (heart rate <60 bpm) occurred, the patient was treated with atropine.
Measurements
Demographic data, including age, height, weight, and gestational age, were recorded. The VAS score (0 = no pain, 10 = maximum pain) was evaluated at baseline (prior to CSE block) and 5, 10, 15, 20, 30, 40, 50, and 60 minutes after the administration of intrathecal drugs. The onset time was defined as the time from intrathecal administration to a VAS <3. The duration of intrathecal injection was defined as the time from intrathecal administration to the time when the epidural analgesia was started. The duration of the three labor stages (the first, the second, and the third stage of labor), Apgar score, umbilical vein pH, and cesarean delivery rate were recorded. Anesthetic consumption, including bolus frequency and total volume of anesthetic solutions, was also recorded. The Bromage scoring system was used to assess motor blockade (1, able to raise legs above table; 2, able to flex knees; 3, able to move feet only; and 4, no movement in legs or feet). 15 The Ramsay sedation scale (RSS) was used to assess the levels of sedation (1, patient anxious, agitated, or restless; 2, patient cooperative, oriented, and tranquil; 3, patient responds to commands; 4, patient asleep, shows a brisk response to a light glabellar tap or loud auditory stimulus; 5, patient asleep, shows a sluggish response to a light glabellar tap or loud auditory stimulus; and 6, patient asleep, no response to a light glabellar tap or loud auditory stimulus. 16 RSS values were recorded at 10, 30, and 60 minutes during labor.
In addition, adverse events, including hypotension (systolic blood pressure <90 mmHg or <30% of the base value), bradycardia (heart rate <60 bpm), nausea, vomiting, shivering, itching, or excessive sedation (RSS value >4), were also observed and recorded.
In the present study, the VAS score was the primary outcome, and the secondary outcomes included the onset time, Apgar score, umbilical vein pH, bolus frequency, total volume of anesthetic solution, Bromage score, RSS score, and adverse events.
Sample size
According to our preliminary data, the VAS score at 10 minutes after intrathecal injection [mean (standard deviation)] was 5.13 (1.78) in Group C, 3.46 (1.99) in Group D, and 3.19 (2.01) in Group S. A sample size of 30 patients in each group was determined with a statistical significance of 0.05 and a power of 95%. To compensate for possible lost or excluded cases, 36 patients were assigned to each group. The sample size was calculated using PASS 11 (www.ncss.com; NCSS, LLC., Kaysville, Utah, USA).
Statistical analysis
Statistical analyses were conducted using IBM SPSS Statistics for Windows, Version 20.0 (IBM Corp., Armonk, NY, USA). The normality of the quantitative data was assessed using the one‑sample Kolmogorov–Smirnov test. Continuous variables were expressed as the mean ± standard deviation, and categorical variables were expressed as numbers and percentages (n, %). One‑way ANOVA was used to analyze continuous variables, followed by the Bonferroni post hoc test. The χ2-test or Fisher’s exact test was used for categorical covariates.
Results
Patient characteristics
A total of 108 parturient women were recruited and assigned to three groups in this study. According to the protocol deviation, one participant was discontinued from the trial owing to a suspected dislodged epidural catheter. Finally, 107 parturient women were analyzed (Figure 1). There were no significant differences in the demographic variables among the three groups (Table 1).
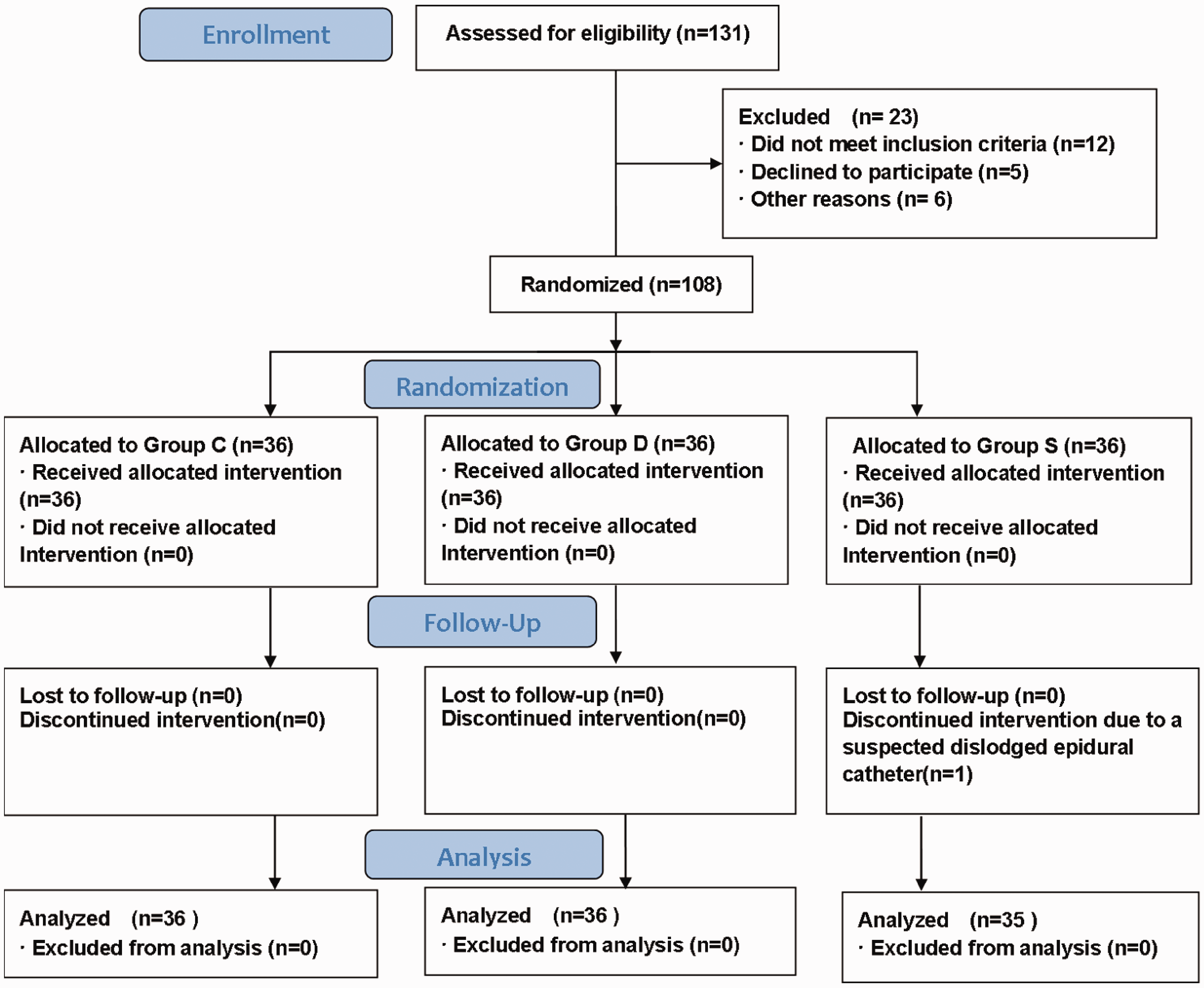
Consolidated Standards of Reporting Trials (CONSORT) flow chart.
Demographic baseline variables.
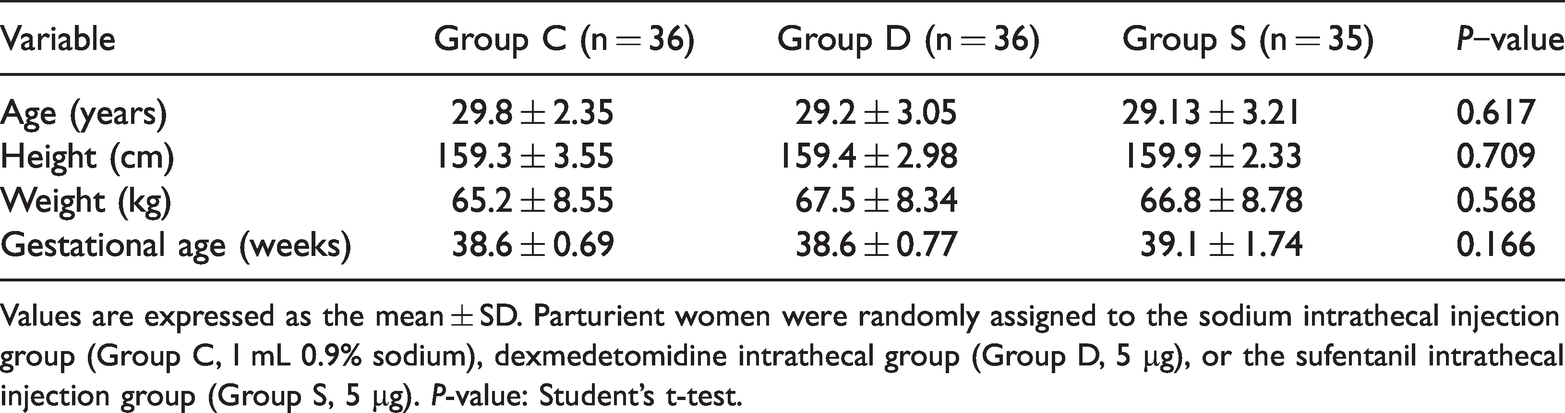
Values are expressed as the mean ± SD. Parturient women were randomly assigned to the sodium intrathecal injection group (Group C, l mL 0.9% sodium), dexmedetomidine intrathecal group (Group D, 5 µg), or the sufentanil intrathecal injection group (Group S, 5 µg).
Primary outcomes
The VAS scores were lower in Groups D and S than in Group C at 5,10, and 15 minutes after intrathecal drug injection (all
Visual Analog Scale score at different time points.

Values are expressed as the mean ± SD. Parturient women were randomly assigned to the sodium intrathecal injection group (Group C, l mL 0.9% sodium), dexmedetomidine intrathecal group (Group D, 5 µg), or the sufentanil intrathecal injection group (Group S, 5 µg).
Secondary outcomes
There was no significant difference in the relative factors among the parturient women in the three groups. These included the duration of each labor stage, the cesarean delivery rate, Bromage score, Apgar score, and umbilical vein pH (Table 3). Furthermore, no patient experienced insufficient analgesia or required additional 5 mL bolus analgesia.
Secondary outcomes.

Values are expressed as mean ± SD or numbers (percentage). Parturient women were randomly assigned to the sodium intrathecal injection group (Group C, l mL 0.9% sodium), dexmedetomidine intrathecal group (Group D, 5 µg), or the sufentanil intrathecal injection group (Group S, 5 µg). The Apgar Score was measured at 1 minute and again at 5 minutes after birth. Compared with Group C: a
Compared with Group C, the onset time was significantly shorter in Groups D and S (
Ramsay sedation scale

Data were presented as mean ± SD. Parturient women were randomly assigned to the sodium intrathecal injection group (Group C, l mL 0.9% sodium), dexmedetomidine intrathecal group (Group D, 5 µg), or the sufentanil intrathecal injection group (Group S, 5 µg). Compared with Group C: a
There was no significant difference among the three groups in the proportion of patients with hypotension, bradycardia, nausea, vomiting, and excessive sedation (Table 3).
Discussion
Persistent labor pain can be harmful to both maternal and fetal physiology by inducing stress pathways. 17 CSE block is an effective and routinely used technique for labor pain relief, which has the advantages of using low doses of local anesthetics and rapid onset of analgesia. 18 The most representative intrathecal adjuvants in clinical practice for labor analgesia are opioids and α2-adrenergic receptor agonists. 19 In this study, compared with the use of 5 µg sufentanil intrathecally combined with 0.1% epidural ropivacaine + 0.2 µg/mL sufentanil, the use of 5 µg Dex intrathecally combined with the epidural drugs described above offered similar pain relief during labor with less shivering and pruritus. Therefore, Dex is a good alternative in labor analgesia as an intrathecal drug combined with epidural analgesia.
Sufentanil has been used as a local anesthetic in spinal blocks for labor analgesia in many previous studies; 4 therefore, intrathecal injection of sufentanil combined with epidural ropivacaine and sufentanil was the positive control in the present study. Dex has also been reported to maintain stability, relieve pain, prevent chills without respiratory depression when used as an intrathecal drug,20,21 and improve intrathecal and epidural blocks.22,23 Mohamed et al. reported that Dex given intrathecally to patients undergoing major abdominal cancer surgery for postoperative analgesia could improve and prolong analgesia. 24 Liu et al. also reported that Dex used as an intrathecal drug for spinal anesthesia achieved prolonged sensory block, motor block, and the time to first analgesic request. 19 Moreover, Minagar et al. reported that Dex administered intrathecally prolonged analgesia and reduced postoperative pain without changes in the hemodynamic parameters and adverse effects. 25 To the best of our knowledge, this study was the first to explore the combination of intrathecal Dex or sufentanil with epidural ropivacaine and sufentanil for labor analgesia. In the present study, the VAS scores were lower at 5, 10, and 15 minutes in Groups D and S compared with Group C, and there were no significant differences in VAS scores at each time point between Groups D and S. The results indicated that using 5 µg Dex or sufentanil intrathecally combined with epidural ropivacaine and sufentanil could offer a similar improved analgesic effect compared with using saline intrathecally combined with the epidural drugs above.
Doses of Dex between 3 and 10 µg were described to be sufficient for spinal administration. 26 Both Dex (5 µg) and sufentanil (5 µg) have been used as intrathecal drugs in clinical practice.19,27 In the present study, the efficiency and safety of 5 µg Dex intrathecally combined with epidural ropivacaine and sufentanil for labor analgesia and the use of 5 µg sufentanil or saline intrathecally combined with the epidural drugs above were compared. The dosage used in the present study was determined according to a preliminary study, which indicated that intrathecal Dex (5 µg) was associated with improved analgesia. 25 Therefore, Dex at a dose of 5 µg was used as an intrathecal drug.
Kiran et al. reported that the onset time of intrathecal Dex was 5 minutes, which was shorter than our results. 28 The inconsistency could be explained by the different Dex doses used in each study. Compared with the use of saline, either 5 µg Dex or 5 µg sufentanil resulted in a faster onset time, improved analgesia, and reduced demand for local anesthetics. Furthermore, the use of 5 µg Dex was associated with fewer side effects compared with the same dose of sufentanil even though each group was given the same epidural drugs. The results indicated that Dex as an intrathecal drug combined with epidural anesthesia could provide rapid and effective analgesia, which was consistent with the clinical results previously reported.28,29
The analgesic effect of Dex is not fully understood. Zhang et al. 11 reported that intrathecal injection of Dex displayed a robust analgesic effect by inhibiting spinal extracellular signal-regulated protein kinase 1/2 signaling via an α2-receptor-dependent manner. Eisenach et al. 30 also reported that Dex produced antinociception rapidly after intrathecal injection and bound to α2‑receptors in the spinal cord. Yang et al. 31 reported that intraperitoneal Dex resulted in a dose-dependent analgesic effect due to the inhibition of hyperpolarization-activated cyclic nucleotide-gated channel currents. Recently, Li et al. 32 demonstrated that the analgesic effect of Dex is likely associated with the inhibition of protein kinase C expression in the spinal dorsal horn. These studies indicated that the analgesic effects of Dex were not only related to α2‑adrenergic receptors but also due to the direct inhibition of the signaling pathway via an α2‑independent mechanism. A systematic review and meta-analysis recently reported that Dex was effective and tolerated as an intrathecal drug. 33
Compared with sufentanil, 5 µg intrathecal Dex reduced the incidence of pruritus, which was consistent with a previous study. 28 Furthermore, the incidence of shivering after Dex was also lower than that of sufentanil in this study, similar to previous studies.20,34 Compared with the placebo, intrathecal Dex increased the sedation scale (10 minutes), which can be attributed to its action on the α2-receptors in the locus coeruleus. However, the sedation score of the control group at 60 minutes was higher than that of intrathecal Dex or sufentanil, possibly because of the elimination of the two drugs. There were no significant differences in the three stages of labor, Bromage score, umbilical vein pH, or Apgar score among the three groups, which was in line with previous studies.28,35
The present study had some limitations. First, the present study was a single‑center clinical trial; therefore, a large‑scale multicenter study is needed to verify the preliminary results. Furthermore, because the present study only investigated the efficiency and safety of 5 µg Dex as an intrathecal drug, further study should be conducted with different doses of intrathecal Dex. Third, one parturient woman in Group C was diagnosed with excessive sedation because she was exhausted after a long period of contractions. When the pain was relieved after CSE block, she rapidly fell into a deep sleep and responded slowly when we gently woke her to measure the RSS score. The results need further confirmation in the future. Finally, although intrathecal Dex is widely used in clinical practice and no obvious side effects have been reported, it is still not licensed for intrathecal use. Importantly, the safety of intrathecal Dex needs to be investigated in large‑scale phase IV clinical trials.
Conclusion
The present study investigated the effects of intrathecal 5 µg Dex or 5 µg sufentanil combined with 0.1% epidural ropivacaine and 0.2 µg/mL sufentanil for labor analgesia. Intrathecal Dex administration displayed improved analgesia, quicker onset times, and less demand for local anesthetics compared with saline administration. In addition, compared with sufentanil, intrathecal Dex administration decreased the incidence of pruritus and shivering. The results of the present study indicated that the use of intrathecal Dex increased the effectiveness and comfort of combined spinal-epidural analgesia in labor.
Footnotes
Availability of data and materials
The datasets used and/or analyzed during the present study are available from the corresponding author on reasonable request.
Author contributions
Conceptualization: Li Y, Huang X.
Data collection: Li G, Wang H, Qi X, Huang X.
Formal analysis: Li G, Li Y
Writing, original draft: Li G, Qi X,
Review and editing: Li Y, Huang X.
Declaration of conflicting interest
The authors declare that there is no conflict of interest.
Funding
This research received no specific grant from any funding agency in the public, commercial, or not-for-profit sectors.
