Abstract
Burkitt lymphoma is a very aggressive B-cell non-Hodgkin lymphoma. Although remarkable progress has been made in the therapeutic scenario for patients with Burkitt lymphoma, search and development of new effective anticancer agents to improve patient outcome and minimize toxicity has become an urgent issue. In this study, the antitumoral activity of Inula viscosa, a traditional herb obtained from plants collected on the Asinara Island, Italy, was evaluated in order to explore potential antineoplastic effects of its metabolites on Burkitt lymphoma. Raji human cell line was treated with increasing Inula viscosa extract concentration for cytotoxicity screening and subsequent establishment of cell cycle arrest and apoptosis. Moreover, gene expression profiles were performed to identify molecular mechanisms involved in the anticancer activities of this medical plant. The Inula viscosa extract exhibited powerful antiproliferative and cytotoxic activities on Raji cell line, showing a dose- and time-dependent decrease in cell viability, obtained by cell cycle arrest in the G2/M phase and an increase in cell apoptosis. The treatment with Inula viscosa caused downregulation of genes involved in cell cycle and proliferation (c-MYC, CCND1) and inhibition of cell apoptosis (BCL2, BCL2L1, BCL11A). The Inula viscosa extract causes strong anticancer effects on Burkitt lymphoma cell line. The molecular mechanisms underlying such antineoplastic activity are based on targeting and downregulation of genes involved in cell cycle and apoptosis. Our data suggest that Inula viscosa natural metabolites should be further exploited as potential antineoplastic agents against Burkitt lymphoma.
Keywords
Introduction
Non-Hodgkin lymphomas (NHLs) are a heterogeneous spectrum of tumors involving the immune system. Taken together, lymphoid neoplasms are the fourth most common tumors, representing a significant public health concern. 1 Even though they are potentially curable diseases, clinical features and survival depend on different factors including histology, biological aggressiveness, age, and stage of disease. Currently, intensive chemotherapy associated to targeted therapy with rituximab has improved treatment outcomes for some NHL subtypes. 2 However, some histological variants such as Burkitt lymphoma (BL) are still characterized by a poor outcome. In fact, in adult BL, a 5-year relapse-free survival, equal to 73%, has been reported in younger and highly selected cohorts, whereas worse results have been reported in the real-world experience with older patients and patients treated outside clinical trials; 3 these findings explain why the development of more effective or synergistic therapeutic approaches undoubtedly represents an unmet clinical need.
B-cell NHL is a paradigm of translocation-based cancers in which deregulated gene expression occurs because of specific balanced translocations of moving key genes under the influence of active lineage-specific promoters or enhancers. In BL, the chromosomal translocation t(8;14) (q24;q32) is observed in 70%–80% of patients. Accordingly, the overexpression and constitutive activation of the c-MYC oncogene is followed by the abnormal transcriptional regulation of downstream genes, resulting in cellular transformation, inhibition of cell cycle checkpoints, and resistance to apoptosis.3–6 Abnormalities of c-MYC oncogene occur in other lymphoma subtypes to confer an adverse prognosis, 7 and they are the main characteristic of B-cell acute lymphoblastic leukemia.8,9
Traditional herbal medicines are a powerful resource of biologically active natural products with strong therapeutic effects which could be used in the treatment of different diseases, including cancer. Inula viscosa, belonging to the genus Inula (Asteraceae), is a perennial plant considered as one of the most essential medicinal plants around the Mediterranean Basin. 10 Different plants of this genus are a source of sesquiterpenoids having a widespread spectrum of biological activities. Specifically, I. viscosa has been applied for a long time in folk medicine for its anti-inflammatory, 11 anthelmintic, antipyretic, antiseptic, and antiphlogistic activities12,13 and in the treatment of diabetes 14 and lung disorders. 15
In the recent past, different studies have elucidated the biological mechanisms underlying the anti-inflammatory effect of I. viscosa, which are based on the inhibition of COX1, COX2, and iNOS enzymatic activities.16,17 Recently, cytotoxic and anticancer activities of I. viscosa have been proved in cell lines of melanoma, 18 cervical cancer, 19 and breast cancer. 20
Interestingly, Li et al. 21 have identified one promising sesquiterpene lactone (SL) derived from a traditional plant, Inula japonica, displaying potent in vitro and in vivo anticancer activities against BL.
This study was planned to establish the potential antitumoral activity on Raji human cell line of the I. viscosa ethanolic extract obtained from plants collected on the Asinara Island, Italy. Furthermore, the aim of this study was to yield insights into the cytotoxic and molecular mechanisms involved in I. viscosa anticancer properties, therefore potentially contributing to the development of new therapeutic agents against BL.
Materials and methods
Plant material
I. viscosa was harvested during February 2018 in remote areas of the Asinara Island, Sardinia, Italy. The plant was authenticated by Prof. Giorgio Pintore, from the Department of Chemistry and Pharmacy, University of Sassari, Italy. The plant was deposited in the herbarium of the same department, under voucher no. 708. The aerial part (leaves and soft branches) of I. viscosa was placed in the shade and air-dried for a week at room temperature (RT), after which it was finely ground. In total, 20 g of powdered plant was extracted by absolute ethanol (100 mL). The obtained extract was subjected to evaporation under vacuum without exceeding the temperature of 40°C, thus obtaining dry residue, and conserved at 4°C until use.
Cell culture and drug treatments
Raji cells (ATCCCL-86, passage 11-25) were cultured in an RPMI 1640 medium (Thermo Fisher Scientific, Waltham, MA, USA), supplemented with 10% fetal bovine serum (FBS; Thermo Fisher Scientific) and 1% penicillin/streptomycin (Thermo Fisher Scientific). The cells were maintained at 37°C in a 95% humidified atmosphere and 5% CO2 in a T25 plastic culture flask. Experiments were performed exposing Raji cell line to increasing concentrations of I. viscosa ethanolic extract (5, 10, 20, 30, 40, 60, and 80 µg/mL) for 24 and 48 h. All experiments were performed in triplicate.
Trypan Blue assay
Raji cells were seeded at the concentration of 40.000/100 µL/well, treated with increasing concentrations of I. viscosa for 24 h as described previously. Subsequently, cell viability was assessed by Trypan Blue assay (0.4%; Sigma-Aldrich, Saint Louis, MO, USA), in which viable cells excluded the blue staining. At the end of the experiment, Trypan Blue was mixed 1:1 with cells and the count was performed using a Burker chamber. Results were expressed as the percentage of cell viability.
Apoptotic analysis
To perform apoptotic analysis, Raji cells were cultured in a six-well plate at the concentration of 1 × 106 cells/well/mL in an RPMI 1640 medium with 10% FBS and maintained at 37°C and 5% CO2. Cells were treated for 24 h with increasing concentrations of I. viscosa: 10, 20, and 30 µg/mL. All experiments were performed in triplicate. After 24 h of exposure, the cells were collected, washed twice with phosphate-buffered saline (PBS; 0.2 µm filtered, adjusted to pH 7.4) solution, and centrifuged at 1400 r/min for 10 min at 4°C. The pellets were re-suspended with binding buffer 1×, containing Annexin V FITC and propidium iodide (BD Pharmingen, San Diego, CA, USA), and incubated in the dark at RT for 15 min. After incubation, the samples were acquired by FACS Canto (FACS Canto II; Becton Dickinson, Franklin Lakes, NJ, USA). 22
Cells cycle analysis
Cell cycle analysis was performed seeding 1 × 106 Raji cells/well/mL treated with 10, 20, and 30 µg/mL of I. viscosa extract. After 24 h, the samples were fixed in 4% paraformaldehyde (PFA; Sigma-Aldrich) for 15 min at RT and then washed with PBS. After fixation, the cells were stained with purified monoclonal mouse Ki-67 antibody and Clone MM1 (Leica Biosystems, Wetzlar, Germany) in permeabilization buffer 1× (BD Pharmingen), washed twice in permeabilization buffer 1×, and incubated for 20 min with secondary antibody (1:200 anti-rabbit FITC conjugated; BD Pharmingen) in permeabilization buffer 1×. After incubation, the cells were washed and stained with 7-aminoactinomycin D (7-AAD; BD Pharmingen) and acquired by FACS Canto (Becton Dickinson). 22 A total of 30,000 events for each sample were acquired, and values are presented as mean ± standard deviation (SD) of the percentage of three independent experiments.
RNA extraction
Total RNA was obtained from 2 × 106 Raji cells treated with 10, 20, and 30 µg/mL of I. viscosa extract and untreated cells, as control. Nucleic acids were extracted with the commercially available RNeasy Lipid Tissue Mini Kit (Qiagen, Hilden, Germany) in accordance with the manufacturer’s instructions. RNA concentration and purity were assessed using the NanoDrop ND-1000 spectrophotometer (Thermo Fisher Scientific).
Quantitative real-time polymerase chain reaction
Total RNA at 2 µg was reverse transcribed to cDNA using the High Capacity cDNA Reverse Transcription Kit (Thermo Fisher Scientific), complying with the manufacturer’s instructions.
Primers for MYC (Hs00153408_m1, 107 bp), CCND1 (Hs 00765553_m1, 57 bp), BCL2 (Hs 00608023_m1, 81 bp), BCL2L1 (Hs 00236329_m1, 65 bp), BCL11A (Hs01093197_m1, 62 bp), and 18S rRNA (Hs99999901_S1, 187 bp) human genes were chosen using Assays-on-Demand™ Products (Thermo Fisher Scientific). Treated and untreated Raji cells were analyzed by quantitative real-time polymerase chain reaction (qRT-PCR), which was performed using TaqMan PCR chemistry and the ABI 7900HT Sequence Detection System (Thermo Fisher Scientific), as described previously. 23 Triplicate reactions were performed for each cDNA sample and the relative mRNA expression level was analyzed according to Applied Biosystems User Bulletin N°2. The 2–ΔΔCt calculation (fold change (FC)) was chosen to represent the level of expression, with FCs of more than 2 and less than 0.5 being considered as overexpression and downregulation, respectively.
Statistical analysis
Experiments are expressed as mean values (95% of confidence interval), and statistical significance between Raji cells treated with 10, 20, and 30 µg/mL of I. viscosa extract and untreated cells was set at p values < 0.05. Data analysis was performed by one-way analysis of variance (ANOVA) test using the GraphPad Prism 5.0 software (GraphPad Software, Inc., San Diego, CA, USA).
Results
Cytotoxicity of I. viscosa: Trypan Blue assay
Trypan Blue assay, performed to evaluate the potential toxicity of increasing concentrations of I. viscosa extract on Raji cell line after 24 h of exposure, allowed to observe a dose-dependent effect of the extract on cell viability at 24 h (Figure 1). The I. viscosa extract did not induce changes in cell viability at low concentrations (5 and 10 μg/mL), whereas it induced cytotoxic effects starting from the concentration of 20 μg/mL (p < 0.05), increasing proportionally the damage with higher concentrations. Based on the obtained data, we decided to use the concentration range of 10–30 μg/mL to study the possible anticancer effect of the I. viscosa alcoholic extract.

Cell viability analysis by Trypan Blue assay. Screening of increasing concentrations of Inula viscosa ethanolic extract on Raji cells at 24 h of exposure.
Apoptotic analysis
The total apoptosis induced by I. viscosa extract is presented in Figure 2(a). The different extract concentrations of 10, 20, and 30 µg/mL induced an increase in cell apoptosis by 40%, 66%, and 74%, respectively, when compared with the control group (CTRL; 10%, p < 0.001). The increase observed in cell apoptosis was in accordance with the proportional decrease in cell viability observed with the rise of the extract concentration (data not shown). Besides, Figure 2(b) shows the apoptotic rates of treated Raji cells measured by flow cytometry using Annexin V FITC in comparison with the CTRL.

Apoptotic analysis and Annexin V and 7-AAD staining of Raji cells after 24 h of Inula viscosa exposure. (a) Apoptosis of Raji cells evaluated after 24 h of incubation at 37°C in 5% CO2 condition. Data represent the percentage of total apoptotic cells (bar histogram); p < 0.001 versus Raji cells untreated (CTRL). (b) Dot spot graph of the apoptotic analysis representative one out of three independent experiments.
Cell cycle analysis
To evaluate the effects of I. viscosa extract on the mitogenic activity of Raji cell line, Ki-67 expression, a nuclear protein used as mitogenic index detected only in proliferative cells, was investigated (Figure 3(a)). Our results showed Ki-67 expression only in untreated cells. The cell cycle analysis was performed to evaluate the effects of I. viscosa extract on cell cycle distribution phases (Figure 3(b)). Our results revealed that all cells treated with I. viscosa extract at different concentrations were blocked in the G2/M phase. Specifically, an increase in cell percentage in the G2/M phase (88.7 ± 3.3) was observed using 10 µg/mL of I. viscosa, depending on accumulation induced by the treatment; however, the cells had the ability to grow (shown by Ki-67 expression). A progressive decrease in cell percentage in the G2/M phase was already observed at 20 µg/mL, reaching the highest value in treatment with 30 µg/mL extract, and was directly proportional to the increase in cell percentage under apoptosis. In fact, at 30 µg/mL, the percentage of apoptosis reached 90% ±2.5%, showing a statistically significant difference with untreated cells (CTRL vs 30 µg/mL; 2 ± 0.764 vs 90 ± 2.5; p < 0.001).
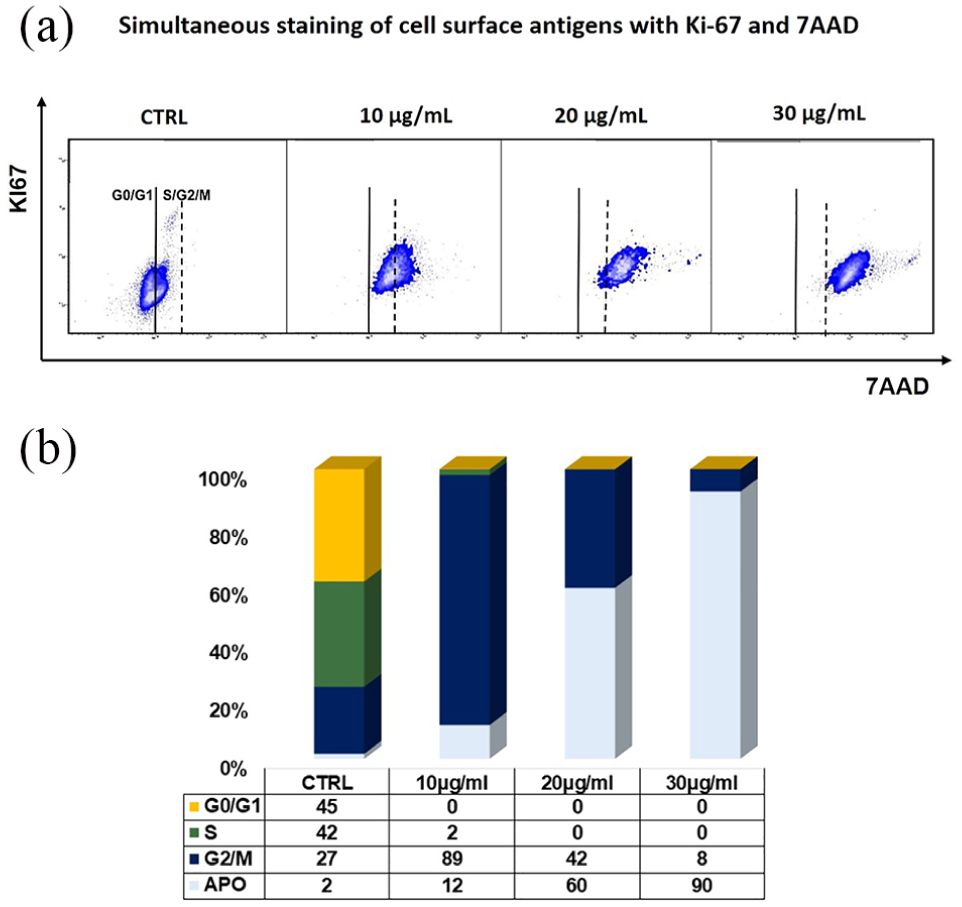
Flow cytometric analysis of Ki-67 and DNA content to analyze cell cycle status. (a) The bar graph describes the cell cycle phases identified by Ki-67 and 7-AAD staining. (b) The Burkitt lymphoma cells, treated with 10, 20, and 30 µg/mL of Inula viscosa for 24 h, have been blocked in the G2/M phase and moved into increasing apoptosis, depending on the concentration of Inula viscosa. All values are presented as mean ± SD of the percentage of three independent experiments.
Gene expression profile analysis
We further investigated whether I. viscosa ethanolic extract could reduce the c-MYC and CCND1 expression in Raji cell line, as the potential mechanisms responsible for cell cycle arrest. After 24 h of treatment with different concentrations of I. viscosa (10, 20, and 30 µg/mL), c-MYC and CCND1 expressions were strongly reduced. Specifically, FC evaluation for each treatment, compared with its untreated counterpart, showed downregulation levels of 23.4 and 12.9 FC for c-MYC and CCND1, respectively, when using 30 µg/mL of I. viscosa extract (p < 0.05). Moreover, expression levels in BCL2, BCL2L1, and BCL11A genes were analyzed to understand the possible role of I. viscosa extract on apoptosis activation, showing a downregulation of all genes, with FC variations of 20.7 for BCL2, 13.6 for BCL2L1, and 25.5 for BCL11A with 30 µg/mL of I. viscosa extract (p < 0.05). Figure 4 shows the qRT-PCR results.

Quantitative real-time PCR analysis for MYC, CCND1, BCL2, BCL2L1, and BCL11A genes. The bar graph describes the distribution of mRNA levels of MYC, CCND1, BCL2, BCL2L1, and BCL11A genes in Raji cells treated with 10, 20, and 30 µg/mL of Inula viscosa extract and untreated cells, as control. Data analysis was performed by one-way analysis of variance (ANOVA) test and considered significant when p values were <0.05.
Discussion
BL is scarcely chemosensitive, mainly in its sporadic and immunodeficiency-associated forms and in adult patients. However, a good response to chemotherapy combination can be obtained at the expense of significant associated toxicities and a high risk of developing tumor lysis syndrome.24,25
A number of chemical compounds characterize the chemical composition of I. viscosa, showing different biological activities. Several flavanones such as sakuranetin, 7-O-methylaromadendrin, and 3-acetyl-7-O-methylaromadendrin were isolated and tested by Hernández et al., 26 who highlighted good efficacy on enzymes involved in the inflammatory response. Therefore, shedding light on anticancer properties of this plant could be a relevant strategy to identify new anticancer agents. Our study reported that I. viscosa collected in an uncontaminated area of the National Park on Asinara Island, Sardinia, showed powerful antiproliferative and cytotoxic activities on Raji human cell line. The treatment of Raji cell lines with increasing concentrations of I. viscosa ethanolic extract showed a dose- and time-dependent decrease in cell viability, showing a reduction in cell proliferation, obtained by an induction of cell cycle arrest in the G2/M phase. Moreover, a dose-dependent increase in cell apoptosis was observed. Specifically, it was observed that cells treated at 10 µg/mL ethanolic extract concentration showed an extensive early apoptosis, whereas at 20 and 30 µg/mL concentrations dead cells were in late apoptosis.
Our results are supported by recent studies, in which several anticancer properties have been attributed to extracts from I. viscosa. Benbacer et al. 19 observed that I. viscosa induces cytotoxic effects on cervical cancer cell lines, by inhibiting proliferation and inducing caspase-dependent apoptosis by a mitochondria-mediated signaling pathway, along with a significant decrease in BCL2 expression. Interestingly, Merghoub et al. 27 have identified a telomerase inhibitory effect of I. viscosa extract able to induce apoptosis on cervical carcinoma cells and that I. viscosa active molecules overcome drug resistance in neoplastic cells. Recently, Bar-Shalom et al. 28 have demonstrated antiproliferative and proapoptotic effects of I. viscosa extract both in vitro and in vivo on colorectal cancer, without side effects in animal models.
In order to analyze the molecular mechanisms involved in I. viscosa anticancer activity, we performed a gene expression analysis on signal transduction and apoptotic pathway members involved in important B-lymphocyte functions. The overexpression of translocated c-MYC was established to play a crucial role in the pathogenesis and prognosis of BL. 29 c-MYC overexpression in BL is due to its translocation to one of the immunoglobulin loci and/or loss of regulatory elements by structural alterations within exon 1, then driving cell proliferation.30,31 Different studies showed that several compounds cause the arrest of proliferation and/or differentiation in Raji cell line by downregulation of c-MYC.32–34 In this study, it was shown that I. viscosa extract is able to strongly reduce c-MYC expression and consequently CCND1 expression, as its targeted gene. Kozar et al. 35 demonstrated that fibroblasts lacking all three D-type cyclins have very moderate defects in cell growth, although they cannot be transformed by MYC and RAS oncogenes. Moreover, cyclin D1 gene is bound by MYC and is induced in different cellular systems. 36
Considering the growth-promoting properties of c-MYC, the paradoxical effect of c-MYC as a potent inducer of apoptosis seems to correlate with different signals received by cells. In BL, this association might explain the typical starry sky appearance, which stems from a high rate of apoptotic cell death. However, specific cytokine survival factor or antiapoptotic proteins, such as those belonging to the BCL2 gene family, suppress the c-MYC apoptotic induction; BCL2 protects cultured cells from c-MYC-induced apoptosis, an activity that might explain their synergism in the induction of aggressive lymphomas in murine models.37,38
Interestingly, in these experiments, the Raji cell treated with I. viscosa displayed a strong downregulation of the antiapoptotic BCL2 gene family, such as BCL2 and BCL2L1, the proteins of which regulate mitochondria-mediated apoptotic pathways. High levels of BCL2 expression are associated with resistance to chemotherapy drugs in tumors and its downregulation is considered as a possible target for cancer therapy.39,40 Recently, Ding et al. 41 showed that using BCL2 siRNA combined with miR-15a oligonucleotides increases methotrexate-induced apoptosis.
Moreover, this study has also exhibited downregulation of BCL11A after the exposure of Raji cell to I. viscosa extract. BCL11A gene abnormalities have been identified in B-cell malignancies and in acute myeloid leukemia subgroups in humans.42–44 Gao et al. 45 have first shown that the suppression of BCL11A by siRNA induces apoptosis in diffuse large B-cell lymphoma and BL cell lines. The combination of BCL11A siRNA with BCL2 siRNA produces a greater induction of apoptotic death in these cell lines. Therefore, data from this work suggest the potential role of I. viscosa as a promising anti-BCL2 and anti-BCL11A therapeutic agent in B-cell lymphoma.
Used as a medicinal plant in different countries, I. viscosa has been characterized for its complex chemical composition, in order to identify bioactive compounds responsible for its biological activities.46,47 Recently, Kheyar-Kraouche et al. 48 analyzed an ethanolic extract from I. viscosa leaves growing in Algeria using high-performance liquid chromatography (HPLC) coupled to photodiode array detection and electrospray ionization mass spectrometry methodologies, and observed 51 phytochemical compounds including phenolic acids, flavonoids, lignans, and terpenoids, 26 of which were identified for the first time and belong to different families of compounds. Different biologically active compounds, belonging to several families, isolated from I. viscosa were tested alone or in association on cancer cell lines49–52 or in “in vivo”53,54 experiments showing strong antineoplastic activity.
Particularly, the SLs are alkylating agents, which cause DNA damage with consequent activation of ATM/ATR kinase and involvement of CDC2, TP53, survivin, and NF-kB, and could induce cell cycle arrest and apoptosis. 18 This mechanism characterizes tomentosin and inuviscolide (SL) purified from I. viscosa extract, which cause cell cycle arrest in the G2/M phase in melanoma cell lines, associated with the appearance of a sub-G0 fraction indicative of apoptotic cell death. 18 Moreover, Japonicone A (SL), derived from the traditional plant I. japonica, exerts anticancer activity against BL cell line. 21 Further studies by Merghoub et al. 55 demonstrated that the antineoplastic effects of I. viscosa in cervical cancer cells depend on tomentosin.
In summary, we demonstrated that I. viscosa ethanolic extract from I. viscosa leaves collected on Asinara Island has a potent antilymphoma activity through inhibition of cell proliferation and induction of cell apoptosis. The potential molecular mechanisms of I. viscosa extract include the downregulation of genes involved in the control of growth and survival pathways, deregulated in BL neoplastic cells. Based on our results and on the huge amount of data available in the literature, we hypothesize that I. viscosa extract contains different biologically active metabolites, with synergistic or additive antineoplastic effects. Further characterization of the plant used in our study might lead to identifying the active metabolites, analyzing their specific pharmacological and toxicological effects, and the functional molecular mechanisms involved, especially in “in vivo” models. Moreover, it will be of great interest to study them in association with common chemotherapy drugs applied in clinical treatment of BL, in order to foster the development of natural drugs with limited toxicity and potential chemotherapy efficiency for BL treatment.
Footnotes
Acknowledgements
L.P. and G.P. share the last authorship. A special thanks to the Asinara National Park, which allowed the collection of Inula viscosa. The authors appreciate Dr Giovanni Satta whose advice about pharmacological properties of Inula viscosa motivated the present study.
Author contributions
P.V., R.M., G.G., M.R.D.M., and L.P. conceived and designed the experiments. P.V., R.M., G.G., S.F., M.P.L.C., I.M., F.P.F., A.d.F., F.S., and M.R.M. performed the experiments. M.R.D.M., P.V., R.M., G.G., L.B., C.F., and L.P. analyzed the data. L.P. contributed the reagents/materials/analysis tools. M.R.D.M., P.V., R.M., L.B., and C.F. wrote the paper. G.P. and G.L.P. authenticated and prepared the plant material. All authors read and approved the final version of the manuscript.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by grants from Fondazione di Sardegna. The authors are grateful to “Associazione italiana contro le leucemie-linfomi e myeloma” (AIL) for the financial support for some reagents and for grant to P.V. and to “Fondazione Umberto Veronesi” for grant to F.P.F. The funders had no role in study design, data collection and analysis, decision to publish, or preparation of the manuscript.
