Abstract
Hepatoblastoma, the most common type of pediatric liver cancer, is treated with a combination of surgery and chemotherapy. An essential drug in the treatment of hepatoblastoma is doxorubicin, which in high doses is cardiotoxic. This adverse effect is due to downregulation of cardiac expression of transcription factor GATA4, leading in turn to diminished levels of anti-apoptotic BCL2 (B-cell lymphoma 2) protein family members. GATA4 is also expressed in early fetal liver, but absent from normal postnatal hepatocytes. However, GATA4 is highly expressed in hepatoblastoma tissue. In this study, we assessed the role of GATA4 in doxorubicin-induced apoptosis of hepatoblastoma cells. Herein, we demonstrate that doxorubicin decreases GATA4 expression and alters the expression pattern of BCL2 family members, most profoundly that of BCL2 and BAK, in the HUH6 hepatoblastoma cell line. Silencing of GATA4 by siRNA prior to doxorubicin treatment sensitizes HUH6 cells to the apoptotic effect of this drug by further shifting the balance of BCL2 family members to the pro-apoptotic direction. Specifically, expression levels of anti-apoptotic BCL2 were decreased and pro-apoptotic BID were increased after GATA4 silencing. On the whole, our results indicate that since high endogenous levels of transcription factor GATA4 likely protect hepatoblastoma cells from doxorubicin-induced apoptosis, these cells can be rendered more sensitive to the drug by downregulation of GATA4.
Introduction
Hepatoblastoma (HB) is a rare liver tumor with an incidence of approximately 1.2 per million individuals, but it accounts for >80% of liver malignancies in childhood. 1 The histology and gene expression profile of HB are reminiscent of fetal liver tissue; characteristic features include extramedullary hematopoiesis within the tumor and production of α-fetoprotein (AFP) by the malignant cells. 2 Serum AFP is currently the most reliable biological marker for HB diagnosis and tumor surveillance, and a complementary tool for predicting prognosis along with liver segment involvement. 3 Activating mutations in the β-catenin-encoding gene CTNNB1 have been implicated in HBs, 4 but also other pathways are likely involved in the pathogenesis of these tumors.
GATA transcription factors are highly conserved zinc-finger DNA-binding proteins. GATA1/2/3 are mainly expressed in hematopoietic cells, 5 whereas GATA4/5/6 are present in mesoderm- and endoderm-derived tissues. 6 GATA proteins are crucial for normal organogenesis and have also important regulatory functions in adult tissues. Alterations in the expression or function of GATA factors have also been linked to developmental disorders, malignancies, and other diseases.7,8
We previously reported that childhood HBs, in contrast to normal liver and hepatocellular carcinomas, abundantly express GATA4. 9 This transcription factor is crucial for the normal development of the mammalian liver. During the mouse embryogenesis, GATA4 is expressed in septum transversum and liver bud at embryonic day e9.5–e10.5. By e14.5, its expression within the developing liver is limited to endothelial cells of the veins, smooth muscle, and epithelial cells lining the liver. 10 GATA4 is also expressed in human fetal hepatocytes at gestational week 8, 11 but its expression declines by gestational week 12. 9 In normal human postnatal liver, GATA4 is expressed in Kupffer cells (liver macrophages), arterial smooth muscle cells, and venous endothelial cells. Another GATA transcription factor crucial for early liver development, but absent from normal postnatal hepatocytes, is GATA6. 12 GATA6 is also expressed in HBs, but to a lesser extent than GATA4. 9
The prognosis of children with HB has remarkably improved due to effective neoadjuvant chemotherapy regimens. 13 A commonly used combination, especially for high-risk patients, is cisplatin and doxorubicin (Dox). 14 Dox is an anthracycline that exerts its effects by intercalating into DNA, interfering with type II topoisomerases, generating free radicals, and inhibiting DNA and RNA synthesis. 15 A well-known side effect of Dox therapy is cardiomyopathy. 16 The molecular mechanisms behind this devastating side effect have been unraveled during the past decade, and GATA4, which is abundantly expressed in mammalian heart, 17 has been implicated in the pathogenesis of this process. Dox suppresses GATA4 DNA-binding activity and decreases GATA4 protein levels in cardiomyocytes.18–20 This decrease in GATA4 expression levels is exerted via caspase-1-mediated cleavage of GATA4 transcription activation domain. 21 Dox-induced apoptosis can be enhanced in cardiomyocytes by silencing GATA4 expression.19,22,23
GATA4 modulates cardiomyocyte apoptosis by directly regulating the expression of genes encoding the B-cell lymphoma 2 (BCL2) family of proteins.22,23 This group of more than 25 proteins includes both anti- and pro-apoptotic members of the intrinsic apoptosis pathway. 24 The anti-apoptotic family members include BCL2 and BCLXL, which inhibit the mitochondrial release of cytochrome c and caspase protein activation. 24 Overexpression of the anti-apoptotic BCL2 family members has been linked to malignant potential and chemotherapy resistance in several human hematologic and solid cancers. 25 Along with myocardium, GATA4 regulates BCL2 and BCLXL expression in other normal and neoplastic tissues including lung vascular smooth muscle 26 and ovarian granulosa cell tumors.22,27 In contrast to most human cell types, normal postnatal hepatocytes lack expression of anti-apoptotic BCL2, presumably due to the slow regeneration of liver tissue. 28 It has been shown that BCL2 is, however, expressed in a subset of HBs. 29 The HB cell line, HUH6, expresses high levels of both BCL2 and BCLXL, further implicating these apoptosis inhibitors in HB pathophysiology. 30 BH3-mimetic drugs, antagonists of the anti-apoptotic BCL2 molecules, have been shown to intensify the intrinsic apoptotic pathway, thus enhancing the effects of cytotoxic drugs, including Dox, in HB cells. 31
In addition to downregulation of the anti-apoptotic BCL2 proteins, Dox treatment has also been shown to activate pro-apoptotic BAK in lymphoma and myeloma cells.32,33 Furthermore, BAK overexpression in a hepatoma cell line sensitizes the cells to Dox-induced apoptosis. 33 BAK expression has also been demonstrated in HB cells. 31 BID is another pro-apoptotic member of BCL2 protein family. The cleavage of BID and its translocation from cytosol to mitochondria is a part of the Fas apoptosis signaling pathway. BID is expressed in normal hepatocytes, and liver-specific BID deficiency protects murine hepatocytes from apoptosis. 34
We hypothesized that GATA4 promotes HB cell survival by acting as an anti-apoptotic factor. The present results show that GATA4 downregulation sensitizes HB cells to Dox-induced apoptosis. GATA4 exerts its effects by regulating the balance between pro- and anti-apoptotic members of the BCL2 protein family by altering the expression levels of BCL2 and BID.
Materials and methods
Cell culture and treatments
An established cell line HUH6, derived from human mixed epithelial/mesenchymal HB, was obtained directly from Health Science Research Bank (Osaka, Japan). Cells were cultured at 37°C in Dulbecco’s Modified Eagle’s Medium (DMEM) (Lonza, Basel, Switzerland) supplemented with 10% fetal calf serum, 2 mM L-glutamine, 100 U/mL penicillin, and 100 µg/mL streptomycin sulfate. The cells were incubated for 48 h with Dox (50–200 ng/mL, Sandoz, Holzkirchen, Germany) diluted in DMEM. It has been demonstrated previously with HUH6 cells that 50% growth inhibition is reached with 200 ng/mL concentration. 35 DMEM only (Dox 0 ng/mL) served as control.
SiRNA transfections
About 70%–80% confluent HUH6 cells were transfected in the absence of antibiotics with a pool of four small interfering RNAs (siRNAs) targeting GATA4 (siGENOME SMART pool reagent) or a non-targeting (Nt) control siRNA (both from Dharmacon, GE Healthcare, Lafayette, CO). GATA4 target sequences were as follows: GGACAUAAUCACUGCGUAA, CGAUAUGUUUGACGACUUC, GAACCUGAAUAAAUCUAAG, UAUCAGAGCUUGGCCAUGG. Lipofectamine RNAiMAX reagent in Opti-MEM medium (both from Life Technologies, Carlsbad, CA) was used for transfections. Final concentration of siRNA was 0.1 µM. After 6 h of incubation with siRNA, normal growth medium was added to stop the transfection. Cells were lysed 72 h after beginning of transfection, and transfection efficacy was measured with quantitative real-time polymerase chain reaction (qRT-PCR) and western blotting. With this method, we reached an approximately 70% reduction in GATA4 messenger RNA (mRNA) levels (Figure 3(a) and (e)).
Adenoviral infections
For adenoviral infections, HUH6 cells were plated on 96-well plates 24 h before transfection. The cells were infected by incubating them with viruses in DMEM containing 0% fetal bovine serum (FBS). The replication-deficient adenoviral construct expressing wild type (WT) rat GATA4 has been previously described. 36 Multiplicity of infection (MOI) used was 40. After 1 h, DMEM with 10% FBS was added to stop the infection. Twenty-four hours after infection, Dox treatment and subsequent analysis were started.
Western blotting
Western blotting was performed as previously described. 27 Primary antibodies used were as follows: goat anti-human GATA4 IgG (sc-1237, Santa Cruz Biotechnologies, Dallas, TX) at dilution 1:1000, mouse anti-human BCL2 IgG (MO887, Dako, Glostrup, Denmark) at dilution 1:500, rabbit anti-human BCLXL IgG (2762S, Cell Signaling Technologies, Danvers, MA) at dilution 1:500, rabbit anti-human BAK IgG (sc-832, Santa Cruz Biotechnologies) at dilution 1:200, mouse anti-human BID IgG (sc-373939, Santa Cruz Biotechnologies) at dilution 1:500, and goat anti-human β-actin IgG (sc-1616, Santa Cruz Biotechnologies) at dilution 1:10,000.
Quantitative real-time PCR
Total RNA was extracted from cell line samples using the NucleoSpin RNA/Protein kit (Macherey-Nagel, Düren, Germany). Reverse transcription reaction was performed by Reverse Transcriptase Core Kit and qRT-PCR by MESA GREEN qPCR MasterMix Plus for SYBR Assay (both from Eurogentec, Seraing, Belgium) according to manufacturer’s instructions. Cyclophilin G served as a reference gene. Primer pairs used are listed in S1 Table.
Apoptosis and cell viability assays
Apoptosis was analyzed from HUH6 cells utilizing Caspase-Glo 3/7 Assay (Promega Corporation, Madison, WI) following manufacturer’s instructions. WST-1 assay (Roche, Basel, Switzerland), based on the cleavage of the tetrazolium salts by mitochondrial dehydrogenases present in viable cells, was utilized for cell viability analyses according to the manufacturer’s instructions. Both experiments were repeated three times, and absolute luminescence values were normalized to control wells containing untreated HUH6 cells.
Statistical analysis of the data
All experiments were performed in triplicate and statistically analyzed with JMP software (JMP, Cary, NC) using one-way analysis of variance (ANOVA), followed by comparison to control using Dunnett’s test. Value of P < 0.05 was considered significant in all experiments.
Results
Expression of BCL2 family members in HUH6 human HB cells after Dox treatment
First, we analyzed the effects of Dox on HUH6 cell viability, apoptosis, and the expression of selected genes of the BCL2 family. Consistent with a previous report, 30 we observed a statistically significant decrease in HUH6 cell viability (Supplemental Figure 1(a)) and an increase in apoptosis (Supplemental Figure 1(b)) after 48 h of treatment with increasing doses of Dox. Nearly all the cells were detached from the plate when a Dox concentration of 200 ng/mL was used; therefore, concentrations ≤ 100 ng/mL were used in subsequent experiments. After 48-h Dox treatment at concentrations of 50–100 ng/mL, we observed a significant decrease in anti-apoptotic BCL2 (Figure 1(a)). Expression of another anti-apoptotic factor BCLXL was not altered, but there was a significant increase in pro-apoptotic BAK expression (Figure 1(a)). After 72 h of treatment, changes in BCL2 and BAK expression were still evident with a concurrent increase in BCLXL expression (Figure 1(b)). At the protein level, no alterations in BCLXL expression were observed at 72 h, but a decrease in BCL2 expression and an increase in BAK expression were evident (Figure 1(c)). Expression of other BCL2 family members (BID, BAD, BAX, BCL-W) was not altered after Dox treatment (data not shown).
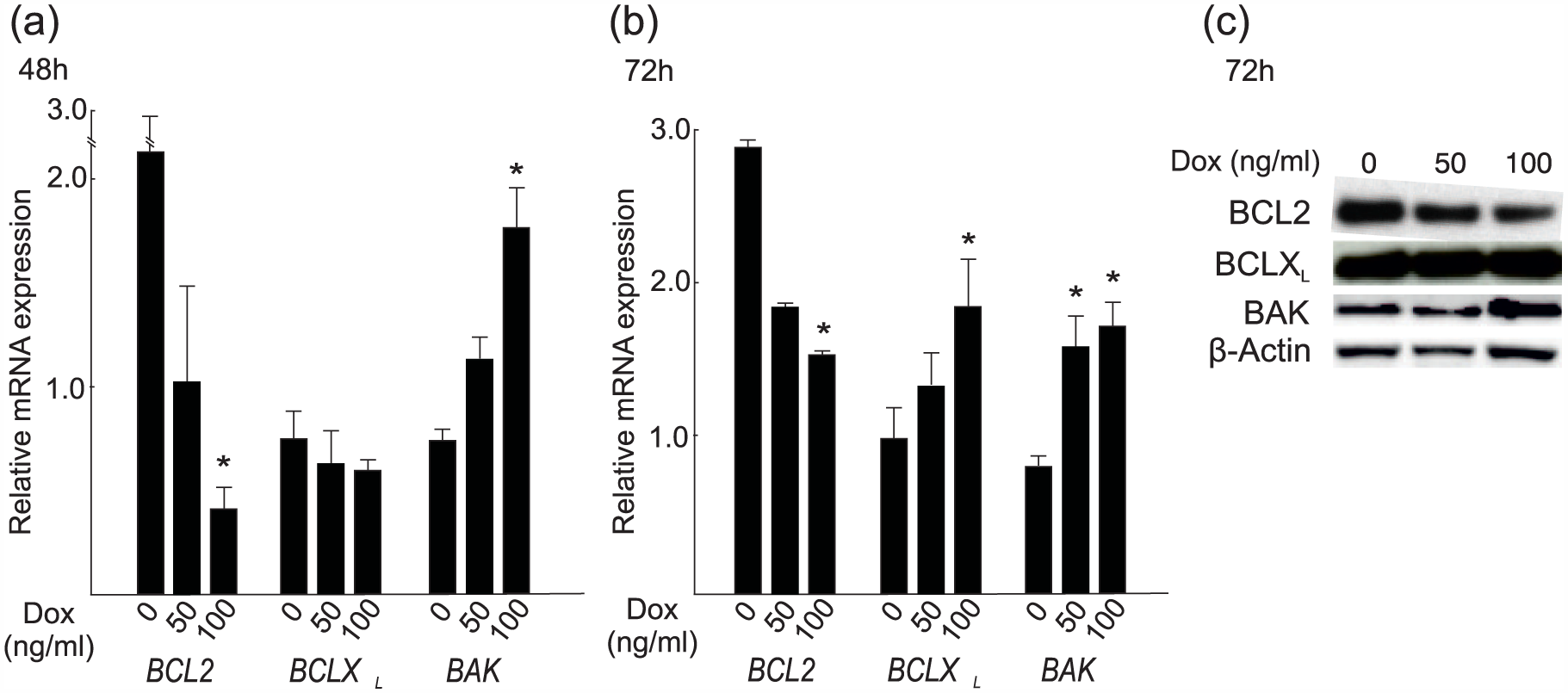
Doxorubicin alters the expression of BCL2 and BCLXL in HUH6 cells. BCL2, BCLXL, and BAK mRNA after (a) 48 h and (b) 72 h of Dox treatment. (c) Western blotting showing protein expression after 72 h of treatment. Dox concentrations 0–100 ng/mL. *p < 0.05 compared to Dox 0 ng/mL.
GATA4 silencing alters expression of BCL2 family members in HUH6 cells
Given that GATA4 is an upstream regulator of BCL2 and BCLXL in some other cell types including cardiomyocytes, ovarian granulosa cell tumor cells, and lung vascular smooth muscle cells,22,27 we hypothesized that the alterations in their expression in HUH6 cells during Dox treatment are due to a reduction in GATA4 expression. To this end, we first studied whether Dox treatment affects GATA4 levels in the human HUH6 cells. In these experiments, we measured GATA4 expression in HUH6 cells after Dox administration and found that Dox treatment significantly reduces the expression of GATA4 mRNA and protein in the cells after 48 h and 72 h (Figure 2).

Doxorubicin reduces GATA4 expression in HUH6 cells. (a) GATA4 mRNA expression after 48 h and 72 h of Dox treatment. *p < 0.05 compared to Dox 0 ng/mL. (b) Western blotting showing GATA4 protein expression after 72 h of Dox treatment (β-actin serves as a loading control). Dox concentrations 0–100 ng/mL.
We next wanted to analyze the effects of a profound GATA4 downregulation in HUH6 cells and used siRNA approach for this purpose. GATA4 siRNA reduced the levels of GATA4 mRNA by 70% compared to Nt siRNA (Figure 3(a)). We did not find statistically significant differences in cell viability or apoptosis between GATA4 siRNA– and Nt siRNA–treated cells (Figure 3(b) and (c)). This is in line with our previous findings with a GATA4-dominant negative mutant in HUH6 cells. 9 We, however, found a significant decrease in the expression levels of anti-apoptotic BCL2 (Figure 3(d) and (e)) in GATA4-silenced HUH6 cells. Furthermore, the expression of the pro-apoptotic BCL2 family member BID increased at both mRNA and protein level in response to GATA4 siRNA treatment (Figure 3(d) and (e)). Expression of BCLXL and several other pro- or anti-apoptotic members of the BCL2 family (BAD, BAX, BAK, BCL-W) remained unaltered after GATA4 siRNA transfections (Figure 3(d) and (e), and data not shown).

Effect of GATA4 silencing on HUH6 cell viability, apoptosis, and expression of BCL2 protein family members. (a) GATA4 mRNA expression, (b) cell viability and (c) caspase 3/7 activity, AND (d) BCL2, BCLXL, and BID mRNA expression 72 h after transfection with non-targeting control siRNA (Nt) and GATA4 siRNA (G4). *p < 0.05 compared to Nt. (e) Western blotting showing GATA4, BCL2, BCLXL, and BID protein expression 72 h after transfection (β-actin serves as a loading control).
Silencing GATA4 enhances the apoptotic effect of Dox in HUH6 cells
Next, we studied whether silencing of GATA4 and treatment with Dox have a synergistic effect on HUH6 cell apoptosis. In these experiments, HUH6 cells were treated with GATA4 siRNA 24 h after plating, and Dox was administered 48 h after plating. After 48 h of Dox and 72 h of siRNA treatment, cell viability and apoptosis were measured. The number of viable cells was decreased in GATA4 siRNA–treated cells versus non-targeted siRNA cells with all Dox concentrations including baseline, but reached statistical significance only with the concentration of 100 ng/mL (Figure 4(a)). Cells devoid of GATA4 underwent Dox-induced apoptosis more readily than control cells, and the differences in the apoptosis rates were highest at a Dox concentration of 100 ng/mL (Figure 4(b)).
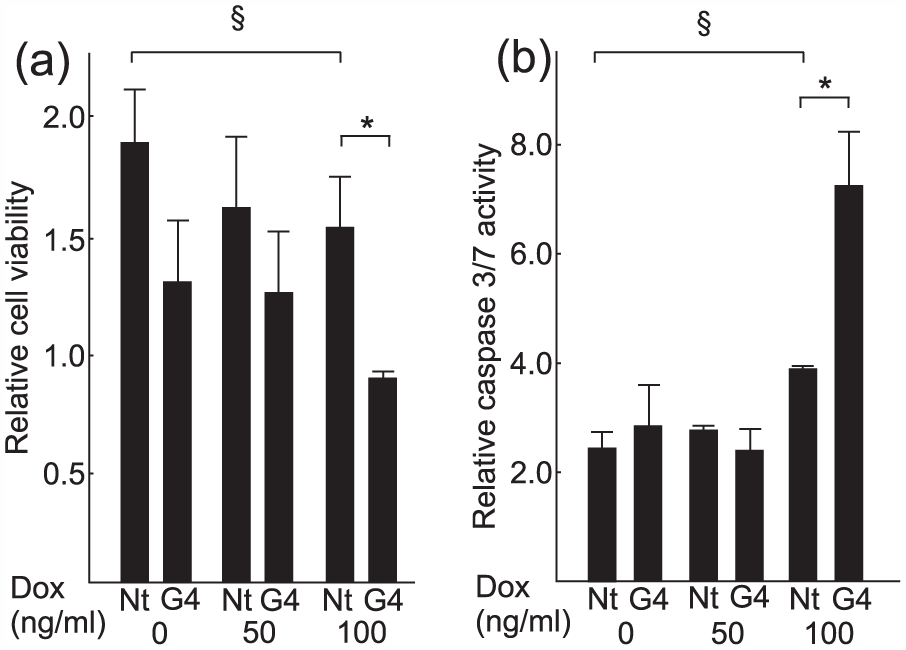
Inhibition of GATA4 expression with siRNA enhances doxorubicin-induced apoptosis in HUH6 cells. (a) Cell viability and (b) apoptosis at 72 h after siRNA transfection (GATA4 and non-target) and after 48 h of Dox treatment (concentrations 0–100 ng/mL). *p < 0.05 compared to Nt. § = p < 0.05 compared to Dox 0 ng/mL.
We also performed analogous experiments with GATA4-overexpressing adenoviral constructs to find out whether increasing the amount of GATA4 could protect the cells from Dox-induced apoptosis. There was no difference in the baseline or Dox-treated apoptosis rates after the overexpression of GATA4 (data not shown). Given the high endogenous GATA4 expression by HUH6 cells, this result was expected, since increasing the amount of GATA4 protein further is unlikely to add more to its function.
Taken together, our results reveal that although Dox itself reduces GATA4 levels in HB cells, further silencing of GATA4 with siRNA intensifies the apoptotic effect of this drug.
Discussion
The present work demonstrates that Dox treatment of human HB cells leads to downregulation of GATA4 and enhanced apoptosis. The effects of Dox can be augmented using siRNA constructs targeting GATA4. Downregulation of GATA4 shifts the balance of expression levels of BCL2 family members to a pro-apoptotic direction, thereby enhancing apoptosis of these malignant cells. Collectively, the results suggest that the high GATA4 expression in HB cells protects these cells from the apoptotic effects of Dox.
It is well known that Dox has several mechanisms through which it exerts its toxic effects. 15 As with other cell types32,37 treatment of HUH6 cells with Dox alone decreased the expression of anti-apoptotic BCL2 and increased the expression of pro-apoptotic BAK. The decrease in BCL2 levels by Dox treatment was accompanied by a delayed reciprocal rise in BCLXL expression. This is in accordance with earlier findings indicating that the anti-apototic BCL2 family members have functional redundancy in other malignant tissues, such as testicular germ cell tumors, melanoma, and colon cancer.38–40
In this study, we tested whether GATA4 protects HB cells from Dox-induced apoptosis by regulating BCL2 family members. Transcription factor GATA4 is absent from normal postnatal hepatocytes, but it is expressed in the majority of HBs. 9 The functional significance of GATA4 in these tumors, however, has remained elusive. Like HBs, ovarian granulosa cell tumors exhibit high levels of GATA4 expression that promote tumor cell survival.36,41 GATA4 also protects normal murine cardiomyocytes from Dox toxicity, and this protective effect is mediated through activation of Bcl2 23 and BclXL.22,23 The binding of GATA4 on Bcl2 promoter in these cells has been demonstrated by electrophoretic mobility shift and chromatin immunoprecipitation assays. 23 Of interest, transactivation studies have shown that GATA4 is a direct regulator of BCL2 also in human granulosa cell tumor cells. 27 In keeping with the aforementioned studies on other cell types, this study revealed that HB cells can be sensitized to Dox by downregulating GATA4 and changing the balance between the pro- and anti-apoptotic BCL2 family members. Our findings implicate BCL2 and BID as the likely targets of GATA4 action in HB cells. Future studies including chip-on-chip assays will ultimately address if the effect of GATA4 on HB cell survival is indeed mediated through the direct binding of this transcription factor to the regulatory regions of these genes in the BCL2 family.
Dox treatment itself is able to somewhat decrease GATA4 expression in HUH6 cells, suggesting that its effects are in part mediated through GATA4 depletion and consequent alterations in the expression of apoptosis-regulating genes. When GATA4 is further silenced with siRNA, the apoptotic effect of Dox is intensified. The effects of Dox and GATA4 on the BCL2 family members and apoptosis in HB cells studied herein are summarized in Figure 5. However, it is likely that effectors/molecules other than BCL2 family members are also involved in GATA4-regulated apoptotic pathways in HB cells.

Schematic model of the effects of (a) doxorubicin and (b) doxorubicin with GATA4 siRNA on the BCL2 protein family expression and apoptosis in HUH6 cells.
HBs are a heterogeneous group of tumors varying in histology and gene expression, and their treatment will likely be more individualized in the future. To achieve this goal, the pathophysiological mechanisms and molecular pathways affected by various treatment approaches need to be elucidated. We have herein identified GATA4 as one factor affecting the response of HB cells to cytostatic treatment. If GATA4 expression levels could be manipulated in HB cells also in vivo, the effects of Dox treatment could be intensified. GATA4-regulated pathways may also offer putative targets for future HB therapy.
Footnotes
Acknowledgements
We thank Drs. Noora Andersson and Jussi Merenmies for critical reading of the manuscript and Ms. Taru Jokinen for technical assistance.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This study was funded by Emil Aaltonen Foundation, Finland; Finnish Cancer Organizations, Finland; Finnish Pediatric Research Foundation, Finland; Helsinki University Central Hospital Research Grants, Finland; and Sigrid Jusélius Foundation, Finland.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
