Abstract
Epithelial ovarian neoplasms are a heterogeneous group including tumor subsets with distinct clinicopathologic and molecular features. Recent evidence from molecular and genomic studies suggests that whereas low-grade serous carcinomas and high-grade serous carcinomas likely develop on two separate pathways, the low-grade serous carcinomas and serous borderline ovarian tumors may represent various stages of the same developmental continuum. The transformation of borderline ovarian tumors into an invasive neoplasm is associated with an array of molecular changes, inter alia controlled by p53 and PI3K/Akt pathway, as well as with a decrease in E-cadherin expression. The latter implies that epithelial–mesenchymal transition is a critical determinant of borderline ovarian tumor invasiveness. The aim of this study was to analyze the expression of transcription factors involved in epithelial–mesenchymal transition: SNAIL, SLUG, TWIST 1, TWIST 2, ZEB 1, and ZEB 2 in borderline tumors and type I ovarian cancers. The study included tissue specimens from 42 patients with histopathologically verified ovarian masses. The expressions for SLUG, TWIST 1, ZEB1, and ZEB 2 were scored based on the nuclear staining, and the expressions of SNAIL and TWIST 2 based on the cytoplasmic and/or nuclear staining. The proportions of ovarian tumors with the immunoexpression of the epithelial–mesenchymal transition transcription factors were 85.7% for SNAIL, 100% for SLUG, 9.5% for TWIST 1, 95.2% for TWIST 2, 23.8% for ZEB 1, and 0% for ZEB 2. The expression patterns of SNAIL, SLUG, TWIST, and ZEB identified in this study suggest that both serous borderline ovarian tumors and type I ovarian cancers undergo dynamic epithelial–mesenchymal interconversions. Our findings obtained in the two groups of tumors which shared some etiopathogenic pathways imply that the expression of the epithelial–mesenchymal transition transcription factors may be activated at early stages of the epithelial–mesenchymal transition, and thus these molecules may play a pivotal role in the development of both serous borderline ovarian tumors and type I ovarian cancer.
Keywords
Introduction
Borderline ovarian tumors (BOTs) represent approximately 10%–20% of all epithelial ovarian masses; the vast majority of BOTs (96%) are serous or mucinous lesions. 1 Serous borderline ovarian tumor (sBOT) is characterized by the presence of hierarchical arborizing edematous papillae, focally covered by stratified epithelium with variable nuclear atypia and few mitoses, as well as by the absence of destructive stromal invasion. 2 BOTs may be precursor lesions for low-grade type I ovarian cancers. 3 The new classification of ovarian malignancies is based primarily on their molecular and genetic characteristics, as well as on the relationship of the latter with clinical and biological properties of the tumors. In line with those criteria, Kurman and Shih 4 classified ovarian malignancies into two types. Type I cancers carry multiple genetic mutations and show relatively high genetic stability. The genes that most commonly undergo mutations in type I ovarian cancers are BRAF, KRAS, and PTEN.5,6 Histopathologically, type I ovarian tumors represent low-grade serous carcinomas (LGSCs), endometrioid carcinomas, mucinous carcinomas, and clear-cell carcinomas. 7 Type II ovarian tumors have a more aggressive phenotype, are genetically instable, and harbor mutations in p53 gene. The most commonly found histopathological variant of type II ovarian tumor is high-grade serous carcinoma (HGSC), which accounts for 70% of all epithelial ovarian malignancies. 8
Epithelial–mesenchymal transition (EMT) is a reversible cellular process, during which the epithelial cells depolarize, lose cell–cell contacts, and gain a spindle–mesenchymal morphology. EMT is characterized by the loss of epithelial morphology and the cytoskeletal reorganization due to which the cells gain migratory potential and become more invasive. 9 EMT is essential for embryonic development and wound healing. During the follicular rupture and at subsequent stages of the ovarian remodeling, the surface epithelium can transform back and forth between the epithelial and mesenchymal phenotypes. 10 EMT may also contribute to a greater invasiveness of tumor cells, which acquire the properties of cancer stem–like cells and secondary tumor-initiating cells, and become chemoresistant.11–13 In solid tumors, the loss of cell–cell adhesion results in a disruption of normal tissue architecture, promoting progression and dissemination of the malignancy. Recent studies demonstrated that EMT plays a critical role in the progression of ovarian carcinomas.14–16 In ovarian malignancies, EMT contributes to greater mobility, invasiveness, and metastatic potential of cancer cells, which gain stem cell–like properties, such as the upregulation of stem cell genes (CD44 and CD133) and the ability to self-renew.17,18 The loss of E-cadherin expression is considered a fundamental event in the EMT. A number of transcription factors, among them SNAIL, SLUG, TWIST, and ZEB, play important roles in EMT regulation, contributing to a decrease in E-cadherin expression.9,19,20 Moreover, EMT is mediated by an array of signaling pathways, among them RTKs, Wnt, TGF-β, and NF-κB, due to the activation of various master transcription regulators from the SNAIL, SLUG, TWIST, and ZEB families. 21 Accurate identification of the tumor subtype based on its morphology and, whenever necessary, also on the basis of its genetic and immunohistochemical profile, is an imperative. Furthermore, it has been suggested that the development of effective strategies to inhibit tumor progression and spread requires better understanding of cellular and molecular pathways present within its microenvironment.
While the relationship between the EMT and the development of an aggressive phenotype in type II ovarian cancer has been studied extensively, still little is known about the role of this process in the etiopathogenesis of borderline tumors and low-grade ovarian carcinomas. The aim of this study was to analyze the expression of the transcription factors involved in EMT: SNAIL, SLUG, TWIST 1, TWIST 2, ZEB 1, and ZEB 2 in borderline tumors and type I ovarian cancers; moreover, we verified whether the expression of the EMT factors correlated with morphological and clinical characteristics of the ovarian malignancies.
Methods
The study included tissue specimens from 42 patients with histopathologically verified ovarian masses, who had been operated on at the Department of Obstetrics and Gynecology, Nicolaus Copernicus University, Collegium Medicum in Bydgoszcz (Poland) between January 2009 and June 2012. The patients underwent surgical resection adequate for a clinical stage of their malignancy and, whenever necessary, received adjuvant platinum-based chemotherapy in line with the current Polish guidelines. 22 Based on histopathological examination of surgical specimens, 8 lesions were classified as sBOTs and 34 as low-grade ovarian carcinomas. The probability of 5-year survival was analyzed in relation to clinicopathological features. Clinicopathological characteristics of the study subjects are summarized in Table 1.
Clinicopathological characteristics of the study subjects.

Analysis of formalin-fixed, paraffin-embedded (FFPE) tissue specimens was carried out at the Department of Clinical Pathology, Nicolaus Copernicus University, Collegium Medicum in Bydgoszcz.
Tissue macroarrays
Representative areas of each tumor were chosen from archival tissue material. Tissue fragments from the donor blocks were cut out and transferred to previously designed and prepared acceptor blocks. Subsequently, the tissues were re-embedded in paraffin, to obtain recipient blocks. Each donor block contained material from five ovarian cancer patients.
The material was cut into 3-µm paraffin sections, using a rotary microtome (Accu-Cut® SRMTM200; Sakura, Torrance, CA, USA). The sections were mounted on microscopic slides providing superior adhesion (“Superfrost Plus”, Menzel Glasser, Braunschweig, Germany) and left for 60 min on a heating plate set at 58°C.
Immunohistochemistry
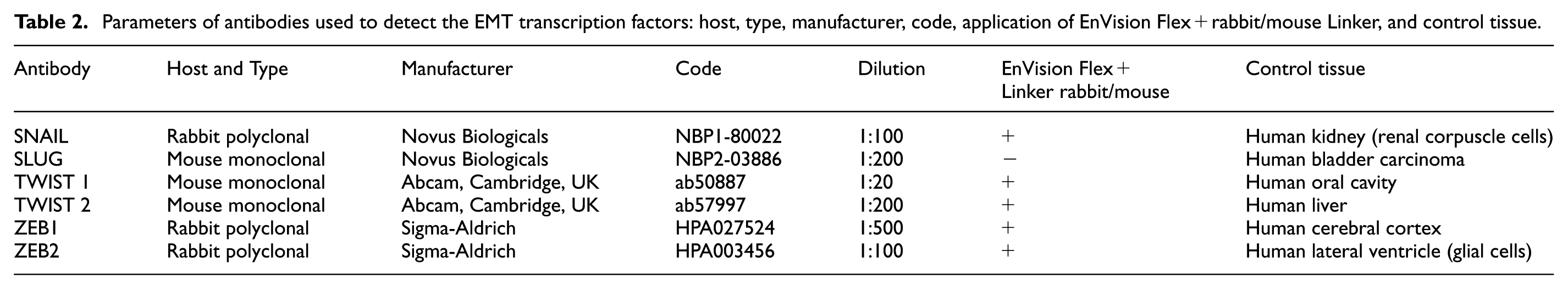
For deparaffinization, rehydration, and antigen retrieval, paraffin sections were pre-treated with a high-pH buffer (Epitope Retrieval Solution) in an automated PT-link system (Dako; Agilent Technologies, Inc., Santa Clara, CA, USA). Then, endogenous peroxidase activity and non-specific binding were blocked by 10 min incubation with 3% H2O2 and 15 min incubation with bovine serum albumin (BSA) at room temperature (RT). Subsequently, the tissue sections were incubated for 30 min with primary rabbit/mouse antibodies (Table 2) at room temperature. In the case of some antibodies, EnVision FLEX + Rabbit/Mouse (Linker; Agilent Technologies, Inc.) was used to enhance the immunohistochemistry (IHC) reaction. Antigen–antibody complexes were detected by a 20 min incubation with EnVision FLEX + HRP (Dako; Agilent Technologies, Inc.) at RT, using 3-3′ diaminobenzidine (DAB) as a chromogen. Finally, the slides were counterstained with hematoxylin, dehydrated in alcohol gradient, cleared in xylene, and mounted (Dako; Agilent Technologies, Inc.).
Parameters of antibodies used to detect the EMT transcription factors: host, type, manufacturer, code, application of EnVision Flex + rabbit/mouse Linker, and control tissue.
Evaluation of immunohistochemical reactions
The antibody-labeled slides were evaluated by two independent pathologists, using ECLIPSE E800 light microscope (Nikon Instruments Europe, Amsterdam, Netherlands) with a low-power objective (×20). The immunohistochemical expression in the ovarian tissues from the tissue macroarrays was quantified based on the Remmele–Stegner (IRS) scoring scale. The IRS score was assigned to each tissue macroarrays spot as the sum of the products of the intensity (0 for negative, 1 for weakly positive, 2 for moderately positive, and 3 for strongly positive) multiplied by the extent of the immunohistochemical expression (1 for 1%–9%, 2 for 10%–50%, 3 for 51%–80%, and 4 for 81%–100%), obtaining the final result between 0 and 12. Statistical analysis included the IRS score for all tissue macroarrays. 23
Statistical analysis
Statistical analysis of the results was carried out with PQStat package, version 1.6.4.121. The significance of intergroup differences in the study variables was verified with Mann–Whitney U test (k = 2) or Kruskal–Wallis test (k > 2). Survival curves were compared with log-rank test, Wilcoxon–Breslow–Gehan test and Tarone–Ware test. The results were considered significant at p < 0.05 and highly significant at p < 0.01.
Ethics
The protocol of the study was approved by the Local Bioethics Committee at the Nicolaus Copernicus University, Collegium Medicum in Bydgoszcz (decision no. KB 413/2016), and written informed consent was sought from each patient or her next of kin.
Results
The expressions of SLUG, TWIST 1, ZEB1, and ZEB 2 were scored based on the nuclear staining, and the expressions of SNAIL and TWIST 2 based on the cytoplasmic and/or nuclear staining (Figures 1–6). The proportions of ovarian tumors with the immunoexpression of the EMT transcription factors were 85.7% for SNAIL, 100% for SLUG, 9.5% for TWIST 1, 95.2% for TWIST 2, 23.8% for ZEB 1, and 0% for ZEB 2.

(a) Microphotograph presenting nuclear SLUG expression in endometrioid ovarian cancer and (b) borderline tumor. (c) The expression was unequivocally strong in both tumor groups. Positive expression in non-cancerous ovarian stroma. (d) Negative expression in basal cell layer of non-cancerous ovarian epithelium. Primary objective magnification ×10.
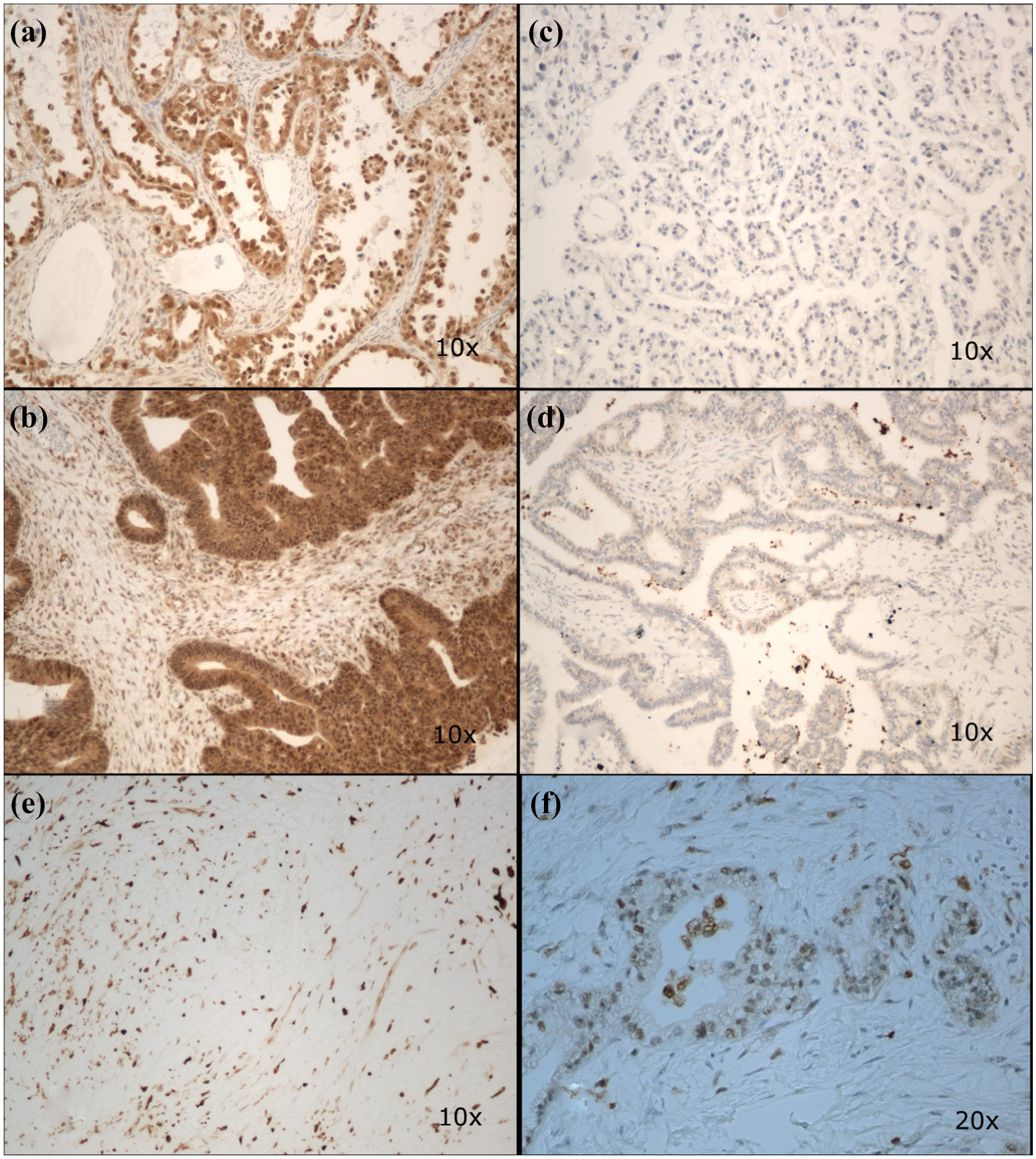
Microphotograph presenting nuclear SNAIL expression in serous ovarian cancer (a) and borderline tumor (b) and negative staining in serous ovarian cancer (c) and borderline tumor (d). Positive expression in non-cancerous ovarian stroma and negative expression in non-cancerous ovarian epithelium (e and f, respectively). Primary objective magnification 10× (a–e) and 20× (f).

Microphotograph presenting nuclear TWIST1 expression in serous ovarian cancer (a, b) and negative staining in mucinous ovarian cancer (c) and borderline tumor (d). Positive expression in non-cancerous ovarian stroma and negative expression in non-cancerous ovarian epithelium (e and f, respectively). There was no presence of positive TWIST1 expression in borderline tumor. Primary objective magnification 10× (a, c, d, e, f) and 20× (b).

Microphotograph presenting cytoplasmic TWIST2 expression ranging from strong to weak in endometrioid ovarian cancers (a and b, respectively). (c) Borderline tumors presented invariably strong expression. Positive expression in non-cancerous ovarian stroma and negative expression in non-cancerous ovarian epithelium (d and e, respectively). Primary objective magnification ×10.

(a) Microphotograph presenting nuclear ZEB1 expression in clear-cell ovarian cancer and(b) borderline tumor and (c) negative staining in serous ovarian cancer and (d) borderline tumor. Positive expression in non-cancerous ovarian stroma and negative expression in non-cancerous ovarian epithelium (e and f, respectively). Primary objective magnification ×10.

(a) Microphotograph presenting negative nuclear ZEB2 expression in serous ovarian cancer and (b) borderline tumor, whereas tumor infiltrating lymphocytes present strong expression. Positive expression in non-cancerous ovarian stroma and negative expression in non-cancerous ovarian epithelium (c and d, respectively). Primary objective magnification ×10.
No statistically significant differences were found in the expression of the EMT transcription factors in borderline tumors and type I ovarian cancers (Figure 7).

Immunoexpression of the EMT transcription factors stratified according to the type of ovarian tumor (sBOT—serous borderline ovarian tumor, OC—ovarian cancer). No statistically significant differences were found in the expression of the EMT transcription factors in borderline tumors and type I ovarian cancers (p > 0.05).
When the immunoexpressions of the EMT transcription factors were stratified according to the menopausal status of the study participants, the only statistically significant difference was found for ZEB 1; the expression of this factor turned out to be stronger in postmenopausal women. The expressions of other transcription factors did not differ significantly between the groups of premenopausal and postmenopausal patients (Figure 8).

Immunoexpression of the EMT transcription factors stratified according to the menopausal status of the study patients. The only statistically significant difference was found for ZEB 1 (p < 0.05); the expression of other transcription factors did not differ significantly between premenopausal and postmenopausal patients.
The immunoexpressions of the EMT transcription factors did not differ significantly depending on the histopathological type, clinical stage, and histological grade of the ovarian tumors (Supplement: Tables 4–6).
Irrespective of the statistical test used, no significant differences were found between the survival of patients with borderline tumors and type I ovarian cancers (Table 3). None of the patients with BOTs (0/8) died during a 5-year follow-up period, whereas four deaths (4/34) were documented among women with type I ovarian cancer. While this was not reflected by a statistically significant difference in 5-year survival rates, it should emphasized that the study groups were relatively small.
Relationship between the type of ovarian tumor and survival.

No statistically significant differences (p > 0.05).
Moreover, no statistically significant differences were observed between the 5-year survival rates for patients with weak and strong immunoexpression of the EMT transcription factors: TWIST 1 (0 vs >0), TWIST 2 (8 vs ≥8), SNAIL (8 vs ≥8), SLUG (8 vs ≥8), ZEB1 (0 vs >0), and ZEB 2 (0 vs >0) (Supplement: Tables 7–11 and Supplement: Figures 9–13).
Discussion
Epithelial ovarian neoplasms are a heterogeneous group including the tumor subsets with distinct clinicopathologic and molecular features. Histopathologically, epithelial ovarian cancers can be divided into high-grade serous, low-grade serous, mucinous, endometrioid, and clear-cell carcinomas. 24 However, the differences in the histological structure do not fully explain the heterogeneity in the clinical phenotypes of ovarian malignancies. Moreover, recent evidence from molecular and genomic studies suggests that whereas LGSCs and HGSCs likely develop on two separate pathways, the low-grade tumors and sBOTs may in fact represent various stages of the same developmental continuum. Both type I ovarian cancers and BOTs are characterized by slower proliferation than HGSCs and higher 5-year survival rates. 25 Although approximately 75% of BOTs are diagnosed at stage I, with the survival rates greater than 90%,26,27 patients with advanced stage disease may die from complications associated with extragonadal spread across the pelvic cavity. Furthermore, approximately 15% of BOTs may recur up to 20 years after the initial diagnosis or later, which is typically associated with a poor outcome. 28 Current standard of care in patients with epithelial ovarian malignancies is maximal cytoreductive surgery and platinum–taxane combination chemotherapy. 29 In the case of BOTs, complete surgical resection is generally considered to be the best curative option, but the extent of the procedure and the use of postoperative treatment, if any, are still a matter of a debate. 2
Transformation of a BOT into an invasive neoplasm is associated with an array of molecular changes, inter alia controlled by p53 and PI3K/Akt pathway, as well as with a decrease in E-cadherin expression. The latter implies that EMT is a critical determinant of BOT invasiveness.30,31 The loss of epithelial adherence and the disruption of tight junctions make EMT a major mechanism involved in the transformation of early-stage tumors into invasive malignancies. Ovarian carcinoma cells frequently undergo EMT prior to their detachment or metastatic spread.32,33 A growing body of evidence suggests that the expression of genes that facilitate EMT and promote formation of metastases is controlled by multiple epigenetic mechanisms. 34 Understanding of the regulatory loops that are involved in close coordination of the EMT program may be crucial for the development of novel strategies aimed at the prevention of metastatic spread.
The cellular changes associated with EMT co-exist with modifications at protein and gene expression levels, such as the downregulation of epithelial intermediate filament forming proteins (cytokeratins), overexpression of type III mesenchymal intermediate filament protein (vimentin), and alterations in cell–cell and cell–matrix adhesion molecules. 35 Another key feature of EMT is the “cadherin switch,” that is, a downregulation of epithelial cadherin (E-cadherin) with a concomitant upregulation of neural cadherin (N-cadherin). 36 This process is postulated to enhance cell mobility and invasiveness36,37 and seems to be regulated by a number of zinc finger transcription factors that negatively modulate the expression of E-cadherin, among them SNAIL, SLUG, TWIST1/2, and ZEB1/2. 38 Recent evidence suggests that the pleiotropic effects of the transcription factors from the SNAIL, ZEB, and TWIST families during cancer initiation and progression extend also on the control of cell fate specification and transition, stem cell and cancer stem cell plasticity, malignant transformation and tumor initiation, cancer cell survival in response to therapy, immune evasion, and shaping of the cancer micro-environment. 39 SNAIL is a transcription factor that promotes EMT by binding to the promoter regions of E-cadherin. Moreover, it was shown to upregulate matrix metalloproteinases, the enzymes that play a role in the degradation of extracellular matrix and facilitate tumor progression.33,40 Two zinc finger transcription repressors implicated in the repression of E-cadherin transcription in vitro, SNAIL and SLUG, 41 together with TWIST participate in mesoderm formation. 42 Similar to SNAIL, also TWIST was shown to downregulate epithelial genes and to stimulate the expression of mesenchymal genes. 43 In cancer cells, TWIST 1 represses E-cadherin and upregulates N-cadherin independently of SNAIL, probably through the association with other proteins.43–45 Activation of TWIST during developmental processes and tumorigenesis occurs on diverse signaling pathways. 43 Under hypoxia, the expression of TWIST is upregulated by hypoxia-inducible factor 1α (HIF1α), which promotes EMT and stimulates dissemination of cancer cells. 46 The two vertebrate ZEB transcription factors, ZEB1 and ZEB2, bind the sequences of regulatory genes at E-boxes and can repress or activate transcription. 47 Therefore, likewise SNAIL and TWIST, ZEBs can bind to E-boxes and function as transcription repressors for some epithelial junction and polarity genes, as well as the activators of mesenchymal genes defining the EMT phenotype. 43 The upregulation of ZEB frequently follows the activation of SNAIL, consistent with SNAIL1 directly targeting the ZEB1 gene. Moreover, TWIST1 was shown to cooperate with SNAIL1 in the induction of ZEB1 expression. 9 ZEB is upregulated by TGF-β and WNT proteins, as well as by the growth factors that activate RAS-MAPK signaling. 43
In our present study, patients with sBOTs and type I ovarian cancers did not differ significantly in terms of SNAIL, SLUG, TWIST 1, TWIST 2, and ZEB 1 expressions. This implies that the expressions of the EMT transcription factors in sBOTs and early-stage type I ovarian cancers may be similar. It should be emphasized that the vast majority of ovarian tumors included in this study (>90%) tested positively for SNAIL, SLUG, and TWIST 2, the key regulators of EMT. The relationship between the overexpression of SNAIL and the greater aggressiveness of ovarian tumors was first reported by Tuhkanen et al., who found a positive nuclear staining for SNAIL protein in both epithelial and stromal compartments. Presence of SNAIL in precursor lesions suggests that this protein may play a role at early stages of ovarian tumorigenesis, before invasion or metastasis. 48 However, another study demonstrated that borderline tumors and ovarian cancers did not differ significantly in terms of their ZEB 1 expressions. 49 In contrast, the expression of another transcription factor, ZEB2, was the weakest in benign ovarian lesions and increased progressively in BOTs, malignant tumors, and metastatic foci. 49 In our present study, we did not find significant relationships between the immunoexpression of the analyzed transcription factors, clinical stage, and histological grade of ovarian tumors. The only statistically significant association was observed in the case of ZEB 1, the expression of which turned out to be stronger in postmenopausal women than in premenopausal patients. We also did not demonstrate statistically significant differences in the survival of patients with borderline tumors and ovarian cancers, but this probably resulted from a relatively small size of the study groups. Similar to our study, Tuhkanen et al. 48 neither found a statistically significant relationship between the expression of SNAIL and the clinicopathological features of ovarian cancers, such as grade and stage, nor demonstrated a prognostic value of this parameter. However, in the study conducted by Takai et al., 50 nuclear expression of SNAIL turned out to be an independent predictor of survival in ovarian cancer patients; the same study did not demonstrate substantial differences in the clinical relevance and prognostic impact of SLUG. According to Davidson et al., 51 the expressions of SNAIL and TWIST 1 were significantly stronger in patients with advanced ovarian malignancies (stage III and IV) than in those with earlier stages of the disease. In turn, Mao et al. 52 demonstrated that the expression of TWIST 2 correlated with the clinical stage (FIGO), but not with the histological grade of ovarian carcinoma. According to Kim et al., 53 the expression of TWIST correlated with the clinicopathological characteristics of ovarian cancer patients and was a predictor of poor survival. Finally, Hosono et al. 54 identified the expression of TWIST as an independent predictor of survival in ovarian cancer patients.
In summary, the expression patterns for SNAIL, SLUG, TWIST, and ZEB identified in this study suggest that both sBOTs and type I ovarian cancers undergo dynamic epithelial–mesenchymal interconversions. Our findings obtained in two groups of tumors sharing some etiopathogenic pathways imply that the expressions of the EMT transcription factors may be activated at early stages of the EMT, and thus these molecules may play a pivotal role in the development of both sBOTs and type I ovarian cancers. The EMT transcription factors not infrequently modulate the expression of each other and functionally cooperate at their target genes; consequently, additional transcription factors are likely to further define the EMT transcription program and to drive the EMT progression. 44 sBOTs are non-invasive tumors considered to be a distinct entity giving rise to invasive LGSCs, and the hereby documented lack of statistically significant differences in the expressions of the EMT transcription factors may constitute a proof for common origin of these two tumor types. Our findings provide a molecular basis for further research on the pathogenesis of sBOTs and type I ovarian cancers and justify the exploration of the EMT transcription factors as potential valuable therapeutic targets.
Supplemental Material
SUPPLEMENT – Supplemental material for Expression of selected epithelial–mesenchymal transition transcription factors in serous borderline ovarian tumors and type I ovarian cancers
Supplemental material, SUPPLEMENT for Expression of selected epithelial–mesenchymal transition transcription factors in serous borderline ovarian tumors and type I ovarian cancers by Pawel Sadlecki, Jakub Jóźwicki, Paulina Antosik and Marek Grabiec in Tumor Biology
Footnotes
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
