Abstract
Epithelial ovarian tumors are a group of morphologically and genetically heterogeneous neoplasms. Based on differences in clinical phenotype and genetic background, ovarian neoplasms are classified as low-grade and high-grade tumor. Borderline ovarian tumors represent approximately 10%–20% of all epithelial ovarian masses. Various histological subtypes of ovarian malignancies differ in terms of their risk factor profiles, precursor lesions, clinical course, patterns of spread, molecular genetics, response to conventional chemotherapy, and prognosis. The most frequent genetic aberrations found in low-grade serous ovarian carcinomas and serous borderline tumors, as well as in mucinous cancers, are mutations in BRAF and KRAS genes. The most commonly observed BRAF mutation is substitution of glutamic acid for valine in codon 600 (V600E) in exon 15. The primary aim of this study was to determine whether fully integrated, real-time polymerase chain reaction–based Idylla™ system may be useful in determination of BRAF gene mutation status in codon 600 in patients with borderline ovarian tumors and low-grade ovarian carcinomas. The study included tissue specimens from 42 patients with histopathologically verified ovarian masses, who were operated on at the Department of Obstetrics and Gynecology, Nicolaus Copernicus University Collegium Medicum in Bydgoszcz (Poland). Based on histopathological examination of surgical specimens, 35 lesions were classified as low-grade ovarian carcinomas, and 7 as borderline ovarian tumors. Specimens with expression of BRAF V600E (VE1) protein were tested for mutations in codon 600 of the BRAF gene, using an automated molecular diagnostics platform Idylla™. Cytoplasmic immunoexpression of BRAF V600E (VE1) protein was found in three specimens: serous superficial papilloma, serous papillary cystadenoma of borderline malignancy, and partially proliferative serous cystadenoma. All specimens with the expression of BRAF V600E (VE1) protein were tested positively for BRAF V600E/E2/D mutation. No statistically significant relationship (p > 0.05) was found between the presence of BRAF V600E mutation and the probability of 5-year survival. BRAF mutation testing with a rapid, fully integrated molecular diagnostics system Idylla™ may be also a powerful prognostic tool in subjects with newly diagnosed serous borderline tumors, identifying a subset of patients who are unlikely to progress.
Introduction
Ovarian cancer is the third most common female genital tract malignancy, with an estimated worldwide annual incidence of about 238,000 cases and ca 151,000 mortality. 1 There are five main histological subtypes of epithelial ovarian cancer: high-grade serous carcinomas, which account for 70% of all epithelial ovarian tumors, low-grade serous carcinomas, endometrioid carcinomas, mucinous carcinomas, and clear-cell carcinomas. 2 Borderline ovarian tumors represent approximately 10%–20% of all epithelial ovarian masses. 3 Previously, treatment of ovarian malignancies and borderline tumors has been selected primarily on the basis of histological grade and clinical stage, but then it became apparent that microscopic structure of the lesion is an equally important therapeutic criterion. Various histological subtypes of ovarian cancers differ in terms of their risk factor profiles, precursor lesions, clinical course, patterns of spread, molecular genetics, response to conventional chemotherapy, and prognosis.4,5 Hence, accurate identification of tumor subtype on the basis of its morphology, and whenever necessary also genetic and immunohistochemical profile, should be a priority. Furthermore, it has been suggested that development of effective strategies to inhibit progression and spread of the tumor requires better understanding of cellular and molecular pathways existing within its microenvironment. Effective control of a malignancy may necessitate an insight into its microenvironment and modification of microenvironmental parameters that promote local growth, invasiveness, and spread of cancer cells.6,7 The most frequent genetic aberrations found in low-grade serous ovarian carcinomas and serous borderline tumors, as well as in mucinous cancers, are mutations in BRAF and KRAS genes. 8 These mutations result in constitutive activation (phosphorylation) of their downstream target, mitogen-activated protein kinase (MAPK), also referred to as extracellular signal–regulated protein kinase (ERK). 9 MAPK pathway is critical for regulation of cancer cell proliferation and survival. 10 BRAF is a cytoplasmic receptor serine/threonine kinase, one out of three members of RAF protein kinase family (ARAF, BRAF, and CRAF). 11 The most commonly observed BRAF mutation is substitution of glutamic acid for valine in codon 600 (V600E) in exon 15. 12 Other common BRAF mutations in codon 600 are BRAF V600E2, V600D, V600K, V600R, and V600M. 13 BRAF V600E mutations are found in virtually all patients with hairy cell leukemia; these aberrations are also quite common in papillary thyroid cancer, malignant melanoma, and colon cancer developing via the alternative pathway. 14 Identification of KRAS and BRAF mutation carriers has gained increasing attention with the advent of targeted therapies, considered a promising treatment for human malignancies with constitutive activation of MAPK pathway and, specifically, with BRAF V600E mutation. 15 However, implementation of such targeted therapeutic strategies requires availability of a highly specific and sensitive diagnostic test for rapid detection of mutant protein and determination of mutation status. One candidate for such test is fully integrated, real-time polymerase chain reaction (RT-PCR)-based Idylla™ system for quick and simple detection of BRAF mutations in formaldehyde-fixed paraffin-embedded (FFPE) tumor samples. 16 The assay is able to identify the presence of ⩾1% BRAF V600 mutated cells in a background of wild-type allele. The Idylla™ BRAF Mutation Test produced results quickly and the closed nature of the cartridge eliminates the risk of PCR contamination. Usefulness of Idylla™ system in detection of BRAF V600E mutation was not evaluated yet in borderline and ovarian cancer patients.
The primary aim of this study was to determine whether fully integrated, RT-PCR-based Idylla™ system may be useful in determination of BRAF mutation status in patients with borderline ovarian tumors and low-grade ovarian carcinomas.
Materials and methods
This study included tissue specimens from 42 patients with histopathologically verified ovarian masses, who were operated on at the Department of Obstetrics and Gynecology, Nicolaus Copernicus University Collegium Medicum in Bydgoszcz (Poland) between January 2009 and June 2012. All patients underwent surgical resection according to clinical stage of the tumor, and whenever necessary, received adjuvant platinum-based chemotherapy in line with current Polish guidelines. 17 Based on histopathological examination of surgical specimens, 35 lesions were classified as low-grade ovarian carcinomas, and 7 as borderline ovarian tumors. Probability of 5-year survival was analyzed in relation to clinicopathological features. Clinicopathological characteristics of the study subjects are summarized in Table 1.
Clinicopathological characteristics of the study subjects.

FIGO: International Federation of Gynecology and Obstetrics.
Immunohistochemical and molecular studies of FFPE specimens were conducted at the Department of Clinical Pathology, Nicolaus Copernicus University Collegium Medicum in Bydgoszcz.
Tissue microarray
Selected archival FFPE tissue specimens (donor blocks) were re-embedded in paraffin-wax blocks and processed routinely to prepare hematoxylin–eosin (H&E)-stained slides. Two representative tumor areas (2 mm in diameter), each containing at least 80% tumor cells, were identified on H&E slides, marked, harvested from donor blocks, and transferred to a recipient tissue microarray (TMA) block using an automated tissue arrayer (TMA Master; 3DHISTECH, Budapest, Hungary). Then, H&E slides were prepared from TMA blocks, to verify that the appropriate tumor site was selected. Control reactions included samples without DNA (negative controls) and with known BRAF mutant status and BRAF V600E (VE1) protein expression (positive controls; lymph node metastasis of malignant melanoma).
Immunohistochemical staining of BRAF V600E (VE1)
Paraffin-embedded TMA blocks were cut into 4 µm-thick sections with a manual rotary microtome (Accu-Cut, Sakura Finetek, Torrance, CA, USA) and mounted on extra adhesive slides (SuperFrost Plus; Menzel-Glaser, Braunschweig, Germany). Immunohistochemical staining, the protocol of which has been described in detail elsewhere, 18 was performed with an automated slide-processing system, BenchMark GX (Ventana Medical Systems, Tucson, AZ, USA). After deparaffinization and rehydration in EZ Prep® (Ventana Medical Systems, Tucson, AZ, USA) solution at 72°C, the slides were incubated for 72 min in Cell Conditioning 1 (CC1; Ventana Medical Systems, Tucson, AZ, USA) solution with pH 9.0 to retrieve the antigens. Then, the activity of endogenous peroxidase was blocked with 3% H2O2 solution, and the slides were incubated at 36°C for 40 min with primary anti-BRAF V600E (VE1) antibody (Ventana Medical Systems). Subsequently, the slides were incubated with OptiView HQ Universal Linker (Ventana Medical Systems, Tucson, AZ, USA) and OptiView HRP Multimer (Ventana Medical Systems, Tucson, AZ, USA), counterstained with hematoxylin and bluing reagent, dehydrated, passed through xylene, and sealed using a mounting medium from Dako (Agilent Technologies, US).
BRAF mutation status in codon 600
Specimens with expression of BRAF V600E (VE1) protein were tested for mutations in codon 600 of the BRAF gene, using an automated molecular diagnostics platform Idylla™ (Biocartis, Mechelen, Belgium). Paraffin-embedded blocks were cut into 4 × 10 µm sections with a manual rotary microtome (Accu-Cut, Sakura, Torrance, USA), placed between qualitative filter papers (10 mm in diameter), and inserted to the Idylla™ BRAF Mutation Test cartridge. Each section contained at least 70% of tumor cells. Each sample underwent deparaffinization, followed by simultaneous rehydration and DNA extraction, using high-intensity focused ultrasound technology. Subsequently, isolated nucleic acids were transferred via microfluidic channels of the cartridge to five separate PCR chambers with pre-deposited PCR reagents. 19 The module combines quantitative allele-specific RT PCR–based reaction with a fluorescent detection system. Molecular test can be used to detect BRAF V600E/E2/D and V600K/R/M mutations, specifically BRAF V600E: c.1799T > A; p.(Val600Glu), V600E2: c.1799_1800TG > AA; p.(Val600Glu), V600D: c.1799_1800TG > AT, c.1799_1800TG > AC; p.(Val600Asp), V600K: c.1798_1799GT > AA; p.(Val600Lys), V600R: c.1798_1799GT > AG; p.(Val600Arg), and V600M: c.1798G > A; p.(Val600Met). 19
Quantification cycle (Cq) value
The presence of a mutant genotype was determined by calculating ΔCq, being the difference between Cq for wild-type BRAF and Cq for V600E/E2/D or V600K/R/M. The samples were considered BRAF V600 mutation-positive if their ΔCq was within a validated range. The samples with a valid wild-type BRAF signal and ΔCq value outside of the validated range were classified as BRAF V600 mutation-negative. 19
The protocol of the study was granted approval from the Local Bioethics Committee at the Nicolaus Copernicus University Collegium Medicum in Bydgoszcz (decision no. KB 413/2016), and written informed consent was sought from each patient or her next of kin. Statistical analysis of the results was carried out with PQStat ver. 1.6 package. Overall survivals were obtained as life-table and Kaplan–Meier estimates and compared with log-rank, Wilcoxon–Breslow–Gehan and Taron–Ware tests. Potential predictors of survival were identified using Cox proportional-hazard regression model. The results were considered significant at p < 0.05 and highly significant at p < 0.01.
Results
BRAF V600E (VE1) protein expression and BRAF mutations in codon 600
The specimens were considered BRAF V600E (VE1)-positive whenever an evident diffuse cytoplasmic immunohistochemical reaction was present. Nuclear immunoexpression of BRAF V600E (VE1) was interpreted as a negative result.20,21
Cytoplasmic immunoexpression of BRAF V600E (VE1) protein was found in 3/42 specimens: serous superficial papilloma (1/42, 2%), serous papillary cystadenoma of borderline malignancy (1/42, 2%), and partially proliferative serous cystadenoma (1/42, 2%). All specimens with the expression of BRAF V600E (VE1) protein (3/42, 7%) tested positively for BRAF V600E/E2/D mutation (protein HGVS: p.(Val600Glu)/p.(Val600Glu)/p.(Val600Asp); base change: c.1799T > A/c.17991800TG > AA/c.1799_1800TG > AT; c.1799_1800TG > AC).
None of the remaining specimens (39/42) showed the expression of BRAF V600E (VE1) protein or the presence of BRAF mutation in codon 600. The input of DNA was sufficient in all cases. Representative PCR curves and corresponding Cq values are shown in Figure 1.

(a–c) Microphotograph presenting H&E-stained specimens and (d–f) corresponding positive immunohistochemical staining of BRAF V600E (VE1) protein in ovarian tumors. Nucleus counterstained with hematoxylin-positive staining for BRAF V600E (VE1) protein (brown) and primary objective magnification 4× to 10×&×46; (g–i) PCR BRAF curves documenting the presence of BRAF V600E mutation.
Influence of selected clinicopathological variables on the probability of 5-year survival
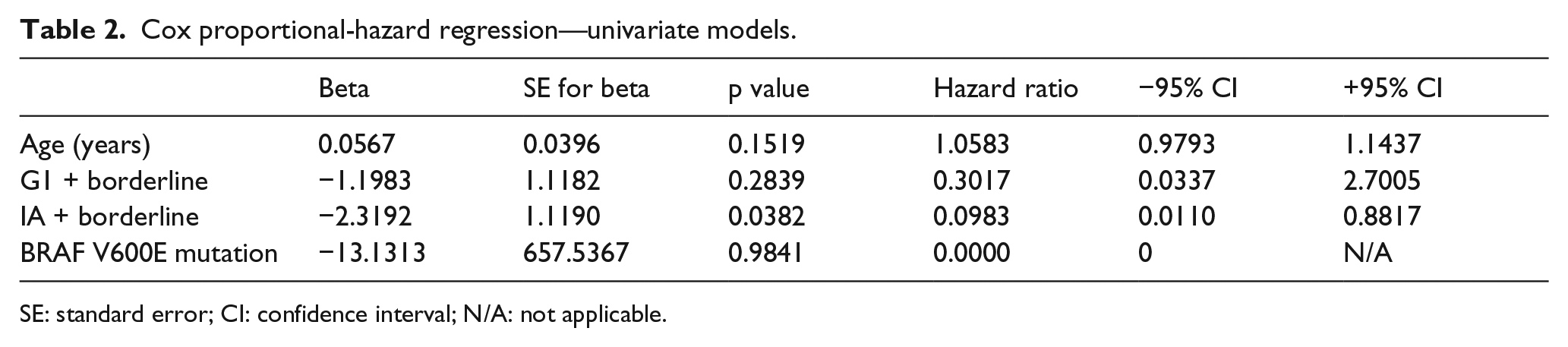
No statistically significant relationship (p > 0.05) was found between the presence of BRAF V600E mutation and the probability of 5-year survival (Figure 2). Histological type of ovarian tumor turned out to be a highly significant (p < 0.01) predictor of 5-year survival in our series; the probability of survival in patients with mucinous carcinomas was significantly lower than in other study subjects (Figure 3, Table 3). Histological grade did not exert a significant effect on 5-year survival (p > 0.05). Survivals differed significantly (p < 0.05) depending on clinical stage (International Federation of Gynecology and Obstetrics (FIGO)); the probability of survival in women with stage IA and borderline tumors turned out to be significantly higher than in patients with other histological types of ovarian carcinomas (Figure 4). Both univariate and multivariate Cox regression analysis (Tables 2 and 3) demonstrated that the only significant positive predictors of survival in our series were clinical stage IA and borderline malignancy of the tumor.

Kaplan–Meier curves for overall survival in patients with ovarian tumors stratified by BRAF V600E mutation. The Kaplan–Meier plots illustrate the lack of statistically significant differences (p > 0.05) in the probability of 5-year survival in patients with and without BRA V600E mutations.
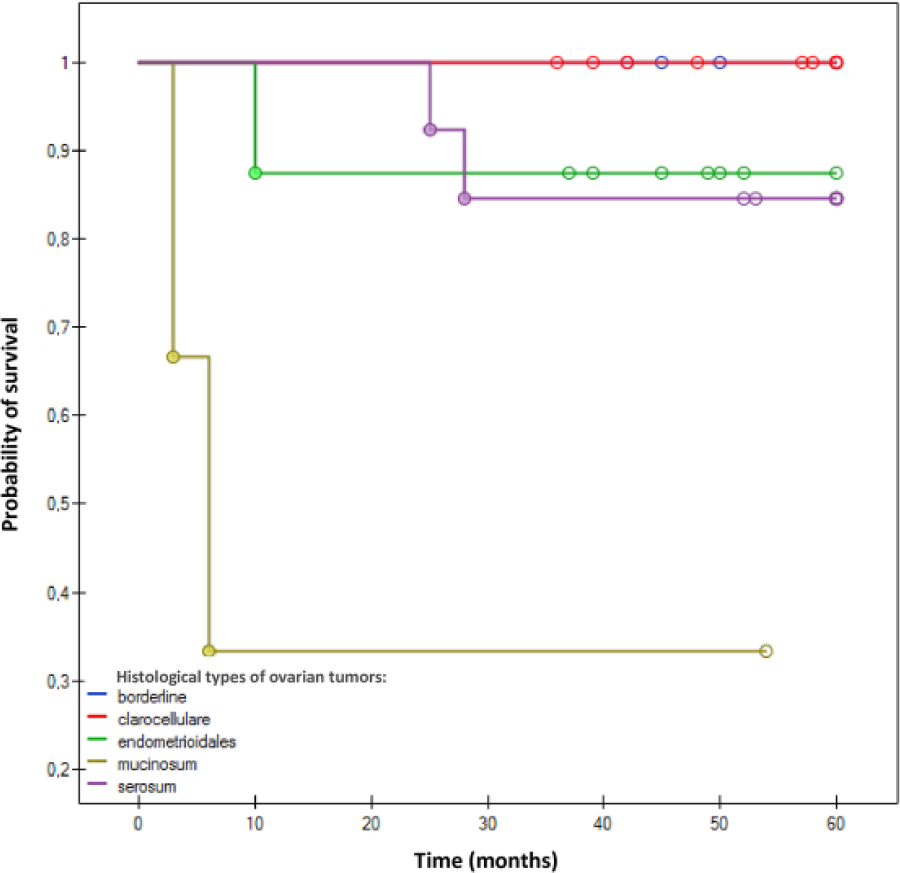
Kaplan–Meier curves for overall survival in patients with ovarian tumors stratified by histological type of ovarian tumors. The Kaplan–Meier curves show the highly significant (p < 0.01) differences in the probability of 5-year survival in patients with various histological types of ovarian tumors. Significantly lower probability of survival was observed in women with mucinous ovarian carcinomas (p < 0.01).
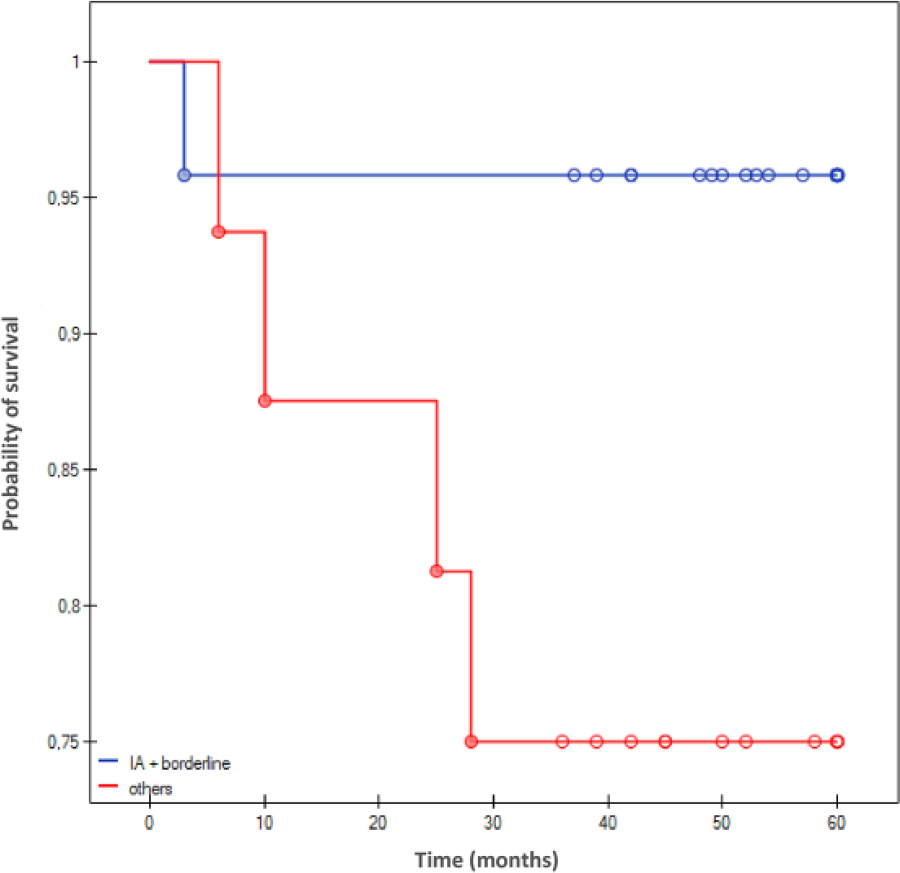
Kap1an–Meier curves for overall survival in patients with ovarian tumors stratified by FIGO stage (lA + borderline versus others). The Kaplan–Meier curves show the significant (p < 0.05) difference in the probability of 5-year survival in patients with ovarian tumors representing various clinical stages according to FIGO classification. Significantly higher probability of survival was observed in women with FIGO stage lA and borderline ovarian tumors.
Cox proportional-hazard regression—univariate models.
SE: standard error; CI: confidence interval; N/A: not applicable.
Cox proportional-hazards regression—multivariate models.

SE: standard error; CI: confidence interval; N/A: not applicable.
Discussion
Epithelial ovarian tumors are a group of morphologically and genetically heterogeneous neoplasms. Based on differences in clinical phenotype and genetic background, ovarian neoplasms are classified as low-grade and high-grade tumors. 22 According to this dualistic model of ovarian carcinogenesis, high-grade tumors arise de novo from distal fallopian tube epithelium, whereas low-grade serous ovarian lesions undergo a step-wise transformation from a benign serous cystadenoma to serous borderline neoplasm and then to invasive low-grade serous carcinoma.23–25 Low-grade ovarian tumors may present histologically as serous, mucinous, endometrioid, or clear-cell carcinomas. 26 Serous borderline neoplasms can be further classified into atypical proliferative serous tumors and non-invasive micropapillary serous carcinomas. A micropapillary serous carcinoma which became invasive is classified as a low-grade serous carcinoma.27,28 The presence of micropapillary features in a serous borderline tumor is more often associated with bilateral ovarian involvement, peritoneal implantation, and recurrent disease than the lack of this histological phenotype. 29 Low-grade serous ovarian tumors typically lack mutations in p53 gene and have low proliferation rate or low mitotic index. As a result, tumors with such characteristics have better prognosis with longer survival, also in advanced stages, even despite lesser sensitivity to chemotherapy. 30 Most serous borderline tumors and low-grade serous ovarian carcinomas are chemotherapy-resistant, with 4% and 2.1%–4.9% reported response rates to cytotoxic chemotherapy in a neoadjuvant and recurrent setting, respectively.30,31 Type I serous borderline tumors and low-grade serous carcinomas, including lesions with a micropapillary phenotype, are characterized by a high frequency of activating mutations upstream of MAPK pathway, primarily BRAF (28%–48%) and KRAS (18%–33%), and occasionally, also ERBB2; in contrast, these mutations are present in <1% of high-grade serous ovarian tumors. 32 Constitutive activation of MAPK pathway can be demonstrated in virtually all serous borderline tumors and low-grade serous carcinomas. 33 Mutations in BRAF and KRAS, components of MAPK cascade, are common in borderline and low-grade serous tumors; furthermore, they can be found in mucinous ovarian carcinomas.34,35 In contrast, these mutations are virtually absent in high-grade serous ovarian carcinomas. V600E is the most common oncogenic mutation in serous ovarian tumors of low malignant potential; this aberration can be also found in low-grade serous carcinomas. 36
To the best of our knowledge, usefulness of Idylla™ system in detection of BRAF V600E mutation was not evaluated in ovarian tumors yet. In this study, expression of VE1 protein and the presence of BRAF V600E mutation were detected in three specimens; all of them were serous ovarian tumors with borderline malignancy. In contrast, we did not find the mutant protein in low-grade serous, mucinous, clear-cell, and endometrioid ovarian carcinomas. The presence of BRAF V600E mutation was not associated with higher probability of 5-year survival. According to the literature, V600E BRAF mutation is associated with early stage at presentation, serous borderline histology, and improved outcome; the presence of a BRAF mutation in patients with serous borderline ovarian tumors was shown to prevent progression to more aggressive disease. 37 However, quite different results were documented in the case of papillary thyroid cancer, in which the presence of V600E BRAF mutation was associated with higher stage and poor prognosis, and in metastatic colon cancer, whereby this genetic aberration was also identified as an unfavorable prognostic factor. This implies that BRAF mutation status has tumor lineage-specific prognostic implications. 38 In this study, only histological type of ovarian tumors and stage IA were significantly associated with 5-year survival rate.
Currently, BRAF mutations are typically detected with molecular methods, such as conventional sequencing, next-generation sequencing, pyrosequencing, and allele-specific PCR with melting curve analysis; however, application of all these techniques requires access to a molecular diagnostic laboratory. 39 As a result, molecular techniques are still considered a secondary screening test in the detection of BRAF mutations. Previous studies showed that immunolabeling with monoclonal mouse antibody (VE1), specifically detecting mutated (V600E) BRAF protein in FFPE specimens, may be an alternative to molecular techniques in screening for BRAF mutations.40,41 Several authors confirmed that anti-BRAF V600E (VE1) antibody provides high reliability and specificity in the detection of mutated BRAF V600E protein. 42 Recently, some new molecular assays for BRAF mutations have become available. These are fully automated molecular diagnostic systems for quantitative allele-specific RT-PCR-based analyses. Unlike current routinely used technologies for BRAF mutation detection, Idylla™ BRAF Mutation Test does not require manual sample preprocessing steps, such as deparaffinization, FFPE tissue digestion, or DNA extraction, since all these procedures are integrated within a single-use cartridge. 43 A complete FFPE tissue section or macro-dissected FFPE specimen is inserted into the cartridge and processed by the Idylla™ system; also further stages, that is, RT-PCR-based mutation detection and result reporting, are fully integrated and automated. 44
In recent years, a plethora of compounds targeting components of Ras/Raf/mitogen-activated protein kinase kinase (MEK) pathway have entered clinical trials, and some of them already demonstrated promising clinical activity, usually in malignancies with BRAF mutations. 45 Testing for activating mutations in BRAF gene is becoming more common, especially in patients qualified to treatment with BRAF inhibitors or other pathway modulators, such as MEK inhibitors. BRAF mutation testing with a rapid, fully integrated molecular diagnostics system Idylla™ may be also a powerful prognostic tool in subjects with newly diagnosed serous borderline tumors, identifying a subset of patients who are unlikely to progress.
Footnotes
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
