Abstract
Liver metastases of colorectal cancer can be operated with a curative intent in selected cases. However, more than half of the patients have a recurrence. The aim of this study was to evaluate the prognostic and predictive value of carcinoembryonic antigen (CEA), carbohydrate antigen 19-9 (CA19-9), human chorionic gonadotropin β (hCGβ) and tumour-associated trypsin-inhibitor (TATI) in colorectal cancer patients before and 3 months after resection of liver metastases. Marker concentrations were determined in blood samples from 168 colorectal cancer patients, who underwent liver resection between the years 1998 and 2007 at Helsinki University Hospital, Finland. The samples were taken before and 3 months after curative resection. Increased concentrations of CEA (>5 µg/L) and hCGβ (>1 pmol/L) 3 months after liver resection correlated with recurrence and impaired overall survival and increased CA19-9 (>26 kU/L) with impaired overall survival, but postoperative TATI was not prognostic. Preoperatively elevated CEA and CA19-9 correlated with impaired overall survival, but not with recurrence. Neither preoperative hCGβ nor TATI was prognostic. In conclusion, CEA is a useful prognostic marker, when measured 3 months after resection of colorectal liver metastases. CA19-9 also has prognostic significance and may have additional value.
Introduction
Colorectal cancer (CRC) is the third most common cancer worldwide and the second leading cause of cancer death in Western countries. 1 About half of CRC patients develop either synchronous or metachronous liver metastases, which can be treated by surgery in selected cases. 2 Resection is the only treatment option that improves long-term survival of patients with liver metastases, but more than half of the resected patients have a recurrence.3,4 Thus, there is an obvious need for prognostic and predictive markers to determine the risk of recurrence early after liver resection and to commence more comprehensive follow-up and possibly more aggressive adjuvant treatment for patients at high risk. It would be valuable if measurement of serum or plasma concentrations of tumour markers could be used to predict the outcome of metastatic CRC after curative resection.
At present, the best marker for CRC is carcinoembryonic antigen (CEA), a glycoprotein belonging to the immunoglobulin superfamily. However, about 20% of CRC tumours have been reported not to produce elevated serum levels of CEA in spite of metastatic disease. 5 According to the European Group on Tumour Markers (EGTM) and American Society of Clinical Oncology (ASCO) guidelines, CEA is the only serum marker that should be measured regularly in CRC patients who are candidates for metastasectomy and systemic therapy,6,7 but so far, there are no guidelines for the use of tumour markers after curative resection of CRC metastases.
Carbohydrate antigen 19-9 (CA19-9) has also been used as a marker for CRC, but it is less sensitive than CEA.8,9 It is not recommended in routine follow-up after surgery,6,7 but it may have prognostic impact on survival and in detection of recurrence, especially in CEA-negative patients.10,11
Human chorionic gonadotropin (hCG) belongs to the family of pituitary glycoprotein hormones comprising luteinizing hormone, follicle-stimulating hormone and thyroid-stimulating hormone. It has been suggested to act as an angiogenic factor in tumour development.12,13 Elevated serum concentrations of the free β subunit of human chorionic gonadotropin (hCGβ) occur in several malignancies including digestive tract cancers. 14 In CRC, serum hCGβ was shown to be an independent prognostic factor, when measured before surgery of the primary tumour.15,16
Tumour-associated trypsin-inhibitor (TATI) is a 6-kDa peptide, which is produced by several tumours and cell lines. It was initially found in the urine of a patient with ovarian cancer, 17 and later it was found to be identical to pancreatic secretory trypsin-inhibitor (PSTI), also called serine peptidase inhibitor Kazal-type 1 (SPINK1), which is also produced in the pancreas. 18 Increased serum concentrations of TATI occur in several types of cancer, including ovarian, bladder, kidney and pancreatic cancers. 19 In primary CRC, high preoperative concentrations of TATI in serum and high expression of TATI in tumour tissue have been shown to correlate with adverse prognosis and increased risk of metachronous liver metastasis.20,21 However, TATI expression in tumour tissue has been found to be associated with better 5-year survival in metastatic CRC. 22 Thus, the role of TATI as a prognostic marker remains to be established.
The aim of this study was to evaluate whether elevated preoperative or 3 months’ postoperative serum or plasma concentrations of CEA, CA19-9, hCGβ or TATI were prognostic for recurrence or survival in metastatic CRC after resection of liver metastases with curative intent. Reliable prognostic markers would help select patients who need more comprehensive follow-up and more aggressive treatment after liver resection.
Methods
Patients
Serum and plasma samples were obtained from 168 CRC patients who underwent their first resection of liver metastases at the tertiary liver surgery clinic of Helsinki University Hospital between the years 1998 and 2007. The primary tumours (all adenocarcinoma) had been operated between 1988 and 2007 at different hospitals in Finland. Patients with extrahepatic metastases were excluded. All patients alive had a minimum follow-up time of 8 years after liver resection. After resection, all patients were referred to consultation of a medical oncologist for consideration of adjuvant treatment and regular follow-up. All patients underwent clinical examination, whole-body computed tomography (CT) or magnetic resonance imaging (MRI) and blood tests at the resection unit at Helsinki University Hospital 3 months after liver resection.
The study was approved by the Ethics Committee of the Helsinki University Hospital. Collection and analysis of blood samples were approved by the National Supervisory Authority for Welfare and Health (Valvira). Clinical data were retrieved from patient records. Information about dates and causes of death were obtained from the Central Statistical Office of Finland.
Plasma samples and assay procedures
Blood samples were drawn before liver resection and 3 months after the operation in all resected patients; 3 month’s blood samples were taken at the postoperative follow-up visit together with whole-body CT or MRI and clinical examination. That time point was considered optimal, since 3 months after operation, the postoperative complications have resolved, liver tissue has regenerated and does not affect the tumour marker levels anymore and possible adjuvant treatment has been initiated. We also estimated that early tumour marker rise most probably would be due to residual disease and not recurrence.
The serum levels of CEA and CA19-9 were determined with an automatic analyser (Bayer, Tarrytown, NY, USA). The upper reference limit for CEA was 5 μg/L and that for CA19-9 was 26 kU/L based on the routinely used upper reference limits. The serum samples were analysed when taken, and the results were retrieved from clinical records.
Plasma samples were stored at −80°C until analysed at the same time. The plasma concentrations of hCGβ were quantified by an immunofluorometric assay (IFMA) based on monoclonal antibodies, and the detection limit was 0.5 pmol/L. 14 The plasma concentrations of TATI were determined by time-resolved IFMA and the detection limit was 0.15 µg/L. 23
Statistics
Survival was calculated from the date of the first liver resection. Endpoints were defined as follows: disease-free survival (DFS): time to any recurrence of CRC or death of any cause; and overall survival (OS): time to death, irrespective of cause. Complete follow-up data concerning OS were available from all patients. Data cut-off date was 17 June 2015.
The tumour markers as non-normally distributed were analysed with non-parametric tests, such as Mann–Whitney U test. Correlations between s-CEA, s-CA19-9, p-hCGβ and p-TATI were calculated using Spearman’s rho test and Cox regression model, and those between the markers and clinical characteristics using chi-square test. Cox regression was also used to estimate the hazard ratio (HR) of s-CEA, s-CA19-9, p-hCGβ and p-TATI. Optimal cut-off values for hCGβ and TATI for 3-year survival were determined by receiver operating characteristic (ROC) analysis. The impact of tumour markers on DFS and OS was estimated using Kaplan–Meier survival analysis and log rank significance test as well as Cox regression model. In Kaplan–Meier log rank analyses, patients with elevated tumour marker values were compared to those with marker values within reference limits. Univariate and multivariate analyses were carried out using Cox regression model.
Statistical analyses were carried out and figures were created using SPSS Statistics 22 (SPSS Inc., Chicago, IL, USA). A p value less than 0.05 was considered significant.
Results
Patient characteristics
Liver resection was performed in 168 patients, of whom 101 (60%) were male and 101 (60%) had their primary tumour in the colon (Table 1). The metastases were synchronous in 89 (53%) and metachronous in 79 (47%) cases. Median age of the patients at the time of liver resection was 64.3 years (range: 36.3–81.5 years). At that time, 48% of the patients were over 65 years old.
Characteristics and outcome of patients (N = 168) operated for colorectal liver metastases in 1998–2007.

Total number, percentage and median time in years (range) presented.
One patient had a recurrence, but died of another reason.
Disease-free and overall survival after liver resection
Median follow-up time for patients alive was 10.8 years (range: 8.1–16.9 years) and 113 (67%) of all patients died during follow-up. During the follow-up time, 115 (68.5%) of the patients had a recurrence, 24 (14%) already within 3 months after resection and 83 (49%) within 3 years. The OS rate of all patients was 68.5% (115/168) at 3 years and 50% (84/168) at 5 years after resection. Median OS was 4.9 years (interquartile range (IQR): 2.4–9.8 years; Table 1).
Markers in serum and plasma before and 3 months after liver resection
The distributions of CEA, CA19-9, hCGβ and TATI measured before and 3 months after liver resection are shown in Table 2. The preoperative concentrations of CEA correlated with CA19-9 (rho = 0.395, p < 0.001), but not with the other markers; 3 months’ postoperative CEA correlated with CA19-9 (rho = 0.181, p = 0.019) and with TATI (rho = 0.164, p = 0.033), but not with hCGβ; 3 months’ CA19-9 correlated with hCGβ (rho = 0.205, p = 0.008), and TATI with hCGβ (rho = 0.180, p = 0.019).
Concentrations of tumour markers before and after resection of colorectal liver metastases.

CEA: carcinoembryonic antigen; CA19-9: carbohydrate antigen 19-9; hCGβ: human chorionic gonadotropin β; TATI: tumour-associated trypsin-inhibitor.
We calculated the optimal cut-off values for CEA, CA19-9, hCGβ and TATI using ROC curve analyses for prediction of 3-year survival after liver resection. The ROC curves showed some difference in optimal cut-off values for preoperative and postoperative samples. We decided to use the cut-off value of 5 µg/L for CEA (preoperative optimal cut-off value 4.35 and postoperative 5.20 µg/L based on ROC analyses) and 26 kU/L for CA19-9 (preoperative optimal cut-off value 80.5 and postoperative 11.5 kU/L based on ROC analyses) according to the routinely used upper reference limits as the ROC analyses did not provide any further improvement of accuracy. The cut-off values of 1.0 pmol/L for hCGβ and 13 µg/L for TATI were chosen based on the 75th percentiles of both preoperative and 3 months’ values.
Before surgery, the concentrations of CEA, CA19-9, hCGβ and TATI exceeded the cut-off limits in 56%, 36%, 24% and 48% of the cases, respectively, irrespective of possible neoadjuvant treatments; 3 months after liver resection, the concentrations exceeded the limits in 16%, 14%, 23% and 46% of the cases, respectively.
Preoperative marker concentrations, DFS and OS
In Kaplan–Meier log rank analyses, elevated preoperative concentrations of CEA (>5 µg/L), CA19-9 (>26 kU/L) or hCGβ (>1.0 pmol/L) did not associate with shorter DFS. Patients with elevated concentrations of TATI (>13 µg/L) had a shorter DFS within 3 years (p = 0.017) and also within the whole follow-up time (p = 0.051).
Elevated concentrations of CA19-9 (>26 kU/L) before liver resection associated with shorter OS (p = 0.006; Figure 1), as did also elevated CEA (>5 µg/L), but only within 3 years’ follow-up (p = 0.010). Preoperative concentrations of hCGβ or TATI did not associate with shorter OS.

Preoperative values of CA19-9 in relation to overall survival after liver resection.
In Cox regression analysis, the results were similar (Table 3), also in multivariate analysis (data not shown).
Results of Cox regression analysis in relation to 3-year DFS and OS.
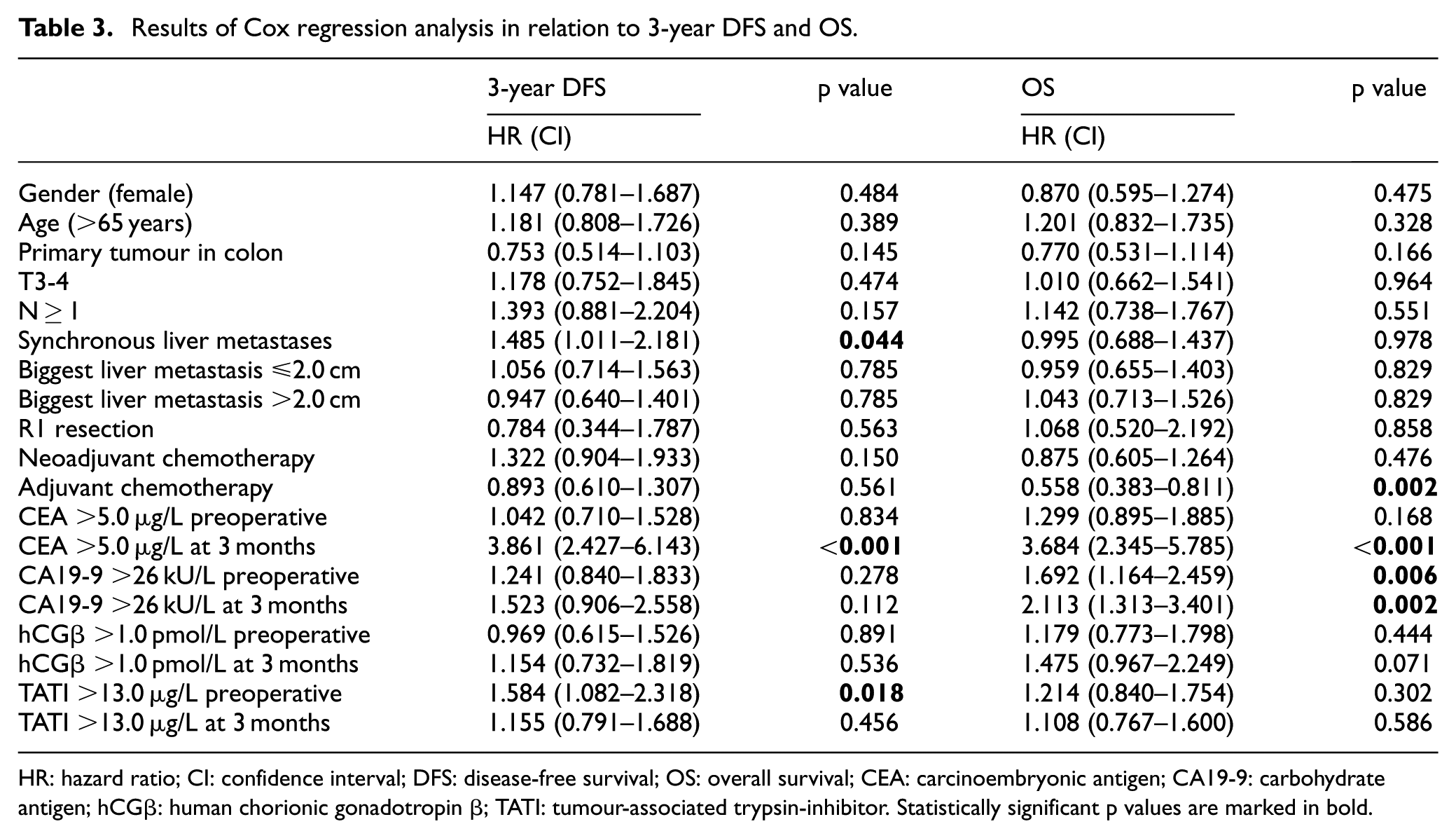
HR: hazard ratio; CI: confidence interval; DFS: disease-free survival; OS: overall survival; CEA: carcinoembryonic antigen; CA19-9: carbohydrate antigen; hCGβ: human chorionic gonadotropin β; TATI: tumour-associated trypsin-inhibitor. Statistically significant p values are marked in bold.
Marker concentrations at 3 months after liver resection, DFS and OS
In Kaplan–Meier log rank analyses, elevated concentrations of CEA (>5 µg/L) and CA19-9 (>26 kU/L) at 3 months after liver resection associated with shorter DFS compared to patients, whose values were within reference limits (p < 0.001; Figure 2(a) and p = 0.011; Figure 2(b), respectively). Elevated concentrations of hCGβ and TATI at 3 months did not associate with shorter DFS.

The 3 months’ values of (a) CEA and 3-year disease-free survival and (b) CA19-9 and long-term disease-free survival.
Elevated concentrations of CEA (>5 µg/L), CA19-9 (>26 kU/L) and hCGβ (>1.0 pmol/L) at 3 months associated with shorter OS within 3 years after liver resection (p < 0.001, p = 0.001 and p = 0.017, respectively), and CEA and CA19-9 also within the whole follow-up time (p < 0.001 and p = 0.002, respectively; Figure 3(a) and (b). TATI did not show correlation to impaired OS.

The 3 months’ values of (a) CEA and (b) CA19-9 in relation to overall survival after liver resection.
The results of Cox regression analysis were in line with these (Table 3), but in multivariate analysis only elevated CEA remained statistically significant (data not shown).
Elevated marker concentrations at 3 months and recurrence
At the 3 months’ control, 24 patients (14.3%) had already a recurrence. When we excluded them from the analyses, there were altogether 144 patients, of whom 91 (54.2%) developed recurrence after 3 months as follows: 85% of the patients with elevated CEA (total N = 13), 80% with elevated CA19-9 (total N = 15), 54% with elevated hCGβ (total N = 28), and 63% of those with elevated TATI (total N = 65). Altogether 131 patients had a normal CEA level, and of those, 80 (61%) had a recurrence later. In Kaplan–Meier log rank analysis, these patients developed recurrence later than those with elevated CEA (p = 0.001). Of the patients who had a normal CEA level but developed recurrence, 9 had elevated CA19-9, 12 elevated hCGβ, and 33 elevated TATI at 3 months.
Univariate and multivariate analyses
In Cox regression analysis, possible neoadjuvant or adjuvant chemotherapy before or after liver resection did not show statistical correlation to DFS, but adjuvant treatment was associated with better OS (p = 0.002; Table 3). Synchronous liver metastases were associated with shorter 3-year DFS compared to metachronous liver metastases (p = 0.044; Table 3). The other results of univariate analyses are shown in Table 3.
Multivariate analyses were carried out with adjustment for synchronous liver metastases or adjuvant chemotherapy (data not shown). In these analyses, elevated CEA (>5.0 µg/L) at 3 months correlated with impaired 3-year DFS and OS (p < 0.001, HR 3.776 and p < 0.001, HR 3.715). Adjuvant treatment remained statistically significant in relation to OS (p = 0.001, HR 0.522). Preoperatively or 3 months postoperatively elevated CA19-9 (>26 kU/L) did not show statistically significant correlations, nor preoperatively elevated TATI (data not shown).
Discussion
To our knowledge, this is the first study to examine the prognostic value of CEA, CA19-9, TATI, and hCGβ 3 months after liver resection of CRC metastases. Our results show that elevated CEA, CA19-9 and hCGβ marker concentrations 3 months after curative-intent liver resection have prognostic value. Preoperatively elevated CEA and CA19-9 were associated with shorter survival.
The strengths of this retrospective study are a quite large patient material, a long and comprehensive follow-up time, and all consecutive, eligible patients at a single institution included. The limitations of our study are the heterogeneity of the patient material, missing information on the BRAF mutation status, and the lack of standardised postoperative measurement point for tumour markers.
To minimise heterogeneity, we only included patients with liver-only metastases, patients with extrahepatic metastases were excluded based on aberrant tumour marker behaviour, and only the first liver resection was taken into consideration. Missing BRAF information is a limitation of this study as it may partly explain the fact that 24 (14.3%) patients had a recurrence already at 3 months after resection. BRAF mutation status was not routinely assessed in 1998–2007, and thus, one significant prognostic factor is missing. In any case, it is likely that recurrences less than 3 months after liver resection are due to residual disease and not due to recurrence.
Very little data about postoperative marker measurement is available and even less on the timing of assessment. Too short an interval postoperatively is affected by postoperative complications like bile duct interference with CA19-9 levels, effects of postoperative growth factors on tumour marker levels, inter-individual variation in postoperative tumour marker half-life, and the timing of adjuvant treatment initiation. Thus, 3 months was considered an optimal time point for predictive tumour marker assessment after liver resection.
Preoperatively elevated CEA and CA19-9 were associated with shorter survival, as shown in previous studies,24,25 also after neoadjuvant chemotherapy in recent studies.26–28
Increased levels of CEA 3 months after liver resection showed the strongest association with early recurrence and impaired OS after liver resection, which is in line with the results of a recent Japanese study. 29 They measured CEA levels before resection of colorectal liver metastases and within 1 month after it and showed that elevated postoperative CEA (>5 µg/L) was a significant risk factor for early recurrence. Elevation of CEA has also been shown to be of value in follow-up after liver resection. 30 In our study, however, 78% of patients (90/115) who experienced a recurrence did not have elevated CEA concentrations at 3 months. That makes it obvious that if we only look at the CEA levels, a notable number of patients with a high risk of recurrence will go undetected. It is still worth noticing that the patients with a normal CEA level at 3 months developed recurrence later than those with elevated CEA, and it is possible that their marker level had not yet risen at our measurement point.
Elevated concentrations of CA19-9 3 months after liver resection correlated not only with impaired OS, but also with shorter DFS in longer follow-up. This is in line with the recent Japanese study evaluating the rising of CA19-9 within a month from liver resection. 29 In metastatic CRC, elevated CA19-9 levels have been associated with poor survival,10,11,31 which corresponds to our results. Interestingly, there were nine patients with elevated CA19-9, but normal CEA levels 3 months after liver resection, who developed a recurrence. Thus, measuring CA19-9 is useful at least for CEA-negative patients. This is in line with the findings in an intensive follow-up programme after liver resection, in which elevations of CEA and CA19-9 were complimentary, but association between tumour markers and prognosis was not presented. 32 It is worth noticing, however, that normal levels of CEA and CA19-9 at 3 months after liver resection do not exclude later recurrence.
The results for hCGβ are interesting. Elevated levels (>1 pmol/L) 3 months after liver resection showed a statistically significant correlation with both impaired DFS and OS. Thus, hCGβ is a marker of adverse prognosis for selected patients as previously shown in preoperative samples, 33 but further research is needed before its use may be recommended in follow-up after liver resection for metastases of CRC.
Previous results on the use of TATI in CRC have been conflicting. High serum levels and tissue expression in the primary tumour have been shown to correlate with the risk of metastases.20,21 Nevertheless, in metastatic CRC, high TATI expression in the primary tumour tissue has been associated with better 5-year survival.15,22 However, in our study, TATI was not associated with impaired survival, and results for recurrence were conflicting. TATI does not seem to be of prognostic or predictive value after liver resection for metastases of CRC. As TATI may also behave as an acute-phase protein, 26 it is possible that elevated concentrations at 3 months are caused by ongoing tissue repair and that TATI may be more informative at later stages.
Measuring tumour markers before liver resection gives important information on OS and the risk of recurrence, which should be taken into account when considering liver resection. Preoperatively high values of CEA and CA19-9 do not correlate directly with high risk of early recurrence, but they are a sign of adverse outcome. Postoperatively elevated values, however, show a clear risk of recurrence. In this patient group, it might be worth considering longer or more intensive adjuvant treatment after resection and more frequent follow-up to find resectable recurrences.
Conclusion
CEA remains the best prognostic and predictive marker for CRC with liver metastases. Elevated serum concentrations of CEA at baseline and 3 months after resection of liver metastases are a sign of adverse prognosis. In patients with normal CEA concentrations, assay of CA19-9 and possibly hCGβ are of additional value. The levels of these markers appear informative for prediction of recurrence, but cannot yet be used as evidence to change the strategies for follow-up or treatment. Measuring tumour markers 3 months after liver resection gives additional information with CEA and CA19-9.
Footnotes
Acknowledgements
The authors thank Marianne Niemelä for marker determinations and Professor Seppo Sarna for advice on the statistical analyses.
Authors’ contributions
Reetta Peltonen collected, analysed and interpreted the data. Pia Österlund participated in the statistical analyses and interpreted results. Marko Lempinen participated in the statistical analyses. Arno Nordin helped to interpret data. Ulf-Håkan Stenman participated in designing the study, analysing data and interpreting results. Plasma samples were analysed in Ulf-Håkan Stenman’s laboratory. Helena Isoniemi designed and directed the study, participated in the statistical analyses and interpreted results. All authors contributed to writing the manuscript. All authors have read and approved the final manuscript.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
