Abstract
Colorectal cancer is among the three most common cancer types for both genders, with a rising global incidence. To date, prognostic evaluation is difficult and largely dependent on early detection and successful surgery. UCHL5/Uch37 is an integral part of the protein homeostasis network as one of the three deubiquitinating enzymes associated with the 26S proteasome. Here, we have investigated in colorectal cancer the possible association of UCHL5 tumor expression and patient survival. UCHL5 tumor expression was evaluated by immunohistochemistry in 779 surgically treated colorectal cancer patients from Helsinki University Hospital, Finland, with assessment of clinicopathological parameters and the effect of UCHL5 expression on patient survival. High and undetectable UCHL5 expression both correlated with increased overall disease-specific survival in the subgroup of patients with lymph-node-positive (Dukes C/stage III) rectal cancer. Within this subgroup of 105 stage-III rectal cancer patients, none of the 7 with high UCHL5 expression died of colorectal cancer within 10 years after surgery (p = 0.012). A similar, though less prominent, survival trend occurred throughout the whole patient cohort. In conclusion, UCHL5 is a promising novel prognostic marker in lymph-node-positive rectal cancer. Our results also advance the currently limited knowledge of biomarkers in colorectal cancer treatment.
Introduction
Colorectal cancer (CRC) is among the three most common cancer types worldwide, with an annual incidence of over one million new cases. Disease recurrence is common, with an overall survival rate of approximately 65% in high-income and less than 50% in low-income countries. 1 Successful treatment and subsequent survival are based largely on a combination of early detection, radical surgery, preoperative radiotherapy (in low rectal cancer), and neoadjuvant or adjuvant chemotherapy or both. Prognostic evaluation of CRC is difficult, and novel prognostic markers are critically needed to aid in treatment decisions.
Proteasome inhibitors such as bortezomib have emerged as attractive therapeutics for refractory multiple myeloma and mantle cell lymphoma, leading the way for a new generation of proteasome inhibitors in clinical trials with hematologic malignancies and solid tumors.2,3 However, occurrence of dose-limiting toxicities and drug resistance has turned attention to identifying alternative targets that modulate the ubiquitin–proteasome system (UPS). More than 40 deubiquitinating enzymes (DUBs), including the three proteasome-associated DUBs POH1(Rpn11), Usp-14, and UCHL5/Uch37 are involved in cancer.3,4 UCHL5 and Usp-14 in particular are tempting cancer therapy targets, due in part to their regulation of the deubiquitination step prior to proteasomal degradation of substrates. UCHL5, a cysteine protease, belongs to the family of ubiquitin C-terminal hydrolases. UCHL5 function is crucial, as UCHL5-knockout mice are embryonically lethal. 5 The Caenorhabditis elegans homolog of UCHL5, UBH-4, tissue specifically regulates proteasome activity in this species, as well as modulates health and lifespan. 6 UCHL5 binds to the proteasome via a reversible 7 and evolutionarily conserved 6 association with the 19S regulatory particle subunit Admr1/Rpn13, which activates its deubiquitinating activity.8–10 UCHL5 also associates with the NFRKB subunit of the INO80 chromatin-remodeling complex, resulting in DUB inactive UCHL5.11,12
UCHL5 shows ubiquitous expression in the majority of normal human tissues with levels ranging from low to high expression, and from undetectable to high levels in cancer tissues. 13 We have shown earlier that in the C. elegans intestine, UBH-4 expression is negatively regulated by the transcription factor DAF-16/FOXO and that in human cancer cell-lines, knockdown of uchl5 by siRNA enhances degradation of proteotoxic proteins. 6 Study of the role of UCHL5 expression in cancer has previously involved esophageal squamous cell carcinoma, hepatocellular carcinoma, and epithelial ovarian cancer, where high UCHL5 expression, analyzed as low versus high expression, correlated with poor survival and increased cancer recurrence.14–16 In contrast, we have shown in another study that high UCHL5 expression associates with better survival in pancreatic ductal adenocarcinoma. 17 Here, we have investigated the possible prognostic role of UCHL5 in CRC.
Materials and methods
Patients
The study cohort comprised 840 successive CRC patients surgically treated between 1983 and 2001 at the Department of Surgery, Helsinki University Hospital, Finland. Follow-up vital-status data came from the Finnish Population Register Centre, and cause of death for those deceased was provided by Statistics Finland. Evaluation of UCHL5 expression was possible in 779 of the patient samples. Median age at diagnosis of all patients was 66 years, with a median follow-up of 5.1 years (range 0–25.8 years); 18 5-year survival after surgery was 58.9% (95% confidence interval (CI): 55.0%–62.8%). 18
Preparation of tumor tissue microarrays
Formalin-fixed and paraffin-embedded tumor samples came from the archives of the Department of Pathology, Helsinki University Hospital. Representative tumor areas had been marked by an experienced pathologist on hematoxylin- and eosin-stained tumor slides. A semiautomatic tissue microarray (TMA) instrument (Beecher Instruments, Silver Spring, MD, USA) served for acquiring and mounting three 1.0-mm-diameter punches from each donor block on the recipient TMA blocks. 19
Antibodies
Rabbit anti-UCHL5 antibody (HPA005908, dilution 1:800; Sigma-Aldrich MO, USA) was used for immunohistochemical staining. A subset of samples was validated with two additional anti-UCHL5 antibodies (SAB1400553, 1:500; Sigma-Aldrich MO, USA; sc-271002, 1:1000; Santa Cruz Biotechnology TX, USA).
Immunohistochemistry
TMA blocks were newly cut into 4 µm sections. The slides were deparaffinized with xylene and rehydrated with decreasing ethanol and distilled water series, followed by treatment in a PreTreatment module (Lab Vision Corp., Fremont, CA, USA) in Tris-HCl (pH 8.5) buffer for 20 min in 98°C for antigen retrieval. The slides were stained with Autostainer 480 (Lab Vision Corp.) by the Dako (Glostrup, Denmark) REAL EnVision Protection system, Peroxidase/DAB+, and Rabbit/Mouse. Tissues were incubated with primary antibody for 1 h at room temperature.
Sample scoring and imaging
UCHL5 expression was scored according to tumor-staining intensity as 0 (undetectable staining, negative), 1 (low staining), 2 (moderate staining), or 3 (strong staining). To ensure reproducibility and precision, the scoring was performed blindly and independently by J.H. and L.A. Occasional differences in scoring were discussed until consensus. The highest score from the triplicate for each sample served for statistical analysis. Representative images of all staining intensities were taken at random, and their brightness was adjusted similarly for all images by the Adobe Photoshop version CS6 (64 bit).
Statistical analysis
The four scoring categories (0–3) of UCHL5 expression were separated for all statistical analysis. Evaluation of the association of UCHL5 expression and clinicopathological parameters was by Fisher’s exact test and linear-by-linear association test (with Monte Carlo estimation of exact p values). Kaplan–Meier survival analysis was used to estimate cumulative survival, and the log-rank test was used to detect differences between groups. If the overall comparison between expression levels of UCHL5 as related to survival was significant, a post hoc analysis was performed by individually comparing UCHL5 expression levels 0–2 to the high expression level 3 one by one. The Šidác correction was applied to multiple comparisons. CIs (95%) were calculated for 5- and 10-year survival rates and mean survival times. Cox proportional hazards regression models served for uni- and multivariate analyses of survival data. The Cox model assumption of constant hazard ratios over time was tested by including a time-dependent variable for each testable variable. In order to fulfill the Cox model assumption, hazard ratios of Dukes D class and differentiation status were analyzed in two time periods with the time-dependent Cox model. Interactions were considered, but no significant interaction emerged. A p value less than 0.05 was considered significant, and two-tailed tests were used. Statistical analysis was performed with IBM SPSS statistical package version 22 (IBM, New York, NY, USA) and SAS version 9.4 (SAS Institute Inc., Cary, NC, USA).
Results
Immunostaining of UCHL5 in CRC
UCHL5 immunostaining was reliably evaluated in 779 (92.7%) of the 840 consecutive patient samples. In the tumor tissue, UCHL5 expression was mostly cytoplasmic and uniform in intensity (Figure 1). The majority of samples showed either low (323, 42%) or moderate (291, 37%) UCHL5 immunoreactivity. Negative and high UCHL5 expression was present in 110 (14%) and 55 (7%) of the samples. For relative distribution of staining intensities and patient numbers in more detail, see Table 1. Normal-appearing cells adjacent to the tumor displayed low or negative staining (data not shown). The immunostaining of UCHL5 was validated with two additional anti-UCHL5 antibodies in a subset of 70–80 samples, and these showed similar staining intensities and patterns (data not shown).

Immunohistochemical staining pattern of UCHL5 in colorectal cancer. Representative images of (a) UCHL5-negative (0), (b) low (1), (c) moderate (2), and (d) strongpositive (3) immunoreactivity. Original magnification was 200×.
Association of UCHL5 expression with clinicopathological parameters.
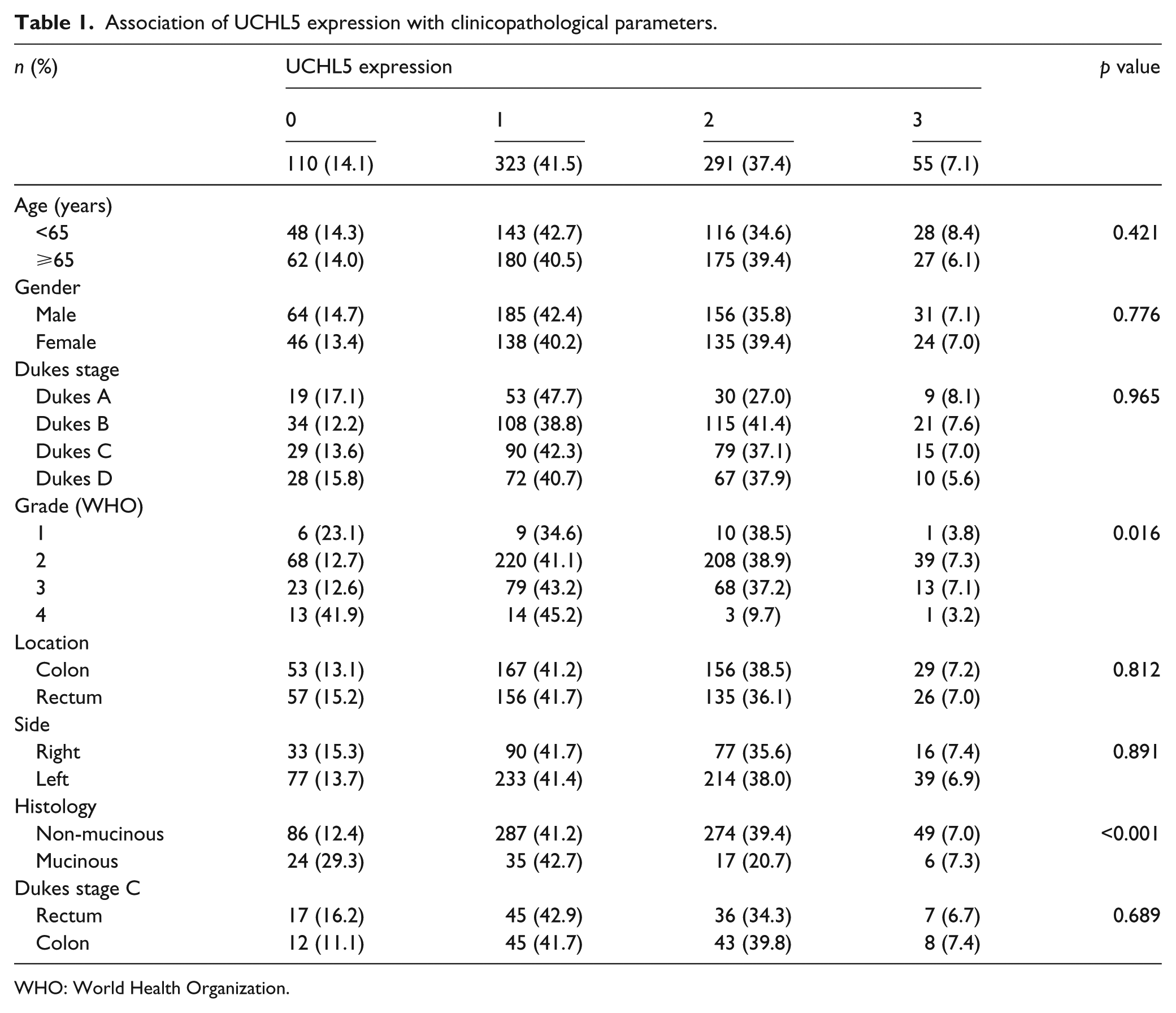
WHO: World Health Organization.
Fisher’s exact test was used for tables with two rows and linear-by-linear association test for tables with more than two rows. Missing data were excluded from the analysis.
Association of UCHL5 expression with clinicopathological parameters
The distribution of UCHL5 expression showed no association with age, gender, stage, or location (right vs left hemicolon, or colon vs rectum; Table 1). However, UCHL5 expression was associated strongly with differentiation status (p = 0.016) and non-mucinous histology (p < 0.001).
Survival analysis
In the overall patient material, no significant difference in disease-specific survival (DSS) was associated with UCHL5 expression, though patients with high UCHL5 expression tended to fare better than patients exhibiting low-to-moderate expression values (Figure 2). When divided by tumor location, this enhanced survival trend in rectal cancer patients was pronounced (Supplementary Figure 1a and b). Further analysis revealed that high UCHL5 expression associated with considerably increased DSS in the subgroup of patients with lymph-node-positive rectal cancer (Dukes C/stage III; p = 0.012; Figure 3). For the total of 81 patients with low or moderate UCHL5 expression the 10-year DSS was 25.8% (95% CI: 13.2%–40.3%) and 35.9% (95% CI: 19.0%–53.2%) respectively, whereas for the 7 patients with high UCHL5 expression the 10-year DSS was 100.0% (Table 2). High UCHL5 expression differed significantly from low in terms of survival (p = 0.0158, log-rank with Šidác correction), when examined over the whole follow-up period of approximately 20 years. A similar survival trend, though not statistically significant, emerged in the subgroup of 3 patients with high UCHL5 expression who had advanced rectal cancer (Dukes D/stage IV; Supplementary Figure 2). In agreement with these results regarding a survival benefit of UCHL5, multivariate survival analysis adjusted for age, Dukes/stage, and differentiation status showed that high UCHL5 expression associated with a lower risk of death in all our rectal cancer patients (Supplementary Table 1). Additionally, lymph-node-positive rectal cancer patients exhibiting negative UCHL5 tumor expression had a 10-year DSS of 56.3% (95% CI: 24.1%–79.3%), which was considerably higher, though not significantly different, than in patients with low or moderate expression (Figure 3 and Table 2). None of the seven Dukes C patients with high UCHL5 expression received preoperative radiotherapy or postoperative cytotherapy. In the same subgroup of Dukes C patients, 4 patients out of 36 with moderate expression, 20 out of 45 with low expression, and 2 out of 17 with negative UCHL5 expression received preoperative radiotherapy. Similarly, postoperative cytotherapy was received by 3 patients with moderate expression, 21 with low expression, and 2 with negative UHCL5 expression.

Patient survival in correlation with UCHL5 tumor expression in colorectal cancer. High and UCHL5-negative expression associate with a trend toward more favorable prognosis in CRC (p = 0.225, overall log-rank test).
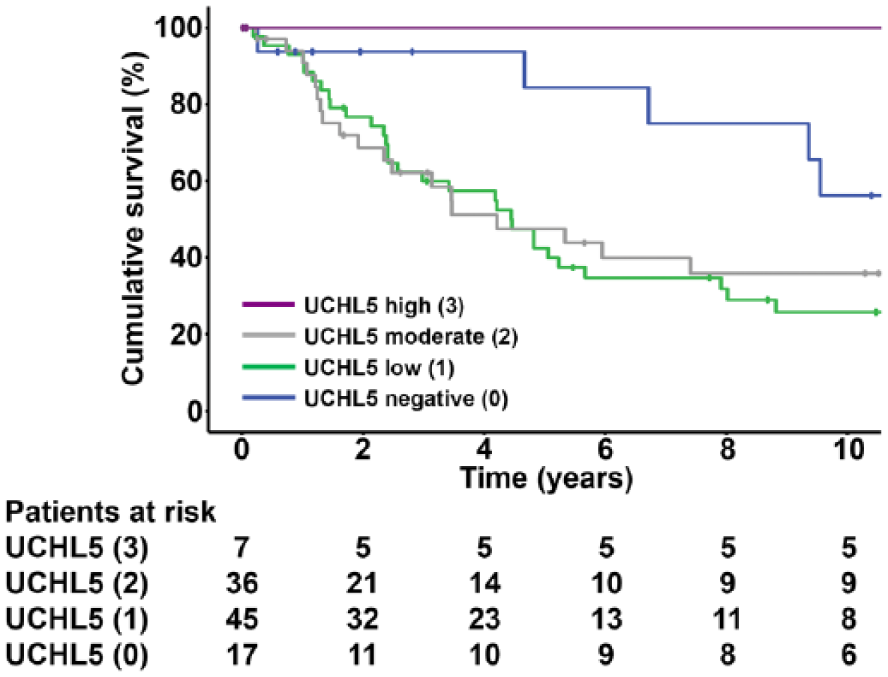
Patient survival in correlation with UCHL5 tumor expression in lymph-node-positive rectal cancer. Patients with strong UCHL5 (3) or UCHL5-negative (0) immunoreactivity exhibit increased survival in lymph-node-positive (Dukes C/stage III) rectal cancer (p = 0.012, overall log-rank test).
Mean and cumulative 5- and 10-year disease-specific survival percent.

CI: confidence interval.
Calculated from all follow-up data (25 years).
Discussion
We show that the proteasome-associated DUB UCHL5 is a novel and promising prognostic marker candidate in lymph-node-positive (Dukes C/stage III) rectal cancer. In this subgroup, patients with high or undetectable UCHL5 immunohistochemical tumor expression, when compared to patients with low or moderate tumor expression, exhibited markedly increased survival both 5 and 10 years after surgery (Figure 3).
Only a few reports have described UCHL5 tissue expression in relation to cancer-patient survival. High UCHL5 expression, as detected by immunohistochemistry or western blotting, correlates with poor survival and increased cancer recurrence in esophageal squamous cell carcinoma, hepatocellular carcinoma, and epithelial ovarian cancer.14–16 One explanation for our contradictory results may be the different manner of expression analysis, that is, comparison of only high versus low expression, as opposed to our method of separately analyzing all four scored expression levels. Considering the nonlinear manner in which UCHL5 expression and survival correlate in our dataset, a survival benefit could be observed only when all four categories of expression levels were kept separate. Alternatively, UCHL5 may also play different roles in cancer, depending on the tissue of origin, which would be in line with our earlier finding that the UCHL5 orthologue UBH-4 regulates proteasome activity in a tissue-specific manner in C. elegans. 6
How does UCHL5 expression mediating survival benefit in lymph-node-positive rectal cancer? Of particular interest is the fact that high UCHL5 expression and undetectable UCHL5 expression each are beneficial for patient survival. We have earlier showed that a small reduction in expression of the UCHL5 orthologue UBH-4 by RNAi leads to an increase in C. elegans lifespan, whereas a stronger knockdown reduces lifespan and brood size. 6 Accordingly, overexpression of UBH-4 in the intestine reduces the lifespan of long-lived daf-2 (insulin/IGF-1 receptor orthologue) mutants. 6 In addition, we have established that the insulin/IGF-1 signaling pathway downregulates UBH-4 expression specifically in intestinal cells. 6 Thus, to maintain normal cellular function and survival, the amount of UCHL5 may vary specifically by cell- and tissue-type. Studies on human histological tissue samples show that UCHL5 expression varies among tissues in both cancer and normal-cell environments. 13 UCHL5 is one of the three proteasome-associated DUBs required for efficient removal of polyubiquitin from proteasomal substrates, leading to substrate degradation by the proteasome. Downregulation of UCHL5 or its orthologue enhances proteasomal activity in C. elegans intestinal cells and in human cancer cell lines.6,20 Moreover, Usp-14, another proteasome-associated DUB, exerts an inhibitory effect on proteasome activity in human cells. 21 One could speculate that a high level of UCHL5 markedly inhibits the proteasome, leading to detrimental accumulation of a particular set of proteasomal substrates. Similarly, lack of UCHL5 may also prevent degradation of certain substrates, which in turn may trigger apoptosis. Recently, UCHL5, along with its proteasomal binding partner Rpn13, is reported to be involved in maintaining cell-cycle progression and DNA replication. 22 In addition, UCHL5 may assert its effect through a proteasome-independent mechanism via its other interacting partner NFRKB of the INO80 chromatin-remodeling complex, which inhibits UCHL5.11,12 Research should focus upon establishing the molecular mechanisms of UCHL5 in cancer.
Proteasome inhibitors are successfully used for treatment of multiple myeloma and mantle cell lymphoma, albeit severe side effects may occur, related to dose-limiting toxicity and drug resistance, especially during cancer recurrence.2,23 Thus, attention has turned to other modulators of the UPS, including DUBs, as promising treatment targets in cancer.4,24 Pharmacological dual inhibition of UCHL5 and Usp-14 induces cytotoxicity, particularly in cancer cells, as well as inhibition of tumor growth.3,25,26 This provides an attractive strategy to overcome resistance to conventional proteasome inhibitors.
CRC has a 5-year relative survival of 65% in high-income countries and below 50% in low-income countries. 1 In Finland, the overall survival rate for colon cancer is 62% for both genders and in rectal cancer 64% and 65% for men and women, respectively. 27 In our material, collected between the years 1983 and 2001 and including both colon and rectal tumors, the survival rate was 60% for men and 57% for women. In the United States, the 5-year survival for rectal cancer is 68%, 28 whereas in our dataset the survival rates were 61% for men and 56% for women. The 5-year survival of Dukes B/stage II CRC patients in the United Kingdom (77%) 29 correlates closely with the survival rate of our patient cohort (78%). The relative 5-year survival rate for colorectal lymph-node-positive cancer (Dukes C/stage III) is 47.7% in the United Kingdom, 29 compared to approximately 55% in our dataset. As correct assessment of cancer stage has improved over time, and considering that our material is relatively old, this could in part explain the difference in survival rates in colorectal lymph-node-positive cancer between our patients and the UK cohort.
To date, the strongest prognostic factor in CRC is cancer stage at diagnosis. Some patients receive adjuvant therapy to improve survival and reduce recurrence. Today, this is routine for all stage-III patients and provides a 10% absolute increase in 5-year survival, although evidence indicates that the benefit from adjuvant treatment is larger in colon than in rectal cancer.30–33 In stage-II disease, the benefit of adjuvant chemotherapy is less obvious, and only 15% to 20% develop recurrence after surgery in the absence of chemotherapy. Interestingly, in our old retrospective series, none of the patients with rectal lymph-node-positive cancer (stage III) and high UCHL5 expression had received adjuvant treatment after surgery. This suggests that UCHL5-mediated survival benefit is not conveyed via improved efficacy of adjuvant treatment. Equally interesting is that none of the patients in this subgroup had received neoadjuvant therapy, indicating that the survival benefit of UCHL5 is derived from changes in the tumor cells.
Our results show that UCHL5 is of an encouraging prognostic value in metastatic rectal cancer. This is especially noteworthy, as there is a distinct lack of effective prognostic CRC markers, and our observations may thus prove to have clinical relevance. Recognizing those patients, who would benefit from or not require adjuvant therapy, would be advantageous in making pre-treatment decisions. However, it should be noted that in our current dataset, only 7 patients exhibited high UCHL5 expression in lymph-node-positive rectal cancer. In support of our finding in stage-III patients, those with advanced rectal cancer (Dukes D/stage IV) and high UCHL5 expression also displayed similar, though not statistically significant, improved survival (Supplementary Figure 2). A total of 40 patients had a survival benefit due to either negative or high UCHL5 expression in combination with Dukes C/stage III or Dukes D/stage IV rectal cancer. The survival benefit from UCHL5 needs to be validated in other CRC patient cohorts, and the prognostic potential of UCHL5 should also be investigated for other types of gastrointestinal cancer. In support of a UCHL5-mediated survival benefit, our concurrent study in pancreatic ductal adenocarcinoma has demonstrated that high UCHL5 expression associates with increased patient survival also in this cancer type. 17
In conclusion, we have linked the prognostic significance of UCHL5 tumor expression in CRC both to tumor location and cancer stage. We propose that UCHL5 is a promising novel prognostic marker in lymph-node-positive rectal cancer. Our findings may also impact the development of new treatment therapies targeting the UPS.
Footnotes
Acknowledgements
The authors thank Päivi Peltokangas and Eveliina Markkula for first-rate technical assistance. C.H. and C.I.H. worked in experimental design and conception. L.A. and J.H. worked in experimentation. L.A., H.M., and M.L. carried out data analysis. C.H. and C.I.H. contributed reagent/material/analysis tool. L.A., H.M., C.H., and C.I.H. worked in writing of the manuscript.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Ethical approval
All procedures performed in studies involving human participants were in accordance with the ethical standards of the Institutional and/or National Research Committee and with 1964 Helsinki Declaration and its later amendments or comparable ethical standards. Our study was conducted with approval from the Surgical Ethics Committee of Helsinki University Hospital (Dnro HUS 266/E6/06, extension TMK02 §66 17.4.2013). Permission to use archived tissue samples without informed individual consent was provided by The National Supervisory Authority of Welfare and Health (Valvira Dnro 10041/06.01.03.01/2012).
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by grants to C.I.H. from the Academy of Finland (259797 and 297776), Medicinska Understödsföreningen Liv och Hälsa r.f., and Ruth and Nils-Erik Stenbäck Foundation and to C.H. from the Sigrid Jusélius Foundation and the Cancer Society of Finland. L.A. was supported by the Doctoral Programme in Biomedicine of University of Helsinki and by grants from the Victoria Foundation and the K. Albin Johansson Foundation.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
