Abstract
Research towards biomarkers that predict patient outcome in colorectal cancer (CRC) is rapidly expanding. However, none of these biomarkers have been recommended by the American Association of Clinical Oncology or the European Group on Tumor Markers. Current staging criteria result in substantial under- and over-treatment of CRC patients. Evasion of apoptosis, a characteristic feature of tumorigenesis, is known to correlate with patient outcome. We reviewed the literature on immunohistochemistry-based studies between 1998 and 2011 describing biomarkers in this pathway in CRC and identified 26 markers. Most frequently described were p53, Bcl-2, survivin, and the Fas and TRAILR1 receptors and their ligands. None of the studies reviewed provided sufficient support for implementing a single marker into current clinical practice. This is likely due to the complex biology of this pathway. We suggest focusing on the combination of key markers within the apoptosis pathway that together represent an ‘apoptotic tumor profile’, which better reflects the status of this pathway in a tumor.
Introduction
Colorectal cancer (CRC) is currently one of the major contributors to cancer-related deaths worldwide.1,2 The amount of data emerging from studies aimed at optimizing the diagnostic process and treatment of this disease is rapidly increasing. This makes the process of tumor development in CRC one of the most thoroughly studied and best characterized models of tumorigenesis. By emphasizing the need of early detection and development of new and improved treatment regimens, an increased understanding of the disease led to decreased mortality rates of nearly 5 percent over the last decade.3–10 However, CRC-related morbidity and mortality affects approximately 800,000 individuals each year worldwide. 2 The survival of CRC patients largely depends on disease stage at the time of diagnosis and varies widely between stages. In clinical practice, however, treatment allocation and outcome prediction is still solely based on the International Union Against Cancer (UICC) Tissue Node Metastasis (TNM) classification. 11 Addition of several pathology-based tumor characteristics is currently used to identify high-risk stage II patients that may benefit from adjuvant chemotherapy. These include perforation of the bowel wall at presentation, tumor invasion at the T4 level, venous tumor invasion, lymph node yield less than 10, and poor or no differentiation of the tumor cells. 12 There is substantial evidence that even with the addition of these risk factors of poor outcome, TNM classification falls short in daily practice and may cause over- or, even worse, under-treatment of patients.11,13–18
In an attempt to improve treatment outcomes for CRC patients, both the American Society of Clinical Oncology's Tumor Markers Expert Panel (ASCO TEMP-2006) and its European counterpart, The European Group on Tumor Markers (EGTM-2007), have reviewed the available literature to determine the clinical applicability of a number of widely studied biomarkers.19–21 Their conclusions were clear and consistent: despite the overwhelming amount of literature, no biomarkers have been recommended for clinical use. Therefore, to improve current staging criteria, new biomarkers must be identified and validated for clinical use. Pepe et al 22 have developed a five-step program that can be used for the development of new biomarkers. The first step is biomarker discovery in a preclinical, exploratory setting. Subsequently, the clinical value of these biomarkers must be determined and verified in a large retrospective study. These results then need to be the validated and eventually confirmed by a prospective randomized controlled trial. It is not until these steps are completed successfully that biomarkers are ready for introduction into clinical practice. The first step, which involves identifying or discovering new biomarkers, can be accomplished by studying the process of tumorigenesis and its related pathways. Cancer cells harbor at least six features that distinguish them from normal cells, one of which is the characteristic ability to evade programmed cell death or apoptosis. 23 In normal tissues, apoptosis plays a pivotal role in the maintenance of tissue homeostasis and the development of the immune system.24,25 Disturbance of this process in tumor cells results in the impaired removal of mutated cells and contributes to tumor progression. In addition, evasion of apoptosis enables malignant cells to escape from tumor immune surveillance and to acquire resistance to cancer therapy. In previous retrospective studies, the status of the apoptotic pathway in a tumor was shown to be of prognostic value in colorectal cancer patients.26–37 Therefore, we focused on this pathway in our search for new potential prognostic biomarkers in colorectal cancer. In this review, we provide an overview of studies designed to determine the prognostic value of biomarkers within the apoptotic pathway in colorectal cancer. Furthermore, we will discuss some of the difficulties and controversies that can arise when studying this tightly regulated and complex process. The goal is to identify key biomarkers in the apoptotic pathway that may be used clinically to determine cancer prognosis. We first discuss the route of apoptosis to identify key proteins in this process and then link this information to studies that examined the prognostic value of these proteins in colorectal cancer. Since immunohistochemistry (IHC) is still the most widely applied and available technique in pathology to determine the expression status of tumor-associated proteins and to study the clinical prognostic relevance of biomarkers, we limited our search to IHC studies.
Data Collection and Analysis
In order to review the literature on prognostic biomarkers related to the pathway of apoptosis and determined using IHC in CRC patients, we performed a search of the PubMed, Embase, and Web of Science databases. We used broad search terms, as recommended in the Stroup guidelines, 38 to identify publications of interest published between January 1998 and June 2011. Key search terms included colorectal cancer, biomarker, apoptosis, prognosis, and immunohistochemistry. The following search strategy (simplified) shows how some of these terms were combined in our Web of Science Search; “TS = ((colorectal or colon or colonic or rectal or rectum) SAME (neoplasm or cancer or tumor or carcinoma)) AND TS = ((prognostic or tumor or cancer or neoplasm or biological or intracellular or signaling or intracellular signaling) SAME (marker or protein or peptide)) AND TS = ((prognosis or prognostic or morbidity or mortality or recurrence or relapse or (disease SAME progression))) AND TS = (immunohistochemistry or immunolabeling or immunocytohistochemistry). After amalgamating the results from the three medical databases and discarding the duplicates, this strategy yielded a total of 2923 unique citations. To extract papers for review, we screened the results for title and abstract. We used the following criteria to determine whether a study was considered eligible for the review:
The study contained data for a marker directly involved in the pathway of apoptosis;
The study was performed in primary tumors from CRC patients;
The study was performed using IHC;
The study contained an analysis of the relationship between expression of the marker and clinical outcome. We selected only studies that used logistic regression or survival curve-based statistical analysis methods to evaluate the impact of a marker;
A full publication in English with details of the method used was available.
Results
Overall, we were able to identify 26 potentially prognostic biomarkers that are directly involved in the apoptotic pathway, which will be discussed in detail below (Fig. 1). These markers were all studied using IHC in the 124 eligible publications that remained after applying our selection criteria from the total of 2923 publications. Expression patterns of these apoptotic (bio)markers were related to patient outcome using logistic regression or survival curve-based analysis methods. Most of the papers of the over 800 were excluded because they described the expression of markers related to the pathway of apoptosis in other types of cancers than colorectal cancer, despite the fact that our search terms included colorectal cancer as a major search term. Over 900 citations were excluded because they did not describe the marker in primary colorectal cancer lesions but rather in metastatic lesions. Table 1 provides an overview of our selection criteria and the corresponding number of citations that were excluded based on these criteria.
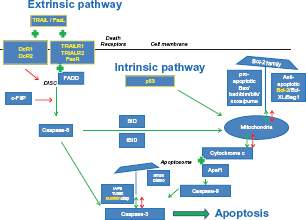
The pathway of apoptosis. A simplified schematic view of the intrinsic and extrinsic pathway of apoptosis and their regulators as described in this review. A green arrow indicates a positive (stimulating or activating) effect of a regulator on a component in the pathway it points to. A green plus sign indicates that there is an interaction between two components. The combination of these two components exerts a stimulating effect on the progression of the apoptotic cascade. A red arrow indicates an inhibitory effect of a regulator on another member or on an activating step within the apoptotic pathway. The markers highlighted in yellow were found to have the most clinical prognostic significance in colorectal cancer patients based on the studies that resulted from our search of the literature. These markers, their functions, and whether they are of true prognostic value are discussed in detail in this review.
Selection of relevant studies on clinical prognosis of apoptosis-related markers.

The general pathway of apoptosis is illustrated in Figure 1 and includes the markers discussed in this review. Although this figure represents a simplified version of the pathway, it shows that the process of apoptosis is highly regulated at multiple levels. Based on the stimulus presented, two pathways initiating the apoptotic process can be identified. 39 The extrinsic pathway is triggered by external death signals that cause the formation of intracellular signaling complexes at the death receptors. This type of apoptosis is typically activated in immune responses. 40 The second pathway, known as the intrinsic pathway, is activated by many different stimuli, including growth factor deprivation and DNA damage, caused by factors such as UV or gamma-irradiation or by chemotherapeutic agents. Exposure of cells to these stimuli initiates a set of intracellular death signals mediated by the p53 protein that activates the apoptotic process. Mitochondria play an important role in the intrinsic pathway with a major regulatory role for the Bel-2 family members. Although already intimately connected via caspase-8 and Bid, both pathways converge at the level of the caspase cascade that eventually leads to the proteolytic activation of executioner members such as caspase-3. 41 The function of caspase proteins is again highly regulated by a group of inhibitor of apoptosis proteins (IAPs). 42 In general, executioner caspases will cleave several substrates, thus acting as a cellular disassembly machine. Cleavage of these substrates is eventually responsible for the morphological features that hallmark apoptotic cell death and include membrane blebbing, cell shrinkage, and chromatin condensation.26,41
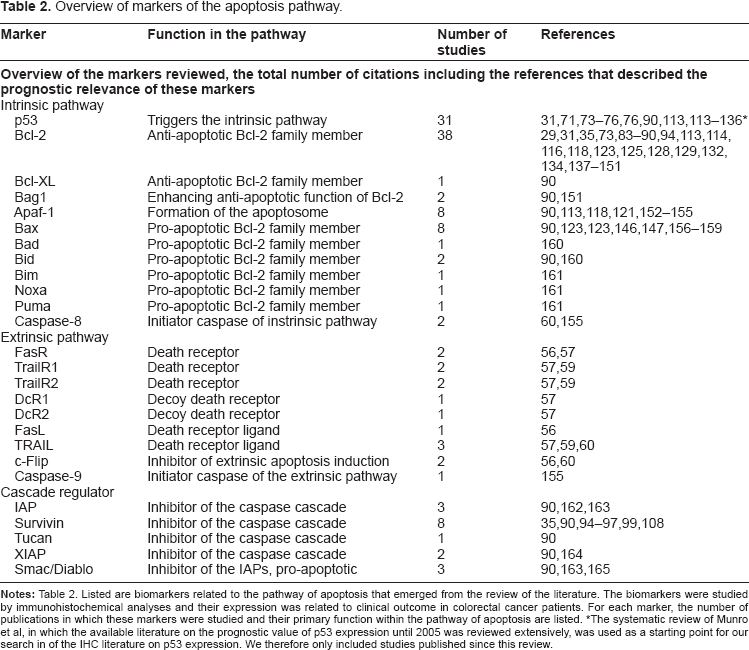
To discuss the results of our literature review, markers are grouped and discussed based on their locations in the pathway as described in Figure 1, starting with the extrinsic pathway of apoptosis and ending with the IAPs. In addition, Table 2 provides a general overview of the number of studies identified that describe the prognostic value of a particular marker, grouped by their function and location in the pathway of apoptosis in the order at which they will be discussed in this review.
Overview of markers of the apoptosis pathway.
The death receptor family and the extrinsic pathway of apoptosis
The most studied signaling pathway in apoptosis is the extrinsic pathway, activation of which is primarily facilitated by the death receptors (DRs). Based on our search, we were able to identify 8 individual biomarkers in this part of the apoptotic pathway for which the prognostic relevance was studied in CRC patients. These markers included Fas receptor (FasR), TRIAL1, TRAILR2, TRAILR3, TRAILR4, FasL, TRAIL, and c-Flip. (Table 2). A unique feature of the extrinsic pathway is that DRs can induce apoptosis independently of the p53 tumor suppressor gene. DRs are members of the tumor necrosis factor receptor superfamily, of which eight family members have been characterized.43,44 The most common receptors are FasR (CD95, DR2) with its ligand FasL and TRAILR1 and -R2 (DR4 and DR5) with their ligand tumor necrosis factor (TNF)-related apoptosis-inducing ligand (TRAIL). All DRs contain cysteine-rich extracellular domains that allow them to recognize their ligands with great specificity. They also harbor intracellular sub-domains better known as the death domains (DDs). 45 DDs allow them to interact with adapter molecules such as FADD (Fas-associated death domain). 46 Signal transduction within the extrinsic pathway starts with the binding of the ligands to the DRs, followed by the formation of multi-protein signaling complexes called death inducing signaling complexes (DISCS) at the intracellular domains of the DRs.47,48 The DISC complex auto-activates pro-caspase 8 through an interaction with a FADD protein. Activated caspase-8 will eventually activate the effector caspase-3 by proteolytic cleavage.48–51 Downregulation of any of the DRs or downstream apoptotic proteins can cause severe limitations in the induction of apoptosis through the extrinsic pathway. There are two other mechanisms involved in the regulation of the extrinsic signaling pathway. First, TRAIL can also bind to two decoy receptors (DcR) in addition to the DRs, including DcR1 and DcR2 (also known as TRAILR3 and TRAILR4, respectively). However, neither decoy receptor can transduce an apoptosis-stimulating signal upon TRAIL binding. The sensitivity of a cell to TRAIL-mediated apoptosis may, therefore, be a function of the ratio of DcR to DR. If there is significant upregulation of the DcRs or downregulation of the DRs, TRAIL will bind to the DcRs instead of the DRs, and the apoptotic signaling is interrupted. 52 Second, cellular Flice-like inhibitory protein (c-FLIP) is, similarly to FADD, a DD-containing protein and can competitively bind to FADD in the DISC formation process instead of the DD domain of the DRs (Fig. 1). This protein, particularly in the c-FLIPL isoform, shows strong structural similarities to pro-caspase-8, and may be a potentially strong inhibitor of the extrinsic apoptotic pathway. There are two other features that are unique to the extrinsic pathway, but contribute considerably to the complexity of its regulation. The first feature is an indirect link with the intrinsic pathway, which can be activated through the formation of tBid, a truncated form of the BID protein. In a subset of cells known as type II cells, DISC formation occurs less frequently, resulting in lower caspase-8 activation and subsequently truncation of the Bid protein into tBid. 53 tBid induces oligomerization of Bax or Bad, upon which the mitochondria release cytochrome c; this eventually induces apoptosis further down the intrinsic pathway. Because of the mitochondrial involvement apoptosis regulation in Type II cells is subject to regulation by the Bcl-2 family proteins. This regulation, which will be discussed in detail in this review, provides the cell with an apoptosis-evading mechanism such as downregulation of DR expression that may occur in a cell during tumorigenesis. 54 A second feature unique to the extrinsic pathway is explained by the so-called ‘Fas-counterattack hypothesis’. 55 In normal tissue homeostasis, the Fas/FasL-induced extrinsic pathway of apoptosis plays a major role in immune surveillance. Activated T lymphocytes express FasL and upon recognition of a tumor cell as a target (via MHC-presented peptides on the tumor cell surface), a tumor cell expressing the FasR may be eliminated by induction of apoptosis. However, it is known that tumor cells can also express FasL and thus are able to counterattack cells from the immune system. 55 By downregulation of FasR expression as well as by upregulation of FasL expression, tumor cells can escape immune surveillance.
In summary, the key players of the extrinsic pathway are the DRs, specifically TRAILR1, R2, and FasR and their ligands. Tumor cells attempt to disrupt signaling through these DRs to overcome apoptosis, which has been widely studied in many types of cancers. Our search identified 12 studies in which one or more of these DRs and their ligands were studied. In 5 of the 12 studies, one of the DR pathway-related markers (FasR, FasL, TRAILR1, and TRAIL) was found to be of significant prognostic value (Table 3).56–60 Hypothetically, based on the biology of the process of tumorigenesis, down-regulation of DR expression or upregulation of expression of their ligands indicate a more aggressive tumor type, and hence worse clinical outcome parameters. Interestingly, most studies reported that upregulation of the expression of Fas and TRAIL was significantly related to worse outcome parameters. The expression of FasL and FasR was studied by both Korkolopoulou et al 56 and Strater et al. 58 In a smaller study by Korkolopoulou et al 56 involving 90 patients, normal cells did not express FasL, but tumor cells showed significant upregulation, which was related to a significantly lower overall survival (OS). Tumor cells also showed expression of the Fas receptor with a mainly cytoplasmatic and granular staining pattern. According to the authors, this indicates that although the Fas receptor was present, it had no true functional properties. Therefore, according to the authors, the seemingly contradictory result of a worse outcome despite upregulation of DR expression could be explained by the Fas-counterattack hypothesis. In the second study by Strater et al, 58 overexpression of the Fas receptor correlated with a significantly better disease-free survival (DFS). Unfortunately, this study did not describe the exact location of FasR expression in the cell. Therefore, it is difficult to determine whether their results confirm Korkolopoulou results or actually oppose them. We were therefore not able to determine whether the Fas-counterattack hypothesis has true clinical value in CRC.
Extrinsic pathway of apoptosis.
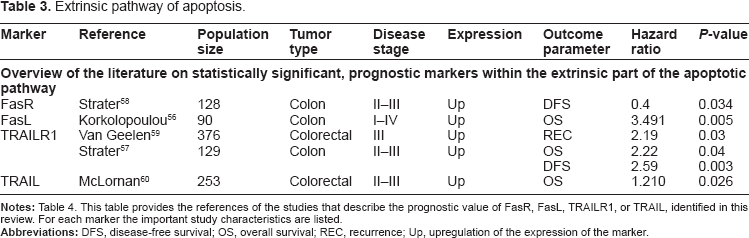
With respect to DR4 and its ligand TRAIL, we could identify three studies reporting the prognostic value of these biomarkers in CRC.57,59,60 In all studies, upregulation of expression of DR4 or its ligand were related to worse outcome parameters, such as higher levels of recurrence and shorter OS. This apparent contradiction with expectations based on biology of tumorigenesis can, according to Van Geelen et al, be explained by the fact that DR4 is also known to have effects on cell proliferation through the activation of nuclear factor kappa B (NF-κB), as described in a number of studies.
In conclusion, we were able to identify five studies reporting FasL, FasR, TrailR1, or TRAIL 2 as significant prognostic markers in colorectal cancer patients. Conclusions varied, which may be due to differences in patient selection and/or study methods. However, more importantly, their conclusions were in contradiction with what is expected based on the biology of the apoptosis pathway. This can be explained by the fact that the functionality of this extrinsic part of the apoptotic pathway in the included studies was only investigated based on changes in protein expression patterns of the tumor cells. The involvement of the immune system was not considered in most of these studies. The process of tumorigenesis attracts many cells that are part of the immune system into the tumor microenvironment. The presence of these cells such as activated CD8+ T cells or Foxp3+ regulatory T cells has been shown to be of prognostic relevance in CRC.64,65 Moreover, activated T cells produce CD95 ligand and can thereby trigger apoptosis in target cells such as tumor cells. 66 As described above, some tumors cells may be able to counteract this mechanism and remove attacking antitumor T cells by increasing their own CD95 L expression. However, this counterattack theory has not yet been conclusively demonstrated in vivo. Therefore, until additional preclinical, exploratory research has been performed to clarify how the pathway of apoptosis and the immune system interact, none of the related markers appear suitable for clinical prognostic application.
p53 Tumor suppressor gene and the intrinsic pathway of apoptosis
The p53 tumor suppressor gene, likely the most well-known protein within the intrinsic pathways of apoptosis, encodes for a transcription factor that regulates the expression of genes involved in the pathway of apoptosis, as well as angiogenesis, cell cycle progression, and genomic maintenance.67,68 Within the intrinsic pathway, it exerts its function at the beginning of the intrinsic apoptotic pathway. p53 causes cell cycle arrest at the G1 phase in response to DNA damage; in case the DNA damage turns out to be irreparable, the p53 protein will activate the appropriate cellular signaling cascades to execute apoptosis. In 50% of human colorectal cancers, p53 is absent or mutated, which has major implications for the execution of apoptosis in colorectal cancer.
69
Mutations in p53 can be determined using IHC since mutated proteins accumulate in the nucleus due to their increased half-life.
70
Different mutations have varying effects and can implicate either loss or gain of function of the p53 protein. Two research groups carried out major systematic reviews on the relationship between p53 abnormalities and outcome in colorectal cancer patients.71,72 Munro et al
71
reviewed a total of 168 IHC-based studies as well as mutation-based studies. Russo et al
72
pooled data from studies analyzing p53 DNA mutations only. Together, these studies reported on p53 expression and mutations in relation to survival in 18,766 patients. Their key finding was that abnormal expression of p53, detected using IHC, was related to an increased risk of death. They concluded that mutations in exon 5 were associated with an adverse outcome, predominantly in proximal, right-sided tumors. Both studies suggested an impact of mutated p53 on clinical outcome, though this relationship was only modest despite the overwhelming amount of data analyzed. The results of these studies were taken into account in the review of prognostic biomarkers in CRC by the ASCO Clinical Oncology's Tumor Marker Expert Panel in 2006. The ASCO panel's recommendation was that with current methods of detection, using either mutation analysis or IHC, p53 status was a poor guide for predicting prognosis in colorectal cancer patients.
19
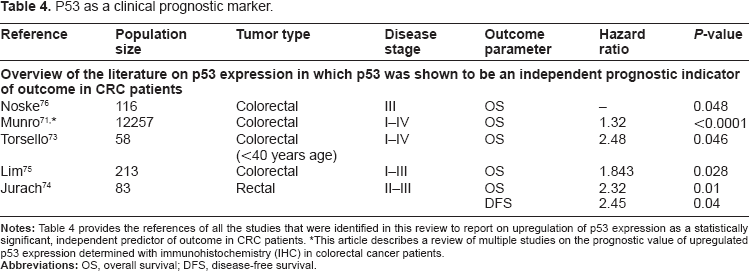
Since the published literature on IHC-based p53 studies until 2005 has already been thoroughly reviewed by Munro et al, we limited our search to reports that have been published since. We identified an additional 30 studies reporting on the expression of p53 determined using IHC that were published after Munro's review in 2005. Four of these 30 papers confirmed the results of Munro et al and claimed p53 to be a significant, prognostic marker in colorectal cancer.73–76 Table 3 provides an overview of their study characteristics. From this table, it can be concluded that although p53 has been widely studied, the studies reporting on statistically significant prognostic relevance show differences in population size, tumor type selection, and disease stage selection. When we examined these publications in more detail, we also noticed that they varied in their IHC methods. Previously, a comparative study by Baas et al
77
demonstrated the monoclonal antibody D07 to be superior over 5 other antibodies for detecting p53 gene protein in archival tissue of colorectal carcinomas, suggesting that this antibody should be used as a gold standard for IHC of p53. This particular antibody was used in 3 out of the 4 studies listed.73,75,76 Furthermore, the sample sizes of all studies rather small at an average of 103 patients, and a closer look at these populations showed that most also seems highly selected on clinical parameters. For example, Jurach et al
73
only included stage II and III rectal cancer patients and the small cohort of Torsello et al
74
consisted only of patients under age 40. The only relatively large study by Lim et al
75
that analyzed the results of 231 stage I, II, and III CRC patients, showed a correlation between upregulation of p53 expression and poor OS. This correlation was more pronounced in their stage III patient selection, but disappeared when only the adjuvant-treated patients were analyzed. Their results appeared to be confirmed by the study of Noske et al.
76
However, in this study, the prognostic value of p53 was only present in a multivariate analysis when expression was analyzed in combination with p21 expression, a major downstream cell cycle inhibitor. The expression of p53 alone in univariate analysis was only borderline-significant at a
Bcl-2 family members and the intrinsic apoptotic pathway
Downstream of p53, the mitochondria play a major role in the initiation and execution of the intrinsic pathway of apoptosis. The B-cell CLL/lymphoma 2 (Bcl-2) family members are mainly responsible for regulating the intrinsic pathway and can be categorized into two groups. The first group consists of anti-apoptotic proteins that are structural and functional homologs of Bcl-2. The most important members of this group are Bcl-2 itself and its splice variant Bcl-2 XL.78, 79 They are mainly bound to the mitochondrial outer membrane (MOM) by their transmembrane (TM) domain, where they stabilize the MOM to prevent cytochrome c release into the cytosol of the cell under normal homeostatic circumstances. 80 Therefore, they can be considered anti-apoptotic proteins. 81
The second group of Bcl-2 family members has proapoptotic capacities. These members include Bcl-2 associated X protein (BAX) and proteins such as Bad, Bid, Bim, Bik, Noxa, and Puma, which are, based on their structures, also known as BH3-only proteins. 78 These proteins are typically bound to the cytoskeleton or cytosol, but upon stimulation they interact with and inhibit their anti-apoptotic counterparts such as Bcl-2. 78 The relative ratio or balance between the expression of both groups of Bcl-2 family members will determine whether stimulation of the intrinsic pathway of apoptosis results in apoptosis as is graphically pointed out in Figure 1. If the pro-apoptotic factors predominate, cytochrome c will be released into the cytosol where it binds to the apoptosis-activating factor 1 (Apaf-1) to form an apoptosome. More downstream in the apoptotic pathway, this apoptosome will form a complex together with an initiator caspase, caspase-9. This caspase will subsequently activate the executioner caspases, caspase-3 and -7.78,82 Deregulation of apoptosis during tumor development can be caused by a disturbance in the homeostatic balance of the Bcl-2 family members.
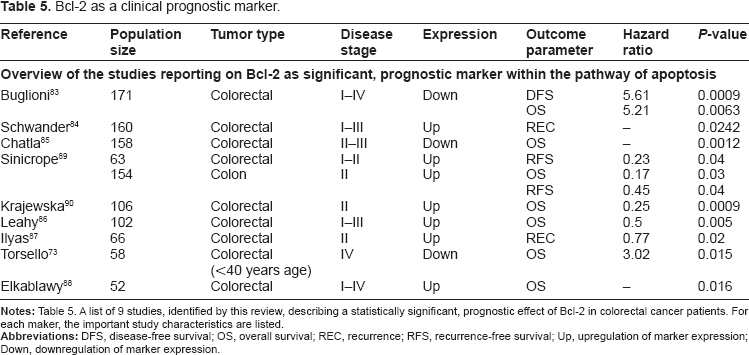
Our search resulted in 55 studies describing the prognostic relevance of markers related to the Bcl-2 protein family. In most of these studies, 38 in total, the expression of Bcl-2 was examined using IHC. In only 9 out of these 38 studies, a statistically demonstrated prognostic relevance of this marker could be established (Table 5).73,83–90 The 9 studies generally used the same methods to determine Bcl-2 expression and all but one were performed on whole paraffin-embedded tissue sections. Furthermore, they were very consistent in their conclusions: in all studies, upregulation of Bcl-2 was related with better survival, as shown for either disease-free, overall, or recurrence-free survival. This is contradictory to is assumed given the anti-apoptotic function of Bcl-2: upregulated Bcl-2 expression should be more likely to be a marker of a more aggressive tumor phenotype. The most plausible explanation for this paradoxical finding is the fact that Bcl-2 not only has an anti-apoptotic function, it can also exert a distinct negative influence on cell cycle progression, which can eventually slow down tumor growth. This may explain the survival benefit for several patients with upregulated Bcl-2 expression.83–85,89 Whether the anti-cell cycle progression or the anti-apoptotic role of Bcl-2 predominates during tumorigenesis may depend on disease stage. In early carcinogenesis, the anti-apoptotic function of Bcl-2 plays a large role, causing genetic alterations to accumulate. In later stages, Bcl-2 functions more as a cell cycle progression inhibitor, lowering the rate of tumor proliferation. This hypothesis is supported by the inverse correlation between Bcl-2 and the percentage of cells in S-phase found by Buglioni et al. 83 The studies describing the prognostic relevance of Bcl-2 would therefore be more informative if disease stage and the expression of other family members were taken into account. This would provide us additional insight in the biological function and effects on the apoptotic pathway of Bcl-2, which will tremendously improve the interpretation of the results.
P53 as a clinical prognostic marker.
Bcl-2 as a clinical prognostic marker.
Inhibitors of apoptosis family proteins and the execution of apoptosis
The actual apoptotic cell death machinery, responsible for the execution of apoptosis and resulting in the morphological features characteristic of apoptosis, consists of a very complex cascade of interacting proteins. The key components include the caspase proteins, as described above. At many levels, regulation takes place to ensure appropriate functioning of the caspase machinery. Key regulators of the caspase cascade are the inhibitors of apoptosis proteins (IAPs) that exert their function through binding of activated caspases. Thus far, 8 IAPs have been identified in mammals, the most well-known being livin, X-linked inhibitor of apoptosis (XIAP), and survivin.26, 91, 92 All IAP family proteins have one or several specific Baculoviral IAP repeats (BIRs). They require at least one BIR to exert their anti-apoptotic function. The function of the IAPs is also strictly regulated by their own set of inhibitors such as Smac/Diablo and Omi/HtrA2. 26 Under normal circumstances, when apoptotic stimuli are present, cells release Smac/DIABLO from their mitochondria into the cytosol, where the complex exerts its pro-apoptotic effect by interacting with the IAPs in order to release bound caspases into the cytosol. 93 The most frequently studied IAP in our search results was survivin, likely because the role of survivin in apoptosis has been the subject of controversy over the last few years. Of the 8 studies describing the prognostic effects of survivin, 4 demonstrated a statistically prognostic effect of survivin (Table 6).35,90,94,99 Initially, it was thought that survivin and the other IAPs selectively bind active caspase-3/-7, and -9, promoting their degradation and thereby inhibiting apoptosis. 100 Survivin, however, lacks the structural motif to bind to caspases and likely only inhibits activated caspase-9 with the help of XIAP.101–103 In contrast to other IAPs, survivin is undetectable in normal adult tissues, but abundantly expressed in transformed cell types and a variety of human cancers, such as cancers originating in the colon, stomach, pancreas, lung, prostate, and breast. 104 Although all four studies were able to show a significant relation between survivin expression and clinical outcome, the direction of this effect was not the same. When looking at these studies in detail it was noticed that they did not apply the same methods of analysis of the IHC results. This is of importance because survivin can be expressed in two cellular compartments: either in the cytoplasm or in the nucleus with different functions.98,105,106 In general, survivin is known to be involved in the regulation of cell viability as well as in the regulation of cell division. It is hypothesized that the nuclear subset is involved in controlling cell proliferation and the cytoplasmatic pool is more involved in regulating cell survival. 107 Sarela et al 94 found a relationship between survivin expression and a shorter DFS when scoring mainly the cytoplasm for survivin positivity. Ponnelle et al 95 showed a positive influence of both cytoplasmatic and nuclear expression on survival in a very small patient population of only 46 patients. This only reached statistical significance for the cytoplasmic group. Fang et al 108 showed a negative effect of survivin expression on OS, disease recurrence, and the development of liver metastasis. The same was true for the study by Sprenger et al 99 in which pretreatment biopsies of rectal cancer patients were analyzed for their survivin expression. In this study, low pre-treatment expression was related to a significantly better DFS. Unfortunately, neither of the groups elaborated on the specific location in the cell at which they scored survivin expression. The image of a tissue microarray (TMA) core that was immunohistochemically stained for survivin expression provided in the publication by Fang et al 108 suggests that the staining pattern was predominantly cytosplasmatic. In conclusion, it appears that localization of survivin expression is in fact of great importance as it is likely related to the protein function and hence should be taken into account in future studies. This may be applicable to all of the other IAP family members since none have been studied widely with standardized scoring methods in large series.
Survivin as a clinical prognostic marker.

Discussion
This review gives an overview of the literature published on IHC-based prognostic biomarkers related to the pathway of apoptosis in colorectal cancer between January 1998 and June 2011. Particularly, we discussed those markers that were proven to be of independent, statistically significant, prognostic value by placing them in the context of their function within the apoptotic pathway. Based on this biological background information, we then analyzed the conclusions drawn by the authors of the studies included to determine whether their conclusions could provide valuable grounds to proceed investigating these markers for prognostic clinical application. The markers we discussed are all major regulatory players in this pathway such as p53, the Fas receptor, and the DR4 with their respective ligands, the Bcl-2 protein family, and survivin. In general, we concluded that none qualified as a single prognostic biomarker for colorectal cancer patients, despite the fact that it has been well-established that the outcome of the pathway of apoptosis is of prognostic value.29–37,99 Based on the information derived from all of the studies discussed, we postulate several explanations for this lack of sufficient clinical prognostic significance for individual markers of the apoptotic pathway. First, study characteristics of investigations of one specific biomarker varied widely. This sometimes provided a marker with prognostic significance, but only in a highly selected group of patients. For example, in the case of p53, some studies were able to reproduce the results of the review of Munro et al, 71 but only when a selected patient cohort was studied which consisted of patients with better survival rates after surgery, as was the case in the study by Lim et al. 75 In this study, the prognostic value of p53 was only present in the stage II/III cohort when both adjuvant and non-adjuvant treated patients were included, which were probably the patients with such good outcome perspectives after surgery that adjuvant treatment was deemed unnecessary. The prognostic value disappeared when only the adjuvant-treated cases were analyzed. This makes the applicability of p53 as a general prognostic marker at time of diagnosis and treatment allocation questionable for the entire colorectal cancer patient population. Second, well-standardized IHC protocols were applied for none of the markers. Possibly even more importantly, there seemed to be no standardized methods for quantifying expression level or a specific location in the cell at which the expression of a marker should be evaluated. This may greatly influence the interpretation of the results of these studies, as for instance the specific location of expression in a cell may be directly related to its biological function and thereby to its effect on the outcome of the apoptotic pathway and patient prognosis. Caution should be taken in the method of quantification, particularly for FasR and survivin expression. In the case of the FasR expression, a high level of expression found in the cytoplasm but not on the cells membrane can indicate the presence of decoy receptors. 56 This may be an indication that the so-called Fas-counterattack hypothesis is indeed true, making it essential to evaluate the subcellular location of expression. 55 In addition, in the case of survivin, staining location appeared to be significant since both nuclear and cytoplasmic expression showed a different relationship with outcome in the studies reviewed.94,95,99,108 We concluded that location of expression may implicate different biological functions of the same protein. Third, we showed that the pathway of apoptosis is strictly regulated at several levels by both stimulatory and inhibitory proteins that highly interact with each other. Using only one marker to describe the outcome of these interactions, therefore, seems inappropriate. To complicate matters even more, the function of a protein can differ depending on progression of the process of tumorigenesis as was shown for Bcl-2 expression. 83 Intervention of key proteins of apoptosis in other pathways makes cellular outcome unpredictable when using expression of single proteins as prognostic biomarkers.
Overall, we can conclude that studying outcome of the pathway of apoptosis, or deciding on a patient's prognosis and treatment, using single markers seems inappropriate given the complexity of this pathway. We already highlighted the delicate balance that exists between expression and effects on apoptosis of the members of the Bcl-2 pathway and of the IAP's and their inhibitors.78,83 To interpret the effect of expression of single proteins without the knowledge of the expression status of any of the others involved seems inappropriate. Therefore, we suggest improving the clinical applicability of these markers. Most importantly, we suggest that the apoptotic profile of a tumor should be determined rather than expression of single markers. This should include several markers that together represent the outcome of all regulatory thresholds within the pathway. An apoptotic profile would better represent the true function of the markers involved and provide insight on the outcome of the apoptotic pathway in individual tumors; thus, an apoptotic profile fulfills the need for prognostic biomarkers in colorectal cancer. 22
In the literature studied for this review, several authors made an attempt to establish such a multi-marker apoptotic phenotype.59,60,83,99 However, in these studies, the selection of biomarkers was based on the markers that showed some prognostic relevance in their series as a single biomarker without any respect to the biology of the pathway. In addition, in most cases, the markers used in these ‘multi-marker phenotypes’ all belonged to just one sub-regulatory unit of the pathway. For example, Van Geelen et al 59 and McLornan et al 60 both studied multiple markers with respect to the DRs, but did not include any of the more downstream regulator proteins. Furthermore, in many of the studies the statistical methods used to analyze the results of these multi-marker phenotypes lacked power. A solution to this problem may be to approach the data as one would do in the case of gene expression array data by performing hierarchical clustering in order to develop a profile. 109 Until then, the biomarkers that will eventually make up this apoptotic profile remain to be determined. In the meantime, we suggest that further studies focus on analyzing the clinical relevance of not only the outcome of (de)-regulation of the apoptotic pathway in colorectal cancer, but also on the outcome of the (de)-regulation of the pathway of proliferation. Under normal circumstances, a key factor in tissue homeostasis is the balance that exists between the level of cell death and the level of cell proliferation.110, 111 Deregulation of either of these pathways can therefore cause disturbance of this balance, which may result in and maintain tumorigenesis. This hypothesis has been studied previously with success in a cohort of 100 colorectal cancer patients in which an AI:PI ratio was determined. 112 This Apoptotic Index:Proliferation Index, based on M30 IHC for the level of apoptosis and Ki67 IHC for the proliferation index, was significantly related to patient outcome. It remains to be determined whether studies in larger patient populations will confirm these results.
In conclusion, to determine the prognostic relevance of biomarkers involved in apoptotic pathways in colorectal cancer using immunohistochemistry, multiple markers that together reflect the apoptotic status in individual tumors should be studied together. The introduction of such a multi-marker apoptotic phenotype or -profile into clinical practice demands standardization of technical assays and quantification methods. For future studies, therefore, we recommend considering the full pathway when beginning an exploratory phase towards the discovery of new biomarkers in colorectal cancer related to outcome of the apoptotic process. Moreover, we recommend applying hierarchical clustering-based statistical analysis and using knowledge regarding the biology of the pathway to identify promising markers. Furthermore, we recommend taking into account markers representing the pathway of proliferation when studying the prognostic effects of the apoptotic pathway in colorectal cancer. These measures will lead to multi-marker profiles that can then be validated in large retrospective studies and can ultimately be introduced into clinical practice.
Author Contributions
Conceived and designed the experiments: EZ. Analyzed the data: EZ, PS. Wrote the first draft of the manuscript: EZ, PS. Contributed to the writing of the manuscript: EZ, AB, MR, PS. Agree with manuscript results and conclusions: EZ, AB, MR, PS, GL, CvdV, PK. Jointly developed the structure and arguments for the paper: EZ, GL, CvdV, PK. Made critical revisions and approved final version: GL, CvdV, PK, EZ, AB. All authors reviewed and approved of the final manuscript.
Funding
Author(s) disclose no funding sources.
Competing Interests
Author(s) disclose no potential conflicts of interest.
Disclosures and Ethics
As a requirement of publication the authors have provided signed confirmation of their compliance with ethical and legal obligations including but not limited to compliance with ICMJE authorship and competing interests guidelines, that the article is neither under consideration for publication nor published elsewhere, of their compliance with legal and ethical guidelines concerning human and animal research participants (if applicable), and that permission has been obtained for reproduction of any copyrighted material. This article was subject to blind, independent, expert peer review. The reviewers reported no competing interests.
Footnotes
Supplementary Data: List of abbreviations (in order of appearance)
| IHC | Immunohistochemistry |
| CRC | Colorectal Cancer |
| ASCO | American Association of Clinical Oncology |
| EGTM | European Group on Tumor Markers |
| Bcl-2 | B-cell Lymphoma 2 |
| P53 | Protein 53 |
| TRAILR1 | Tumor necrosis factor Related Apoptosis Inducing Ligand Receptor 1 |
| UICC | International Union Against Cancer |
| TNM | Tissue Node Metastasis |
| ASCO-TEMP | American Association of Clinical Oncology Tumor Markers Expert Panel |
| DNA | Deoxyribonucleic Acid |
| UV | Ultraviolet |
| IAP | Inhibitor of Apoptosis Protein |
| DR | Death Receptor |
| FasR | Fas Receptor |
| TRAILR2/R3/R4 | TRAIL Receptor 2/3/4 (see TRAILR1) |
| FasL | Fas Ligand |
| c-Flip | Cellular FADD Like Interleukin 1 beta-converting Enzyme Inhibitory Protein (see FADD) |
| TNFR | Tumor Necrosis Factor Receptor |
| DD | Death Domain |
| FADD | Fas-Associated Death Domain |
| DISC | Death Inducing Signalling Complexes |
| DcR1/2 | Decoy Receptor 1/2 |
| MHC | Major Histocompatibility Complex |
| OS | Overall Survival |
| DFS | Disease-Free Survival |
| NF-κB | Nuclear Factor kappa-light-chain-enhancer of activated B cells |
| CD8 | Cluster of Differentiation 8 |
| CD95 | Cluster of Differentiation 95 |
| T cell | Thymus cell |
| CD95 L | CD 95 ligand (see CD 95) |
| Bcl-2 XL | Bcl-2 Extra Large (see Bcl-2) |
| MOM | Mitochondrial Outer Membrane |
| TM | Transmembrane |
| BAX | Bcl-2-Associated X protein (see Bcl-2) |
| BAD | Bcl-2-Associated Death Promoter (see Bcl-2) |
| Bim | Bcl-2 like interacting mediator of cell death (see Bcl-22) |
| Bid | BH3 interacting domain death agonist (see BH3) |
| Puma | P53 upregulated modulator of apoptosis |
| TUCAN | Tumor-upregulated CARD containing antagonist of caspase 9 (see CARD) |
| CARD | Caspase Recruitment Domain |
| BH3 | Bcl-2 Homology 3 (see Bcl-2) |
| Apaf-1 | Apoptosis-Activating factor 1 |
| S-phase | Synthesis phase |
| XIAP | X-linked IAP (see IAP) |
| BIR | Baculovirus IAP Repeat (see IAP) |
| SMAC | Second Mitochondria-Derived Activator of Caspases |
| DIABLO | Direct IAP Binding protein with Low pI (see IAP) |
| HtrA2 | High temperature requirement protein A2 |
| TMA | Tissue Micro Array |
| N | Number of patients in study population |
| Bag1 | Bcl-2-associated Athanogene 1 |
| REC | Recurrence |
| RFS | Recurrence-Free survival |
