Abstract
Current guidelines do not systematically recommend 18F-FDG PET/CT for breast cancer staging; and the recommendations and level of evidence supporting its use in different groups of patients vary among guidelines. This review summarizes the evidence about the role of 18F-FDG PET/CT in breast cancer staging and the therapeutic and prognostic impact accumulated in the last decade. Other related aspects, such as the association of metabolic information with biology and prognosis are considered and evidence-based recommendations for the use of 18F-FDG PET/CT in breast cancer staging are offered. We systematically searched MEDLINE for articles reporting studies with at least 30 patients related to clinical questions following the Problem/Population, Intervention, Comparison, and Outcome framework. We critically reviewed the selected articles and elaborated evidence tables structuring the summarized information into methodology, results, and limitations. The level of evidence and the grades of recommendation for the use of 18F-FDG PET/CT in different contexts are summarized. Level III evidence supports the use of 18F-FDG PET/CT for initial staging in patients with recently diagnosed breast cancer; the diagnostic and therapeutic impact of the 18F-FDG PET/CT findings is sufficient for a weak recommendation in this population. In patients with locally advanced breast cancer, level II evidence supports the use of 18F-FDG PET/CT for initial staging; the diagnostic and therapeutic impact of the 18F-FDG PET/CT findings is sufficient for a strong recommendation in this population. In patients with recently diagnosed breast cancer, the metabolic information from baseline 18F-FDG PET/CT is associated with tumor biology and has prognostic implications, supported by level II evidence. In conclusion, 18F-FDG PET/CT is not recommended for staging all patients with early breast cancer, although evidence of improved regional and systemic staging supports its use in locally advanced breast cancer. Baseline tumor glycolytic activity is associated with tumor biology and prognosis.
Introduction
Among females worldwide, breast cancer (BC) is the most frequently diagnosed cancer, with a 5-year prevalence of 31.4% and is the leading cause of cancer death, accounting for 15.5% of all cancer deaths.1,2 The diagnosis of BC is based on clinical examination in combination with imaging and is confirmed by histopathological assessment. Imaging includes bilateral mammography and ultrasound of the breast and regional lymph nodes.
Accurate staging is important for management decisions and prognosis in patients with newly diagnosed BC. All patients with newly diagnosed BC undergo regional staging including axillary and internal mammary lymph node evaluation and those with a high risk of early distant metastases also undergo systemic staging including sites beyond the regional lymph nodes. Magnetic resonance imaging (MRI) of the breast and fluorine-18 fluorodeoxyglucose (18F-FDG) positron emission tomography/computed tomography (PET/CT) are not routinely recommended for initial staging.3–8
Apart from imaging, in patients with clinically positive axilla, pre-treatment disease staging includes histopathological examination of the primary tumor and cytology/histology of the axillary lymph node (ALN) specimens obtained using ultrasound-guided fine-needle aspiration biopsy or core biopsy to determine whether ALN dissection is needed. In patients with clinically negative axilla, the current standard of ALN staging in early-stage BC is sentinel lymph node biopsy.5,6
According to BC guidelines such as National Comprehensive Cancer Network (NCCN), European Society for Medical Oncology (ESMO), Spanish Society of Medical Oncology (SEOM), and the National Institute for Care Excellence (NICE), 18F-FDG PET/CT is not indicated in women with apparently early-stage (I or II) BC or even in those with operable stage III BC;3–8 it is considered optional for women with suspicious or equivocal findings on CT or MRI and for women with locally advanced BC, especially those with advanced axillary nodal disease, because the risk of early distant metastases is high enough to justify systemic staging as part of the initial evaluation. In this setting, 18F-FDG PET/CT can detect metastases that are not visible on other modalities, and these findings can change treatment options. However, the levels of evidence and the recommendations for the use of 18F-FDG PET/CT vary among guidelines, basically because different guidelines use different quality evaluation scales and most are designed for the assessment of treatment-related studies rather than for diagnosis. Table 1 summarizes the current guidelines recommendations and level of evidence for using 18F-FDG PET/CT in BC staging.
Evidence level and grade of recommendations for 18F-FDG PET/CT in staging breast cancer as reported in current clinical guidelines.

18F-FDG PET/CT: fluorine-18 fluorodeoxyglucose positron emission tomography/computed tomography; NCCC: Norris Cotton Cancer Center.
Strength of recommendation (five categories, A–E)/quality of evidence (three categories, I–III).
Grade of recommendation (six categories, 1A–2C).
Categories of evidence and consensus (four categories, 1, 2a, 2b and 3).
Given the evolving role of 18F-FDG PET/CT in the management of BC patients and the low level of evidence for its use in initial staging reported in the guidelines, we reviewed and summarized the current evidence to offer evidence-based recommendations for the use of PET/CT in this setting.
Materials and methods
The search strategy was structured following the PICO framework, in which
We searched MEDLINE to identify studies published in English between January 2006 and March 2016 that contained the above-mentioned PICO terms or synonymous terms in titles and/or abstracts. To select studies, one investigator reviewed the titles of all the studies identified in the search and excluded those not related with the main objective: initial PET (staging). Next, we reviewed the abstracts of the included titles and obtained the complete articles of those that reported systematic reviews or original studies using 18F-FDG PET, 18F-FDG PET/CT, or both techniques in a sample at least of 30 patients.
Two investigators examined all the papers that fulfilled these criteria and excluded duplicated studies and reviewed articles or meta-analyses that were superseded by more recent ones. Moreover, studies included in systematic reviews or meta-analyses were excluded when a systematic review or meta-analysis answered the question they explored.
We critically reviewed the selected articles and elaborated evidence tables, structuring the summarized information into methodology, results, and limitations. We assessed the level of evidence for the use of 18F-FDG PET in each situation and established the recommendation grades following the NICE adaptation 9 of the levels of evidence for diagnostic studies of the Oxford Centre for Evidence-Based Medicine and the Centre for Reviews and Dissemination (Table 2). Recommendations were simplified to consider grade A or B as a strong recommendation and grade C or D as weak recommendation.
Levels of evidence and grades of recommendation established in the National Institute for Clinical Excellence guidelines.

Cost-effectiveness studies were evaluated using the Scottish Intercollegiate Guidelines Network (SIGN) 10 scale, which ranges from (++), when the study fulfilled all or most of the quality criteria, to (−), when it fulfilled few or none of the quality criteria.
In the assessment of each study, the gold standard considered valid was histological confirmation of lymph node or distant metastases.
We reviewed all the information from the literature searches to answer the following main question:
Moreover, we considered other, secondary questions to answer the main question:
Finally, we considered other questions about 18F-FDG PET/CT that were not strictly related with initial staging:
Results
The levels of evidence and grades of recommendations for the answers to the questions were as follows:
1a 18F-FDG PET/CT imaging is not recommended in early-stage BC. Level of evidence II, strong recommendation.11–28 For more information see Tables 3–5.
2a. 18F-FDG PET/CT should be used in locally advanced BC for initial staging. Evidence level II, strong recommendation.11–28 For more information, see Tables 3–5.
3a. 18F-FDG PET/CT should be used in high-risk patients, such as those with inflammatory BC. Evidence Level II, strong recommendation. In triple-negative BC, there is limited evidence to recommend the systematic use of the technique.29–35 For more detailed information, see Table 6.
4a. Patients’ age at diagnosis is not in itself, independent of clinical stage or molecular biology, a sufficient criterion to indicate 18F-FDG PET/CT. 35 Evidence level IV, weak recommendation.
In patients with recently diagnosed BC, the diagnostic effectiveness of 18F-FDG PET/CT is superior to that of other diagnostic techniques.
In patients with recently diagnosed BC, the percentage of cases in which 18F-FDG PET/CT changed the staging with respect to conventional imaging is high enough to recommend it.
In patients with recently diagnosed BC, 18F-FDG PET/CT results in a significant enough change in the therapeutic management to recommend it.
In patients with recently diagnosed BC, the baseline glycolytic activity on 18F-FDG PET/CT is associated with tumor biology.
In patients with recently diagnosed BC, the metabolic information from a baseline 18F-FDG PET/CT has implications for the patient’s prognosis.
There is not enough evidence to address the cost-effectiveness of 18F-FDG PET/CT62,63 (Table 10).
Locoregional breast cancer staging: FDG PET/CT lymph node assessment in early and locally advanced disease.
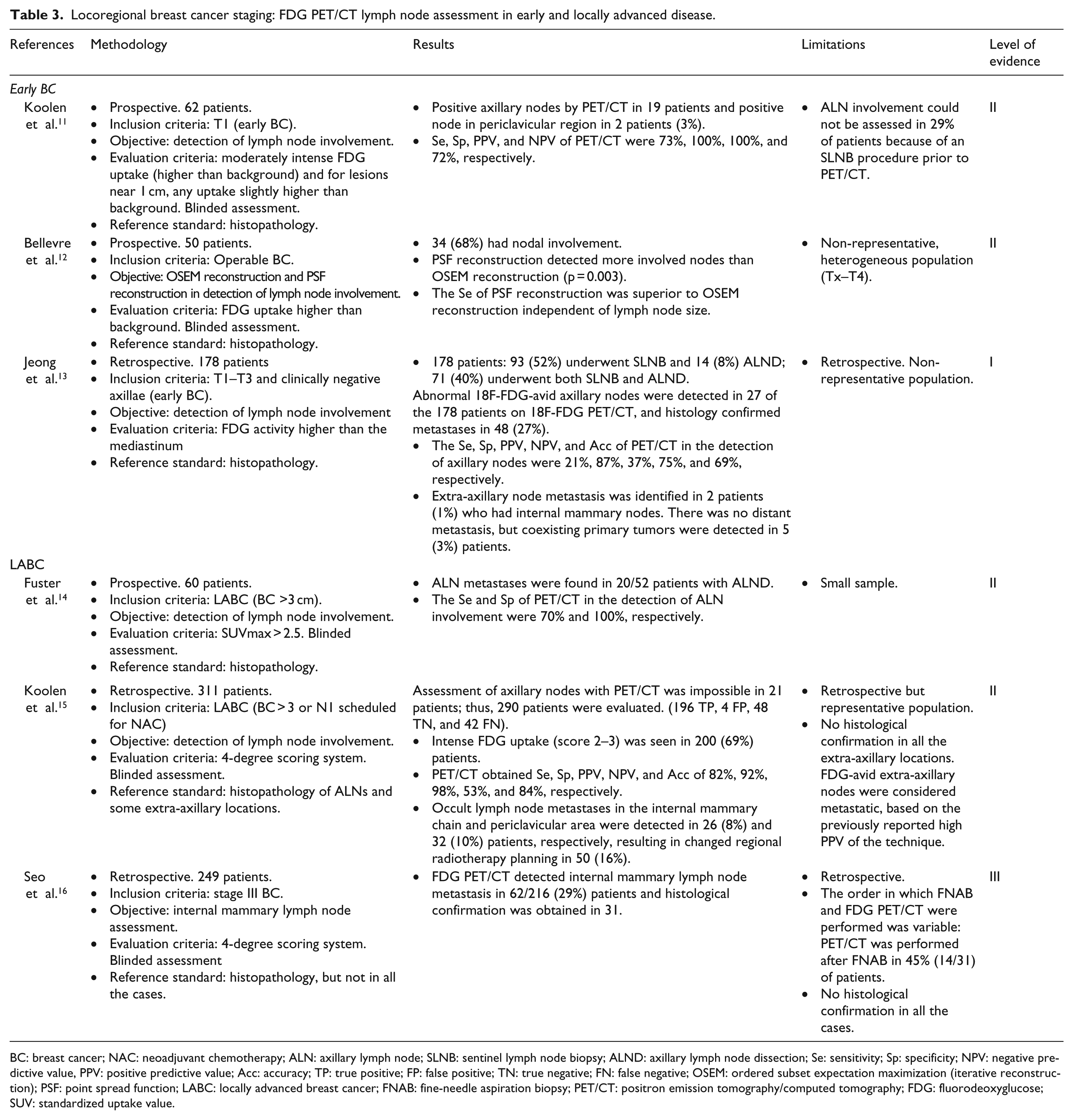
BC: breast cancer; NAC: neoadjuvant chemotherapy; ALN: axillary lymph node; SLNB: sentinel lymph node biopsy; ALND: axillary lymph node dissection; Se: sensitivity; Sp: specificity; NPV: negative predictive value, PPV: positive predictive value; Acc: accuracy; TP: true positive; FP: false positive; TN: true negative; FN: false negative; OSEM: ordered subset expectation maximization (iterative reconstruction); PSF: point spread function; LABC: locally advanced breast cancer; FNAB: fine-needle aspiration biopsy; PET/CT: positron emission tomography/computed tomography; FDG: fluorodeoxyglucose; SUV: standardized uptake value.
Locoregional breast cancer staging: comparison of FDG PET/CT lymph node assessment in early and locally advanced disease with other diagnostic techniques.

Se: sensitivity; Sp: specificity; PPV: positive predictive value; NPV: negative predictive value; Acc: accuracy; SLNB: sentinel lymph node biopsy; ALNM: axillary lymph node metastases; ALND: axillary lymph node dissection; LABC: locally advanced breast cancer; US: ultrasonography; cMRI: contrast-enhanced magnetic resonance imaging; DWI-MRI: diffusion-weighted magnetic resonance imaging; CI: conventional imaging; PET/CT: positron emission tomography/computed tomography; BC: breast cancer; 95% CI: 95% confidence interval.
Distant metastases detection: FDG PET/CT compared with other diagnostic techniques.

CS: conventional staging; BS: bone scintigraphy; Se: sensitivity; Sp: specificity; DOR: diagnostic odds ratio; PLR: positive likelihood ratio; NRL: negative likelihood ratio; CI: confidence interval; AUC: area under curve; M1: distant metastases; US: ultrasound; PET/CT: positron emission tomography/computed tomography; LABC: locally advanced breast cancer.
Staging in function of groups of risk: inflammatory breast cancer, triple-negative breast cancer, and young patients.

IBC: inflammatory breast cancer; CS: conventional staging (breast magnetic resonance imaging, chest CT, liver ultrasonography, bone scintigraphy); DCE-CT: dynamic contrast-enhanced CT; LABC: locally advanced breast cancer; TNBC: triple-negative breast cancer; PET/CT: positron emission tomography/computed tomography.
Changes in stage and treatment resulting from 18-F-FGD PET findings.
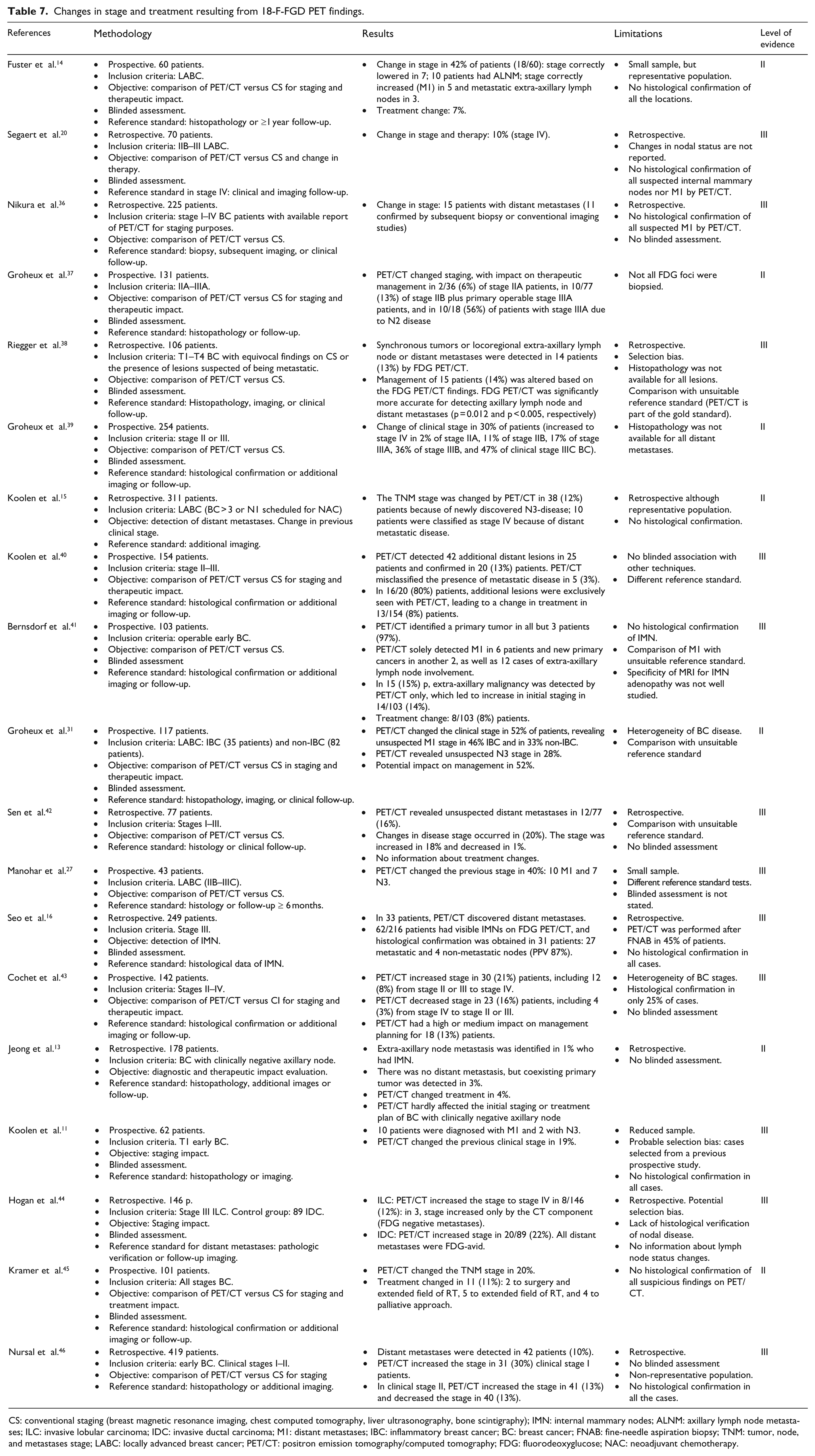
CS: conventional staging (breast magnetic resonance imaging, chest computed tomography, liver ultrasonography, bone scintigraphy); IMN: internal mammary nodes; ALNM: axillary lymph node metastases; ILC: invasive lobular carcinoma; IDC: invasive ductal carcinoma; M1: distant metastases; IBC: inflammatory breast cancer; BC: breast cancer; FNAB: fine-needle aspiration biopsy; TNM: tumor, node, and metastases stage; LABC: locally advanced breast cancer; PET/CT: positron emission tomography/computed tomography; FDG: fluorodeoxyglucose; NAC: neoadjuvant chemotherapy.
Relation of metabolic variables with tumor biology (histopathological prognostic factors and molecular phenotypes).

68Ga-RGD: 68Ga-labeled arginine–glycine–aspartic acid; ER: estrogen receptor; PR: progesterone receptor; HER2: human epidermal growth factor receptor 2; RI: retention index; TN: triple negative; SUVmax: maximum standardized uptake value; SUVavg: average standardized uptake value; RI: retention index; IDC: invasive ductal carcinoma; ILC: invasive lobular carcinoma; LABC: locally advanced breast cancer; NAC: neoadjuvant chemotherapy; BC: breast cancer; TNM: tumor, node, and metastases; ALN: axillary lymph node.
Relation of metabolic information derived from basal 18F-FDG PET/CT with prognosis.

PFS: progression-free survival; MTV: metabolic tumor volume; TLG: total lesion glycolysis; IDC: invasive ductal carcinoma; ALN: axillary lymph node; DFS: disease-free survival; EFS: event-free survival; OS: overall survival; ROC: receiver-operating-characteristic curve; AUC: area under the curve; IBC: inflammatory breast cancer; DCE-CT: dynamic contrast-enhanced computed tomography; TN: triple negative; ER: estrogen receptor; HR: hazard ratio; BC: breast cancer; 95% CI: 95% confidence interval; PR: progesterone receptor; PET/CT: positron emission tomography/computed tomography; FDG: fluorodeoxyglucose; RFS: recurrence-free survival; SUVmax: maximum standardized uptake value.
Cost-effectiveness of basal 18-F FDG PET/CT.

BC: breast cancer, ALND: axillary lymph node dissection, QALY: quality-adjusted life years, SLNB: sentinel lymph node biopsy; PET/CT: positron emission tomography/computed tomography; MRI: magnetic resonance imaging.
Discussion
18F-FDG PET/CT in regional and distant staging
In the preoperative work-up of early-stage BC with clinically negative axilla, 18F-FDG PET/CT can be omitted because it hardly affects the initial staging and treatment planning in most patients. In regional staging, 18F-FDG PET/CT is less sensitive than sentinel lymph node biopsy in assessing ALN involvement. The low prevalence of distant metastases and the risk of false-positive findings retract from the usefulness of 18F-FDG PET/CT for distant staging in these patients.11–13
By contrast, in patients with positive axilla, especially those with locally advanced BC, 18F-FDG PET/CT can be useful prior to surgery or neoadjuvant chemotherapy, based on the high rate of detection of distant metastases, ranging from 6% to 26%.16,20,23,27 The percentage of patients with extra-ALN involvement detected by PET/CT in locally advanced BC ranges from 10% to 29%.14–16 Moreover, in this setting, given the high prevalence of lymph node involvement (up to 80%), 18F-FDG PET/CT’s high negative predictive value in lymph nodes adds to its usefulness.12,15
Patients younger than 40 years usually have more aggressive phenotypes of BC, which are more easily detected by 18F-FDG PET/CT.30,31 Thus, PET/CT can be useful if younger age is associated with higher risk disease.
Comparison of 18F-FDG PET/CT with other diagnostic techniques
Several studies have found that 18F-FDG PET/CT is more accurate than conventional imaging (e.g. US) in the detection of axillary and extra-axillary (internal mammary) lymph node metastases and bone metastases.16–18,20,23,24,36,38 However, methodological shortcomings in some of these studies reduce their value in addressing the real diagnostic impact of 18F-FDG PET/CT. The superiority of 18F-FDG PET/CT with respect to conventional imaging for detecting extra-ALNs and metastatic disease acquires more relevance in locally advanced BC.20,21,23 Unfortunately, the three meta-analyses that examined 18F-FDG PET/CT’s value in detecting distant metastases were limited by the inclusion of patients regardless of their treatment status.24–26
Diagnostic and prognostic impact of 18F-FDG PET/CT
18F-FDG PET/CT imaging changes the initial staging by conventional imaging in 5% to 52% of cases; the percentage is lower in patients with early-stage or operable BC 13 and higher in those with locally advanced BC or inflammatory BC.11,41,42,46 18F-FDG PET detects distant metastases in more than 20% of patients with locally advanced BC14,16,20,27,28,31,37,39,40 and changes the initial staging in up to 52% of patients with inflammatory BC.29–32 Other authors have reported changes in N stage based on the diagnosis of unsuspected metastatic extra-ALNs (N3). These findings are significantly more likely in patients with at least stage IIB BC (considered locally advanced BC by some authors or at least BC in which neoadjuvant chemotherapy is indicated), in whom PET/CT detected extra-ALN in 5%–29%.14,16,37,39,40,43,46 Although the evidence seems stronger for stages IIB and III BC, the heterogeneous populations and the predominantly retrospective design used in the studies may explain the differences obtained and the lack of reproducibility of the results.
Another limitation is that most of the studies evaluated did not histologically confirm all the suspicious or pathological lesions detected by 18F-FDG PET/CT; some studies used other imaging techniques to confirm the lesions, although they are less accurate than 18F-18F-FDG PET/CT, especially for bone and morphologically normal structures such as small-size lymph nodes. However, despite our strict reference standard, we recognize that it would be unethical to obtain specimens for histological confirmation from all the lesions detected by PET. Given the greater accuracy of 18F-FDG PET/CT with respect to the most morphological imaging techniques, pathologic findings on PET/CT should be considered suspicious for metastases in the absence of other explanations.
Therapeutic impact consists of changes from curative to palliative treatment, changes to the extent of the radiotherapy field, or additional surgical treatment. 40 18F-FDG PET/CT changes the initial treatment in 1%–8% of patients with early-stage BC, in 7%–13% of those with locally advanced BC,13,14,20,37,40,41,43 and in up to 52% of those with more aggressive tumors such as inflammatory BC. 31 Nevertheless, the therapeutic impact of 18F-FDG PET/CT in early-stage BC is controversial. Jeong et al. 13 reported that the impact of 18F-FDG PET/CT on the initial staging and treatment in patients with early BC with clinically negative axillary nodes was practically null. By contrast, Bernsdorf et al. 41 found a substantial impact on initial staging and on clinical management in patients with early-stage BC and tumors ≥2 cm. Thus, there is insufficient evidence to establish the therapeutic impact of 18F-FDG PET/CT in early-stage BC. By contrast, 18F-FDG PET/CT has a greater therapeutic impact in locally advanced BC.14,20,31,37 The retrospective nature and lack of histological confirmation were the main limitations of most of the studies included, reducing their evidence level, despite the large number of patients evaluated in some. However, the evidence seems stronger for stages IIB and III of BC.
Glycolytic activity with 18F-FDG PET/CT, tumor biology, and prognosis
Semiquantitative metabolic parameters obtained with 18F-FDG PET/CT provide information about tumor biology. Maximum standardized uptake value (SUVmax) increases with the biological aggressiveness of the tumors; high-grade, hormone receptor–negative, and triple-negative tumors have higher SUVmax than low-grade and hormone receptor–positive tumors.47–56 However, a reproducible SUVmax cutoff that would predict tumor biology has yet to be established. A mean SUVmax close to 10 has been reported in triple-negative BC, although the wide dispersion makes it impossible to define an effective cutoff.51–53 Differences in SUVmax values are mainly due to the enormous biological heterogeneity of BC, even among tumors of the same molecular phenotype, although differences in PET acquisition (supine vs. prone position) and in the way the region of interest was selected (automatically vs. manually) may also contribute to variation among studies.49,52 Another factor making comparisons among studies difficult is that molecular subtype based on immunohistochemistry might not correspond with the molecular subtype determined by the gene expression profile; furthermore, especially in advanced stages where neoadjuvant treatment is indicated, immunohistochemistry is done on imaging-guided core-needle biopsy specimens, so the results are not as reliable as analyses of the entire tumor. 56 However, despite these limitations, 18F-FDG-PET has demonstrated a good correlation with the molecular phenotypes.51,53,55,56
18F-FDG-PET can have an impact on the patient’s prognosis in two ways: by detecting distant metastases occult to other techniques and by detecting glycolytic activity in the primary tumor and ALN. Various authors have compared prognostic stratification at initial staging by 18F-FDG-PET versus conventional imaging.31,43 The detection of occult distant metastases on 18F-FDG PET/CT is associated with shorter survival independent of the molecular phenotypes.28,32,33,39 With respect to the semiquantitative information from 18F-FDG PET/CT, the prognostic impact of the SUVmax of the primary tumor is controversial. Whereas some authors found no association between tumor 18F-FDG uptake and prognosis,32,58 others reported that patients with high tumor uptake had worse outcomes.28,47,59,61 Furthermore, a single and reproducible SUVmax has yet to be established; cutoff values range from 3 to 6.28,59,61 The variability among studies is probably due to the different immunohistochemical characteristics of the tumors rather than to differences in methodology. However, it is noteworthy to consider how this semiquantitative metric affects the prognosis of both operable and advanced BC, regardless of the biology of the tumor.28,32,33,39,47,57–61 The lack of reproducibility of SUV has led to explorations of other metrics, such as total lesion glycolysis, although more studies are necessary to assess their impact. 60 The evidence for the prognostic value of SUVmax in ALN is limited, although higher values have been associated with higher recurrence rates.28,57,58
One important aspect is the applicability of the prognostic information derived from 18F-FDG PET. Whereas 18F-FDG PETs increasing the stage initially diagnosed with conventional imaging may modify the planned treatment, the use of information about tumor glycolysis is more controversial, although patients with tumors with high SUVmax should perhaps undergo stricter follow-up than those with tumors with low SUVmax. Thus, despite the high heterogeneity among the studies with respect to clinical stages, tumor biology (inflammatory BC, triple-negative BC), end-points (overall survival (OS), progression-free survival (PFS), disease-free survival (DFS)), methodology (breast, ALN), and design (retrospective or prospective), all the studies found that PET/CT has prognostic value in BC.28,32,33,39,47,57–61
Cost-effectiveness of 18F-FDG PET/CT
The evidence on the cost-effectiveness of 18F-FDG-PET or PET/CT in BC is limited. Meng et al. 63 concluded that MRI was the most cost-effective strategy to replace sentinel lymph node biopsy, but also pointed out that further studies using up-to-date techniques in larger samples are required to obtain more accurate data on the sensitivity and specificity of MRI. Sloka et al. 62 used PET/CT to calculate the cost savings for lymph node staging in early BC against ALN dissection, a context with reduced applicability. In summary, more studies are needed to evaluate cost-effectiveness of 18F-FDG-PET in patients with BC.
Conclusion
18F-FDG PET/CT is not recommended in early BC, although evidence supports its use in locally advanced BC based on improved regional and distant staging. The evidence for systematically recommending 18F-FDG PET/CT in triple-negative BC is more limited, and further studies are necessary to assess the real diagnostic and therapeutic impact. There is insufficient evidence to address the cost-effectiveness of 18F-FDG PET/CT. Baseline tumor glycolytic activity is associated with biology and prognosis. Well-designed prospective studies are necessary to determine the real impact of 18F-FDG PET/CT in the more controversial settings of BC staging.
Footnotes
Acknowledgements
All the authors have participated in the writing and revision of this article and take public responsibility for its content. The present publication is approved by all authors and by the responsible authorities where the work was carried out. All the authors confirm that the article is not under consideration for publication elsewhere.
Compliance with ethical standards
Neither animals nor human subjects were used in this study.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Ethical approval
This review article does not report any new studies done by any of the authors.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: Spanish Society of Nuclear Medicine and Molecular Imaging has supported the publication and english review expenses of this article.
