Abstract
Aim
To assess the diagnostic value of pre-surgery axillary ultrasound for nodal staging in patients with primary breast cancer and to identify clinical/histopathological factors impacting diagnostic performance.
Study Design
Single-center, retrospective chart analysis. We assessed sensitivity, specificity, and positive and negative predictive value of clinical examination as well as axillary ultrasound vs. clinical examination alone. The histopathological results were the standard of truth. In addition, we analyzed clinical and histopathological factors regarding their potential to impact sensitivity and specificity.
Results
We enrolled a total of 172 women in the study. Sensitivity of clinical examination plus ultrasound was significantly higher than for clinical examination alone (58% vs. 31.6%). Specificity and positive predictive value were similar while the negative predictive value increased from 63.4% to 73% when additionally applying ultrasound. Sensitivity and specificity of axillary ultrasound were impacted by tumor size (
Conclusion
Since pre-surgery axillary nodal staging is currently used to determine disease management, axillary ultrasound is a useful add-on tool in the diagnostic armamentarium for breast cancer patients. Tumor size, suspicious axillary palpation, number of affected lymph nodes and distant metastases increase diagnostic performance of this diagnostic modality.
Introduction
Thoroughly performed pre-surgery diagnostics are essential for all disease management decisions. In breast cancer patients, axillary lymph node status is of paramount importance as a prognostic factor and predictor of survival 1 as there is a direct correlation between primary tumor size and risk of axillary lymph node metastases as well as between the number of affected axillary lymph nodes and the recurrence risk. 2
Today, sentinel node biopsy and/or conventional axillary lymph node dissection (ALND, level I + II) are considered the gold standard for nodal staging. 3 While for clinically node-positive patients ALND is the standard of care, clinically node-negative patients need to be first evaluated by sentinel node biopsy (SLNB). In case of identification of affected lymph nodes, an ALND is recommended in order to avoid local tumor spread. 3 In order to shorten surgery time, attempts should be undertaken to apply noninvasive techniques for nodal staging, such as axillary ultrasound.
Ultrasound has been shown to be a useful tool in the diagnostic armamentarium for nodal staging. 4 Vaidya et al reported the combination of clinical palpation complemented by axillary ultrasound to increase the sensitivity for axillary lymph node detection from 70% to 82%. 5 However, accuracy strongly depends on technical skills and experience of the operator. 6 In our study we aimed to confirm this finding with the data generated in our population by our staff.
Sensitivity achieved by ultrasound, however, is inferior to those achieved by surgical procedures although fine needle aspiration cytology or biopsy might improve US diagnosis. In the study reported, here we attempted to identify clinical/histopathological factors that might impact diagnostic performance of the combination of palpation and axillary ultrasound.
Materials and Methods
Study Design
We conducted a retrospective chart analysis on women with primary breast cancer who were treated at the department of OB/GYN at Saarland University Hospital, 66421 Homburg/Saar, Germany over a period of two years. Our clinic is a certified breast cancer center that strictly follows the international diagnostic and therapeutic guidelines postulated by the European Society of Mastology, so called EUSOMA criteria. 7 According to the guidelines of the Helsinki Declaration, ethics approval was not necessary for this retrospective analysis.
Study Population
Datasets of women of all ages presenting a primary invasive breast cancer stage pT1a or higher were eligible for inclusion. Only complete cases that included a thoroughly documented medical history, records of clinical palpation findings, axillary ultrasound outcomes, and histopathological results of primary tumor and axillary lymph nodes were accepted. Patients, who had performed their pre-surgery diagnostic procedures externally or who presented noninvasive tumors (DCIS, CLIS) or a recurrent disease, were excluded.
Study Assessments
We used a special template for recording medical history comprising age, height, weight, family history, parity, age at first delivery, and duration of lactation. The clinical examination included inspection and palpation of both breasts and armpits providing the clinical nodal status.
Mammographies were performed at our local department of radiology and evaluated by at least two physicians (four-eyes-principle).
The axillary ultrasound examinations were carried out by specially trained gynecologists using a Hitachi EUB-8000 with a linear 13 MHz probe. The patient was lying in supine position with widely angled arms above the head. Breasts and armpits were studied in transversal and sagittal planes, with special focus on the region around the axillary vessels and the region above and below the collarbones. Lymph nodes size > 2 cm in diameter, round shape, Solbiati-Index < 2 (longitudinal-transverse diameter ratio), loss of central echo, disrupted lymph node capsule, and increased intranodal vascularization or atypical vessels were considered as morphologic ultrasound criteria of lymph node malignancy. The histopathological assessments were done by the in-house department of pathology (Dept. of Pathology).
Statistical Methods
Continuous variables are presented as mean ± standard deviation (SD). For categorical variables frequency resp. contingency tables were calculated. In case of missing data, the calculations were based on the available data set. We calculated sensitivity, specificity, ppv, and npv for the two diagnostic methods (palpation alone and palpation plus axillary ultrasound).
We used Pearson's Chi-Square-test resp. Fisher's exact test to compare sensitivity and specificity depending on certain clinical and pathological factors. A
All data analyses were performed with SPSS™ (SPSS Inc. Chicago, Illinois, Version 17).
Results
Demography of study population (n = 172).

Tumor characteristics of study population (n = 172).

The diagnostic performance of the axillary palpation alone and the combination of palpation plus axillary ultrasound are depicted in Figure 1. The palpation alone had a low sensitivity of 31.6% while both modalities combined reached 58%, a statistically significant increase in diagnostic efficacy ( Prediction of nodal status by palpation only vs. palpation and sonography.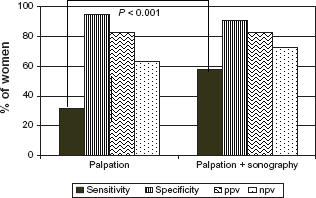
Impact of clinical and pathological factors on sensitivity and specificity of palpation plus axillary sonography.
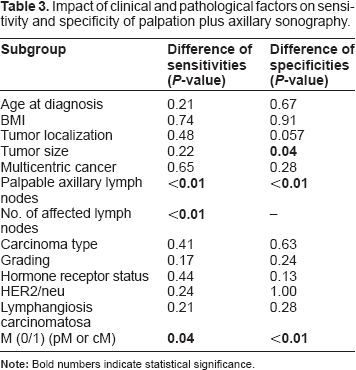
While for tumor size, no clear-cut trend depending on size categories could be detected, positive or negative axillary palpation significantly increased sensitivity and specificity, respectively. With increasing numbers of affected lymph nodes, sensitivity of ultrasound increased (
Factors significantly influencing sensitivity and specificity of the combination of palpation plus axillary ultrasound.
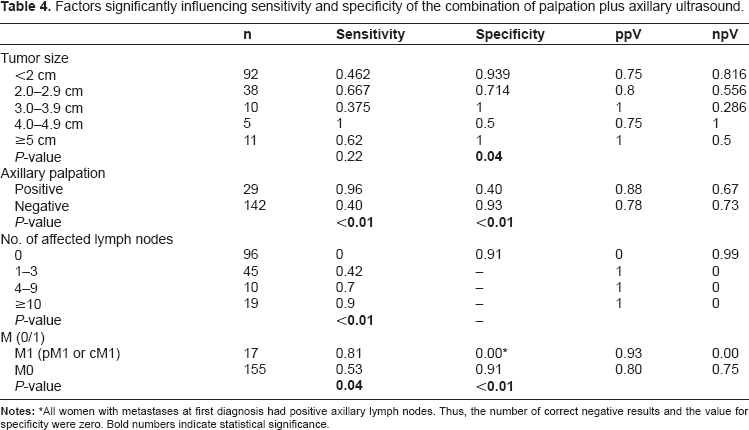
All women with metastases at first diagnosis had positive axillary lymph nodes. Thus, the number of correct negative results and the value for specificity were zero. Bold numbers indicate statistical significance.
Discussion
We conducted this retrospective chart analysis to assess the diagnostic value of pre-surgery axillary ultrasound for nodal staging in breast cancer patients and to identify clinical/pathological factors impacting diagnostic performance. Limitations of this study include the retrospective study design, a somewhat small sample size, as well as the inter-reader variability in ultrasound results due to three participating gynecologists. In addition, the study population showing relatively advanced tumor stages and large primary tumors might not be representative for other centers.
Our results show that clinical palpation alone had a low sensitivity of 31.6% while when combined with axillary ultrasound, both modalities together reached 58%, a statistically significant increase in sensitivity (
We could show that clinical/pathological parameters like age at diagnosis, BMI, tumor localization, multi-centricity, carcinoma type, grading, hormone receptor status, HER2/neu status, and presence or absence of lymphangiosis carcinomatosa had no impact on sensitivity and specificity of palpation plus axillary ultrasound. This result is in line with the findings of Bedrosian et al, who conducted a very similar study on 208 patients. 14 In contrast to Bedrosian et al, we could provide evidence for four factors increasing either sensitivity (number of affected lymph nodes), specificity (tumor size), or both (axillary palpation, distant metastases). It appears quite conclusive that sensitivity increases parallel to the number of affected lymph nodes. Unfortunately, we were not able to demonstrate a clear-cut relationship between tumor size and sensitivity: While sensitivity increased from the tumor size category < 2 cm (46.2%) to 2.0–2.9 cm (66.7%), no further increase for larger tumors could be detected. This is probably due to the small sample size of 10, 5, and 11 patients in the tumor categories of 3.0–3.9 cm, 4.0–4.0 cm and >5 cm, respectively (Table 4). It is noteworthy that the findings of Aitken et al provide strong evidence that tumor size > 5 cm is one of the strongest predictors of lymph node metastases. 18 Susini et al found tumor localization in the outer quadrants to be a significant independent predictor of axillary lymph node metastases. 19 In our population, we were not able to confirm any impact of tumor localization. The fact that suspicious axillary palpation and presence of metastases affect axillary lymph node status is obvious.
Conclusion
Since pre-surgery axillary nodal staging is determining disease management, ie, SNLB or ALND, axillary ultrasound is a useful add-on tool in the diagnostic armamentarium for breast cancer patients. Tumor size, suspicious axillary palpation, number of affected lymph nodes, and distant metastases increase diagnostic performance of this diagnostic modality.
Author Contributions
Conceived and designed the experiments: KE, CL, AL, MFO. Analysed the data: KE, CL, AL, MFO, JE. Wrote the first draft of the manuscript: JE. Contributed to the writing of the manuscript: KE, AL, ES. Agree with manuscript results and conclusions: KE, CL, AL, MFO, JE, ES. Jointly developed the structure and arguments for the paper: JE, KE, ES. Made critical revisions and approved final version: KE, ES. All authors reviewed and approved of the final manuscript.
Funding
Authors disclose no funding sources.
Competing Interests
Author(s) disclose no potential conflicts of interest.
Abbreviations
ALND, axillary lymph node dissection; BMI, body mass index; CLIS, lobular carcinoma in situ; DCIS, ductal carcinoma in situ; npv, negative predictive value; ppv, positive predictive value; SLNB, sentinel lymph node biopsy.
Disclosures and Ethics
As a requirement of publication author(s) have provided to the publisher signed confirmation of compliance with legal and ethical obligations including but not limited to the following: authorship and contributorship, conflicts of interest, privacy and confidentiality and (where applicable) protection of human and animal research subjects. The authors have read and confirmed their agreement with the ICMJE authorship and conflict of interest criteria. The authors have also confirmed that this article is unique and not under consideration or published in any other publication, and that they have permission from rights holders to reproduce any copyrighted material. Any disclosures are made in this section. The external blind peer reviewers report no conflicts of interest.
