Abstract
Although the oncogenic role of PPFIA1 (liprin-α1) in breast cancer has been reported, whether its dysregulation is associated with metastasis risk or survival outcomes in breast cancer patients is not clear. Our primary data showed that PPFIA1 expression was significantly higher in liver metastatic breast tumors than in the primary tumors. Then, we tried to pool previous annotated genomic data to assess the prognostic value of PPFIA1 in distant metastasis–free survival, the risk of metastatic relapse, and metastatic relapse–free survival in breast cancer patients by data mining in two large databases, Kaplan–Meier plotter and bc-GenExMiner 4.0. Results from Kaplan–Meier plotter showed that although high PPFIA1 expression was generally associated with decreased distant metastasis–free survival in estrogen receptor+ patients, subgroup analysis only confirmed significant association in estrogen receptor+/N− (nodal negative) group (median survival, high PPFIA1 group vs low PPFIA1 cohort: 191.21 vs 236.22 months; hazard ratio: 2.23, 95% confidence interval: 1.42–3.5, p < 0.001), but not in estrogen receptor+/N+ (nodal positive) group (hazard ratio: 1.63, 95% confidence interval: 0.88–3.03, p = 0.12). In estrogen receptor− patients, there was no association between PPFIA1 expression and distant metastasis–free survival, no matter in Nm (nodal status mixed), N−, or N+ subgroups. In bc-GenExMiner 4.0, Nottingham Prognostic Index– and Adjuvant! Online–adjusted analysis validated the independent prognostic value of PPFIA1 in metastatic risks in estrogen receptor+/N− patients. Based on these findings, we infer that high PPFIA1 expression might be an independent prognostic indicator of increased metastatic relapse risk in patients with estrogen receptor+/N− breast cancer, but not in estrogen receptor+/N+ or estrogen receptor− patients.
Introduction
Breast cancer is the most common malignant disease in women. 1 Metastases at distant sites, such as bone, lung, and liver, are the main cause of death.2,3 Therefore, it is necessary to look for reliable predictors for site of distant metastasis and impact on survival. Since breast cancer is a clinically heterogeneous disease, gene-expression signatures of specific subtypes of primary tumors might be a useful prognostic tool to identify the patients with high risk of metastasis.4,5
PPFIA1 is a gene encoding liprin-α1 in human, which belongs to the liprin family of cytosolic scaffold proteins and includes four liprin-α, two liprin-β members, and liprin-γ/kazrinE. 6 The functions of liprin-α proteins have been mainly studied in neurons, such as regulating active zone morphogenesis, synapse maturation, and trafficking of synaptic vesicles.7,8 In breast cancer, PPFIA1 is frequently amplified and exerts regulative effect on cell adhesion, signal transduction, and organization of the cytoskeleton.9–11 Some recent studies suggest that PPFIA1 is an important gene regulating cell edge dynamics during motility and is required for migration and invasion of some breast cancer cells.12,13 Mechanistically, liprin-α1 can form a complex with liprin-β1, ERC1/ELKS, and LL5 proteins, which maintains the highly polarized and dynamic cytoplasmic structures of cancer cell front and drives tumor cell migration.13,14 Although the functional role of PPFIA1 in tumor cell invasion has been well characterized, whether its dysregulation is associated with metastasis risk or survival outcomes in breast cancer patients is not clear. One recent study explored the prognostic value of TMEM16A, PPFIA1, and FADD in invasive ductal breast carcinoma. 15 Their results showed that although individual expression of these genes did not have significant association with disease-free survival, their combined expression was significantly associated with disease-free survival (p = 0.034). 15 However, the number of patients included in this study is relatively small (N = 98) and the heterogeneous nature of breast cancer was not considered in their analysis.
In this study, we tried to pool previous annotated genomic data to assess the prognostic value of PPFIA1 in distant metastasis–free survival (DMFS), the risk of metastatic relapse (MR), and MR-free survival in breast cancer patients by data mining in two large databases.
Materials and methods
Patient tissue collection
This study was approved by the Ethics Committee of the First Affiliated Hospital of Guangxi University of Chinese Medicine and the Affiliated Tumor Hospital of Guangxi Medical University. Primary and metastatic tumor tissues from 34 breast cancer patients with liver metastasis were obtained from tissue bank of these two hospitals. The samples were obtained during surgical resections and were immediately frozen in liquid nitrogen and stored at −80°C until further use.
Quantitative real-time polymerase chain reaction analysis
Total RNA was extracted from cells using the TRIzol Reagent (Invitrogen, Carlsbad, CA, USA) and converted into cDNA using a First-Strand Synthesis Kit (Invitrogen). To detect PPFIA1 messenger RNA (mRNA) expression, quantitative real-time polymerase chain reaction (qRT-PCR) analysis was performed on an ABI 7700 sequence detector using gene-specific primers and TaqMan Universal PCR Master Mix according to the directions of manufacturer (Applied Biosystems, Foster City, CA, USA). The primers used for PPFIA1 were as follows: F, 5′-ACAGATGGAGTGCTGGACAT-3′; R, 5′-CACATGACTGGAAAGGGAAG-3′. Glyceraldehyde 3-phosphate dehydrogenase (GAPDH) was used as the housekeeping gene to normalize PPFIA1 expression. The primers used for GAPDH were as follows: F, 5′-GAAGGTGAAGGTCGGAGTCAAC-3′; R, 5′-CAGAGTTAAAAGCAGCCCTGGT-3′. The 2−ΔΔCt method was used to calculate the relative quantities of PPFIA1. All reactions were performed in triplicate.
Western blot analysis
Total protein from cell samples were extracted using a radioimmunoprecipitation assay (RIPA) lysis buffer (Beyotime, Shanghai, China), and the protein concentration was measured using the bicinchoninic acid (BCA) method. Each sample containing 25 µg protein was separated by 10% sodium dodecyl sulfate–polyacrylamide gel electrophoresis (SDS-PAGE) and then was transferred to a polyvinylidene difluoride (PVDF) membrane. After washing, the membranes were incubated with primary anti-PPFIAI (1:250, ABT352; Merck Millipore, Billerica, MA, USA) and anti-GAPDH (1:2500, ab9485; Abcam, Cambridge, UK). The second horseradish peroxidase (HRP)-conjugated secondary antibodies were obtained from Abcam. The signal intensity of the protein bands was visualized using the enhanced chemiluminescence (ECL) Western blotting substrate (Promega, Madison, WI, USA). The signal intensity was quantified using ImageQuant TL software (GE Healthcare, Piscataway, NJ, USA).
Exploring microarray data
The expression of PPFIA1 in one previous microarray data (GDS5666) 16 that compared gene-expression profiles among the parental mammary tumor cell line 4T1 and 4T1-derived metastatic populations isolated from liver, lung, or bone were reviewed in Gene Expression Omnibus (GEO) datasets with the online tool provided by the database.
Analysis of the association between PPFIA1 expression and metastasis risk and metastasis-free survival
The association between PPFIA1 expression and DMFS was assessed using Kaplan–Meier plotter (http://kmplot.com/analysis/), an online database that provides assessment of the effect of 54,675 genes on survival using 10,293 cancer samples, including 22,277 genes in 5143 breast cancer samples.17,18 The survival curve was generated using the JetSet best probe set with median as the cutoff. Subgroup analysis was further performed according to the estrogen receptor (ER) status and lymph nodal status. Long-rank test was performed to detect difference between the curves.
To assess the association between PPFIA1 expression and MR risk and MR-free survival, pool study was performed using bc-GenExMiner 4.0, which is a database of published annotated genomic data including 5609 breast cancer patients.19,20 Subgroup analysis was performed according to the ER status and the lymph nodal status. The prognostic impact of PPFIA1 was first evaluated by means of univariate Cox proportional hazards model and a forest plot and Kaplan–Meier curves (for the median-split pool). To evaluate the independent prognostic impact of PPFIA1 relative to the well-established breast cancer prognostic indexes, including Nottingham Prognostic Index (NPI)21,22 and Adjuvant! Online (AOL), 23 adjusted Cox proportional hazards models were systematically performed.
Statistical analysis
Data were presented as mean ± standard deviation (SD) with at least three repeats. Statistical analyses and graphical representations were performed with GraphPad Prism 5 (San Diego, CA, USA) software. The Student’s
Results
PPFIA1 upregulation might be associated with distant metastasis of breast cancer
In this study, we first investigated the expression profiles of PPFIA1 in 4T1 parental cells, 4T1-derived primary tumor, and the metastatic populations isolated from liver, lung, or bone in one previous microarray (GDS5666). The results showed that the bone and liver metastatic populations had increased expression of PPFIA1 (Figure 1(a)). In addition, the rank of PPFIA1 within the bone and liver metastatic populations was also elevated (Figure 1(a)). To further verify this trend, we compared PPFIA1 expression in 34 cases of paired primary breast tumors and liver metastatic tumor samples. QRT-PCR analysis confirmed that PPFIA1 expression was significantly higher in liver metastatic tumors than in the primary tumors (Figure 1(b)). Then, we detected PPFIA1 expression at protein level in cases 5, 9, 25, and 30, which had significantly upregulated PPFIA1 mRNA expression in liver metastasis than in primary tumors (Figure 1(c) and (d)), and in cases 7, 13, 23, and 32, which had similar PPFIA1 mRNA expression between liver metastasis and primary tumors (Figure 1(e) and (f)). Western blotting data confirmed significantly upregulated PPFIA1 expression in liver metastasis compared to primary tumors in cases 5, 9, 25, and 30 (Figure 1(d)). In comparison, PPFIA1 expression was similar between liver metastasis and primary tumors in cases 7, 13, 23, and 32 (Figure 1(f)). These results suggest that PPFIA1 expression is consistent at mRNA and protein levels. Since PPFIA1 is generally upregulated in liver metastasis, we further analyzed its association with DMFS in breast cancer patients. Data mining in Kaplan–Meier plotter showed that among 1746 patients with breast cancer, high PPFIA1 expression was significantly associated with decreased DMFS (hazard ratio (HR): 1.6, 95% confidence interval (CI): 1.31–1.96, p < 0.001; Figure 1(g)). The median DMFS in low PPFIA1 expression cohort was 142.39 months, but it was significantly dropped to 68.4 months in high PPFIA1 expression cohort (Figure 1(g)).
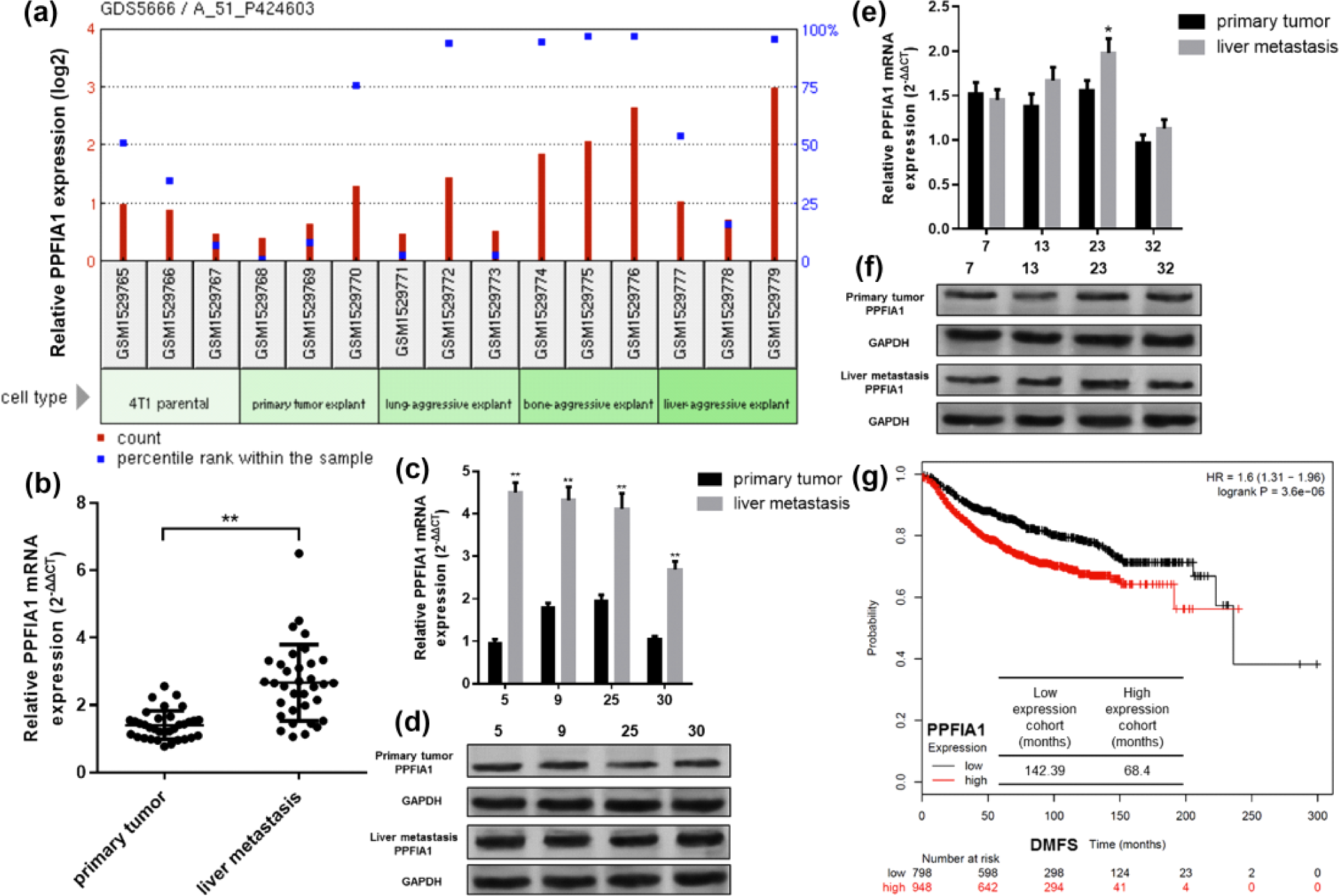
PPFIA1 upregulation might be associated with distant metastasis of breast cancer. (a) PPFIA1 expression in 4T1 parental cells, 4T1-derived primary tumors, and in metastatic populations isolated from liver, lung, or bone. Data were obtained using one previous microarray (GDS5666). The bar chart was obtained using the tool provided by GEO datasets. (b) QRT-PCR analysis of PPFIA1 mRNA expression in 34 cases of primary breast tumors and the metastatic breast tumors in liver. (c–f) qRT-PCR analysis of PPFIA1 mRNA expression (c and e) and western blot analysis of PPFIA1 protein expression (d and f) in case 5, 9, 25 and 30 (c and d) and in cases 7, 13, 23 and 32 (e and f) among the 34 paired tissues. (g) Kaplan–Meier plots of the association between PPFIA1 expression and DMFS in breast cancer patients. Data were obtained using Kaplan–Meier plotter.
High PPFIA1 expression might be associated with unfavorable DMFS in ER+ breast cancer
Then, we further investigated the association between PPFIA1 expression and DMFS in different subgroups of breast cancer. Interestingly, in ER+ breast cancer, we found that high PPFIA1 expression was associated with significantly shorter DMFS (median survival, high PPFIA1 group vs low PPFIA1 cohort: 191.21 vs 236.22 months; HR: 2.13, 95% CI: 1.49–3.04, p < 0.001; Figure 2(a)). To further investigate whether lymph nodal status has an influence on the association, subgroup analysis was further performed in lymph nodal positive and negative groups. Results showed that the association was only observed in ER+/N− (nodal negative) group (median survival, high PPFIA1 group vs low PPFIA1 cohort: 191.21 vs 236.22 months; HR: 2.23, 95% CI: 1.42–3.5, p < 0.001; Figure 2(b)), but not in ER+/N+ (nodal positive) group (HR: 1.63, 95% CI: 0.88–3.03, p = 0.12; Figure 2(c)). However, in ER− patients, there was no association between PPFIA1 expression and DMFS, no matter in Nm (nodal status mixed), N−, or N+ subgroups (Figure 2(d)–(f)).
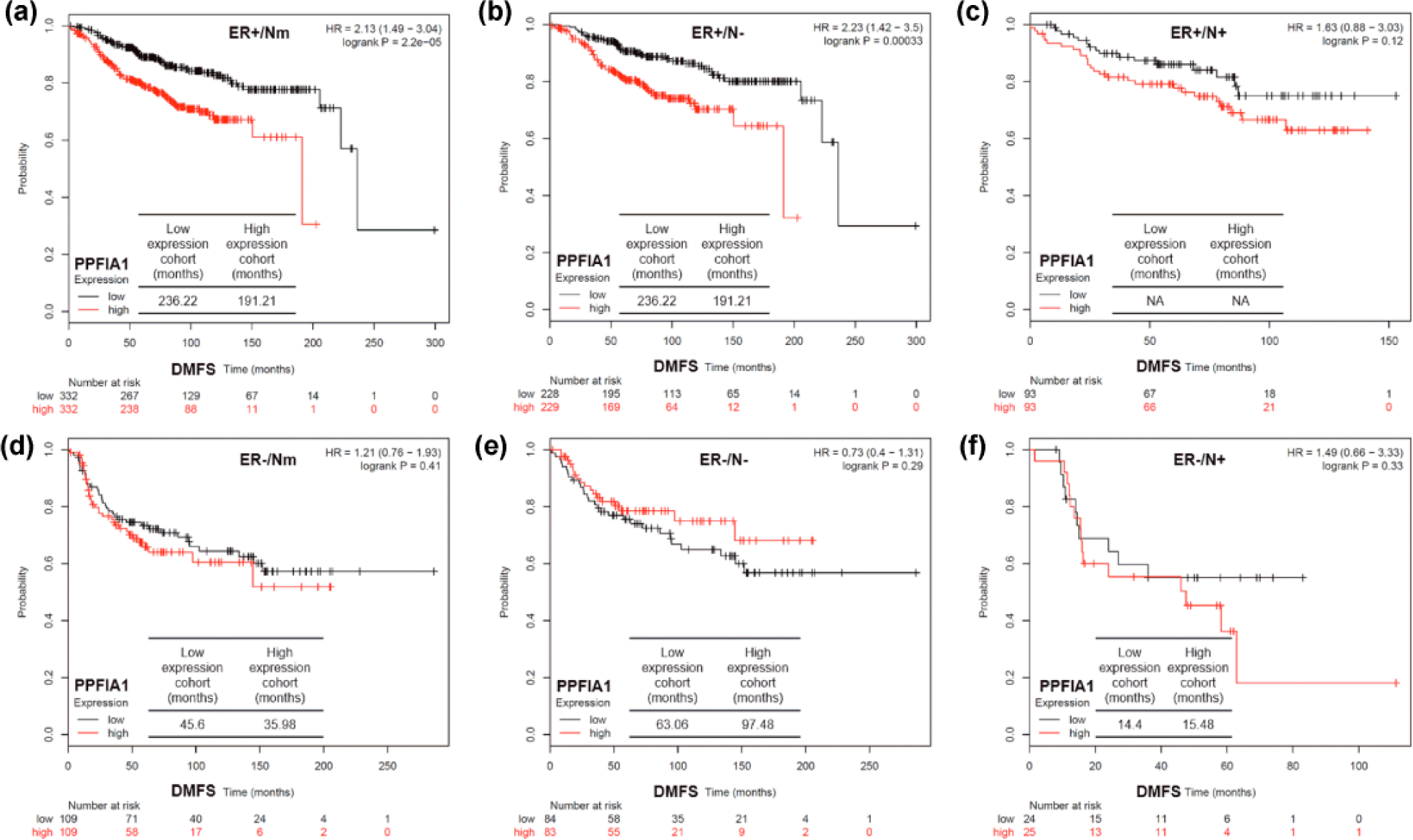
Kaplan–Meier plots of the association between PPFIA1 expression and DMFS in breast cancer patients grouped by ER and lymph nodal status: (a–f) Kaplan–Meier plots of the association between PPFIA1 expression and DMFS in different group of breast cancer patients (A: ER+ and Nm; B: ER+ and N−; C: ER+ and N+; D: ER− and Nm; E: ER− and N−; F: ER− and N+). The median survival (months) was calculated and given in the tables within the images. N (+, −, m): lymph nodal status (+: positive, −: negative, m: mixed; data were obtained using Kaplan–Meier plotter).
High PPFIA1 expression might be an independent prognostic indicator of increased MR risk in patients with ER+/N− breast cancer
To further investigate the prognostic value of PPFIA1 in metastasis, data mining was further performed in bc-GenExMiner 4.0. In this database, we found that 36 previous studies including 5861 breast cancer patients reported the association between PPFIA1 expression and MR risk and MR-free survival (Supplementary Table 1). Forest plot and Kaplan–Meier curves showed that in ER+/Nm patients, PPFIA1 was associated with increased risk of MR (HR: 1.24, 95% CI: 1.16–1.33, p < 0.001; Figure 3(a) and Table 1) and shorter MR-free survival (HR: 1.40, 95% CI: 1.21–1.63, p < 0.001; Figure 3(b)). NPI- and AOL-adjusted studies confirmed that high PPFIA1 expression was an independent indicator of increased MR risk in this group of patients (p = 0.011 and p = 0.014, respectively; Figure 3(c) and (d)). To test robustness of the findings, subgroup analysis was further performed in ER+ patients with negative or positive lymph nodal metastasis, respectively. In ER+/N− patients, high PPFIA1 expression was also significantly associated with increased risk of MR (HR: 1.25, 95% CI: 1.13–1.40, p < 0.001; Figure 4(a) and Table 1) and shorter MR-free survival (HR: 1.42, 95% CI: 1.43–1.78, p = 0.0023; Figure 4(b)). NPI- and AOL-adjusted results confirmed that high PPFIA1 expression was an independent indicator of increased MR risk in this group of patients (p = 0.0003 and p = 0.0001, respectively; Figure 4(c) and (d)). However, although the univariate Cox analysis showed that high PPFIA1 expression was associated with increased risk of MR in ER+/N+ patients (Figure 5(a) and (b)), but NPI- and AOL-adjusted results failed to confirm the association (p = 0.984 and p = 0.398, respectively; Figure 5(c) and (d)).

The association between PPFIA1 expression and MR risk and MR-free survival in ER+/Nm breast cancer patients: (a) and (b) Forest plots displaying univariate Cox’s analysis of PPFIA1 expression and the risk of (a) MR and (b) MR-free survival in ER+/Nm breast cancer patients. (c) Summary of original studies with data used for NPI-/AOL-adjusted analysis and (d) the results of NPI-/AOL-adjusted analysis of the association between PPFIA1 expression and the risk of MR in ER+/Nm breast cancer patients. Data were obtained using bc-GenExMiner 4.0.
PPFIA1 univariate Cox analysis of MR risk in bc-GenExMiner 4.0.

HR: hazard ratio; 95% CI: 95% confidence interval; ER: estrogen receptor status; MR: metastatic relapse; N (+, −, m): lymph nodal status (+: positive, −: negative, m: mixed).
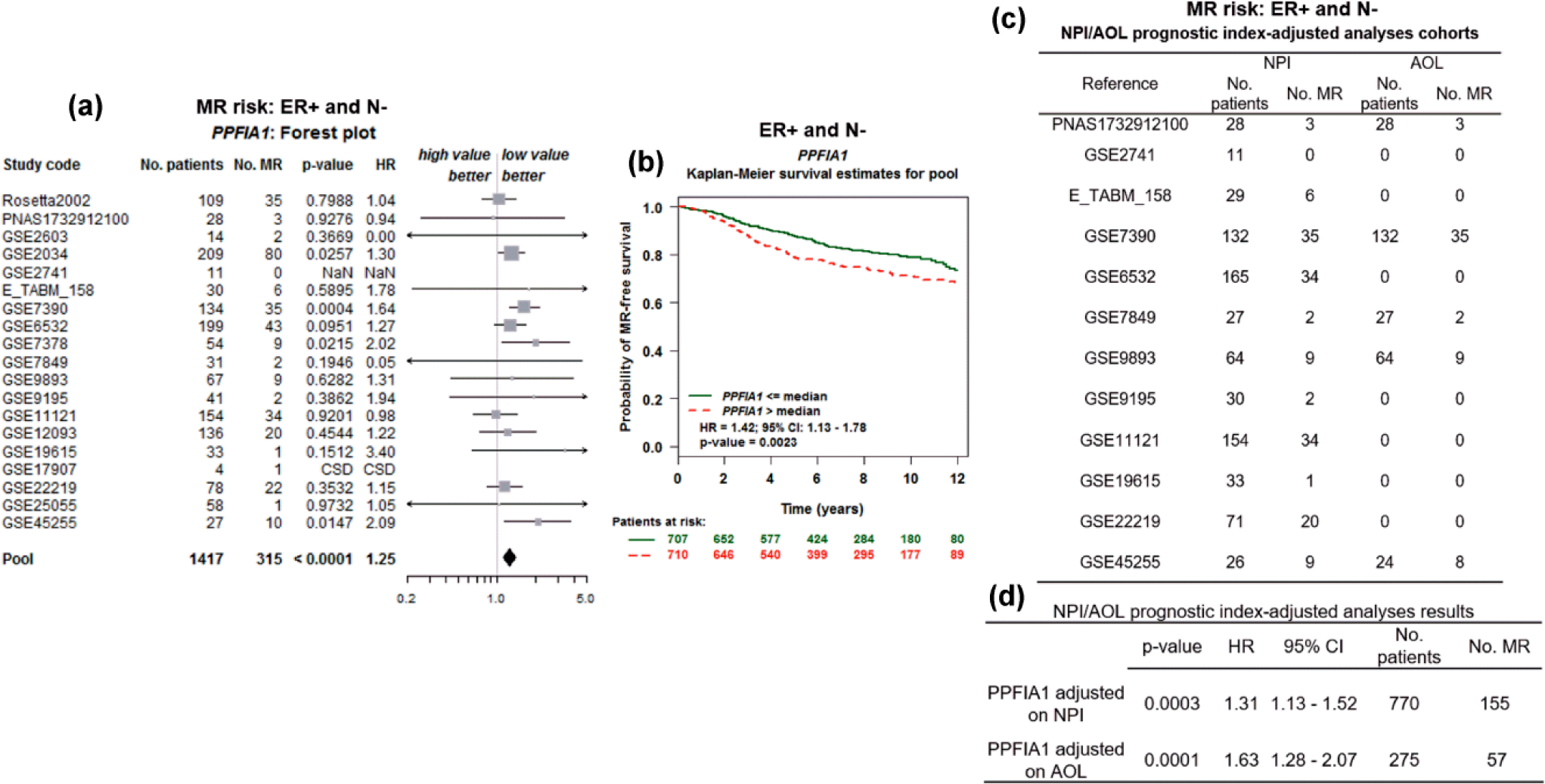
The association between PPFIA1 expression and MR risk and MR-free survival in ER+/N− breast cancer patients. (a and b) Forest plots displaying univariate Cox’s analysis of PPFIA1 expression and the risk of (a) MR and (b) MR-free survival in ER+/N− breast cancer patients. (c) Summary of original studies with data used for NPI-/AOL-adjusted analysis and (d) the results of NPI-/AOL-adjusted analysis of the association between PPFIA1 expression and the risk of MR in ER+/N− breast cancer patients. Data were obtained using bc-GenExMiner 4.0.

The association between PPFIA1 expression and MR risk and MR-free survival in ER+/N+ breast cancer patients: Forest plots displaying univariate Cox’s analysis of PPFIA1 expression and the risk of (a) MR and (b) MR-free survival in ER+/N+ breast cancer patients. (c) Summary of original studies with data used for NPI-/AOL-adjusted analysis and (d) the results of NPI-/AOL-adjusted analysis of the association between PPFIA1 expression and the risk of MR in ER+/N+ breast cancer patients. Data were obtained using bc-GenExMiner 4.0.
High PPFIA1 expression is not an independent prognostic indicator of MR risk in patients with ER− breast cancer
Then, we assessed the prognostic value of PPFIA1 in ER− breast cancer patients. No matter in ER−/Nm, ER/N−, or ER/N+ patients, the results of univariate Cox analysis and NPI- and AOL-adjusted analysis did not find any significant association between PPFIA1 expression and MR risk (Supplementary Figures 1–3).
Discussion
The PPFIA1 gene is located in chromosome 11q13, the region of which is frequently amplified in breast cancer. 15 One previous microarray based on 2000 human breast cancer cases demonstrated that PPFIA1 is amplified in 15% of the cases and is one of the most upregulated genes in breast cancer.10,24 A series of studies observed that liprin-α1 can positively enhance cell migration and invasion of some types of cancer cells, such as colon cancer cells 25 and breast cancer cells. 12 In colon cancer cell line RKO cells, liprin-α1 interacts with ING4, which is a tumor suppressor that inhibits cell proliferation via interacting with p53 and the histone acetyltransferase p300. 25 Liprin-α1 overexpression or ING4 silencing enhances tumor cell proliferation and migration. 25
In breast cancer, the role of liprin-α1 as a positive regulator of cancer cell invasion and migration has been reported. Mechanistically, liprin-α1 is required for stabilization of lamellipodial protrusions, which are morphological structures enabling effective migration of breast cancer MDA-MB-231 cells and other types of tumor cells. 6 Another recent study also found that liprin-α1 can positively enhance the morphological changes such as cellular junctions and front-rear cell polarity as well as changes in focal adhesion morphology in invasive breast cancer cells. 6 In in vitro cell model, knockdown of liprin-α1 significantly reduces invasion capability by reducing efficient motility and extracellular matrix (ECM) degradation. 12 The results of time-lapse studies indicated that the reduced ECM degradation might be a result of impaired stability of the invadopodia. 12 In in vivo animal model, liprin-α1 silencing strongly impairs the metastatic potential of breast tumors. 13 These findings suggest that liprin-α1 is an important protein contributing to tumor cell invasion. In this study, based on 34 cases of paired breast cancer liver metastasis and primary tumors, we also confirmed significantly upregulated PPFIA1 expression in the distant metastatic tissues. Therefore, it is meaningful to assess its prognostic value in breast cancer.
A very recent study explored the prognostic value of PPFIA1 expression alone or in combination with TMEM16A and FADD, two genes coding a chloride channel and an adaptor protein, respectively, in invasive ductal breast carcinoma. 15 Their results suggest that PPFIA1 expression alone could not predict poor disease-free survival, but their combined expression was significantly associated with perineural invasion and poor disease-free survival. 15 However, the statistical power of this study is low due to small number of patients included (N = 98). In addition, subgroup analysis was not performed due to the small number of patients. Our data mining in Kaplan–Meier plotter showed that among 1746 patients with breast cancer, high PPFIA1 expression was significantly associated with decreased DMFS in ER+ patients, but not in ER− patients. In addition, subgroup analysis showed that the association was only significant in ER+/N− group, but not in ER+/N+ group.
To further verify these findings and to assess the independent prognostic value of PPFIA1 in tumor metastasis, data mining was further performed in bc-GenExMiner 4.0, in which we found that 36 previous studies including 5861 breast cancer patients reported the association between PPFIA1 expression and MR risk and MR-free survival. Forest plot and Kaplan–Meier curves showed that in ER+/N− patients, high PPFIA1 expression was significantly associated with increased risk of MR and shorter MR-free survival. The following NPI- and AOL-adjusted results confirmed the independent prognostic value of PPFIA1 in ER+/N− patients. However, although the univariate cox analysis showed that high PPFIA1 expression was associated with increased risk of MR in ER+/N+ patients, NPI- and AOL-adjusted results failed to confirm the association. In ER− breast cancer patients, no significant association between PPFIA1 expression and MR risk was observed.
Conclusion
PPFIA1 upregulation might be associated with distant metastasis of breast cancer. High PPFIA1 expression might be an independent prognostic indicator of increased MR risk in patients with ER+/N− breast cancer, but not in ER+/N+ or ER− patients.
Footnotes
Acknowledgements
J.Y. and N.N.W. have contributed equally to this study.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
