Abstract
Background:
Studies in the United States are scarce that assess the survival differences between breast-conserving surgery plus radiation (Breast-Conserving Therapy; BCT) and mastectomy groups using population-based data while accounting for sociodemographic and clinical factors that affect the survival of women with early-stage breast cancer (ESBC).
Objective:
To assess whether BCT provides superior long-term overall survival (OS) and breast cancer-specific survival (BCSS) compared with mastectomy in women with ESBC, while considering key factors that impact survival.
Design:
Cohort study.
Methods:
We analyzed data on women aged 20 years and older diagnosed with stage I-II breast cancer (BC) in 2004 who received either BCT or mastectomy. The data were collected by 5 state cancer registries through the Centers for Disease Control and Prevention-funded Patterns of Care study. Multivariable Cox proportional hazard models, accounting for sociodemographic and clinical factors, were used to calculate hazard ratios (HRs) with 95% confidence intervals (CI). Sensitivity analysis involved optimal caliper propensity score (PS) matching to address residual confounding.
Results:
Of the 3495 women, 41.5% underwent mastectomy. The 10-year OS and BCSS were 82.7% and 91.1% for BCT and 72.3% and 85.7% for mastectomy, respectively. Adjusted models showed that mastectomy recipients had a 22% higher risk of all-cause deaths (ACD) (HR = 1.22, 95% CI = [1.06, 1.41]) and a 26% higher risk of breast cancer-specific deaths (BCD) (HR = 1.26, 95% CI = [1.02, 1.55]) than BCT recipients. Sensitivity analysis demonstrated that mastectomy was associated with a higher risk of ACD (P < .05) but did not exhibit a statistically significant risk for BCD. Women with HR+/HER2+ (luminal B) or invasive ductal carcinoma BC who underwent mastectomy had higher risks of ACD and BCD compared with BCT recipients, while the hazards for ACD in triple-negative BC did not remain significant after adjusting for covariates.
Conclusion:
ESBC BCT recipients demonstrate superior OS and BCSS compared with mastectomy recipients.
Plain Language Summary
This study compared the long-term survival outcomes of breast-conserving therapy (BCT) and mastectomy for early-stage breast cancer. Analyzing data from over 3400 women diagnosed in 2004, researchers found that BCT recipients had higher 10-year overall and breast cancer-specific survival rates compared with mastectomy recipients. Adjusted models showed a 22% higher risk of all-cause death
Introduction
Breast cancer (BC) is the most commonly diagnosed cancer and the second leading cause of cancer death among American women. 1 In 1980s, randomized clinical trials for early-stage breast cancer (ESBC) demonstrated that breast-conserving surgery plus radiation (breast-conserving therapy [BCT]) yielded equivalent survival as mastectomy.2-4 Consequently, a National Institutes of Health (NIH) Consensus Development Conference in June 1990 recommended BCT for most stage (I-II) BC cases, 5 leading to increase BCT adoption. 6
Despite earlier studies have confirmed that BCT has comparable overall survival than mastectomy,7-12 recent research suggests that BCT provides superior overall survival or disease-free survival.13-20 This improved survival with BCT is likely attributed to advancements in BC management, including enhanced patient selection for BCT, meticulous attention to surgical margins, and widespread adoption of 3-dimensional radiation treatment planning. 21
Notably, 10-year breast cancer-specific survival (BCSS) for women diagnosed with ESBC from 1998 to 2008, using SEER registry data, was 94% for BCT and 90 % for mastectomy. 17 In a study using 2004-2016 diagnosed ESBC cases in Louisiana, the 10-year BCSS was 92.7% for BCT and 88.8% for mastectomy, with 28.6% higher hazards of all-cause death (ACD) and 29.8% higher hazards of breast cancer-specific death (BCD) observed in the mastectomy group compared with BCT group. 16
Factors that have been associated with poorer BC survival are older age, lack of insurance or Medicaid coverage, advanced tumor stage, poorly differentiated tumor grade, obesity, higher comorbidity score, advanced cancer stage, hormone receptors negativity, and residing in rural areas.16,22-25 In addition, African American women experienced worse survival outcomes than White women.26,27 Various treatments, including chemotherapy 28 and hormone therapy, 29 can enhance BC survival, 16 and surgery choice independently influences BC survival. 16 However, an ongoing debate persists regarding whether BCT or mastectomy is superior or equivalent.
Studies conducted outside of the United States, such as in the Netherlands, 30 Germany, 13 and Norway, 14 have demonstrated better survival outcomes for BCT recipients. However, these studies often lack information on comorbidities13,14,30 and human epidermal growth factor receptor 2 (HER2) status,14,30 both of which can influence survival. Furthermore, one of these studies, based on the registry data, restricted the analysis to the 50- to 69-year age group, 14 thus limiting the generalization of study findings.
Similar studies conducted in the United States using cancer registry data lack information on comorbidity, HER2 status, and complete adjuvant therapy. 17 Two other studies15,31 based on National Cancer Database (NCDB) only included data from hospitals accredited by the American College of Surgeons, which might not represent all BC cases from nonhospital settings or those hospitals that were not part of NCDB.
Overall, prior US studies often omitted key factors influencing survival among ESBC women underwent BCT and mastectomy. To address these research gaps, our study aims to determine whether BCT provides superior long-term survival (overall survival [OS] and BCSS) compared with mastectomy using data from the Centers for Disease Control and Prevention (CDC) funded Patterns of Care (PoC) study. This data set includes complete information on adjuvant treatment (chemotherapy and hormone therapy), hormone receptors (HRs), HER2 status, and comorbidity severity, which enhances our understanding of ESBC treatment outcomes and aiding patient decision-making.
Material and Methods
Data source and study population
Our study used 2004 diagnosis year female BC data collected by five state cancer registries (California, Georgia, Kentucky, Louisiana, and Wisconsin) for the “Breast and Prostate Cancer Data Quality and Patterns of Care (PoC) study,” a CDC funded research project. 32 The CDC PoC study was specially designed to acquire complete adjuvant therapy data and gather additional data that were not routinely collected by US cancer registries for cancer cases diagnosed in 2004, including comorbidity severity. 33
Eligibility criteria
The eligibility criteria were women who were (1) aged 20 years or older diagnosed with microscopically confirmed early-stage (American Joint Committee on Cancer [AJCC] I-II) BC; (2) recipients of either BCT, defined as breast-conserving surgery (BCS) or lumpectomy (which involves removing the tumor or cancerous tissue while preserving the breast followed by radiation therapy to eliminate any remaining cancer cell in the breast, including site-specific surgery codes 19-24) or mastectomy only, defined as a removal most of or all breast tissue based on the definition of site-specific surgery codes 30 to 80 for mastectomy. The detailed BC surgery codes can be found on this website (https://seer.cancer.gov/archive/manuals/2010/AppendixC/breast/surgery_codes.pdf); and (3) possessing complete information on the BC diagnosis date, follow-up date, and cause of death. All eligible patients were followed through December 2015 if alive.
We excluded BC cases with an unknown race (n = 2), unknown urban-rural status (n = 5), unknown tumor size/no mass found (n = 4), unknown chemotherapy (n = 46), and unknown hormone therapy (n = 84). In addition, we excluded the non-Hispanic American Indian/Alaska Native (AI/AN) race (n = 15) due to the small number counts. Cases identified from autopsy or death certificate in the parent CDC PoC study were excluded.
The study data were collected by population-based cancer registries, which are authorized by law to access information on cancer patients from medical records for cancer surveillance purposes. The data set used for analysis was de-identified. This study received ethical approval from the Louisiana State University Health Science Center Institutional Review Board (IRB#2368), ensuring compliance with ethical standards and appropriate research procedures.
Outcome variable
The primary outcomes of interest were OS and BCSS. Survival was defined as the time elapsed between the date of initial diagnosis in 2004 and the date of death or date of last contact. The underlying cause of death was obtained from state death files or National Death Index (NDI) data. The event for OS was recoded as death from any cause. The event for BCSS was defined based on the Surveillance, Epidemiology, and End Results (SEER) cause-specific death classification 34 using ICD-10 SEER cause-specific death to differentiate BC and Non-BC deaths.
Exposure variable
The exposure variable was treatment type, which was categorized as mastectomy (site-specific surgery codes 30-80) and BCS (site-specific surgery codes 19-24) along with postsurgery radiation, referred as breast-conserving therapy (BCT), with BCT as the reference. 35
Covariates
Covariates included race/ethnicity (Non-Hispanic White, Non-Hispanic Black, Non-Hispanic Asian/Pacific Islander, and Hispanic), age at diagnosis (20-40, 40-49, 50-59, 60-69, ⩾70 years), marital status at diagnosis (single/separated/divorced/widowed, married, unknown), insurance status at diagnosis (no insurance, Medicaid [includes Medicare with Medicaid eligibility], private (include Medicare with private Supplement), Medicare, other public health insurance and unknown), area-level urban/rural (urban, rural, and urban-rural mix), diagnosis state (California, Georgia, Kentucky, Louisiana, and Wisconsin), and body mass index 36 (calculated in kg/m2 and classified as underweight <18.5, normal weight 18.5-<25, overweight 25-<30, obesity ⩾30, and unknown).
We used the Adult Comorbidity Evaluation-27 (ACE-27) developed by Piccirillo et al. This validated comorbidity index is tailored for cancer patients with twenty-six comorbidity categories that categorize specific diseases and conditions into one of the three levels of comorbidity: grade 1 (mild), grade 2 (moderate), or grade 3 (severe), and unknown based on the severity of individual organ decompensation and prognostic impact. 37 To determine a patient’s overall comorbidity score, individual diseases or comorbid conditions are classified, and the highest-ranked single ailment is used as the basis. In cases where 2 or more moderate ailments are present in different organ systems or disease groupings, the overall comorbidity score is designated as severe (grade 3).37,38 For user convenience, a user-friendly comorbidity-coding program designed by cancer registrars and health care professionals at Washington University is accessible on this website: http://cancercomorbidity.wustl.edu/web_courses_support/ComorbidityCoding/. 37
ACE-27 index is more fine-tuned than Charlson Index because it incorporates the concept of “decompensation” which assess the relative severity of the patient’s condition for each of the 26 specified comorbidities. These comorbidities include (1) myocardial infarction; (2) angina/coronary artery disease; (3) congestive heart failure; (4) arrhythmias; (5) hypertension; (6) venous disease; (7) peripheral arterial disease; (8) respiratory system disease; (9) hepatic disease; (10); stomach/intestinal disease; (11) pancreatic disease; (12) end-stage renal disease; (13) diabetes mellitus; (14) stroke; (15) dementia; (16) paralysis; (17) neuromuscular disease; (18) psychiatric disorders; (19) rheumatologic disease; (20) AIDS; (21) solid tumor including melanoma; (22) leukemia and myeloma; (23) lymphoma; (24) alcohol abuse; (25) illicit drugs; and (26) obesity.
Specific cancer-related covariates included AJCC stage (I, II), tumor size (<1, 1-<2, 2-<3, ⩾3 cm), tumor grade (I-well, II-moderate, III/IV-poorly differentiated/undifferentiated and unknown grade), and the subtype of hormone receptors (HR) and human epidermal growth factor receptor 2 (HER2) coded as HR+/HER2+, HR+/HER2−, HR−/HER2+, and HR−/HER2−. The hormone receptors (HR) status for BC are based on the status of estrogen receptor (ER) and progesterone receptor (PR); they were classified as HR+ (ER+ and/or PR+) or HR− (ER− and PR−). 39 The borderline ER or PR is grouped with ER+ or PR+. However, for HER2, the positive includes those women who tested positive at least from one of the fluorescence in situ hybridization (FISH) or immunohistochemistry (IHC) test, and the negative includes those women who tested negative in both tests or at least tested negative in one test but unknown/test not done for another test. Treatment-related covariates included chemotherapy (yes, no), and hormone therapy (yes, no).
Statistical analysis
We performed descriptive statistics on patients and tumor characteristics and conducted a chi-square test to assess the associate between covariate and surgery type. We used the Kaplan-Meier method and log-rank test for the survival curves. We used Cox proportional hazard regression model to assess the differences in OS and BCSS among the women who received mastectomy versus BCT. Hazard ratios (HRs) and 95% confidence interval (CI) were calculated for both OS and BCSS through Cox regression analysis. We tested the proportionality hazard (PH) assumption using the method proportionality test and cumulative martingale residuals (supremum test). Multicollinearity of the covariates was detected using the variance inflation factors (VIF), and VIF > 5 indicates multicollinearity. An alpha level of .05 was used to determine statistical significance. All data analyses were performed using SAS 9.4 and R 4.2.0.
For sensitivity analysis, we used the optimal caliper propensity score (PS) matching method 40 to effectively address selection bias and addresses residual confounding. To create the PS, we considered baseline covariates, including race, age at diagnosis, marital status, insurance, diagnosis state, urban rural residence, and stage at diagnosis. These variables were selected due to their minimal variation between patients who underwent BCT and mastectomy, mitigating concerns of over-adjustment concerns. During the PS matching process, we set the caliper at 0.001 for nearest neighbor matching without replacement. Following matching, our sample size was reduced to 2900, ensuring a well-balanced comparison between the BCT and mastectomy.
Results
Of 3495 women diagnosed with early-stage (I-II) BC, approximately 41.5% underwent a mastectomy, while 58.5% opted for BCT (Table 1). The majority of patients were Non-Hispanic White (NHWs) (59.2%), aged above 60 years (45.8%), married (57.4%), privately insured (62.1%), urban residents (53.3%), and overweight/obese (53.4%). More than four-fifths of patients had either none or mild comorbidities (86.7%). HR+/HER2− BC accounted for 37.8% of the cases, while triple-negative breast cancer (TNBC) represented for 11.4%. Among all deaths (24.4% of total), 11.4% were attributable to BC, and 13.0% to non-BC deaths. There were no differences in receipt of BCT or mastectomy based on race (P = .14) and chemotherapy (P = .18) (Table 1).
Early-stage (I-II) breast cancer patients and tumor characteristics by surgical approach, CDC PoC 2004 data.
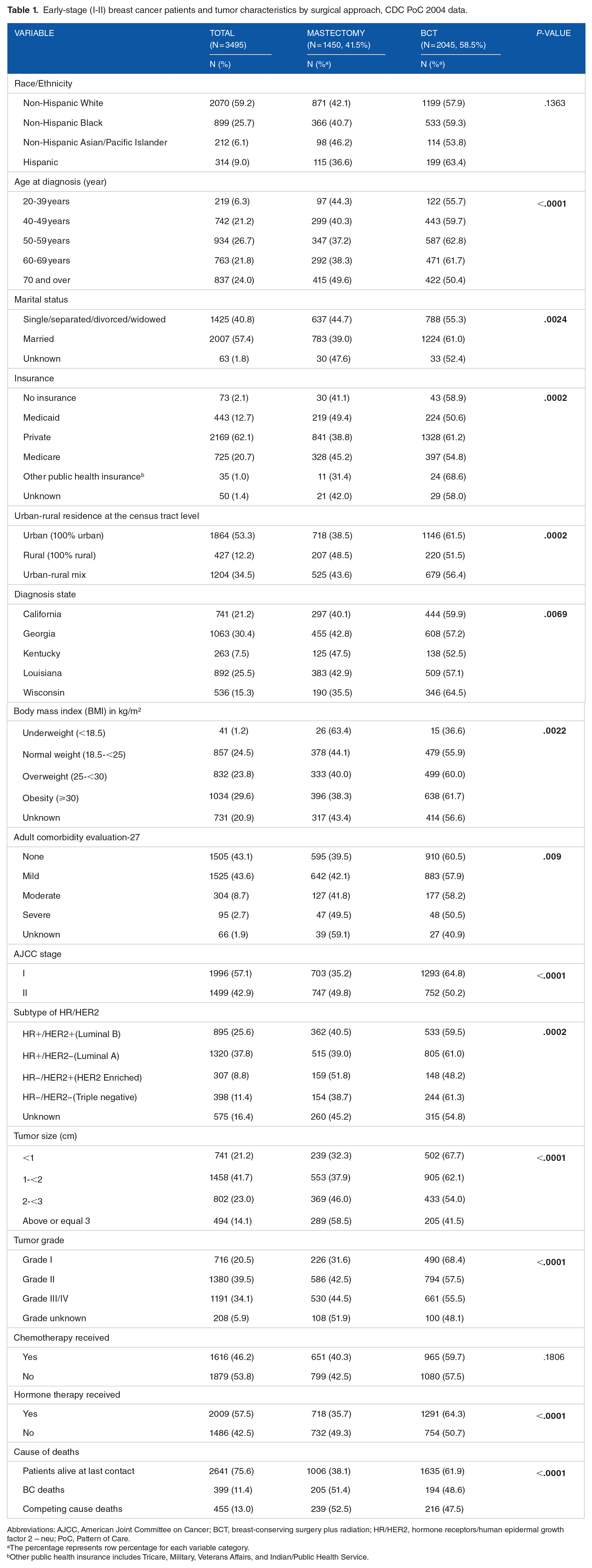
Abbreviations: AJCC, American Joint Committee on Cancer; BCT, breast-conserving surgery plus radiation; HR/HER2, hormone receptors/human epidermal growth factor 2 – neu; PoC, Pattern of Care.
The percentage represents row percentage for each variable category.
Other public health insurance includes Tricare, Military, Veterans Affairs, and Indian/Public Health Service.
10-year OS and BCSS
The median follow-up time for the entire cohort was 137 months. The proportionality assumption was met for surgery in both OS (P = .48) and BCSS (P = .81). Figure 1A and B demonstrate the differences in OS (P < .0001) and BCSS curves (P < .0001) between BCT and mastectomy. The 10-year OS was 82.7% (95% CI = [81.0%, 84.3%]) for BCT recipients, which was statistically significantly higher than that for mastectomy, 72.3% (95% CI = [69.9%, 74.6%]). Similarly, the 10-year BCSS was also statistically significantly higher for BCT recipients with a rate of 91.1% (95% CI = [89.7%, 92.3%]) compared with 85.7% (95% CI = [83.7%, 87.5%]) for mastectomy recipients (Table 2). Moreover, the 10-year OS for BCT recipients was higher across all sub-categories of variables compared with mastectomy recipients. For instances, among Non-Hispanic Black (NHB) women, the 10-year OS was 79.6% (95% CI = [75.9%, 82.8%]) for BCT and 68.3% (95% CI = [63.3%, 72.9%]) for mastectomy (MST). For women aged 70 and above at diagnosis, the 10-year OS was 61.4% [56.6%, 65.9%] for BCT and 47.0% (42.1%, 51.7%) for mastectomy. Furthermore, among women with mild comorbidities, the 10-year OS was 80.9% [78.1%, 83.3%] for BCT and 67.5% (63.7%, 71.0%) for MST. In addition, for women with AJCC stage II, the 10-year OS was 79.2% (76.1%, 81.9%) for BCT and 67.6% [64.1%, 70.8%] for MST.

Overall survival (A) and breast cancer-specific survival (B) curves for all patients with early-stage (I-II) breast cancer, CDC PoC 2004 data.
10-year overall survival (OS) and 10-year breast cancer-specific survival (BCSS) by surgical approach, CDC PoC 2004 data.

Abbreviations: AJCC, American Joint Committee on Cancer; BCT, breast-conserving surgery plus radiation; HR/HER2, hormone receptors/human epidermal growth factor 2 – neu; PoC, Pattern of Care.
Other public health insurance includes Tricare, Military, Veterans Affairs, and Indian/Public Health Service.
Factors associated with OS and BCSS for women with stage I-II BC
There was no severe (VIF < 5) multicollinearity among the covariates. In the unadjusted model, patients who underwent mastectomy were 66% (HR = 1.66; 95% CI = [1.45, 1.90]) more likely to die from overall causes compared with those who received BCT. After adjusting for sociodemographic and clinical covariates listed in Table 3, patients receiving a mastectomy were 22% more likely to die of overall causes compared with patients who received BCT (HR 1.22; 95% CI = [1.06, 1.41]). In addition, other factors significantly associated with the worse OS were older age groups (70 years and older), being single/separated/divorced/widowed, being underweight, having comorbidities, larger tumor size (⩾1 cm), higher tumor grade III/IV, and not receiving of chemotherapy and hormone therapy.
Factors associated with overall survival (OS), CDC PoC 2004 data.

Abbreviations: AJCC, American Joint Committee on Cancer; BCT, breast-conserving surgery plus radiation; HR, Hazard ratios; HR/HER2, hormone receptors/human epidermal growth factor 2 – neu; PoC, Pattern of Care.
Other public health insurance includes Tricare, Military, Veterans Affairs, and Indian/Public Health Service.
Similarly, for BC-specific deaths, in the unadjusted model, patients who underwent mastectomy were 61% more likely to die of BC compared with patients who received BCT (HR = 1.61; 95% CI = [1.33, 1.96]) (Table 4). After adjusting for all covariates listed in Table 4, patients receiving a mastectomy were 26% more likely to die of BC compared with patients who received BCT (HR = 1.26; 95% CI = [1.02, 1.55]). Other factors significantly associated with the worse BCSS included comorbidities, higher stage (stage II), larger tumor size (⩾1 cm), and higher tumor grade III/IV (Table 4).
Factors associated with breast cancer-specific survival (BCSS), CDC PoC 2004 data.

Abbreviations: AJCC, American Joint Committee on Cancer; BCT, breast-conserving surgery plus radiation; HR, Hazard ratios; HR/HER2, hormone receptors/human epidermal growth factor 2 – neu; PoC, Pattern of Care.
Other public health insurance includes Tricare, Military, Veterans Affairs, and Indian/Public Health Service.
In our sensitivity analysis, after PS matching, the study population consisted of 2900 BC women, with 1450 received mastectomy and 1450 received BCT. There were no significant differences in the distribution of matched covariates between BCT and mastectomy (Supplementary Table 1). In the adjusted optimal caliper PS matching model, patients receiving a mastectomy were 18% more likely to die of overall causes compared with those who received BCT (HR = 1.18; 95% CI = [1.01, 1.36]; P = .03) (Supplementary Table 2). Similarly, Patients receiving a mastectomy were 18% more likely to die of BC compared with patients who received BCT, although this difference did not reach statistical significance (HR = 1.18; 95% CI = [0.96, 1.47]; P = .11) (Supplementary Table 2).
In addition, in the subgroup analysis based on HR/HER2 status, the differences in OS and BCSS between mastectomy and BCT recipients were more pronounced for the HR+/HER2+ BC compared with other subtypes of HR/HER2 (Table 5). In the unadjusted model, women with HR+/HER2+ BC who underwent mastectomy had a 91% higher risk of overall deaths (HR = 1.91; 95% CI = [1.46, 2.50]) and 116% higher risk of BC-specific deaths (HR = 2.16; 95% CI = [1.46, 3.20]) compared with those who received BCT. After adjusting for other covariates, the risk of overall and BC-specific deaths was reduced to 53% (HR = 1.53; 95% CI = [1.15, 2.04]) and 78% (HR = 1.78, 95% CI = [1.18, 2.71]), respectively. In the case of triple-negative BC (HR−/HER2−), the hazard risk for overall deaths was significantly higher in the unadjusted model (HR = 1.61; 95% CI = [1.13, 2.30]) but not for BC-specific deaths (HR = 1.42; 95% CI = [0.91, 2.22]). However, after adjusting for other covariates, the hazard risk was reduced to 26% (HR = 1.26; 95% CI = [0.83, 1.92]) for overall causes of death and 12% (HR = 1.12; 95% CI = [0.67, 1.88]) for BC-specific deaths. No significant differences were observed in OS and BCSS among patients in the HR-/HER2+ group between mastectomy and BCT recipients (Table 5).
Association of surgery received (mastectomy vs breast-conserving therapy) with overall survival and breast cancer-specific survival based on subtype of HR/HER2 status.
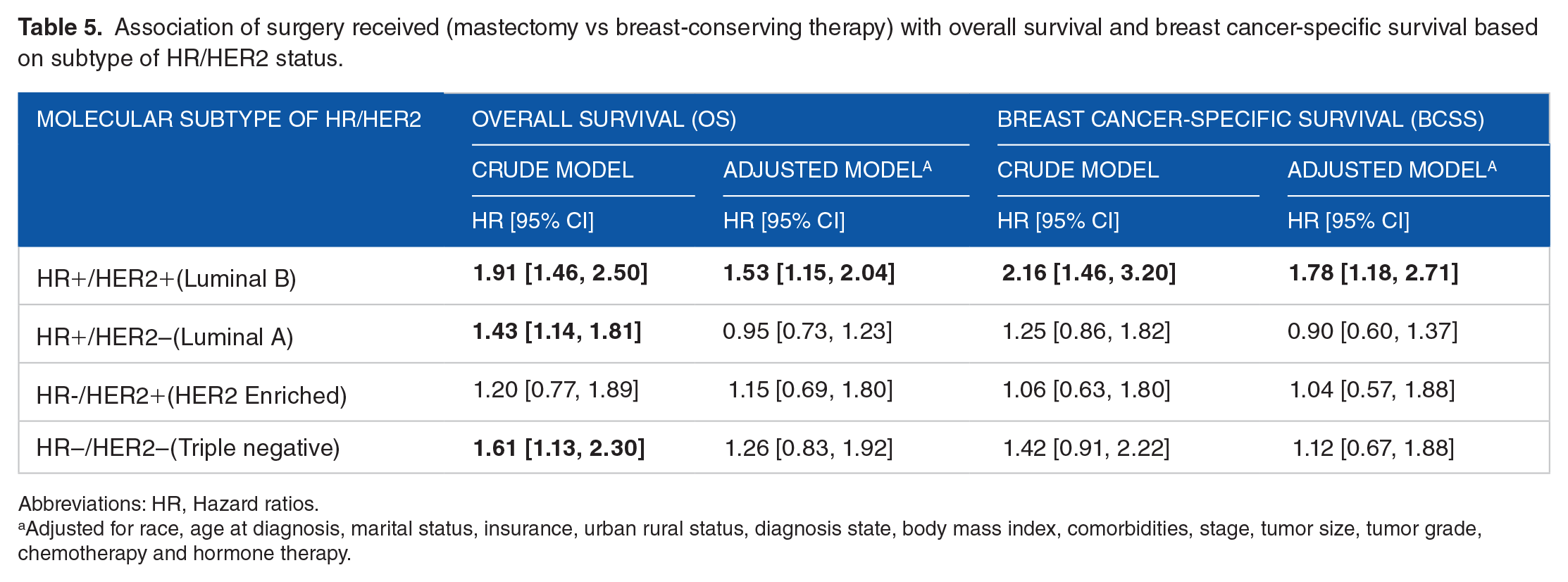
Abbreviations: HR, Hazard ratios.
Adjusted for race, age at diagnosis, marital status, insurance, urban rural status, diagnosis state, body mass index, comorbidities, stage, tumor size, tumor grade, chemotherapy and hormone therapy.
Furthermore, in our histological subgroup analysis, we found invasive ductal carcinoma cases who received mastectomy had a 31% higher risk of overall deaths (HR = 1.31, 95% CI = [1.11, 1.55]) and a 38% higher risk of BC-specific deaths (HR = 1.38, 95% CI = [1.09, 1.76]) compared with their counterparts who received BCT in the adjusted models. Conversely, among women with invasive lobular carcinoma, there was a tendency toward a higher risk of overall deaths (HR = 1.28, 95% CI = [0.63, 2.61]) and BC-specific causes (HR = 1.70, 95% CI = [0.51, 5.65]), although these associations did not reach statistical significance. Detailed results can be found in Supplementary Table 3.
Discussion
The significant finding from our study is that the 10-year survival is significantly higher for ESBC women (stage I-II) who received BCT compared with those who underwent mastectomy, for both -all cause of deaths and BC-specific deaths. The differences persist, even after adjusting for sociodemographic factors, comorbidity severity, adjuvant treatment, and molecular subtype of HR/HER2. Our findings provide robust support to conclusions drawn in a prior study 16 involving ESBC patients in Louisiana, which similarly observed a significantly higher risk of deaths from all-causes and BC among women who underwent mastectomy compared with BCT.
Moreover, our results are consistent with SEER study findings,41,42 and other European studies,13,14,30,43 where patient’s populations might be less diverse compared with those in the US. In contrast, an Italian study for ESBC cases diagnosed between 2001 and 2013 19 did not find a significant difference in overall deaths between the mastectomy and BCT recipients. However, this discrepancy might be attributed to the relatively short 5 year follow-up duration and a lack of comorbidities information in the Italian study. 19 Therefore, the superior outcomes observed in patients undergoing BCT in recent years may be related to advancements in breast imaging, widespread adoption of 3-dimensional treatment planning, and a reduction in the extent of surgery.5,44
In addition, our study underscores a significant improvement in 10-year BCSS associated with BCT, with an absolute difference of 5.4% (91.1% BCT vs 85.7% mastectomy). This improvement in BCSS is slightly higher than the findings of a US-based study that used the SEER database, which reported an absolute difference of 4% (94% BCT vs 90% mastectomy). 17
Furthermore, our findings are supported by other studies that conducted specific group analyses and recommend evaluating surgery options, considering BCS whenever feasible. For instance, a systematic review by Christiansen et al 45 showed that BCS is associated with improved survival compared with mastectomy in early BC (T1-2N0-1M0). In another study by Sagona et al, 46 it was found that the BCS resulted in significantly higher overall survival rates at 3, 5, and 10 years compared with mastectomy for ipsilateral BC recurrence, although there was no difference in disease- free survival rates. In addition, a clinical trial study (ACOSOG Z11102) 47 reported that women with multiple ipsilateral breast cancer (MIBC) who underwent BCT had a 5 year estimated local recurrence rate of 3.1%, which is below clinically acceptable threshold of 8%, further supporting BCT as a reasonable surgical option for these BC patients.
In our subgroup analysis, we observed that women with HR+/HER2+ (luminal B) BC who underwent mastectomy had a significantly higher risk of overall cause of deaths and BC-specific deaths compared with those who received BCT, even after adjusting for other covariates. However, for triple-negative BC (HR-/HER2-), the hazard risk for overall causes of death initially showed a higher risk in the unadjusted model but did not remain statistically significant after accounting for other covariates. Furthermore, Kunkler et al 48 highlighted the significance of radiotherapy following BCS, as the omission of radiotherapy increased the local recurrence of BC among women aged 65 years and older with early HR-positive BC. Another SEER study 42 conducted among young patients (aged ⩽ 40 years) with a median follow-up 30 months reported that BCT resulted in significantly improved prognosis for patients with luminal and triple-negative subtypes. However, no significant difference was observed among patients with the HER-2 enriched subtype, which is consistent with our findings to some extent. These discrepancies in the results regarding triple-negative subtypes results may be attributed to longer follow-up periods and adjustments for comorbidities carried out in our study.
Despite the clear evidence of BCT offering better survival outcomes, a nationwide US study focusing on ESBC cases diagnosed between 1998 and 2011 from the National Cancer Data Base revealed a concerning trend of increasing proportion of BCS-eligible patients opting mastectomy, particularly among women with node-negative and in-situ disease. 49 Several factors contribute to this trend, including women’s preferences for mastectomy due to perceived peace of mind, 50 and logistical challenges such as transportation issues 51 especially affecting older women accessing radiation therapy. 52 In addition, factors such as fear of cancer recurrence and belief that mastectomy is a more aggressive treatment, 5 desire for control over cancer care decisions, 53 and surgeon preferences all play significant roles. 54 Surgeons with higher case volumes tend to proactively recommend BCS54,55 but face challenges when patients express a preference for mastectomy, 55 potentially due to lack of knowledge about BC, 50 and these factors. Furthermore, underutilization of BCT following neoadjuvant chemotherapy can be attributed to various clinical, socioeconomic and psychological factors. 56 To address these issues, interdisciplinary teams should collaborate to evaluate the patient journey, facilitate informed treatment counseling, and support shared decision-making. 56 Targeted financial programs can also help reduce socioeconomic disparities and improve access to BCT services.
In addition, some women expressing a preference for mastectomy may not align their treatment goals with recommended approaches, and in many cases, health care professionals might not even inquire about patient preferences. This underscores the need for improvement in the decision-making quality in BC treatment selection. 50 Therefore, it is crucial that patients are well informed 43 about their treatment options, including the pertinent evidence related to OS and BCSS. This knowledge empowers them to make informed decisions when opting for mastectomy over BCT, provided that BCT is a viable option for them. This informed decision-making process is essential to ensure that patients receive the most appropriate and effective treatment for their specific circumstances.
The strengths of our study lie in its larger geographic scope, being a population-based study utilizing data from five state registries of CDC PoC study, which included complete information on the chemotherapy, hormone therapy, and HER2 status, aspects that were lacking in prior population-based studies. In addition, we had 90% follow-up rate for alive cases within 10 years. Furthermore, we employed the ACE-27 tool, which considers the severity of comorbidities 57 providing better control for the influence of comorbidities on the treatment received and survival. Our sensitivity analysis using optimal caliper PS matching adds an additional layer of control for potential confounding factors, ensuring a more balanced distribution of covariates among treatment groups. The subgroup analysis of hormone receptors and pathological type of BC provided more insights on the survival by surgery choices.
Nevertheless, we acknowledge several limitations of this study, including a small number of loss to follow-up cases, and lack information on lifestyle factors such as smoking that might be more prevalent in the deprived socioeconomic group 58 that could negatively affect systemic therapy treatment and survival. Furthermore, we lack information on BC recurrence and corresponding treatment. Another limitation is the exclusion of cases with unknown chemotherapy (n = 46), which could possibly introduce selection bias into the analysis. In addition, we cannot ignore the limitation of losing samples using the optimal caliper propensity score matching.
Overall, our study emphasizes the importance of considering BCT over mastectomy for eligible patients to improve survival outcomes. This recommendation is particularly significant for women with severe comorbidities, higher tumor size, and tumor grade. In addition, our findings suggest that BCT may be a preferred treatment modality in specific patient populations, such as women with HR+/HER2+ BC or invasive ductal carcinoma BC, as these groups showed elevated risks after undergoing mastectomy. The model discussed in our study provides evidence-based information on survival outcomes and risk factors associated with various treatment options. Clinicians can consider incorporating this information to personalize treatment plans based on patients’ characteristics, including cancer subtype, severity of comorbidities, and tumor histology such as invasive ductal carcinoma. However, it is important to note that relying solely on our retrospective study for clinical treatment decisions should be avoided. Additional research is necessary to further validate these findings and explore additional pathological types beyond hormone receptor subtypes, invasive ductal, or lobular carcinoma.
Tailoring treatment decisions based on patient characteristics, and BC pathological types is crucial in clinical practice when discussing treatment options with patients. Implementing these findings requires interdisciplinary collaboration, guidelines development, education campaigns, and addressing patient preferences through informed decision-making. This approach can ultimately improve patient outcomes and ensure treatment choices align with evidence-based practices. Future research should focus on understanding the factors that influence patient preferences for mastectomy. This knowledge can help health care providers better support patients in making informed decisions and provide appropriate counseling to maximize the potential benefits of BCT.
Conclusion
The improved OS and BCSS are evident for ESBC women who underwent BCT compared with those who received mastectomy, even after adjusting sociodemographic factors, adjuvant treatment, HR/HER2 status, and comorbid severity. Thus, our study underscores the importance of encouraging ESBC women to consider a BCT whenever it is a viable option. Shared decision-making between healthcare providers and patients can lead to more personalized and favorable treatment choices, ultimately improving patient outcomes.
Supplemental Material
sj-docx-1-bcb-10.1177_11782234241273666 – Supplemental material for Higher 10-Year Survival with Breast-Conserving Therapy over Mastectomy for Women with Early-Stage (I-II) Breast Cancer: Analysis of the CDC Patterns of Care Data Base
Supplemental material, sj-docx-1-bcb-10.1177_11782234241273666 for Higher 10-Year Survival with Breast-Conserving Therapy over Mastectomy for Women with Early-Stage (I-II) Breast Cancer: Analysis of the CDC Patterns of Care Data Base by Pratibha Shrestha, Mei-Chin Hsieh, Tekeda Ferguson, Edward S Peters, Edward Trapido, Qingzhao Yu, Quyen D Chu and Xiao-Cheng Wu in Breast Cancer: Basic and Clinical Research
Footnotes
Acknowledgements
The data used for this publication were collected by the Centers for Disease Control and Prevention’s (CDC) National Program of Cancer Registries (NPCR) Patterns of Care Study for Female Breast and Prostate (BP) Cancers, which was funded by CDC through cooperative agreements with the participating state cancer registries. This manuscript is written on behalf of the Patterns of Care Study BP Group. The findings and conclusions in this report are those of the authors and do not necessarily represent the official position of CDC. The authors thank Trevor Thompson for his contribution on sample design, the creation of analytic data files, and sample SAS programs.
We thank the Centers for Disease Control and Prevention (CDC) and Louisiana Tumor Registry (LTR) for providing cancer registry data and continuous support for the successful completion of this study. We also like to show our extreme gratitude to the National Cancer Institute—Surveillance Epidemiology and End Results Program and CDC-National Program of Cancer Registries for funding the LTR
Declarations
Supplemental material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
