Abstract
Early recurrence is common after curative hepatectomy for hepatocellular carcinoma and is associated with poor prognosis. This study aimed to identify risk factors of early recurrence after curative hepatectomy in hepatocellular carcinoma. Overall, 63 patients who underwent curative hepatectomy for hepatocellular carcinoma were enrolled. Patients were divided into the early recurrence group, who developed recurrence within 12 months after hepatectomy (n = 10), and the non-early recurrence group (n = 53). Clinicopathological factors of early recurrence were retrospectively analyzed. Among the 63 patients, 10 (15.9%) patients experienced early recurrence. Univariate analysis showed tumor necrosis (p = 0.012), level of PIVKA-II (prothrombin induced by vitamin K absence or antagonist-II; p = 0.002), and microvascular invasion (p = 0.029) to be associated with early recurrence. By multivariate analysis, there were significant differences in high PIVKA-II (p < 0.001) and tumor necrosis (p = 0.012) in patients with early recurrence. The optimal cutoff values of PIVKA-II and tumor necrosis were 46 mAU/mL and 3% of total tumor volume, respectively. Patients with a high preoperative PIVKA-II level and extent of tumor necrosis, which are independent risk factors for early recurrence, should be actively treated and monitored closely after hepatectomy.
Keywords
Introduction
Hepatocellular carcinoma (HCC) is the fifth most common cancer and the third leading cause of cancer mortality worldwide. 1 Although many treatment modality such as transarterial chemoembolization (TACE), radiofrequency ablation (RFA), percutaneous ethanol injection (PEIT), and radiation treatment are available for HCC, hepatic resection is still regarded as the first-line treatment option for curative treatment for HCC, together with liver transplantation, particularly in early HCC. 2
Despite recent improvements in overall survival after hepatic resection, long-term prognosis after hepatic resection is unsatisfactory, because of the high recurrence rate. In particular, the high early recurrence (ER) rate is a major obstacle in HCC patients who have undergone resection. 3 Therefore, numerous studies have been performed to identify important predictive factors for ER after curative hepatic resection. The proposed risk factors for ER are classified into three categories: (1) tumor factors such as tumor size, number, Edmondson–Steiner (ES) grade, and preoperative tumor markers; (2) host factors such as grade of hepatitis activity, presence of cirrhosis, and hepatitis viral load; and (3) surgical factors such as surgical margin and extent of liver resection. In this retrospective study, we aim to identify risk factors of ER after curative hepatectomy in HCC.
Materials and methods
Patients
A total 63 patients who underwent curative hepatectomy for HCC at Pusan National University Yangsan Hospital between June 2013 and April 2015 were included in this study. Curative resection was defined as resection with a tumor-free margin and no residual tumors in the remnant liver, and ER was defined as recurrence within 12 months after hepatectomy. The medical records of all patients were reviewed retrospectively, and data for demographics, laboratory results including tumor markers, tumor pathology, and recurrence were collected for each patient.
Patients follow-up
After surgery, patients were followed in the outpatient clinic every 3 months for the first 2 years and every 6 months thereafter. We evaluated recurrence on the basis of laboratory measurements including alpha-fetoprotein (AFP) and prothrombin induced by vitamin K absence or antagonist-II (PIVKA-II) and dynamic computed tomography or magnetic resonance imaging at each visit. Recurrence was defined as the appearance of a new lesion compatible with HCC on radiologic examination during the follow-up period.
Clinicopathological analysis
Patient demographics and laboratory test findings were reviewed, including age, gender, hepatitis status, tumor markers (AFP and PIVKA-II), indocyanine green retention at 15 min (ICG R15), liver stiffness (LS) value, and neutrophil-to-lymphocyte ratio (NLR). The AFP changes were classified into two groups based on the change in the AFP from the preoperative level to 3 months after hepatectomy (absent: AFP decreased <50%; present: AFP decreased ≥50%).The NLR was defined as the absolute neutrophil count divided by the absolute lymphocyte count. Histopathological findings of the HCC such as tumor size, tumor number, tumor differentiation, micro- or macrovascular invasion, intrahepatic metastasis, extent of tumor necrosis, and non-tumor liver pathology were reviewed. Histologic tumor differentiation was determined according to the criteria of ES. Grades I and II were considered low-grade HCC, and grades III and IV were considered as high-grade.
Statistical analysis
Continuous variables were expressed as mean (± SD) or median (range). The chi-square test was used to compare categorical variables. Independent prognostic factors affecting ER were identified using univariate analysis, and stepwise multivariate regression was performed using a Cox proportional hazard model to evaluate the prognostic factors predicting ER. Receiver operating characteristic (ROC) curve analysis was used to define the optimal cutoff values of PIVKA-II and tumor necrosis, which were defined by maximizing the sum of sensitivity and specificity. P values of less than 0.05 were considered to be statistically significant.
Results
Demographics and clinicopathological features
The patient’s demographics and clinicopathological features are summarized in Table 1. Among the 63 patients, 49 were male ( 77.8%) and 14 were female (22.2%). The mean patient age was 57.5 years. Overall, 49 patients (77.8%) tested positive for hepatitis B virus (HBV), while 5 (7.9%) tested positive for hepatitis C virus (HCV). The mean preoperative AFP and PIVKA-II were 634.4 ng/mL and 1003.3 mAU/mL, respectively. The median LS value was 8.6 kPa and the mean ICG R15 was 11.6%. In total, 55 patients (87.3%) had a single tumor, and mean tumor size was 3.2 cm. Macrovascular invasion was identified in 2 patients (3.2%), and microscopic vascular invasion was identified in 11 patients (17.5%). According to ES tumor differentiation grade, 47 patients (74.6%) had grade I–II and 16 (25.4%) had grade III–IV. Tumor necrosis was identified in 18 patients (28.6%).
Baseline clinicopathological characteristics.
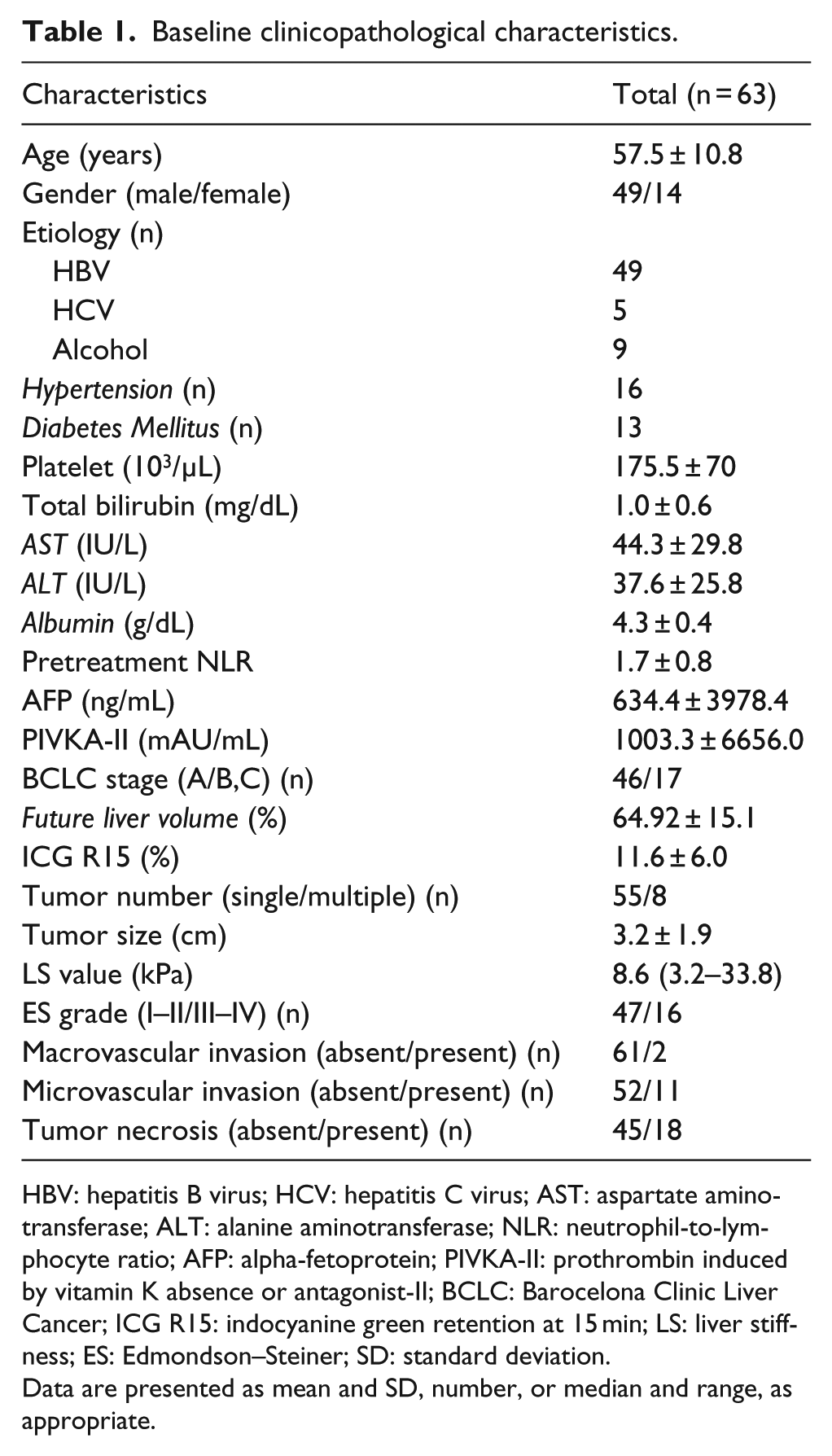
HBV: hepatitis B virus; HCV: hepatitis C virus; AST: aspartate aminotransferase; ALT: alanine aminotransferase; NLR: neutrophil-to-lymphocyte ratio; AFP: alpha-fetoprotein; PIVKA-II: prothrombin induced by vitamin K absence or antagonist-II; BCLC: Barocelona Clinic Liver Cancer; ICG R15: indocyanine green retention at 15 min; LS: liver stiffness; ES: Edmondson–Steiner; SD: standard deviation.
Data are presented as mean and SD, number, or median and range, as appropriate.
Risk factors for ER HCC after hepatectomy
Univariate analysis to identify prognostic factors affecting ER showed that PIVKA-II (p = 0.002), microvascular invasion (p = 0.029), and tumor necrosis (p = 0.012) were significant risk factors. After these variables were included in multivariate Cox proportional hazard model analysis, PIVKA-II (p < 0.001) and tumor necrosis (p = 0.012) were found to be independently predictive for ER (Table 2).
Risk factors for early recurrence (⩽12 months) after curative hepatectomy in HCC patients.
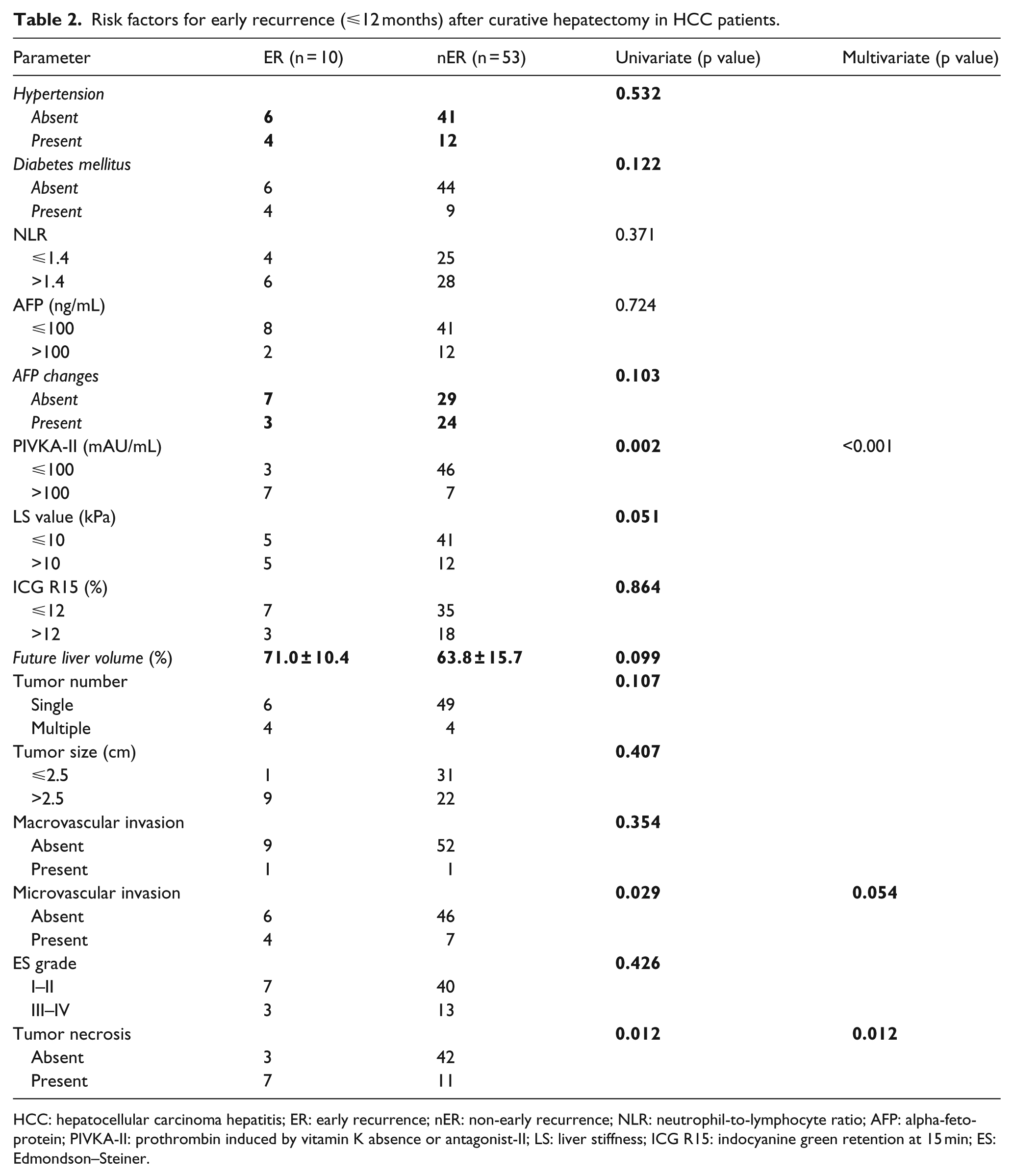
HCC: hepatocellular carcinoma hepatitis; ER: early recurrence; nER: non-early recurrence; NLR: neutrophil-to-lymphocyte ratio; AFP: alpha-fetoprotein; PIVKA-II: prothrombin induced by vitamin K absence or antagonist-II; LS: liver stiffness; ICG R15: indocyanine green retention at 15 min; ES: Edmondson–Steiner.
Determination of the optimal cutoff value of PIVKA-II and tumor necrosis
ROC curves were used to identify the PIVKA-II value and extent of tumor necrosis with highest predictive power to discriminate patients with and without recurrence. According to the ROC curve, the optimal cutoff value of PIVKA-II was 46 mAU/mL (area under the curve (AUC) = 0.804, 95% confidence interval (CI) = 0.684–0.893, p = 0.0003). The optimal cutoff value of tumor necrosis was 3.0% (AUC = 0.769, 95% CI = 0.646–0.866, p = 0.0029; Figure 1).
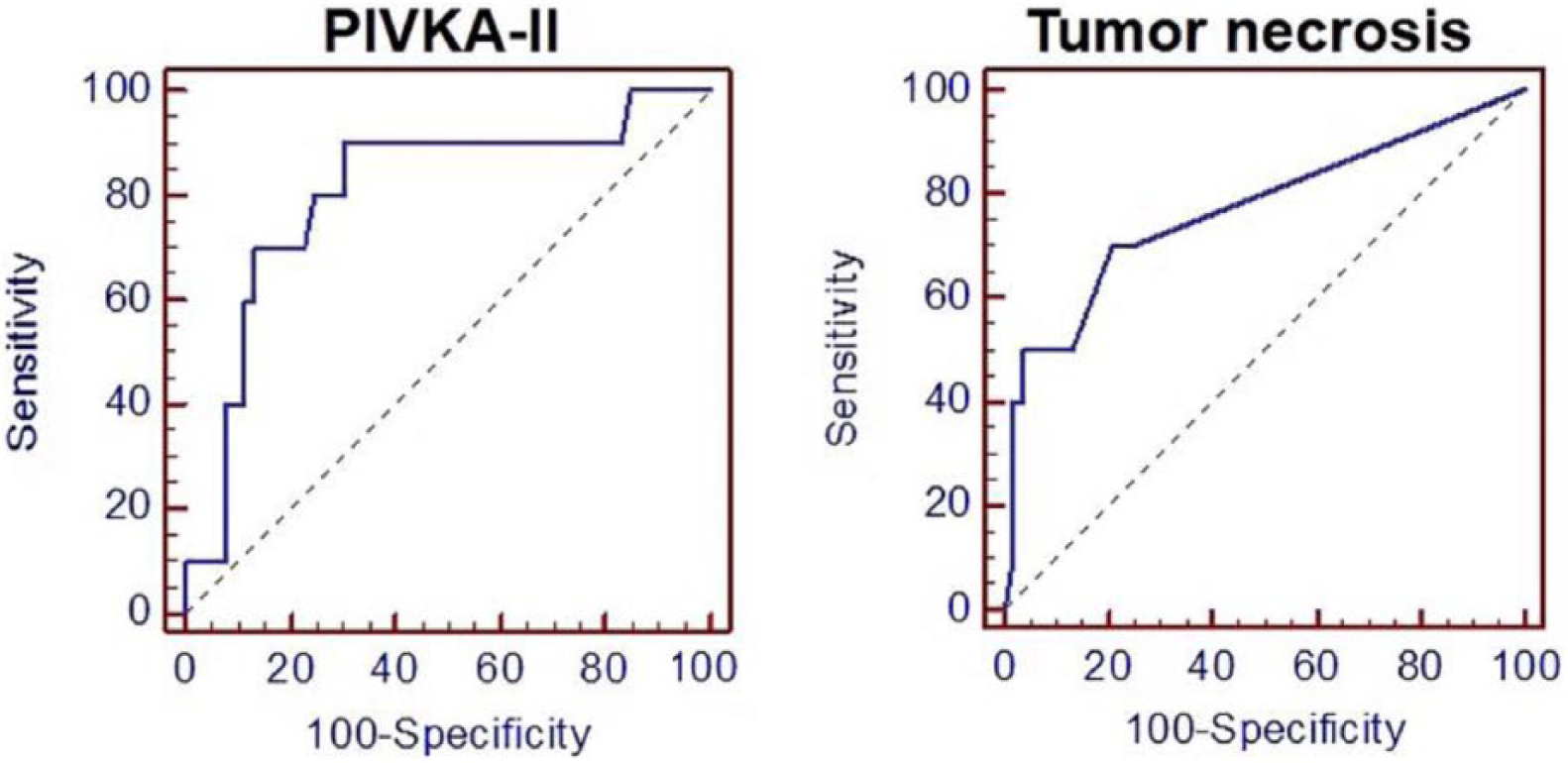
The ROC curves of PIVKA-II and tumor necrosis for predicting early recurrence after curative hepatectomy.
Correlation between clinicopathological factors and PIVKA-II/tumor necrosis
Clinicopathological factors of patients with different PIVKA-II level and tumor necrosis status are summarized in Table 3 and Table 4, respectively. Significant differences in tumor size (p = 0.002) and tumor necrosis status (p = 0.030) were observed between the groups with low and high PIVKA-II levels. Significant differences in LS value (p = 0.016), tumor number (p = 0.005), and tumor size (p = 0.024) were observed depending on tumor necrosis status. No significant differences were found in other clinicopathological variables (p > 0.05 for all).
Comparisons of clinicopathological characteristics of HCC patients with PIVKA-II.

Comparisons of clinicopathological characteristics of HCC patients with tumor necrosis.
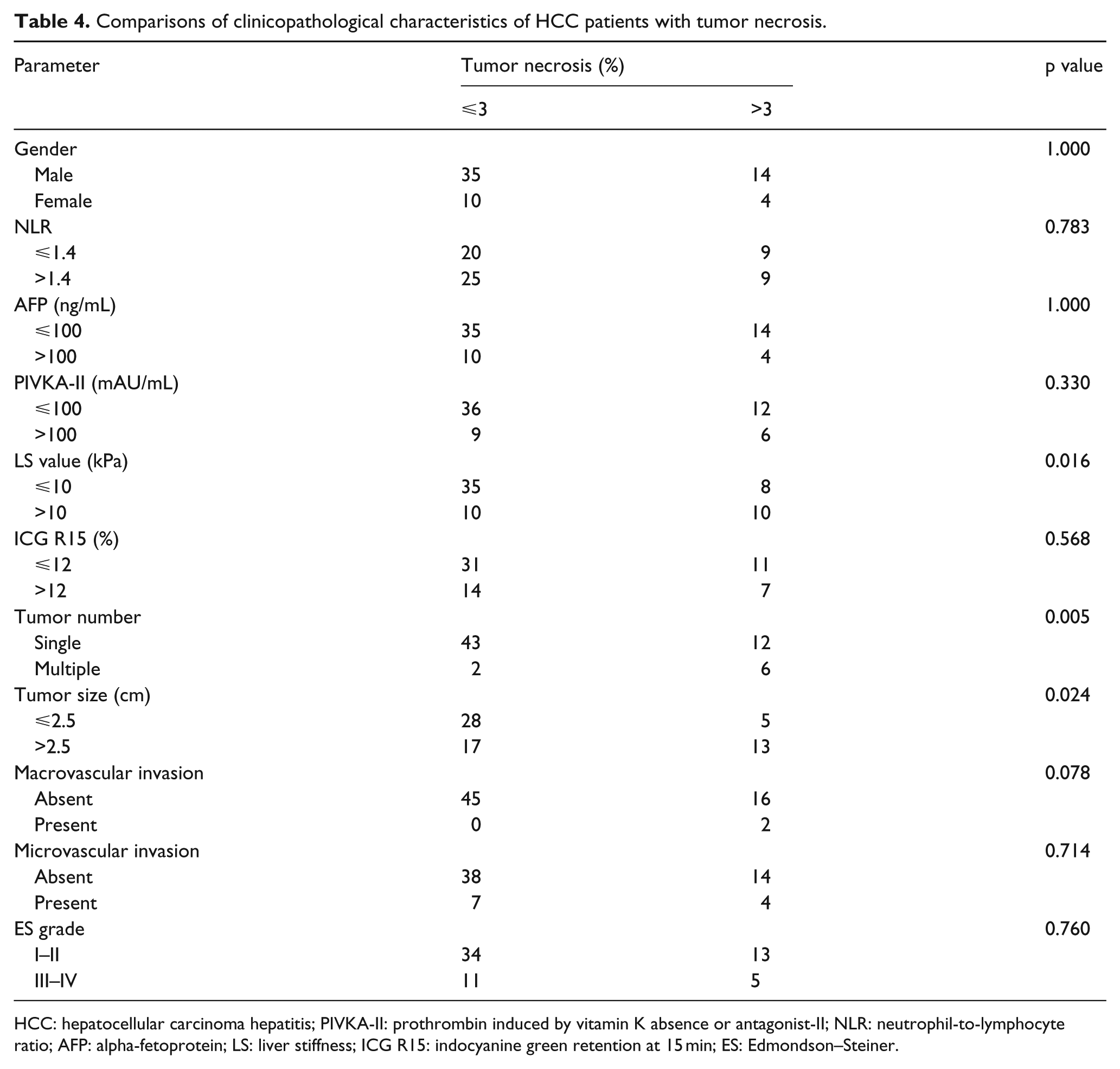
HCC: hepatocellular carcinoma hepatitis; PIVKA-II: prothrombin induced by vitamin K absence or antagonist-II; NLR: neutrophil-to-lymphocyte ratio; AFP: alpha-fetoprotein; LS: liver stiffness; ICG R15: indocyanine green retention at 15 min; ES: Edmondson–Steiner.
Discussion
Although surgical resection of HCC is a safe procedure with low mortality rates and represents a potentially curative treatment to HCC, the high recurrence rate after hepatectomy results in a poor prognosis.4–6 Therefore, understanding and predicting recurrence is important to improve the prognosis of HCC. HCC may recur through different mechanisms: it can originate from intrahepatic metastasis of primary tumor or can be de novo tumor occurring in the remnant liver. 6 The pattern of HCC recurrence could be categorized into early and late. In other studies, the period from 6 months to 2 years after hepatectomy was defined as the ER period.7–12 However, in this study, we defined ER as within 12 months after hepatectomy.
Our findings showed that the rate of ER HCC after curative hepatectomy was 15.9% and the PIVKA-II level (>100 mAU/mL), microvascular invasion, and tumor necrosis were risk factors for ER. In addition, we identified the cutoff value of PIVKA-II and the extent of tumor necrosis, had the highest predictive power to discriminate patients with and without recurrence, to be 46 mAU/mL and 3.0%, respectively.
Numerous studies identified the risk factors for ER to include AFP, PIVKA-II, microvascular invasion, tumor size, tumor number, and tumor differentiation.7–12 Several recent studies have shown that various inflammation-based markers such as NLR have been associated with recurrence and overall and disease-free survival in patients with cancer, including HCC.13–16 Nonetheless, the prognostic value of these inflammation-based markers is controversial and requires further research.
Our study identified the PIVKA-II as a risk factor determining ER. PIVKA-II is an abnormal form of the coagulation protein, prothrombin induced by vitamin K absence or antagonist-II. PIVKA-II is well known as an effective serum biomarker for HCC diagnosis and prognosis. Furthermore, a recent study found a relationship between PIVKA-II production and tumor angiogenesis. 17 Previous studies have reported that high serum levels of PIVKA-II are related to portal vein invasion or intrahepatic metastasis after hepatic resection.18,19 In addition, there is now a large body of evidence indicating that PIVKA-II has a p prognostic and predictive value for recurrence after hepatic resection.20–23
Most studies use a predefined value of PIVKA-II based on existing knowledge to evaluate risk factors for ER after hepatectomy. To our knowledge, only one study has reported the role the cutoff value of PIVKA-II determined using ROC curve analysis in predicting ER after hepatectomy. 20 In this study, we found that an area under the ROC curve of 0.804 (cutoff value = 46 mAU/mL) for PIVKA-II showed a good predictive performance regarding ER after hepatectomy. However, it remains to be confirmed whether the predictive value of the PIVKA-II level can be generalized to patients undergoing hepatectomy.
Tumor necrosis is a common histological feature of solid tumors and arises from chronic ischemic injury due to rapid tumor growth. Therefore, the extent of necrosis is considered to represent the level of tumor hypoxia, and greater hypoxia is related to metastasis and poor prognosis.24,25 In addition, tumor necrosis could result in release of a large number of proinflammatory cytokines, which stimulate tumor growth, and therefore correlate with a poor prognosis.26,27 Tumor necrosis has been reported to have prognostic value in many solid caner types, such as colon, lung, 25 breast, 28 and renal malignancies, including both renal cell carcinoma29,30 and upper urinary tract urothelial carcinoma. 24 Although several studies have evaluated tumor necrosis as a prognostic factor for recurrence or survival in HCC, these studies have not shown a significant correlation between tumor necrosis and ER.31–33 We found that tumor necrosis was an independent predictor of ER after hepatectomy. Moreover, we used ROC curve to identify the ideal cutoff value of tumor necrosis for predicting ER (3%).
LS measurement using transient elastography (TE; FibroScan) has been used to assess liver fibrosis non-invasively. Various studies reported that LS value could be used to predict the development of complication of liver cirrhosis, including HCC development and postoperative complication such as hepatic insufficiency.34–38 Furthermore, recent studies show that LS is useful for predicting late recurrence after curative resection of HCC.34,39 Although we also evaluated the correlation between the LS value and ER, we found no association probably because the mechanism underlying ER is usually intrahepatic metastasis of primary tumor. Recently, among various biomarkers, NLR has been found to be associated with recurrence and survival after treatment, including hepatectomy.14,40,41 The NLR is thought to be an inflammation marker: a high NLR reflects the imbalance in the immune response favoring neutrophils over lymphocytes and may result in week antitumor immunity and a pro-tumoral microenvironment. However, we found no relationship between NLR and ER after hepatectomy.
Our study has some limitations. First, the sample size was small, because this was a retrospective single-center study. In addition, we found that HBV is the most common cause of HCC, which is not the case in most Western countries. Second, selection bias was inevitable and may have influenced the analysis, because we selected only patients who had undergone TE. Despite these limitations, we identified the ideal cutoff value of PIVKA-II and the extent of tumor necrosis for predicting ER after hepatectomy in HCC. To our knowledge, this is the first report to do so.
In summary, our study examined prognostic factors that affect ER after hepatectomy and identified the ideal cutoff values of risk factors. Patients with high preoperative PIVKA-II and a high extent of tumor necrosis on pathological examination should be actively treated and monitored closely to prevent ER. We hope that our findings will help identify patients at risk of ER after hepatectomy and help them receive the treatment required to improve survival. Further biological studies with large cohorts are needed to validate the clinical usefulness of PIVKA-II and tumor necrosis as predictive markers for ER in HCC.
Footnotes
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
