Abstract
Hepatocellular carcinoma is the major form of primary liver cancer, which is the second and sixth leading cause of cancer-related death in men and women, respectively. Extensive research indicates that Wnt/β-catenin signaling pathway, which plays a pivotal role in growth, development, and differentiation of hepatocellular carcinoma, is one of the major signaling pathways that is dysregulated in hepatocellular carcinoma. Cyclin D1 is a proto-oncogene and is one of the major regulators of Wnt signaling pathway, and its overexpression has been detected in various types of cancers including hepatocellular carcinoma. Using several validated bioinformatic databases, we predicted that the microRNAs are capable of targeting 3′-untranslated region of Cyclin D1 messenger RNA. According to the results, miR-20a was selected as the highest ranking microRNA targeting Cyclin D1 messenger RNA. Luciferase assay was recruited to confirm bioinformatic prediction results. Cyclin D1 expression was first assessed by quantitative real-time polymerase chain reaction in HepG2 cell line. Afterward, HepG2 cells were transduced by lentiviruses containing miR-20a. Then, the expression of miR-20a and Cyclin D1 was evaluated. The results of luciferase assay demonstrated targeting of 3′-untranslated region of Cyclin D1 messenger RNA by miR-20a. Furthermore, 238-fold decline in Cyclin D1 expression was observed after lentiviral induction of miR-20a in HepG2 cells. The results highlighted a considerable effect of miRNA-20a induction on the down-regulation of Cyclin D1 gene. Our results suggest that miR-20a can be used as a novel candidate for therapeutic purposes and a biomarker for hepatocellular carcinoma diagnosis.
Introduction
Hepatocellular carcinoma (HCC), also called hepatoma, is the most common form of primary liver cancer in adults.1–3 HCC is much more common in sub-Saharan African countries and Southeast Asia than in the United States.4,5 HCC is also one of the leading cause of cancer-related deaths worldwide, accounting for more than 600,000 deaths annually with more prevalence in men. 6
Recently, researchers have considered signaling pathways in HCC as the new source of targets for novel therapeutic strategies. Wnt/β-catenin signaling pathway plays a pivotal role in growth, development, and differentiation of cells and is one of the notable signaling pathways dysregulated in cancers such as HCC.2,6–9 Cyclin D1 is one of the main proteins of Wnt/β-catenin pathway encoded by the CCND1. 10 This proto-oncogene plays a key role in the regulation of G1 to S phase progression in a variety of cell types. 11 Increased expression of CCND1 in various cancers has been reported, and it is confirmed that inhibiting CCND1 expression in cancer cells results in declined tumorigenesis, increased apoptosis, and drug sensitivity.12,13 As far as known, a principle cause of HCC is epigenetic alterations including deoxycytosine methylation, histone modification, and microRNAs (miRNAs) dysregulation. 14 Several studies have demonstrated the role of these small non-coding RNAs, 19–22 nucleotide length, in development and progression of HCC.15–19 Therefore, they have a great potential to be used as diagnostic and therapeutic targets. Different studies concerning HCC had elucidated that the expression of some miRNAs is associated with different stages of cancers and is applicable for monitoring response to therapy.
Regarding the small size of miRNAs, a variety of approaches such as deep sequencing, microarray, and computational prediction are applicable for miRNA expression analysis. Considering advances in bioinformatic algorithms and computational modeling systems, it has been facilitated to predict miRNAs targeting specific messenger RNAs (mRNAs).20,21 In comparison with the expensive instruments required for microarray and sequencing, bioinformatic software tools are more accessible and cost-effective for identification of miRNA targets. Bioinformatic algorithms mainly predict miRNA targets with high accuracy based on matching between the seed region of a miRNA and the sequence of its target(s) and thermodynamic stability of the miRNA:mRNA complex.22–24
The aim of this study was to exploit different bioinformatic prediction tools to find the highest ranking miRNAs targeting 3′-untranslated region (UTR) of Cyclin D1 mRNA. After approval of bioinformatic predictions by luciferase assay, a lentiviral vector-containing target miRNA was recruited to overexpress the miRNA and to find the effect of its induction on target gene in HCC cells, HepG2.
Materials and methods
Bioinformatic prediction of miRNAs
The nucleotide sequence of the 3′-UTR of Cyclin D1 was retrieved from NCBI database (http://www.ncbi.nlm.nih.gov). Then, we used miRNA databases such as TargetScan, DIANA, PicTar, miRanda, microcosm, and mirPath to predict miRNAs targeting 3′-UTR of Cyclin D1. In addition, miRWalk database, providing validated results in conjunction with bioinformatic predictions for genes, pathways, organs, transcription factors, and diseases, was also used. 25 After selecting miRNAs that target Cyclin D1 with the highest probability, mirZ database, which predicts both miRNA targets and compares their expression in different cell lines, 26 was exploited. Furthermore, Qiagen database, which provides a list of miRNAs in cancers, was applied. Finally, DIANA TOOLS-mirPath was used to determine the role of the predicted miRNAs in Wnt signaling pathway. DIANA TOOLS-mirPath gathers data from DIANA-microT-CDS and DIANA-TarBase (version 6.0) and incorporates these data by meta-analysis to predict miRNA targets. 27
Cloning
The sequence of Cyclin D1 and the selected miRNA were retrieved from NCBI and miRBase (www.mirbase.org), respectively. Primers for cloning these sequences were designed using AlleleID7 (PREMIER Biosoft, USA) and Gene Runner version 5.1 (Informer Technologies Inc, Spain). Subsequently, it was validated using Oligo6 software (Molecular Biology Insights, Inc, USA) and NCBI BLAST. The sequences were amplified using the designed specific primers (Table 1). The amplicons were electrophoresed and extracted from agarose gel using QIAquick Gel Extraction kit (QIAGEN, Germany). Then, the polymerase chain reaction (PCR) fragments were ligated into suitable vectors, and each ligation product was separately used to transform DH5α Escherichia coli-competent cells. Accurate cloning of the target sequences was confirmed by colony PCR, restriction enzyme digestion, and sequencing.
The sequences of the primers used in the study.

Restriction sites are indicated in italics.
Cell culture
HepG2 cells were purchased from National Iranian Cell Bank (NCBI, Pasteur Institute of Iran, Tehran, Iran) and cultured in Dulbecco’s modified Eagle’s medium (DMEM) containing 5% fetal bovine serum (FBS), 2 mM glutamine, and 100 U/mL penicillin/streptomycin at 37°C in a humidified atmosphere with 5% CO2.
Quantitative reverse transcription polymerase chain reaction
Total RNA was extracted from HepG2 cells using RNX™-Plus (Cinnagen, Tehran, Iran). The quality and quantity of the extracted RNA were assessed by agarose gel electrophoresis and UV absorbance. Primer design for complementary DNA (cDNA) synthesis of the miRNA was performed based on the method developed and described previously. 14 In addition, AlleleID7 and Gene Runner software applications were used to design primers for Cyclin D1 expression. β-actin and SNORD 47(U47) were used to normalize the expression data of the target gene and the miRNA, respectively. Quantitative reverse transcription polymerase chain reaction (qRT-PCR) was prepared in a final volume of 20 µL containing 10 µL of 2 × SYBR Premix Ex Taq II (Tli RNase H Plus, Takara, Japan), 0.4 µM of each primer, 0.4 µL of 50 × ROX™ passive dye, 2 µL of cDNA (<100 ng), and 6.8 µL of distilled water. The reaction was performed in triplicates using StepOne™ instrument (Applied Biosystems, USA). The results were analyzed using ΔΔCT method.
Luciferase assay
HEK 293T cells were cultured in DMEM containing 10% FBS at 37°C in humidified atmosphere with 5% CO2 and then sub-cultured into 24-well plates. After 24 h, when 80% of confluency was obtained, the cells were transfected with p-LentiIII-eGFP (Applied Biological Materials Inc, Canada) or p-LentiIII-eGFP-miR-20a-containing vector separately, using calcium phosphate method. After 48 h, the rate of transfection was analyzed by fluorescent microscopy. Successfully transfected cells were again transfected with Cyclin D1 3′-UTR-containing psiCHECK™-2 Vector (Promega, Madison, USA) using calcium phosphate method. After 48 h of transfection, the culture medium was removed, cells were washed with cold phosphate-buffered saline (PBS), and 30 µL of cell lysis buffer (Promega) was added to the cells. The lysates were then transferred to 1.5-mL microtubes, and 5 µL of each sample was mixed with 20 µL of LARIITM and then Stop & Glo solutions (Promega), respectively. Using Sirous illuminator (Berthold, Germany), the ratio of the Renilla to firefly luminosity intensity was measured. In addition, to analyze the off-target effects, glyceraldehyde 3-phosphate dehydrogenase (GAPDH) 3′-UTR was also cloned in the psiCHECK™-2 Vector to be used as an internal control.
Packaging and lentivirus production
HEK 293T cells were cultured in 10-cm plates. Thereafter, at 70% confluency, helper plasmids (pMD2.G and psPAX2, Addgene, Cambridge, MA, USA) and p-LentiIII-eGFP-empty or miR-20a-containing vectors were used to transfect the cells using calcium phosphate method. After 24 h of transfection, we started to collect viruses from the culture media every 24 h for 3 days, and viruses were concentrated using ultracentrifugation at 23,000 r/min (40,000g) for 2.5 h at 4°C.
HepG2 cell transduction
Concentrated viruses mixed with Polybrene (8 µg/mL) were added to HepG2 cells cultured in DMEM containing 5% FBS. After 4 h, the culture medium was replaced with fresh medium to avoid the toxic effects of Polybrene. After 48 h, the cells were treated with puromycin (4 µg/mL) for 3 days. The rate and quality of transduction procedure were assessed using fluorescent microscopy.
Gene expression analysis after transduction
Total RNA was extracted using RNX™-Plus kit (Cinnagen, Tehran, Iran). cDNA synthesis and qRT-PCR reactions were performed according to the previously mentioned protocol to analyze the relative expression of Cyclin D1 and miR-20a.
Statistical analysis
Luciferase assay data were analyzed using Student’s t-test by SPSS software (Version 16, IBM, USA) to determine any significant relationship between the test and control groups. p-value less than 0.05 was considered as statistically significant. All qRT-PCR data were analyzed by StepOne® software v2.2.2 using ΔΔCT method.
Results
Bioinformatic prediction of miRNAs
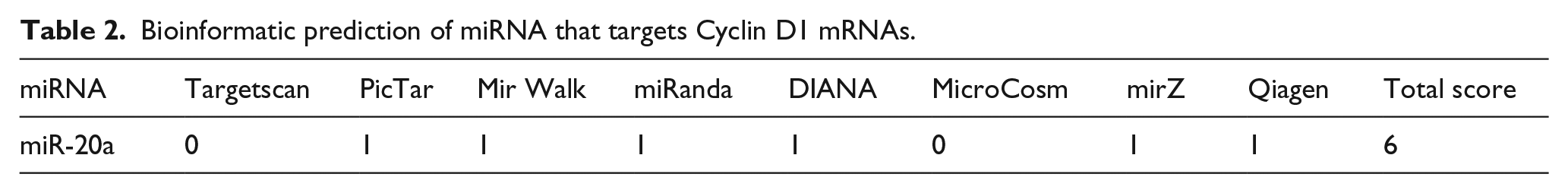
Since there is the probability of false-positive results in any individual database and online software applications that predict target mRNAs of miRNAs, we exploited several applications including DIANA, Targetscan, Miranda, PicTar, mirPath, microCosm, miRWalk, and mirZ to reduce false-positive results. Then, a score table was prepared based on binding affinity, the score of mRNA:miRNA interaction, the number of databases that confirmed the mRNA:miRNA interaction, and experimentally validated mRNA:miRNA interactions. Based on the score table, miR-20a was considered to interact with 3′-UTR of Cyclin D1. To further confirm the bioinformatic predictions, we performed a reverse analysis, that is, using candidate miRNA (miR-20a), we searched databases for verified miR-20a target genes. The results verified that Cyclin D1 was the target of miR-20a (Table 2).
Bioinformatic prediction of miRNA that targets Cyclin D1 mRNAs.
DIANA TOOLS-mirPath
This web server was used to find the role of miR-20a in signaling pathways. The results showed that miR-20a targets key genes including Cyclin D1 in WNT signaling pathway (Figure 1).
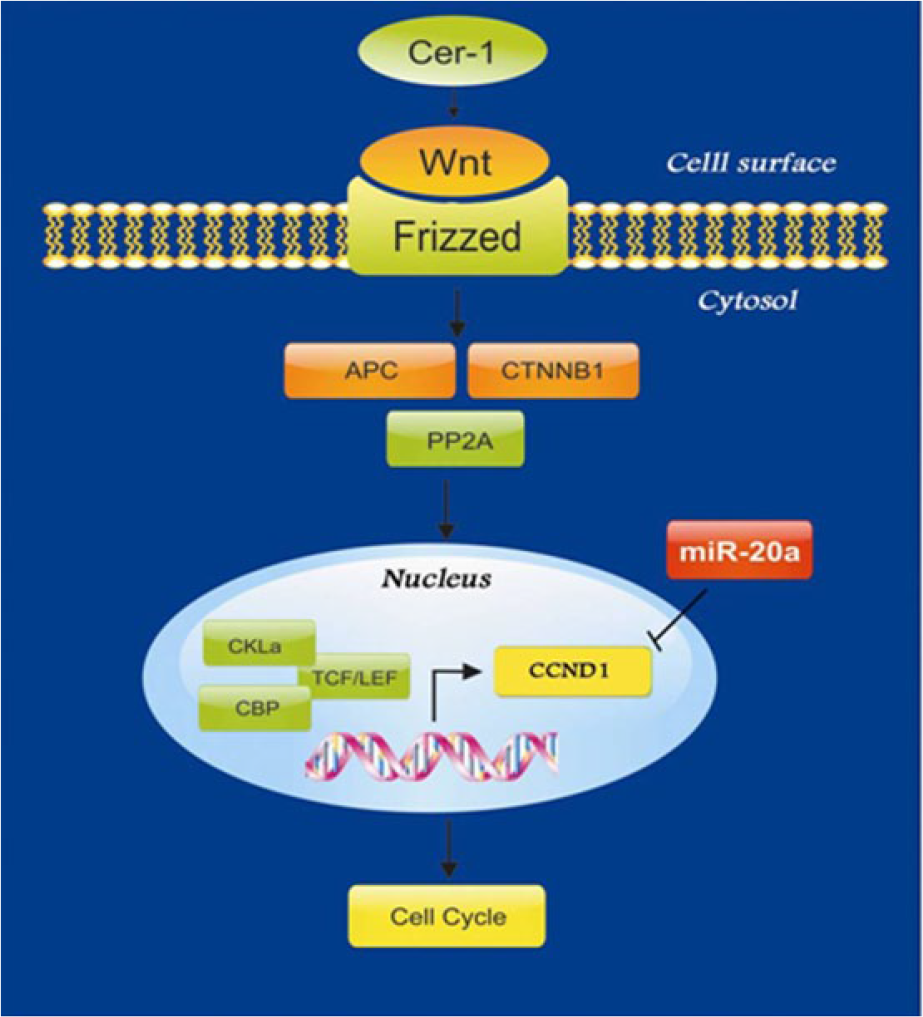
miR-20a targets Cyclin D1 (CCND1) and other genes specified by green color in WNT signaling pathway.
Luciferase assay confirmed miR-20a targets Cyclin D1
To verify that miR-20a targets 3′-UTR of Cyclin D1 mRNA, luciferase assay was performed. Based on the results, a significant decrease in Renilla to firefly luminosity intensity (p-value < 0.05) confirmed that the 3′-UTRs of Cyclin D1 is directly targeted by miR-20a. The expression of relative luciferase intensity decreased by the factor of 1.2 (Figure 2).

Luciferase assay performed on HEK 293T cell line to confirm miRNA–mRNA interaction using psiCHECK™-2 vector, assessing the interaction of miRNA and 3′-UTR of target gene.
HepG2 cells transduction
Concentrated viruses containing miR-20a mixed with Polybrene were added to HepG2 cells cultured in DMEM with 5% FBS. Fluorescent microscopy showed successful transduction of HepG2 cells (Figure 3).

HepG2 cells were transduced with miR-20a-containing vector p-LentiIII-eGFP: (a) normal and (b) green fluorescent image.
Target gene and miRNA expression analysis after transduction
After transduction of HepG2 cell line, the expression of miR-20a increased 1500 folds (Figure 4). In addition, qRT-PCR data indicated that Cyclin D1 gene expression decreased 238 folds (Figure 5).

miR-20a expression after HepG2 cell transduction with pLenti-III-eGFP vector containing miR-20a.

Cyclin D1 (CCND1) expression after HepG2 cell transduction with pLenti-III-eGFP vector containing miR-20a.
Discussion
HCC is a malignancy that its death and incidence rate are equal. The best therapeutic interventions against HCC are surgery and transplantation, while other strategies do not seem to be helpful. 28 Furthermore, prognosis and diagnosis of HCC is only possible in the late stages of the disease when therapeutic options are least effective. New therapeutic approaches can be achieved by better understanding the molecular mechanisms underlying HCC 29 such as Wnt/β-catenin signaling pathway. 30 We focused on targeting Wnt/β-catenin signaling pathway via Cyclin D1 inhibition. Cyclin D1, which can be the target of different miRNAs, has been suggested as a target for HCC treatment.1,7,12,13,31,32 miRNAs are small molecules with high impact on signaling pathways of cells. Identification of miRNAs which play key roles in targeting and regulating genes in signaling and metabolic pathways is a new approach in cancer treatment. 33
In 2005, Murakami et al. showed an abnormal expression profile of miRNAs in HCC for the first time. They found that precursor miR-18, miR-20, and miR-92 interfere with cell differentiation in HCC. In 2006, they examined miRNA expression profiles in clinical samples. Their results presented that 30 miRNAs were differentially expressed between tumor and normal samples. 34 Other studies demonstrated that miR-21 and miR-221 are also up-regulated in liver cancer and are involved in cancer initiation and progression.35,36 In addition, in various research studies, miR-122, miR-26a, miR-29, and miR-375 were used for inhibition of HCC in mice, and a significant reduction in tumor size was achieved. 15 In 2008, Sun et al. 37 showed that transfection of A549 cells (non–small cell lung carcinoma) with miR-34 resulted in a decline in the expression of Cyclin D1 mRNA and protein. Two years later, Jung et al. transfected HepG2 cells with miR-34. They also found a noticeable change in the expression of some proteins like Cyclin D1. In their experiment, transfection of HepG2 cells with miR-34 led to a reduction in proliferation and invasion of the cells. 38
In the recent years, there have been tremendous advances in bioinformatic algorithms, biological/molecular databases, and computational modeling systems that have helped scientists to predict mRNA targets of miRNAs.20,22,33,39 Bioinformatic approaches can overcome the obstacles confronted by experimental methods. Concerning miRNA prediction, TargetScan, DIANA, mirZ, miRWalk, microcosm, mirPath, PicTar, and miRanda are the best suggested software applications. In this study, we exploited these bioinformatic algorithms to predict miRNAs targeting Cyclin D1 3′-UTR. We also recruited RT stem-loop primers, which were approved to be cost-effective and as reliable as commercially available primers, developed by Mohammadi-Yeganeh et al. 14 for miR-20a reverse transcription and qRT-PCR. 26 The expression patterns of Cyclin D1 and miR-20a were in line with our hypothesis considering Cyclin D1 overexpression in this type of tumor and probable down-regulation of miR-20a which was selected as the miRNA targeting CCND1 3′-UTR. miRNA expression analysis after transduction in HepG2 cell line containing p-LentiIII-eGFP-miR-20a compared to the control vector-containing cells showed a significant increase in miRNA expression level which can prove the efficiency of our viral construct. Interestingly, luciferase assay results finally confirmed the bioinformatic prediction results and showed that miR-20a targets 3′-UTR of Cyclin D1 and regulates its expression.
Different studies have indicated the role of miRNAs such as miR-195, miR-20, miR-19a, and miR-20a in regulating the expression of Cyclin D1 and some other related genes in different cancers.40–43 In a study conducted by Chen et al. 44 in 2010, it was demonstrated that overexpression of miR-193b in melanoma cells caused decreased expression of Cyclin D1. Gene expression analyses in our study also revealed that miR-20a significantly decreased Cyclin D1 expression. Previous studies concerning miR-20a in angiogenesis showed controversial results. Although some studies had indicated its role in the induction of the angiogenesis,42,43,45,46 a few others indicated that it inhibits angiogenesis. 43
Finally, in this study, we indicated that Cyclin D1 is one of the contributing factors in tumorigenesis and metastasis of HCC. These results are in agreement with previous studies on HCC. Since there are still a lot of limitations in miRNA studies and experiments, more investigations are needed to confirm the role of miR-20a as a new therapeutic agent for HCC.
Conclusion
We suggest that by thoroughly studying signaling pathways and molecular mechanism underlying HCC, it would be possible to use miR-20a as a new therapeutic to prevent Cyclin D1 oncogene overexpression in order to treat HCC. However, since one single miRNA can target and inhibit several mRNAs, more research is needed to carefully detect their targets. Furthermore, one single mRNA can be regulated by several miRNAs, and therefore, it seems necessary to assess the role of other miRNAs in the regulation of Cyclin D1. Nonetheless, it sounds promising that in the near future we can benefit from miRNAs, which are among the regulators of cell fate determination, for the treatment of cancers such as HCC.
Footnotes
Acknowledgements
The authors thank Stem Cell technology Research Center, Tehran, Iran, Cellular and Molecular Biology Research Center, Shahid Beheshti University of Medical Sciences, and Department of Biotechnology, School of Advanced Technologies in Medicine, Shahid Beheshti University of Medical Sciences, Tehran, Iran and Pasteur Institute of Iran, Tehran, Iran for providing technical supports. The authors would like to thank Mr Vahid Kia for final proofreading of the manuscript.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
