Abstract
The incidence of thyroid cancer has increased significantly in the last decade, and the most frequent type of this cancer is papillary thyroid carcinoma. MicroRNAs have been demonstrated to be abnormally expressed in tumors and associated with the development of the tumors. Our aim was to analyze the role and molecular mechanisms of tumor suppressor let-7b in the papillary thyroid carcinoma. Expression of let-7b and high-mobility group A2 in papillary thyroid carcinoma tissues and cell lines was assessed using quantitative reverse transcription polymerase chain reaction and western blot analysis. To explore the role of let-7b or high-mobility group A2 in the BCPAP and TPC-1 cells, 3-(4,5-dimethylthiazol-2-yl)-2,5-diphenyltetrazolium bromide and Transwell methods were used. Let-7b expression was significantly downregulated while expression of high-mobility group A2 was upregulated dramatically in papillary thyroid carcinoma tissues and cells compared with that in normal thyroid tissues and cells. In addition, overexpression of let-7b or knockdown of high-mobility group A2 inhibited cell migration and invasion compared with that of control. Besides, high-mobility group A2 was negatively regulated by let-7b in BCPAP cells. Moreover, high-mobility group A2 reintroduction reversed the anti-proliferation, anti-migration, and anti-invasion roles of let-7b. Let-7b might function as a tumor suppressor in papillary thyroid carcinoma by suppressing the expression of high-mobility group A2, and therefore might provide a promising therapeutic target for patients with papillary thyroid carcinoma.
Introduction
Thyroid cancer is the most common type of endocrine malignancy, which shows increasing incidence in the last decade in many countries. 1 Among thyroid carcinoma, papillary thyroid carcinoma (PTC) which is named for its papillary histological architecture, accounts for about 80%. 2 Though the 5-year survival rate of patients with PTC is over 95%, PTCs are with more aggressive phenotypes, which can disseminate into lymph nodes or distant tissues and even dedifferentiate into more deadly thyroid cancers. 3 Conventional treatments of PTC, including hormone therapy, surgery, and radioiodine ablation therapy are not effective in metastatic cases. 4 Thus, there is urgent need to provide novel therapeutic targets into this malignant disease.
MicroRNAs (miRNAs) are short non-coding (16–24 nucleotides) and single-stranded regulatory RNAs which inhibit the expression of target genes by binding to the 3′ untranslated region (3′-UTR) of their target messenger RNAs (mRNAs). 5 The aberrant expression and dysregulation of some miRNAs in PTC have been associated with the prognosis and development of the disease. 6 Moreover, miRNA could give rise to the downregulation of tumor suppressor gene expression or the upregulation of oncogene expression. 7
Let-7 was first discovered in Caenorhabditis elegans to promote differentiation. 8 Later, it has been identified to play an important role in regulating gene expression in lots of organisms. 9 Besides, the let-7 family is a key miRNA which contains let-7a, let-7b, let-7c, let-7d, let-7e, let-7f, let-7g, and let-7i and acts as a tumor suppressor in cancers.10–12 Moreover, it has been demonstrated to be highly expressed in normal thyroid glands. 13 However, the role of let-7b in PTC has not been fully explored.
High-mobility group A2 (HMGA2), a member of the HMGA family, is a non-histone chromatin protein. 14 In addition, it can alter chromatin architecture to promote or repress the action of transcriptional enhancers by binding to AT-rich regions of DNA. 15 HMGA2 is highly expressed in many malignant neoplasms including pituitary, 16 lung, 17 ovary, 18 and PTC 19 but lowly expressed in normal tissues (NT). 20 In addition, more evidences have shown that HMGA2 plays a vital role in the development of tumor via regulating cell metastasis and epithelial–mesenchymal transition (EMT).21,22 Based on these findings, we hypothesized that the HMGA2 played a key role in PTC.
In this study, we aimed to investigate the expression of let-7b miRNA and HMGA2 mRNA in different PTC tissues and cell lines. Moreover, the role of let-7b and HMGA2 in cell proliferation, migration, and invasion of BCPAP and TPC-1 cells was analyzed. Furthermore, we demonstrated that the role of let-7b in the cell proliferation, migration, and invasion might depend on its regulation of HMGA2. Our findings could provide a novel insight into the role of let-7b and HMGA2 in PTC and propose a novel therapeutic strategy for PTC.
Materials and methods
Tumor samples
A total of 20 pairs of PTC tissues, and corresponding paracancerous thyroid (PT) tissues, and 10 cases of NTs from adjacent thyroid benign lesions were obtained from patients who underwent surgical resection at the Department of Thyroid Surgery in Henan Provincial People’s Hospital between 2009 and 2014. The PT tissues were ⩾2 cm away from the edge of the PTC in 20 patients. Besides, the NTs were ⩾2 cm away from the edge of the thyroid benign lesions in 10 patients. The study was approved by the Human Ethics Committee of Henan Provincial People’s Hospital. Written informed consent was obtained from all patients. None of the patients recruited in this study had radiotherapy or chemotherapy before the surgery. The diagnosis of PTC was histopathologically confirmed. The resected tissue samples were snap-frozen in liquid nitrogen immediately and stored at −80°C for further study. The clinicopathological features of 20 patients are summarized in Tables 1 and 2.
Correlations between clinicopathological features and let-7b expression levels in patients.
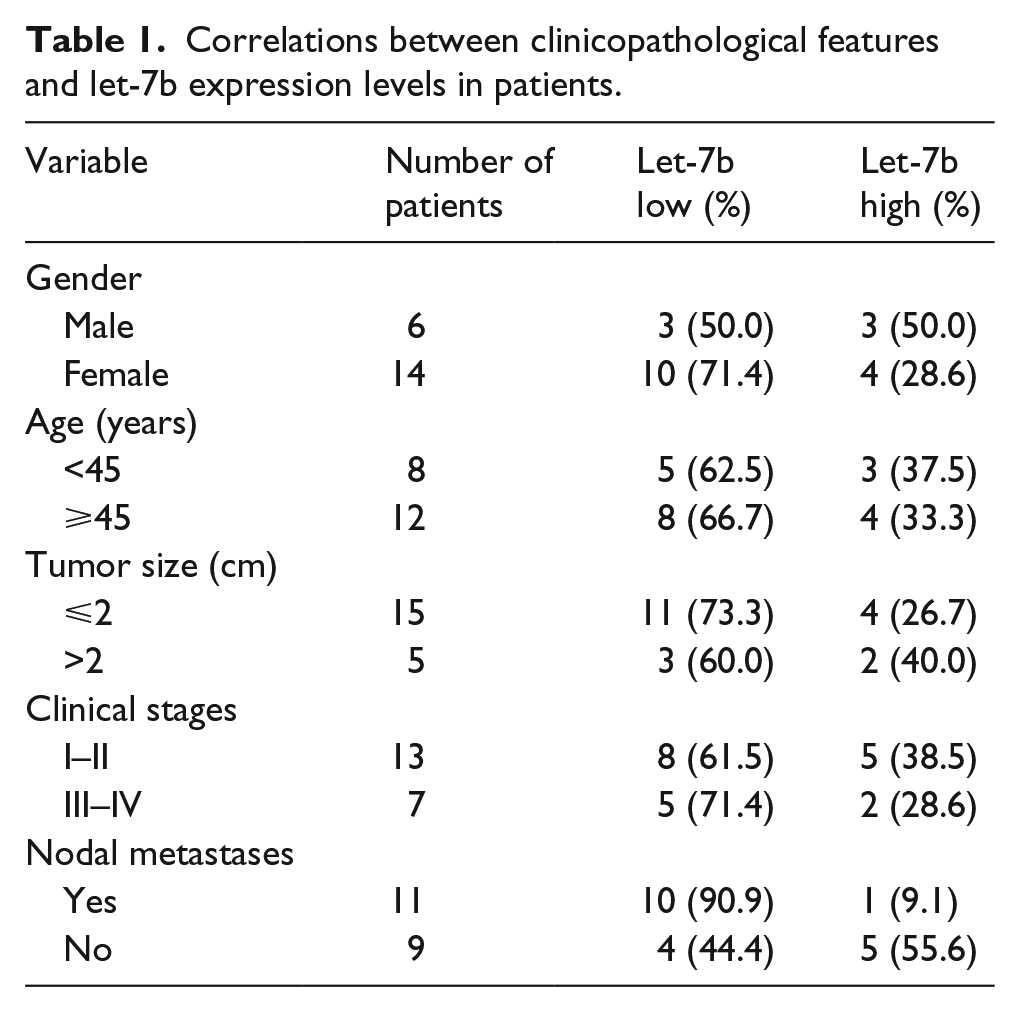
Correlations between clinicopathological features and HMGA2 expression levels in patients.

HMGA2: high-mobility group A2.
Cell culture
In total, four PTC cell lines (BCPAP, IHH4, TPC-1, and CGTHW-3) and a normal human thyroid epithelial cell line (PCCL3) were obtained from the Institute of Biochemistry and Cell Biology at the Chinese Academy of Sciences (Shanghai, China). All cell lines were cultivated in RPMI-1640 medium (Gibco) containing with 10% fetal bovine serum (FBS; Gibco, USA) in 5% CO2 chamber at 37°C.
Cell transfection
To establish overexpression of let-7b stable clone, BCPAP and TPC-1 cells were transfected with let-7b mimics or the non-targeting (scramble oligonucleotides) control. To overexpress the expression of HMGA2, the cells were transfected with pcDNA3.1-HMGA2, and the blank vector was used as a control. To generate HMGA2 constructs, regions of human HMGA2 CDS (NM_003483.4) were generated by polymerase chain reaction (PCR) amplification and then cloned into the pcDNA3.1 plasmid. To silence the expression of HMGA2, the cells were transfected with si-HMGA2 or scramble siRNA. For the downregulation of let-7b, the cells were transfected with let-7b inhibitor or the scramble control. All transfection were performed using Lipofectamine 2000 (Sigma, USA) according to the manufacturer’s instructions, and all the vectors were designed and synthesized by GeneChem (Shanghai, China).
3-(4,5-Dimethylthiazol-2-yl)-2,5-diphenyltetrazolium bromide assay
To investigate biological changes of the thyroid cancer cell lines, the cell proliferation was analyzed using the 3-(4,5-dimethylthiazol-2-yl)-2,5-diphenyltetrazolium bromide (MTT) assay as previously described. 23 In brief, the cells were incubated with 0.5 mg/mL of MTT for 4 h. Then, the formazan crystals were dissolved with 100 mL of dimethyl sulfoxide. After that the absorbance was measured using a 96-well plate reader (ELX800; Biotek Instruments, USA) at 570 nm at different time points (1, 2, 3, and 4 days).
Transwell migration and invasion assays
Cell migration was measured using a Transwell chamber (8 μm pores; Corning, USA). Transfected cells were harvested and then seeded into the upper chambers, and the lower chambers contained 500 μL of Dulbecco’s Modified Eagle’s Medium (DMEM) supplemented with 10% FBS. Cells were then incubated for 24 h at 37°C. Then, cells which migrated into the reverse side of the Transwell membrane were fixed with methanol and stained with 0.5% crystal violet (Sigma). Then, cells were counted at 100× magnification under a light microscope (CX31; Olympus, Japan), and the five visual fields were examined. For the cell invasion assay, the Transwell membrane was pre-coated with 30 μL of Matrigel (mixed in the ratio of 1:3 with phosphate-buffered saline (PBS); Corning) and the following protocols were the same as described above.
Quantitative reverse transcription PCR
Total RNA was extracted from transfected cells using TRIzol reagent (Invitrogen, USA). The reverse transcription PCR (RT-PCR) experiments were carried out as described previously. 24 Gene expression levels were analyzed with the 2−ΔΔCt method, and U6 or glyceraldehyde 3-phosphate dehydrogenase (GAPDH) was used as an internal control. Primers were listed below—Let-7b: 5′-GGGGTGAGGTAGTAGGTTG-3′ (forward) and 5′-CAGTGCGTGTCGTGGA-3′ (reverse); HMGA2: 5′-ACCGGTGAGCCCTCTCCTAA-3′ (forward) and 5′-CGTTTTTCTCCAGTGGCTTCTG-3′ (reverse); U6: 5′-GCTTCGGCAGCACATATACTAAAAT-3′ (forward) and 5′-CGCTTCACGAATTTGCGTGTCAT-3′ (reverse); and GAPDH: 5′-ACCCACTCCTCCACCTTTGA-3′ (forward) and 5′-CCCTGTTGCTGTAGCCAAATTC-3′ (reverse).
Western blotting
Cells were lysed in radioimmunoprecipitation assay (RIPA) buffer supplemented with a protease and phosphatase inhibitor cocktail (Roche, USA). Then, total protein lysates were separated by 10% sodium dodecyl sulfate–polyacrylamide gel electrophoresis (SDS-PAGE) and transferred onto polyvinylidene difluoride (PVDF) membranes. After blocking with 5% non-fat milk, the membranes were probed with primary antibody against HMGA2 or GAPDH (Cell Signaling Technology, USA) for overnight at 4°C. Then, the membranes were incubated with specific secondary antibodies at room temperature for 2 h. The protein bands were visualized by enhanced chemiluminescence (ECL) chromogenic substrate with horseradish peroxidase (HRP). GAPDH was used as an internal control.
Luciferase reporter assay
This assay was performed as previously described. 25 Briefly, the HMGA2 3′-UTR was cloned into pGL3 luciferase reporter (Promega, USA), generating pGL3-HMGA2. Mutations were introduced in potential let-7b binding sites using the QuikChange Site-Directed Mutagenesis Kit (Stratagene, USA). Then, 1 μg of the constructs were co-transfected with 1 μg of let-7b mimic or scramble control into the BCPAP cells. After 24 h of transfection, the dual-luciferase reporter assay was performed using the Dual-Luciferase Reporter Assay Kit (Promega) according to the manufacturer’s protocol.
In vivo animal studies
The animal studies were approved by the Henan Provincial People’s Hospital. Moreover, all protocols were performed according to the guidelines of the Care and Use of Laboratory Animals by the National Institute of Health, China. Male Balb/c nude mice of 3–5 weeks old (weighing 18–20 g) were obtained from the Experimental Animal Center of Zhengzhou University. The mice were anesthetized with chloral hydrate before any invasive operations. To establish PTC in vivo, BCPAP cells (1 × 107) were resuspended in 50 μL of PBS and injected into the cortex of the anterior tuberosity of the right tibia. After implantation for 7 days, the mice were divided into two groups randomly with six mice per group. The let-7b group was intratumorally treated twice per week with the let-7b mimics, and the negative control (NC) group was injected with the NC miRNA. Tumor volumes were measured using caliper and calculated according to the formula: tumor volume (mm3) = L × W2/2, where L is the length and W is the width. 26 After 19 days, the mice were sacrificed by CO2 asphyxiation. Tumors were excised immediately and weighed.
Statistical analysis
All data were expressed as mean ± standard deviation (SD). All statistical analyses were performed using GraphPad Prism 6.0. (GraphPad Software, USA). The statistical significance between two groups was assessed using the Student’s t test. p < 0.05 was considered as statistically significant.
Results
Expression of let-7b and HMGA2 mRNAs in PTC tissues and cell lines
To determine let-7b and HMGA2 mRNA expression in PTC tissues, total RNA extracted from 20 pairs of PTC tissues, and corresponding PT tissues, and 10 cases of NT from adjacent thyroid benign lesions. As shown in Figure 1(a) and (b), let-7b expression was downregulated significantly in the PTC compared with the NT. However, the relative mRNA expression of HMGA2 was upregulated dramatically in the PTC than that in the NT. There was no significant difference between PT and NT.

Expression of let-7b and HMGA2 mRNA in different tissues and cells. (a and b) Relative expression levels of let-7b miRNA and HMGA2 mRNA in NT, PT, and PTC tissues examined by qRT-PCR. Besides, expression of let-7b and HMGA2 mRNA in PTC cell lines (BCPAP, IHH4, TPC-1, and CGTHW-3) and a normal human thyroid epithelial cell line (PCCL3; *p < 0.05 compared with the NT group or PCCL3 cell line).
The expression levels of let-7b and HMGA2 from four PTC cell lines (BCPAP, IHH4, TPC-1, and CGTHW-3) and one normal human thyroid epithelial cell line (PCCL3) were also evaluated. Compared to PCCL3 cells, let-7b expression was downregulated, and HMGA2 mRNA was upregulated significantly in BCPAP, IHH4, TPC-1, and CGTHW-3 cells.
Let-7b overexpression inhibited cell proliferation, migration, and invasion
Because cell lines BCPAP and TPC-1 possessed of marked let-7b changes, the two cell lines were used in cell transfection to alter let-7b levels, and the results showed effective upregulation of let-7b by transfecting let-7b mimics (Figure 2(a) and (f)). MTT assay showed that the proliferation of let-7b overexpressing cells was impaired than that of control cells (Figure 2(b) and (g)). In addition, Transwell assays showed that let-7b overexpression decreased migrated and invasive cell numbers in the two cell lines (Figure 2(c)–(e), (h)–(j)), indicating that let-7b might suppress cell proliferation, migration, and invasion in PTC cell lines.

Overexpression of let-7b inhibited cell proliferation, migration, and invasion of BCPAP and TPC-1 cells. (a) BCPAP cells were transfected with let-7b mimics and scramble control and were confirmed by qRT-PCR. (b) MTT assays were performed to determine cell proliferation of BCPAP cells. (c) Representative image of cell migration and invasion of let-7b-overexpressed BCPAP cells. (d and e) Quantitative results of migration and invasion cell numbers of BCPAP cells. (f) The expression levels of let-7b in TPC-1 cells transfected with let-7b mimics or scramble control. (g) Cell proliferation of TPC-1 cells. (h) Representative image of cell migration and invasion of TPC-1 cells. (i and j) Quantitative results of migration and invasion cell numbers of TPC-1 cells (*p < 0.05 compared with the control group).
Knockdown of HMGA2 inhibited cell proliferation, migration, and invasion
Next, BCPAP and TPC-1 cells were transfected with HMGA2 siRNA to repress the expression levels of endogenous HMGA2. As a result, the relative expression of HMGA2 was downregulated, which was inhibited by HMGA2 siRNA (Figure 3(a) and (f)). Besides, we found that cells transfected with HMGA2 siRNA reduced the cell proliferation, migration, and invasion abilities of BCPAP and TPC-1 cells compared with that of scramble siRNA–transfected cells (Figure 3(b)–(e), (g)–(j)).
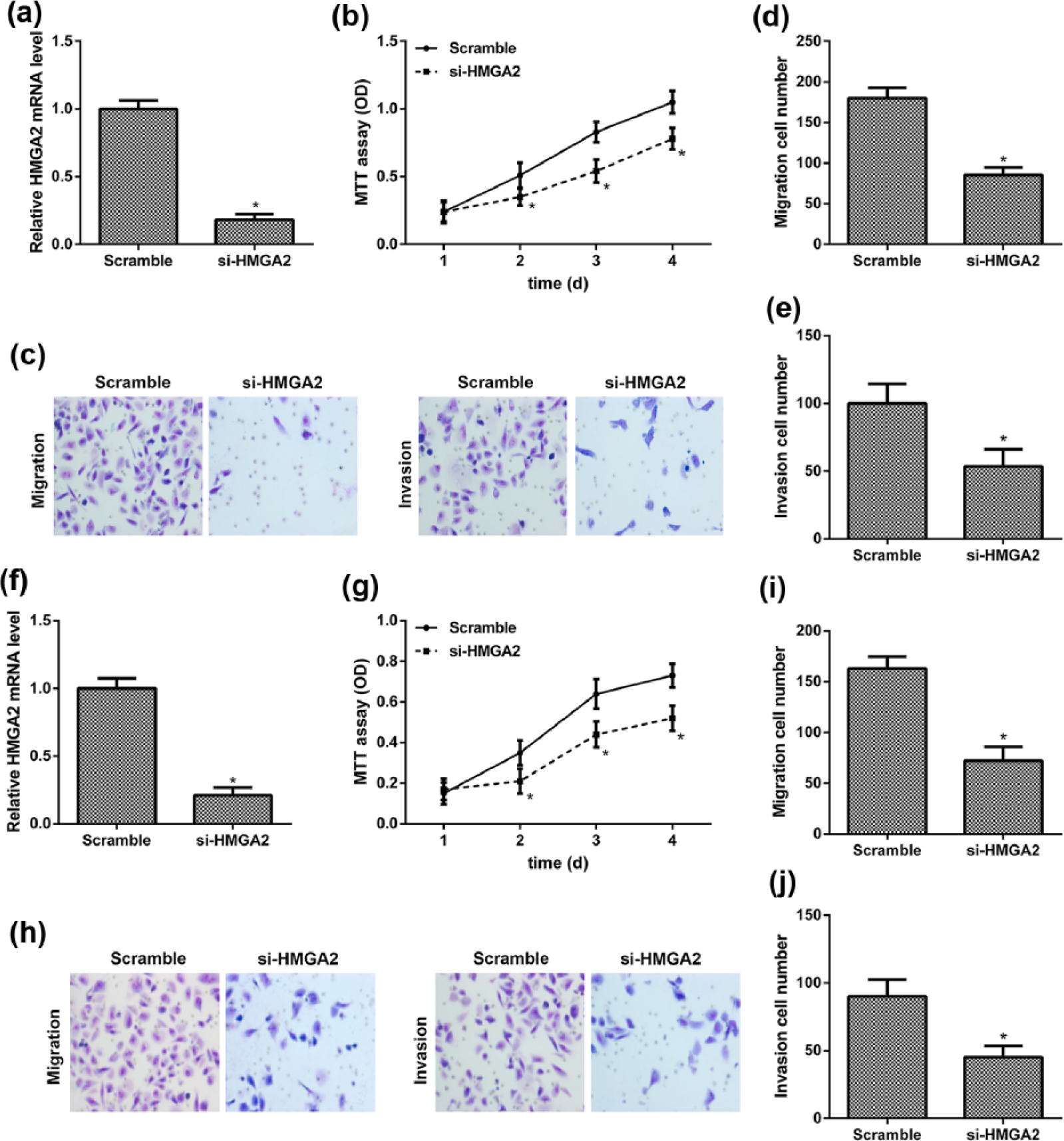
Knockdown of HMGA2 inhibited cell proliferation, migration, and invasion of BCPAP and TPC-1 cells. (a) Relative expression levels of HMGA2 in the BCPAP cells transfected with HMGA2 siRNA and the scramble siRNA detected by qRT-PCR. (b) Cell proliferation of BCPAP cells transfected with HMGA2 siRNA and the scramble siRNA determined by MTT assays. (c) Representative image of cell migration and invasion of BCPAP cells. (d and e) Quantitative results of migration and invasion cell numbers of BCPAP cells. (f) The expression levels of HMGA2 in TPC-1 cells transfected with HMGA2 siRNA or scramble siRNA. (g) Cell proliferation of TPC-1 cells. (h) Representative image of cell migration and invasion of TPC-1 cells. (i and j) Quantitative results of migration and invasion cell numbers of TPC-1 cells (*p < 0.05 compared with the scramble group).
HMGA2 was negatively regulated by let-7b in BCPAP cells
To investigate whether HMGA2 was a direct target of let-7b in BCPAP cells, a dual-luciferase reporter assay was performed. Co-expression of let-7b significantly inhibited the luciferase reporter activity of the wild-type HMGA2 3′-UTR but not of the mutant HMGA2 3′-UTR (Figure 4(a)), indicating that let-7b may suppress HMGA2 expression by binding to its 3′-UTR.

Let-7b negatively regulated the expression of HMGA2 in BCPAP cells. (a) Prediction of let-7b binding sites in the 3′-UTR of HMGA2 gene. Potential binding site was highlighted in red, and luciferase reporter assays were conducted in BCPAP cells. (b) qRT-PCR and western blot analysis of HMGA2 expression in BCPAP cells transfected with scramble control or let-7b mimics. (b) HMGA2 expression in BCPAP cells transfected with scramble control or let-7b inhibitor detected by qRT-PCR and western blot analysis (*p < 0.05 compared with the control group).
To further confirm that HMGA2 was regulated by let-7b in BCPAP cells, quantitative RT-PCR (qRT-PCR) and western blotting were performed. The mRNA and protein expression of HMGA2 were downregulated significantly in the let-7b-overexpressed cells (Figure 4(a)). However, suppression of let-7b resulted in upregulation of HMGA2 mRNA and protein expression levels (Figure 4(c)). Thus, the results indicated that let-7b may negatively regulate the expression of HMGA2 in BCPAP cells.
HMGA2 overexpression blocked the roles of let-7b
To identify the functional connection between let-7b and HMGA2, BCPAP cells were transfected with indicated constructs. The protein expression levels of HMGA2 were downregulated in the cells transfected with let-7b mimics. However, the effects were blocked by HMGA2 expression vector while were enhanced by si-HMGA2 (Figure 5(a) and (e)). In addition, HMGA2 reintroduction reversed the anti-proliferation, anti-migration, and anti-invasion roles of let-7b (Figure 5(b)–(d)). However, si-HMGA2 promoted these abilities of let-7b (Figure 5(f)–(h)). Therefore, our results suggested that the roles of let-7b in the cell proliferation, migration, and invasion of PTC partly depended on its regulation of HMGA2.

HMGA2 reintroduction blocked the anti-proliferation, anti-migration, and anti-invasion roles of let-7b. (a) HMGA2 protein expression was determined by western blotting in BCPAP cells. Cells were transfected with let-7b mimics or scramble control alone or co-transfected with let-7b mimics and HMGA2 expression vector. (b) Cell proliferation was measured by MTT assays in BCPAP cells. (c) Representative images of cell migration and invasion of BCPAP cells. (d) Quantitative results of migration and invasion cell numbers of BCPAP cells. (e) The protein expression levels of HMGA2 in BCPAP cells. Cells were transfected with let-7b mimics or scramble control alone or co-transfected with let-7b mimics and si-HMGA2. (f) Cell proliferation of BCPAP cells. (g) Representative images of cell migration and invasion of BCPAP cells. (h) Quantitative results of migration and invasion cell numbers of BCPAP cells (*p < 0.05 compared with the control group; #p < 0.05 compared with the let-7b-overexpressed group).
Let-7b suppressed in vivo tumor growth
To investigate the treatment effect of let-7b in vivo, the models of PTC on nude mice were established. For the BCPAP-originated tumor, compared with the NC-treated group, the therapeutic effect of let-7b began to show after 14 days. The average tumor volume was significantly smaller in the BCPAP-originated tumor injected with let-7b mimics than that injected with NC (Figure 6(a) and (b)). Moreover, the BCPAP-originated tumor weight of the let-7b group weighed extremely lighter than that of the NC group (Figure 6(c)).

Let-7b suppressed tumor growth in animal models of PTC. (a) The average BCPAP-originated tumor volume during a 19-day period of therapy in the nude mice injected with let-7b mimics or the negative control (NC). (b) The average BCPAP-originated tumor volume during a 19-day period of therapy in the nude mice injected with let-7b mimics or the NC. (c) Statistical graphs of BCPAP-originated tumor weight in the nude mice injected with let-7b mimics or the NC (*p < 0.05 compared with the control group).
Discussion
It has been reported that miRNAs can act as activators or inhibitors in carcinogenesis. 27 The let-7 family of miRNAs is highly conserved in function and sequence across different species, 28 and it is one of the first discovered tumor suppressor miRNAs. 29 Long et al. 30 demonstrated that let-7a may be a tumor suppressor in laryngeal cancer through reducing cell growth, promoting cell apoptosis, and decreasing the oncogene expression. Esquela-Kerscher et al. 10 found that downregulation of let-7 expression played a role in tumor formation and lung cancer progression. Han et al. 31 revealed that overexpression of let-7b can inhibit migration and invasion of gastric cancer cells by directly targeting ING1. In addition, let-7b has been found to be notably downregulated, thus negatively regulates the expression of multiple oncogenes, such as c-Myc, 32 RAS, 33 and HMGA2. 34 In this study, let-7b expression was downregulated significantly in the PTC tissues and cells. Moreover, functional study suggested that overexpression of let-7b inhibited the proliferation, migration, and invasion of BCPAP and TPC-1 cells, suggesting that let-7b is a tumor suppressor factor in PTC.
In this study, it showed that HMGA2 might be a downstream target of let-7b in PTC. HMGA2 belongs to the HMGA family and plays a key role in chromatin organization. 35 Besides, HMGA2 was related to the proliferation, invasion, and prognosis of a lot of malignant tumors. 36 In addition, very high expression level of HMGA2 was observed in the malignant thyroid tumors compared with the benign thyroid tumors. 19 However, the biological effects of HMGA2 on PTC remain unclear. Here, we showed that the relative mRNA expression of HMGA2 was upregulated dramatically in the PTC tissues and cells. Knockdown of HMGA2 reduced the proliferation, migration, and invasion of BCPAP and TPC-1 cells, indicating the role of HMGA2 in PTC.
It has been demonstrated that the HMGA2 3′-UTR has seven conserved sites complementary to the let-7 miRNA. 37 Moreover, let-7 was reported to be downregulated while HMGA2 was upregulated in lung cancers together with the repression of cell proliferation after ectopic expression of let-7, and overexpression of HMGA2 rescued the growth inhibition by let-7. 14 Besides, let-7 was lowly expressed and inhibited cell proliferation in esophageal squamous cell carcinoma (ESCC), and HMGA2 protein expression was highly expressed in ESCC and may be negatively regulated by let-7. 38 Furthermore, there was a significant inverse correlation between the expression of let-7 and HMGA2 in human pituitary adenomas, and let-7 reduction may contribute to overexpression of HMGA2. 39 Our results indicated that let-7b may suppress HMGA2 expression by binding to its 3′-UTR. Moreover, HMGA2 reintroduction reversed the roles of let-7b in cell proliferation, migration, and invasion, and these abilities were abolished by si-HMGA2. According to our findings, it strongly revealed a causal link between let-7b and HMGA2 during the development of PTC.
Previous studies showed that the significant overexpression of HMGA2 can be attributed to an inhibition of its interaction with let-7 miRNAs caused by the long non-coding RNA H19, which has been demonstrated in pancreatic cancer. 40 In addition, it was reported that HMGA2 knockdown markedly inhibited cell proliferation and migration via suppressing the transforming growth factor beta (TGF-β)/Smad and Wnt/β-catenin signaling pathway in bladder cancer cells. 41 Therefore, the underlying mechanism of the progression of PTC driven by let-7b-HMGA2 pathway still needs to be investigated.
In conclusion, our results demonstrated that overexpression of let-7b effectively suppressed the proliferation, migration, and invasion of PTC cells. More importantly, let-7b could decrease HMGA2 expression levels, and HMGA2 could affect the biological function of let-7b in PTC cells. Therefore, our findings provide a new insight for understanding the molecular mechanisms of let-7b in PTC, and let-7b might be an effective therapeutic approach for PTC treatment.
Footnotes
Acknowledgements
H.L. and L.Z. are the co-first authors.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by the Henan Provincial Department of Science and Technology Foundation (132300410367).
