Abstract
C-X-C chemokine receptor type 4 and stromal cell–derived factor-1 were proven to play important roles in several types of cancer and in many biological processes connected with tumor growth, invasion, angiogenesis, and metastasis. However, the clinical significance of C-X-C chemokine receptor type 4 and stromal cell–derived factor-1 expression in colorectal cancer remains inaccurate. The purpose of this systematic meta-analysis is to investigate the role of C-X-C chemokine receptor type 4 and stromal cell–derived factor-1 as prognostic factors for survival and the association between C-X-C chemokine receptor type 4/ stromal cell–derived factor-1 and clinicopathology in colorectal cancer. Databases including PubMed, EMBASE, and Cochrane Library were searched for relevant literatures updated till January 2017. Review Manager 5.3 was used for data analysis. In our meta-analysis, C-X-C chemokine receptor type 4 expression is related to tumor–node–metastasis stage, tumor differentiation, liver metastasis, lymph node metastasis, distant metastasis, and diagnosis, and no correlation of C-X-C chemokine receptor type 4 expression with tumor size, gender, preoperative carcinoembryonic antigen, age, or vascular invasion has been observed. Stromal cell–derived factor-1 expression has no relationship with tumor–node–metastasis stage, lymph node metastasis, vascular invasion, age, gender, distant metastasis, or diagnosis. The expression of stromal cell–derived factor-1 has association with tumor differentiation. Moreover, the pooled hazard ratio for disease-free survival/overall survival showed that overexpression of C-X-C chemokine receptor type 4/stromal cell–derived factor-1 reduced disease-free survival/overall survival in colorectal cancer. Therefore, High expression of C-X-C chemokine receptor type 4/stromal cell–derived factor-1 which is essential in tumor progression can predict poor survival that may provide more advance prognostic clues to colorectal cancer patients.
Keywords
Introduction
Colorectal cancer (CRC) is the third most common cancer and the third leading cause of cancer-related death around the world. In developed countries, CRC is the second cause of cancer-related death. CRC death rate is 20%–50% related with liver metastasis. However, the overall cure rate of CRC has not been significantly improved in recent years. Many CRCs are at an advanced stage when first diagnosed and some show metastasis to distant organs. There is no agreement yet on the most effective therapy for advanced CRCs, especially those with liver metastasis.1,2 The early detection of distant metastasis in CRC is very important. Therefore, looking for the factors which impact the development and metastasis of CRC is crucial for individualized treatment which will enhance the cure rate of CRC.
Chemokines, cytokines with molecular masses of 8–10 kDa, are classified into four groups (CXC, CXC3, CC, and C) according to the positions of the four conserved cysteine residues. 3 Chemokine stromal cell–derived factor-1 (SDF-1), also known as CXCL12, is expressed in stromal cells, including fibroblasts and endothelial cells, 4 and interacts specifically with the seven-transmembrane G protein-coupled receptor, C-X-C chemokine receptor type 4 (CXCR4). 5 CXCR4 and SDF-1 were proven to play important roles in several types of cancer and in many biological processes that are connected with tumor growth, invasion, angiogenesis, and metastasis.6,7 However, the clinical significance of CXCR4 and SDF-1 expression in CRC remains unclear. The purpose of this systematic review is to investigate the role of CXCR4 and SDF1 as prognostic factors for survival and the association between the expression of CXCR4/SDF1 and clinicopathological factors in CRC.
Materials and methods
Search strategy
Search terms were as follows: CXCR4 or C-X-C chemokine receptor type 4, SDF-1 or chemokine stromal-derived factor 1, CRC, prognosis, survival, and so on. We searched relevant literatures updated till January 2017 in multiple databases including PubMed, EMBASE, Cochrane Library, Medline, Google Scholar, and Sino Med. The search strategy follows the Cochrane Review Manual.
Criteria of inclusion and exclusion for study selection
Criteria of inclusion and exclusion for study selection were established based on the principle of PICOS. Criteria of inclusion were as follows: (1) all patients were diagnosed as having primary colorectal neoplasms, and all of them were confirmed by pathology postoperative; (2) The expression status of CXCR4/SDF-1 was detected by real-time polymerase chain reaction (RT-PCR) or immunohistochemistry (IHC); (3) the relevance between the expression of CXCR4/SDF1 and clinicopathological factors was revealed; (4) disease free survival (DFS) or overall survival (OS) was used for survival analysis; and (5) the hazard ratios (HRs) and 95% confidence interval (CI) were used for evaluating the association between the expression of CXCR4/SDF-1 and survival status. Criteria of exclusion were as follows: (1) studies with cell lines or animals, (2) studies without original data, and (3) repeated reports, reviews, case reports, and so on.
Data extraction
Two reviewers selected and extracted the data independently and then cross-checked the results of the studies included. Disagreement between the two reviewers was settled by a third reviewer. The information extracted includes first author’s name, publication time, country, total number of cases, research methods, clinicopathological factors, and data of survival analysis and other related events.
Quality assessment
The quality of all studies included was evaluated independently by reviewers by Newcastle–Ottawa Scale. The standard consists of three aspects: (1) the selection method of case group and control group, (2) the comparability between case group and control group, and (3) the evaluation method of exposure factors. The highest score is 10 points. The higher the score, the better the quality of articles.
Data analysis
Analysis was performed using the Review Manager 5.3 (Cochrane Collaboration, Oxford, UK). Count data were calculated by odds ratios (ORs) and 95% CI. I2 was used for heterogeneity analysis. If I2 ⩾ 50% or p ⩽ 0.05, there is heterogeneity, and the random effects model was chosen for pooling ORs. Otherwise, the fixed effect model was selected. The potential publication bias was examined by funnel plots; p < 0.05 was considered to be statistically significant.
Results
Study selection
Initially, 59 literatures were retrieved using the above-mentioned search strategy. In all, 34 articles were excluded due to the method of study, incompleteness of data, and so on. Finally, 25 studies which met the criteria were selected for further study and analysis. The flow graph of article selection was shown in Figure 1.

Flow graph of articles selected.
Study characteristics and quality
Among all the articles included, nine were conducted in China, five in Japan, three in America, two in Netherlands, two in Italy, two in Germany, one in Tunisia, and one in Norway. All of them were published in the last 12 years (2005–2016). The total number of patients enrolled was 3796. The expression status of CXCR4/SDF-1 was evaluated by IHC or RT-PCR. Quality assessment for articles included was conducted by Newcastle–Ottawa Scale. All the studies included scores higher than 5 points, belonging to high-quality literature. The characteristics and scoring of studies are shown in Table 1.
The characteristics and scoring of studies.

IHC: immunohistochemistry; RT-PCR: real-time polymerase chain reaction.
Meta-analysis of CXCR4 and SDF-1
There were two papers that evaluated the association of the expression status between CXCR4 and SDF-1. There was no significant association observed between the expression status of CXCR4 and SDF-1 (OR = 3.74, 95% CI = 0.71–19.63, p = 0.12; Figure 2).

The association of the expression status between CXCR4 and SDF-1.
Meta-analysis of CXCR4/SDF-1 and clinicopathological feature
The correlation of CXCR4 with clinicopathological factors such as age, gender, diagnosis, tumor size, vascular invasion, and status of lymph node metastasis was shown in all the studies included. The results of this meta-analysis show that CXCR4 expression is related to tumor–node–metastasis (TNM) stage (OR = 0.55, CI = 0.40–0.76, p = 0.0003; Figure 3(a)), tumor differentiation (OR = 0.63, CI = 0.51–0.78, p < 0.0001; Figure 3(b)), liver metastasis (OR = 2.08, CI = 1.40–3.09, p = 0.0003; Figure 3(c)), lymph node metastasis (OR = 2.25, CI = 1.51–3.36, p < 0.0001; Figure 3(d)), distant metastasis (OR = 2.17, CI = 1.39–3.40, p = 0.0007; Figure 3(e)), and diagnosis (OR = 0.57, CI = 0.38–0.86, p = 0.008; Figure 3(f)). However, no correlation of CXCR4 expression with tumor size (OR = 1.09, CI = 0.86–1.39, p = 0.49; Figure 4(a)), gender (OR = 1.01, CI = 0.87–1.17, p = 0.90; Figure 4(b)), preoperative carcinoembryonic antigen (CEA; OR = 1.55, CI = 0.72–3.32, p = 0.26; Figure 4(c)), age (OR = 1.18, CI = 0.97–1.43, p = 0.10; Figure 4(d)), or vascular invasion (OR = 1.31, CI = 0.66–2.62, p = 0.44; Figure 4(e)) has been observed.
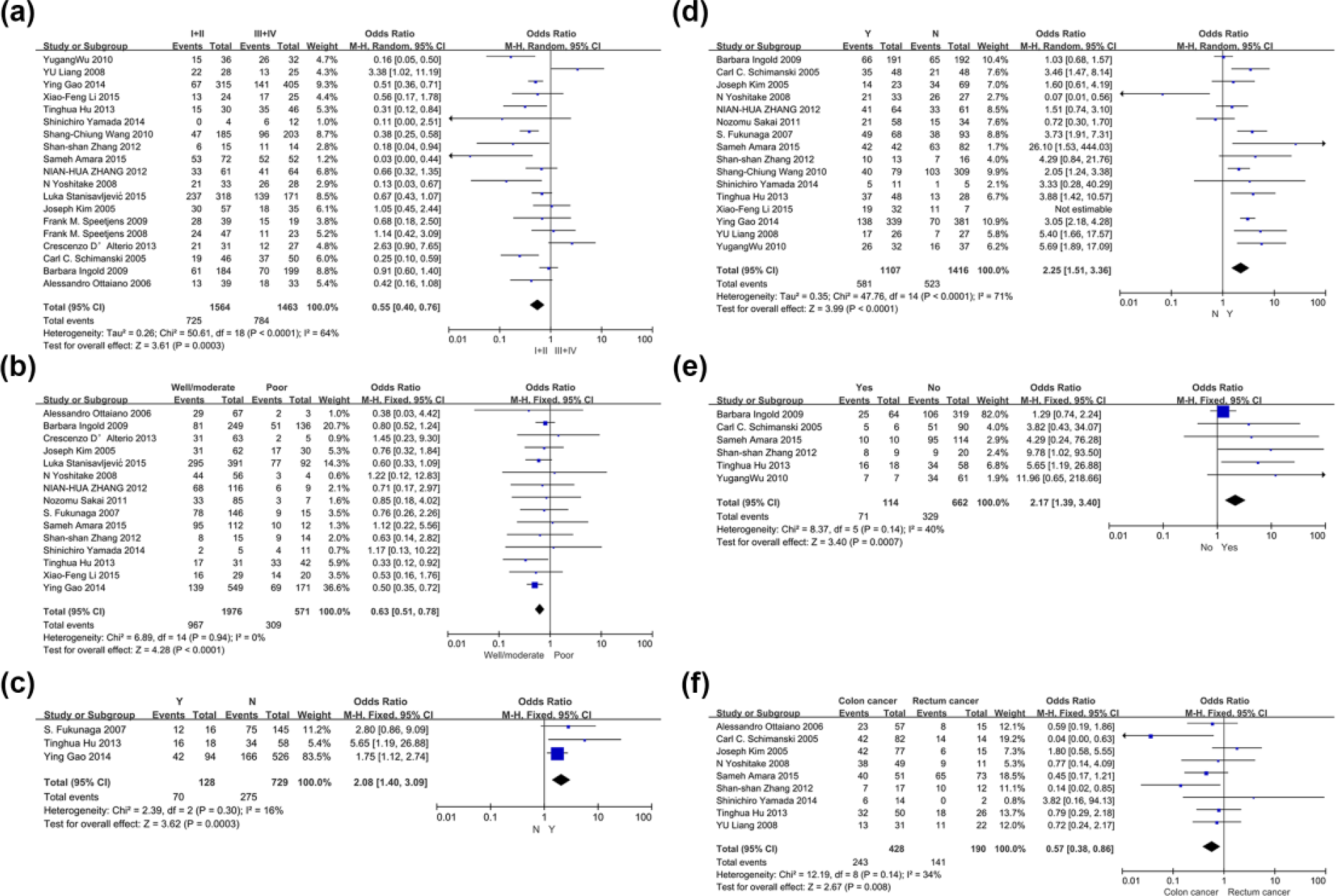
CXCR4 expression is related to (a) TNM stage, (b) tumor differentiation, (c) liver metastasis, (d) lymph node metastasis, (e) distant metastasis, and (f) diagnosis.

No correlation of CXCR4 expression with (a) tumor size, (b) gender, (c) preoperative CEA, (d) age, or (e) vascular invasion.
Only 6 of 25 studies evaluated the association between SDF-1 expression and clinicopathological factors. The result of this analysis indicates that SDF-1 expression has no relationship with TNM stage (OR = 0.84, CI = 0.48–1.49, p = 0.55; Figure 5(a)), lymph node metastasis (OR = 0.64, CI = 0.27–1.52, p = 0.31; Figure 5(b)), vascular invasion (OR = 1.36, CI = 0.21–8.76, p = 0.74; Figure 5(c)), age (OR = 5.00, CI = 0.66–37.78, p = 0.12; Figure 5(d)), gender (OR = 1.12, CI = 0.89–1.41, p = 0.32; Figure 5(e)), distant metastasis (OR = 1.02, CI = 0.61–1.70, p = 0.095; Figure 5(f)), or diagnosis (OR = 1.05, CI = 0.53–2.08, p = 0.90; Figure 5(g)). The expression of SDF-1 has association with tumor differentiation (OR = 2.12, CI = 1.12–4.01, p = 0.02; Figure 5(h)).

SDF-1 expression has no relationship with (a) TNM stage, (b) lymph node metastasis, (c) vascular invasion, (d) age, (e) gender, (f) distant metastasis, or (g) diagnosis. (h) Expression of SDF-1 has association with tumor differentiation.
The heterogeneity was observed in the analysis of CXCR4 expression with vascular invasion (p = 0.01; I2 = 63%), TNM stage (p < 0.0001; I2 = 64%), and lymph node metastasis (p < 0.0001; I2 = 71%) and SDF-1 expression with TNM stage (p = 0.009; I2 = 70%), tumor differentiation (p = 0.04; I2 = 58%), lymph node metastasis (p = 0.002; I2 = 80%), vascular invasion (p < 0.00001; I2 = 91%), and age (p = 0.02; I2 = 89%), so a random-effect model was selected.
Meta-analysis of CXCR4/SDF-1 and disease-free survival/OS
Table 2 and Figure 6(a)–(d) show the relationship between disease-free survival (DFS)/OS and the expression status of CXCR4/SDF-1. The pooled HR for DFS/OS showed that overexpression of CXCR4 reduced DFS/OS in CRC (HRDFS = 1.88, 95% CIDFS = 1.30–2.71, pDFS = 0.0008; HROS = 1.67, 95% CIOS = 1.29–2.18, pOS = 0.0001) as well as SDF-1 (HRDFS = 1.79, 95% CIDFS = 1.19–2.69, pDFS = 0.005; HROS = 2.08, 95% CIOS = 1.09–4.00, pOS = 0.03). The results above indicate that the prognosis of CRC patients with positive CXCR4/SDF-1 expression is poor.
The relationship between DFS/OS and the expression status of CXCR4/SDF-1.

HR: hazard ratio; CI: confidence interval; DFS: disease-free survival; OS: overall survival; CXCR4: C-X-C chemokine receptor type 4; SDF-1: stromal cell–derived factor-1.

(a) The relationship between DFS and the expression status of CXCR4, (b) the relationship between OS and the expression status of CXCR4, (c) the relationship between DFS and the expression status of SDF-1, and (d) the relationship between OS and the expression status of SDF-1.
Discussion
As CRC prognosis and treatment are currently based on a classification system, CRC still shows large heterogeneity in clinical outcome. 32 Numerous patients with CRC have been at an advanced stage when they were first diagnosed. Up to 30%–50% patients with stage II and III CRC will develop recurrence or distant metastasis even they undergo radical surgery. 33 CXCR4 and SDF-1 have been shown to play vital role in several types of cancer and in many biological processes, including tumor growth, invasion, angiogenesis, and metastasis. SDF-1, also known as CXCL12, interacts specifically with CXCR4. Some studies showed that the expression of CXCR4/SDF-1 was apparently correlated with TNM stages in many types of cancer, such as non-small-cell lung carcinoma (NSCLC), 34 esophageal cancer, 35 CRC, 8 triple-negative breast cancer, 36 and pancreatic adenocarcinoma. 37
In order to investigate the role of CXCR4 and SDF1 as prognostic factors for survival and the association between CXCR4/SDF-1 and clinicopathology in CRC, we analyzed the outcomes of 3808 patients with CRC from 25 eligible individual literatures to perform this systematic meta-analysis. This analysis concluded that high expression of CXCR4/SDF-1 can predict poor survival which may provide more advance prognostic clues to CRC patients. In addition, all the studies included met the standards of Newcastle–Ottawa Scale. Our meta-analysis showed that CXCR4 expression is related to TNM stage, tumor differentiation, liver metastasis, lymph node metastasis, distant metastasis, and diagnosis, and no correlation of CXCR4 expression with tumor size, gender, preoperative CEA, age, or vascular invasion has been observed. Moreover, we observed that SDF-1 expression has no relationship with TNM stage, lymph node metastasis, vascular invasion, age, gender, distant metastasis, or diagnosis. Meanwhile, our analysis showed that the expression of SDF-1 has association with tumor differentiation. Surprisingly, there was no significant association observed between the expression status of CXCR4 and SDF-1, though CXCR4 is the specific receptor of SDF-1. Our results could be essential for defining powerful prognostic factors which may be involved in disease progression and metastasis.
The role of CXCR4/SDF-1 signaling in cancer progression is related to a large variety of cancer signaling pathway. In the blood system diseases, JAK2 cooperates with CXCR4/SDF-1 signaling to increase the chemotactic response of human cell lines and primary CD34+ cells through PI3K pathway.38,39 In CRC, SDF-1 activates extracellular signal–regulated protein kinase (ERK)-1/2, stress-activated protein kinase (SAPK)/c-Jun N-terminal kinase (JNK), Akt, matrix metallopeptidase (MMP)-9, and vascular endothelial growth factor (VEGF). 40 These SDF-1-induced signals mediate increased cancer cell migration and invasion. Here, we found that CXCR4/SDF-1 overexpression was associated with a bad prognosis in CRC patients. Therefore, we speculated that CXCR4/SDF-1 axis might act in the development of migration, invasion, and metastasis of CRC via signaling pathways mentioned above. CXCR4 antagonist could provide a greater survival benefit to tumor patients. Hz515H7, a monoclonal antibody (mAb) which binds to human CXCR4, strongly inhibits cell migration and proliferation and induces both antibody-dependent cellular cytotoxicity and complement-dependent cytotoxicity against neoplastic cells. 41 Studies about AMD3100, a CXCR4 antagonist, showed that CXCR4 blockade could inhibit glioblastomamultiforme (GBM) growth and invasion.42,43 And in CRC, AMD3100 also decreased invasion and metastasis of human CRC cells in vitro. 44 Other potential molecular prognostic factors of CRC such as PUM2 and CBX8 that affect the disease progression and metastasis will be of great importance. The mechanisms above would help identify personalized treatment decisions based on individual tumor characteristics to develop potential targets for the effective treatment of CRC.
This study has several limitations needed to be recognized. The number of literatures and patients included was relatively small especially in the studies of SDF-1. This may be a cause of the result that no significant association was observed between the expression status of CXCR4 and SDF-1. Currently, there is no accurate definition of the cut-off values about the expression of CXCR4/SDF-1. And the variety of concentrations of antibody can be a source of potential bias. Additionally, not all the studies we reviewed offered complete information, and heterogeneity was found in analysis. All the factors above should be considered when evaluating the results.
The pooled HR for DFS/OS showed that overexpression of CXCR4/SDF-1 reduced DFS/OS in CRC. In conclusion, those findings of the present analysis confirmed the prognostic value of CXCR4/SDF-1 in CRC. Moreover, joint examination of CXCR4/SDF-1 can provide more advance prognostic clues to CRC patients. These joint examinations may provide assistance in the selection of patients with a worse prognosis; these patients can then be directed to the more intensive treatment of adjuvant chemotherapy to achieve better prognosis.
Footnotes
Declaration of conflicting interest
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This study was financially supported by grants from the Development Center for Medical Science and Technology, Ministry of Health, P.R. China (no. W2013R02); Applied Basic Research Projects of Shanxi Science and Technology Department, P.R. China (no. 201601D011128); and Research Subject of Shanxi Health and Family Planning Commission, P.R. China (no. 2015049); Fund Program for the Scientific Activities of Selected Returned Overseas Professionals in Shanxi Province, China (2014-779).
