Abstract
C-X-C chemokine receptor type 4 (CXCR4) is a receptor for a pleiotropic chemokine CXCL12. Previous studies have shown that the acute administration of the CXCR4 antagonist AMD3100 reduced neuroinflammation in stroke brain and mobilized bone marrow hematopoietic stem cells (HSCs). The purpose of this study was to characterize the neuroprotective and neurotrophic effect of a novel CXCR4 antagonist CX549. We demonstrated that CX549 had a higher affinity for CXCR4 and was more potent than AMD3100 to inhibit CXCL12-mediated chemotaxis in culture. CX549 effectively reduced the activation of microglia and improved neuronal survival after injury in neuron/microglia cocultures. Early poststroke treatment with CX549 significantly improved behavioral function, reduced brain infarction, and suppressed the expression of inflammatory markers. Compared to AMD3100, CX549 has a higher affinity for CXCR4, is more efficient to mobilize HSCs for transplantation, and induces behavioral improvement. Our data support that CX549 is a potent anti-inflammatory agent, is neuroprotective against ischemic brain injury, and may have clinical implications for the treatment of stroke.
Introduction
Chemokine ligand 12 [CXCL12; also called stromal cell-derived factor-1 (SDF-1)], a pleiotropic chemokine in the CXC subfamily, is constitutively expressed in the central nervous system (CNS) and periphery. CXCL12 interacts with a G protein-coupled receptor, C-X-C chemokine receptor type 4 (CXCR4). CXCR4 is present in neurons, astrocytes, microglia 1 , bone marrow-derived cells, and neural progenitor cells 2 4 . The expression of CXCL12 and CXCR4 is regulated in several pathological conditions, such as stroke. In clinical stroke patients, the level of serum CXCL12 was inversely correlated with infarct volume and neurological severity5,6. In experimental stroke mice, CXCL12 was transiently downregulated at 6 h after permanent middle cerebral artery occlusion (MCAo), but continued to increase at 2 to 4 days postischemia 3 . The expression of CXCL12 7 , mainly in the penumbra region, peaked within 3-7 days after the MCAo in rats 8 . Similar to CXCL12, CXCR4 was upregulated in the ischemic brain. The increase in CXCR4 mRNA expression was found in the ischemic penumbra at 2-4 days after MCAo3,7.
CXCR4/CXCL12 has multiple functions in the CNS 9 . Treatment with lipopolysaccharide (LPS) increased CXCR4 expression in primary cultured microglia. Inactivation of CXCR4 attenuated activation and proliferation of microglia 1 . The interaction of CXCR4 and microglia was also found in the stroke brain. Enhanced immunoreactivity of CXCR4 and microglia markers was found in the area surrounding the infarct core at 96 h after MCAo 1 . Early posttreatment with the CXCR4 antagonist AMD3100 (or Plerixafor) reduced the number of microglia in the peri-infarct area 10 , attenuated infiltration of CD3/CD4 T cells into the ischemic hemisphere, and improved functional recovery in stroke animals11,12. Taken together, these data suggest that CXCR4/CXCL12 signaling plays a significant role in neuroinflammation, and AMD3100 is neuroprotective through anti-inflammatory effects in the brain following stroke.
In the periphery, CXCR4/CXCL12 regulates the trafficking of bone marrow hematopoietic stem cells (HSCs) 13 and mesenchymal stem cells (MSCs) 14 . CXCL12 retains stem cells in the bone marrow, while CXCR4 antagonists release these cells from the bone marrow 15 . A rapid increase in CD34+ (an HSC marker) cells in the peripheral blood was found within 4 h after AMD3100 injection in patients 16 or healthy volunteers 17 . Increasing evidence has also supported that HSCs, transplanted peripherally, provided trophic factors 18 and induced functional recovery and neurogenesis in stroke animals 19 21 . Since CXCR4 antagonists mobilize HSCs into circulation, these chemicals may indirectly promote trophic responses from HSCs to the lesioned tissue. For example, the mobilization of HSCs is associated with protective or reparative effects of CXCR4 antagonists in animal models of acute ischemic kidney injury 22 and myocardial infarction 23 .
CXCR4/CXCL12 also controls the homing of grafted HSCs or MSCs to the lesioned site in ischemic brain. CXCL12 in the penumbral regions 8 is a chemoattractant for the bone marrow-derived HSCs or MSCs 18 . Overexpressing CXCR4 in the MSC transplants increased cell migration to the ischemic boundary and improved behavioral recovery in stroke rats24,25. These data suggest that CXCL12/CXCR4 has a dual modulatory effect on the homing of HSCs in stroke animals. Inhibition of CXCR4 increased the migration of the HSCs from bone marrow to peripheral blood, while it also reduced the migration of bone marrow-derived MSCs (BM-MSCs) into the lesioned brain for repair 8 .
The purpose of this study was to characterize the neuroprotective and cell-mobilizing effects of a novel and potent CXCR4 antagonist CX549 (Fig. 1) in cultured cells and in an animal model of stroke. We demonstrated that CX549 has anti-inflammatory effects in primary neuronal cultures and in vivo. Early posttreatment with CX549 reduced brain damage and enhanced behavioral recovery in the stroke animals. CX549 mobilized bone marrow HSCs to the peripheral blood, and transplantation of peripheral mononuclear cells collected from the donor rats receiving CX549 improved behavioral recovery in stroke animals.

Comparison of binding affinity, chemotaxis, and cell mobility of AMD3100 and CX549. (A) Chemical structure of CX549 and AMD3100. (B) CX549 had a significantly high binding affinity to the CXCR4 receptors in the HEK293 cells overexpressing CXCR4. (C) CX549 was more potent than AMD3100 in inhibiting CXCL12-mediated chemotaxis of CCRF-CEM cells, a human cell line with high CXCR4 expression *p < 0.005, two-way ANOVA + Newman-Keuls test. HEK, human embryonic kidney; CXCR4, C-X-C chemokine receptor type 4; CXCL12, motif chemokine ligand 12; CCRF-CEM, T-cell acute lymphoblastic leukemia; ANOVA, analysis of variance.
Materials and Methods
Animals and Chemicals
Adult Sprague-Dawley rats and C57BL/6 mice were used in this study. Animals were purchased from the National Laboratory Animal Center (Taipei, Taiwan). All study protocols were approved by the Animal Research Committee at the National Health Research Institutes. All animals were treated in accordance with the Guide for the Care and Use of Laboratory Animals published by the US National Institutes of Health (NIH). AMD3100 was purchased from Sigma-Aldrich (St. Louis, MO, USA). CX549 was supplied by Dr. K.-S. Shia at the National Health Research Institutes, Taiwan.
Flow Cytometry Analysis
Male C57BL/6 mice (8-10 weeks old) were treated with vehicle (saline), CX549, or AMD3100 [5 mg/kg, subcutaneously (SC)]. Blood samples (300 μl) were collected from the heart at 2 h after injection. Cells were labeled with allophycocyanin-conjugated anti-CXCR4 (clone 2B11; 1:80 dilution; Affymetrix/eBioscience, San Diego, CA, USA), fluorescein isothiocyanate (FITC)-conjugated anti-CD34 (clone RAM34; 1:200; Affymetrix/eBioscience), and phycoerythrin-conjugated anti-CD133 (clone 13A4; 1:200; Affymetrix/eBioscience) and were then washed and characterized by flow cytometry (Guava Technologies, Hayward, CA, USA) as previously described 22 .
CXCR4 Binding Assay
Human embryonic kidney (HEK) 293 cells overexpressing CXCR4 (for details, please see Wu et al. 22 ) were homogenized. Cell membranes were incubated with 0.16 nM [125I]CXCL12 (PerkinElmer, Waltham, MA, USA) along with vehicle, CX549, or AMD3100 in incubation buffer [50 mM 4-(2-hydroxyethyl)-1-piperazineethanesulfonic acid (HEPES)-NaOH (pH 7.4), 100 mM NaCl, 5 mM MgCl2, 1 mM CaCl2, 0.5% bovine serum albumin (BSA; Sigma-Aldrich)]. Nonspecific binding was defined in the presence of 50 μM AMD3100. The reaction mixtures were incubated for 1.5 h at 30°C and were then transferred to a 96-well GF/B filter plate (Millipore Corp., Billerica, MA, USA). The reaction mixtures were terminated by manifold filtration and washed with ice-cold wash buffer (50 mM HEPES-NaOH, pH 7.4) four times. The radioactivity bound to the filter was measured by Topcount (PerkinElmer Inc.).
Chemotaxis Assay
CCRF-CEM (T-cell acute lymphoblastic leukemia) cells were suspended in Rosewell Park Memorial Institute (RPMI)-1640 medium (Gibco, Thermo Fisher Scientific, Waltham, MA, USA) containing 10% fetal bovine serum (FBS; Biowest, Nuaillée, France) and then preincubated with vehicle, CX549, or AMD3100 for 10 min at 37°C. The assay was performed in Millicell Hanging Cell Culture Inserts (EMD Millipore, Bedford, MA, USA). Compounds containing 10 nM CXCL12 were plated in the lower chambers of the inserts, and cells with compounds were plated in the upper chamber of the inserts at a density of 2.5 × 105 cells/well. After 2.5 h of incubation at 37°C, cells in both chambers of the inserts were measured by flow cytometry.
Primary Rat Cortical Neuron (PCN) and Microglia (BV2) Cell Coculture
Primary cultures (around 65% neurons + 35% glia) were prepared from embryonic (E14-15) cortex tissue obtained from fetuses of timed pregnant Sprague-Dawley rats. After removing the blood vessels and meninges, pooled cortices were trypsinized (0.05%; Invitrogen, Carlsbad, CA, USA) for 20 min at room temperature. After rinsing off trypsin with prewarmed Dulbecco's modified Eagle's medium (DMEM; Invitrogen), cells were dissociated by trituration, counted, and plated into 96-well (5.0 × 104/well) cell culture plates precoated with polyethylenimine (Sigma-Aldrich). The culture plating medium consisted of neurobasal medium (Invitrogen) supplemented with 2% heat-inactivated FBS, 0.5 mM
Stroke Surgery and Drug Administration
Adult male Sprague-Dawley rats were anesthetized with chloral hydrate [400 mg/kg, intraperitoneally (IP)]. The right distal brain of the MCA was ligated (MCAo) with a 10-0 suture using the methods previously described 26 . The ligature was removed after 60 min of ischemia to generate reperfusion injury. Core body temperature was monitored and maintained at 37°C. After recovery from the anesthesia, body temperature was maintained at 37°C using a temperature-controlled incubator. CX549 (1 mg/kg/day, IP), AMD3100 (1 mg/kg/day, IP), or vehicle was administered to the animals at 10 min after the onset of reperfusion and then daily for 4 days.
Behavioral Measurement
Two behavioral tests were used to analyze stroke behavior as previously described. Body asymmetry was analyzed using an elevated body swing test27,28. Neurological deficits were evaluated by the Bederson's test 29 .
Measurement of Cerebral Infarction
Five days after MCAo, animals were killed by decapitation. The brains were removed, immersed in cold saline for 5 min, and sliced into 2.0-mm-thick sections. The brain slices were incubated in 2% triphenyl tetrazolium chloride (TTC; Sigma-Aldrich), dissolved in normal saline for 10 min at room temperature, and then transferred into a 4% PFA solution for fixation. The area of infarction on each brain slice was measured double blind using a digital scanner and the Image Tools program (University of Texas Health Sciences Center, San Antonio, TX, USA), as previously described 30 .
Interleukin-6 (IL-6) and Tumor Necrosis Factor-α (TNF-α) Measurement
Cerebral cortical tissue was collected on day 5 after MCAo. Tissues were lysed in radioimmunoprecipitation assay (RIPA) lysis buffer (#20-188; Millipore, Darmstadt, Germany) containing protease inhibitor cocktails (#78442; Thermo Fisher Scientific) and then centrifuged at 16,000 × g for 10 min at 4°C. The protein concentration of the supernatant was determined by Bradford protein assay (Bio-Rad Laboratories, Hercules, CA, USA). IL-6 and TNF-α protein levels were measured using enzymelinked immunosorbent assay (ELISA) kits (#BMS625 and #BMS622; eBioscience, Waltham, MA. USA). The intensity of the horseradish peroxidase (HRP) enzyme reaction was measured at 450 nm using a microplate reader (BioTek Synergy™ HT, Winooski, VT, USA).
Cell Transplantation
Two sets of animals were used in the cell transplantation study. (A) The donor rats were treated with vehicle, AMD3100 (1 mg/kg), and CX549 (1 mg/kg). Two hours after drug administration, peripheral blood, collected through cardiac puncture, was mixed with αMEM/Hank's balanced salt solution (HBSS; Gibco, Thermo Fisher Scientific) mixture (2:1). Peripheral blood mononuclear cells (PBMCs) were separated on a Ficoll gradient by centrifugation at 400 × g for 20 min at room temperature. The PBMC pellet was resuspended in PBS at 1 × 107 cells/ml concentration, and each milliliter of cell suspension was labeled with chlormethylbenzamide-1,1′-diotadecyl-3,3,3′,3′-tetramethylindocarbocyanine perchlorate (CM-Dil; 1 μg/ml; Invitrogen) for 10 min at room temperature. (B) The recipient rats received a 60-min MCAo. The CM-Dil-labeled PBMCs (107 cells/ml) were transplanted through the right femoral vein at 5 min after the onset of reperfusion. A 12-h locomotor activity and Bederson's neurological test were examined on days 2 and 5 poststroke.
Immunohistochemistry
Animals were anesthetized and perfused transcardially with saline followed by 4% PFA in phosphate buffer (PB; 0.1 M; pH 7.2). The brains were dissected, postfixed in PFA for 48-72 h, and transferred to 20% sucrose in 0.1 M PB for at least 16 h. Serial sections of the entire brain were cut at 35-μm thickness on a freezing cryostat (model: CM 3050 S; Leica, Heidelberg, Germany). After blocking with 4% BSA and 0.5% Triton X-100 in 0.1 M PB, brain slices were incubated with antibodies against CD34 (monoclonal 1:100; Cat: 11-0341; eBioscience) at 4°C overnight. Sections were rinsed in 0.1 mol/L PB and were mounted on slides and coverslipped. Control sections were incubated without primary antibody. Confocal analysis was performed using a Nikon D-ECLIPSE 80i microscope (Nikon Instruments, Inc., Tokyo, Japan) and the EZ-C1 3.90 software (Nikon). The optical density of CD34 immunoreactivity or CM-Dil fluorescence was quantified in three consecutive brain sections with a visualized anterior commissure in each animal. Five to six photomicrographs were taken along the perilesioned region per brain slice. All immunohistochemical measurements were done by blinded observers.
Statistical Analysis
Data were expressed as mean ± standard error of the mean (SEM). Two-tailed unpaired Student's t-tests were performed to compare two groups. One-way analysis of variance (ANOVA) was used to compare three or more groups followed by Fisher's least significant difference (LSD) post hoc test. When two time points or two variables were included, a two-way ANOVA was applied followed by Fisher's LSD post hoc test. A value of p < 0.05 was considered significant. All analyses were performed with the SigmaPlot ver. 12.5 software (Systat Software Inc., Chicago, IL, USA).
Results
In Vitro Characterization of CX549 and AMD3100
The affinity for CXCR4 receptors was examined using a radioligand binding assay. Both AMD3100 and CX549 inhibited the specific binding of [125I]CXCL12 to the CXCR4 receptors. CX549 had a significantly high binding affinity to the CXCR4 receptor than did AMD3100 (p < 0.001, two-way ANOVA) (Fig. 1B). Similarly, CX549 was more potent than AMD3100 to inhibit CXCL12-mediated chemotaxis of CCRF-CEM cells, a human cell line with high CXCR4 expression (p = 0.009, two-way ANOVA) (Fig. 1C).
Neuroprotection in Cultured Neuronal Cells
The neuroprotective effect of CXCR4 antagonists was examined in primary cortical neuronal and microglia (BV2 cells) mixed cultures. Cells were treated with glutamate (100 μM) followed by CX549 or AMD3100 on DIV 10 and were fixed 2 days later. Glutamate significantly reduced MAP2 immunoreactivity (p < 0.001, F3,28 = 47.118) (Fig. 2A1 and A2) but increased IBA1 immunoreactivity (p < 0.001, F3,28 = 33.208) (Fig. 2B1 and B2). These responses were significantly antagonized by AMD3100 or CX549 (MAP2: p < 0.002; IBA1: p < 0.001) (Fig. 2A and B). These data suggest that AMD3100 or CX549 has neuroprotective and anti-inflammatory actions in the neuron/microglia mixed culture.
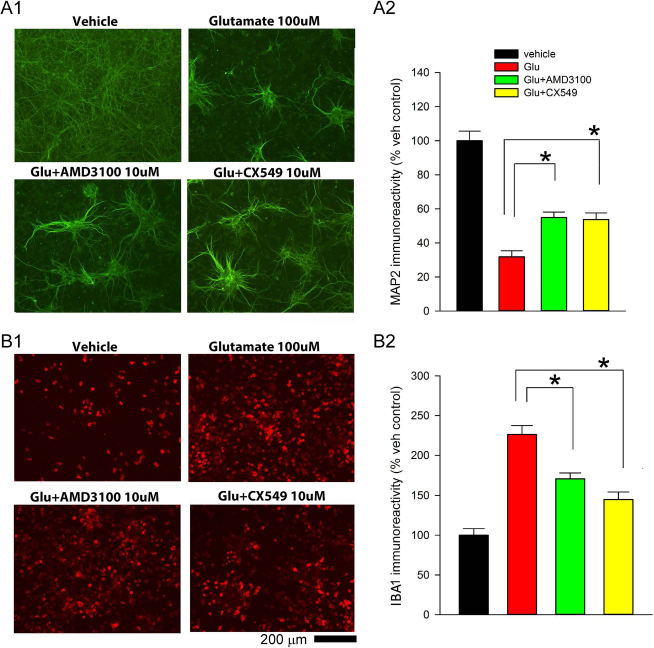
Neuroprotective and anti-inflammatory action of CX549 or AMD3100 in primary neuronal/microglia mixed cultures. Treatment with glutamate (A1) reduced MAP2, while (B1) increased IBA1, immunoreactivity. Coadministration with AMD3100 or CX549 significantly antagonized glutamate-mediated changes in (A2) MAP2 and (B2) IBA1 immunoreactivity. *p < 0.001. MAP2, microtubule-associated protein 2; IBA1, ionized calcium-binding adaptor molecule 1.
Early Posttreatment with CX549 or AMD3100 Improved Behavioral Functions in Stroke Rats
A total of 36 adult rats were used for this part of the study. Of these, 28 rats were subjected to a 60-min MCAo, and 8 rats were used as a nonstroke control. Stroke animals received CX549 (1 mg/kg/day, IP, n = 7), AMD3100 (1 mg/kg/day, IP, n = 7), or vehicle (n = 14) after MCAo (day 0). Animals were placed in activity chambers for 1 h on day 2. All stroke animals showed a significant reduction in locomotor activity, compared to the nonstroke controls (p < 0.05) (Fig. 3A-E). Treatment with CX549, compared to vehicle, significantly improved the locomotor activity in the stroke rats (horizontal activity: p = 0.039, F3,32 = 12.165; vertical movement number: p = 0.009, F3,32 = 12.602; total distance traveled: p = 0.044, F3,32 = 9.511; vertical activity: p = 0.013, F3,32 = 8.798; movement time: p = 0.043, F3,32 = 10.966) (Fig. 3A-E). In contrast, AMD3100 did not show significant improvement (horizontal activity: p = 0.309; vertical movement number: p = 0.219; total distance traveled: p = 0.208; vertical activity: p = 0.501; movement time: p = 0.255) (Fig. 3A-E). The neurological tests were conducted on days 2 and 4. CX549 and AMD3100 significantly reduced neurological deficits in the stroke rats (p = 0.002, F2,38 = 7.541) (Fig. 3F).
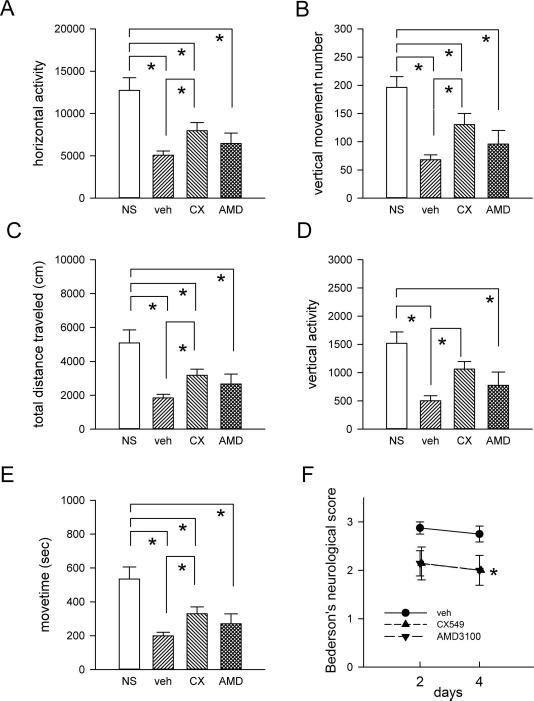
Early posttreatment with CX549 or AMD3100 improved behavioral functions in stroke rats. (A-E) Locomotor activity was recorded for 1 h at 2 days after MCAo. Posttreatment with CX549 significantly increased (A) horizontal activity (p = 0.039), (B) vertical movement number (p = 0.009), (C) total distance traveled (p = 0.044), (D) vertical activity (p = 0.013), and (E) movement time (p = 0.043). (F) Bederson's neurological test was conducted at 2 and 4 days after MCAo. Both CX549 and AMD3100 significantly reduced neurological deficits in the stroke rats (p = 0.002). MCAo, middle cerebral artery occlusion.
Posttreatment with CX549 or AMD3100 Reduced Brain Infarction
A total of 21 rats were treated with CX549, AMD3100, or vehicle after MCAo for 5 days. Animals were sacrificed on day 5. The brains were removed and sliced into 2.0-mm-thick sections for TTC staining. Typical brain infarction from each group was demonstrated in Figure 4A. Treatment with AMD3100 or CX549 significantly reduced the total infarct volume (p < 0.05, F2,18 = 5.045) (Fig. 4B). To examine the topographic relationship of protection, the area of infarction in each slice was compared with animals receiving the vehicle. Using a two-way ANOVA, we found that treatment with CX549 or AMD3100 significantly lowers infarction (p < 0.001, F2,126 = 24.764). Post hoc analysis revealed that the significant differences in brain infarction were localized mainly on slices 4 to 7 among the CX549, AMD3100, and vehicle groups (p < 0.05) (Fig. 4C).
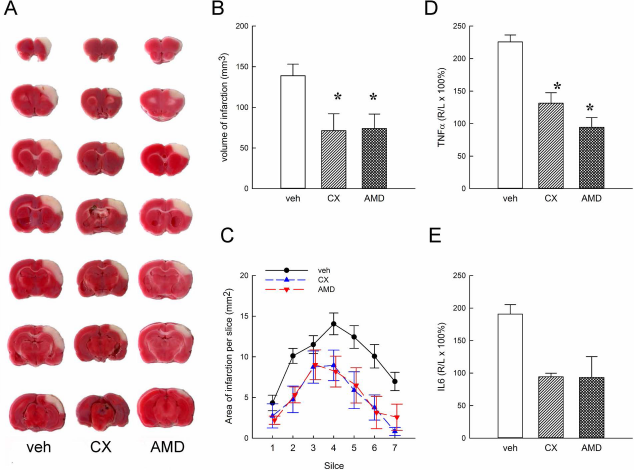
Systemic administration of CX549 and AMD3100 reduced infarction volume and the expression of TNF-α and IL-6 in stroke brain. Stroke rats received vehicle, CX549, or AMD3100 injection. Animals were sacrificed 5 days after MCAo. (A) Representative TTC-stained brain sections from each group. (B) CX549 and AMD3100 significantly reduced infarction volume compared to vehicle. (C) Area of infarction per 2-mm section (seven sections per each brain; anterior to posterior) was significantly reduced in the CX549 and AMD3100 groups. Treatment with CX549 or AMD3100 reduced (D) TNF-α and (E) IL-6 levels. *p < 0.05. TNF-α, tumor necrosis factor-α; IL-6, interleukin-6; TTC, triphenyl tetrazolium chloride.
CX549 and AMD3100 Reduced IL-6 and TNF-α in the Ischemic Cortex
Cortical tissue from animals receiving drug treatment was harvested 5 days after MCAo. A twofold increase in TNF-α and IL-6 expression was found in the lesioned side (R) cortex, compared to the nonlesioned side (L) cortex (Fig. 4D and E). Treatment with CX549 or AMD3100 significantly reduced TNF-α levels (p < 0.01, F2,5 = 16.905) (Fig. 4D). A similar trend in the reduction of IL-6 was found in animals receiving CX549 or AMD3100 (p = 0.059, F2,5 = 5.239) (Fig. 4E).
CX549 and AMD3100 Mobilized Hematopoietic Stem Cells to Peripheral Blood
We previously demonstrated that AMD3100, at a dose of 5 mg/kg, mobilized CD34+/CD45+ and CD34+/CD45+/CXCR4+ stem cells in C57BL/6 mice 31 . To examine the mobilizing effect, AMD3100, CX549 (5 mg/kg), or vehicle was administered to adult C57BL/6 mice (n = 12). Blood samples were collected at 2 h after injection for flow cytometry. AMD3100 or CX549 significantly increased circulating CXCR4+/CD34+ stem cells (p < 0.02, F2,9 = 11.121) (Fig. 5A) and CXCR4+/CD133+ stem cells (p < 0.05, F2,9 = 9.260) (Fig. 5B). A significant increase in the circulating CXCR4+/CD133+ stem cells was found after CX549 injection compared to AMD3100 injection, suggesting that CX540 was more efficient to mobilize HSCs to the peripheral blood.

CX549 and AMD3100 mobilized hematopoietic stem cells. Application of CX549 and AMD3100 significantly increased circulating (A) CXCR4+/CD34+ and (B) CXCR4+/CD133+ cells in mice *p < 0.05; #p = 0.015. CXCR4, C-X-C chemokine receptor type 4.
Behavioral Recovery After Transplantation of PBMCs From Animals Receiving CX549 or AMD3100
PBMCs were collected from 17 donor rats 2 h after receiving vehicle, CX549 (1 mg/kg, SC), or AMD3100 (1 mg/kg, SC). Cells were labeled with CM-Dil, resuspended in PBS at 1 × 107 cells/ml concentration and then transplanted intravenously to the stroke rats (n = 17) at 5-10 min after reperfusion. Locomotor activity was examined 4 days after MCAo for 12 h. Stroke animals receiving transplants from the CX549 or AMD3100 donors, compared to vehicle donors, had a significant improvement in horizontal activity (p < 0.005, F2,42 = 5.774, two-way ANOVA + Fisher's LSD test) (Fig. 6A), vertical movement number (p < 0.02, F2,42 = 5.935) (Fig. 6B), total distance traveled (p < 0.05, F2,42 = 3.558) (Fig. 6C), and movement time (p < 0.01, F2,42 = 5.369) (Fig. 6E). Vertical activity was significantly improved after transplantation of PBMCs from donors receiving AMD3100 (p = 0.045) (Fig. 6D). Bederson's neurological test was conducted at 2 and 4 days after MCAo. Both CX549 and AMD3100 significantly reduced neurological deficits in the stroke rats (p = 0.011, F2,34 = 5.112, two-way ANOVA) (Fig. 6F).

Transplantation of PBMCs from the donors receiving CX549 or AMD3100 induces behavior recovery. PBMCs were collected from donor rats receiving vehicle, CX549, or AMD3100. These cells were transplanted to stroke rats after reperfusion. (A-E) Locomotor activity was examined 4 days after MCAo for 12 h. Stroke animals receiving transplants from the CX549 or AMD3100 donors had significant improvement in horizontal activity (A) (p = 0.006), vertical movement number (B) (p = 0.005), total distance traveled (C) (p = 0.037), and movement time (E) (p = 0.008). Vertical activity was significantly improved after transplantation of PBMCs from donors receiving AMD3100 (D) (p = 0.045). Bederson's neurological test was conducted at 2 and 4 days after MCAo. Both CX549 and AMD3100 significantly reduced neurological deficits in the stroke rats (F) (p = 0.011). PBMCs, peripheral blood mononuclear cells; MCAo, middle cerebral artery occlusion.
Migration of Grafted Hemopoietic Stem Cells in the Host Brain
At 5 days after transplantation, the recipient animals were sacrificed for histological examination. CM-Dil+ cells were found in the penumbra of the lesioned cortex. No CM-Dil+ cells were found in the contralateral hemisphere. Since an equal amount of PBMCs was transplanted to each stroke rat, the density of CM-Dil+ cells in the lesioned brain was not significantly different among the three recipient groups (p = 0.212) (Fig. 7A and B). In contrast, the density of CD34 immunoreactivity, representing the HSCs, was significantly enhanced in the lesioned cortex in animals receiving the PBMC grafts from the CX549 or AMD3100 donors (p < 0.05) (Fig. 7C and D).

Migration of CD34 cells to the stroke brain. The PBMCs were collected from the donor animals receiving vehicle, CX549, or AMD3100. These cells were labeled with CM-Dil dye and transplanted into the stroke rats. Animals were sacrificed at 5 days after grafting for histological analysis. (A) CM-Dil+ cells were found in the penumbra of the lesioned cortex in all recipient animals. (B) Density of CM-Dil+ cells in the lesioned brain was not significantly different among the three groups of recipients (p = 0.212). (C) Cells expressing CD34 immunoreactivity were found in the lesioned cortex in animals receiving PBMC transplants. (D) CD34 cell density in the lesioned cortex was significantly increased in the host animals receiving the PBMC graft from the CX549 or AMD3100 donors (p < 0.05). PBMCs, peripheral blood mononuclear cells; CM-Dil, chlormethylbenzamide-1,1′-diotadecyl-3,3,3′,3′-tetramethylindocarbocyanine perchlorate.
Discussion
In this study, we characterized the pharmacological properties of a novel CXCR4 antagonist, CX549, and its protective effect against stroke. CX549 had a high affinity to CXCR4 and was more potent than AMD3100 to inhibit CXCL12-mediated chemotaxis in culture. CX549 effectively reduced the activation of microglia and improved neuronal survival after injury in neuron/microglia cocultures. Early poststroke treatment with CX549 significantly improved behavior function, reduced the brain infarction, and suppressed the expression of inflammatory markers. Our data supported that CX549 is a potent anti-inflammatory agent and is neuroprotective against ischemic stroke.
It has been shown that AMD3100, given from day 2 to day 5 after MCAo, reduced neurological score in adult rats on day 5 12 . Early posttreatment with AMD3100 reduced brain infarct and edema 11 , attenuated the number of microglia or inflammatory cells in the peri-infarct zone, and reduced neurological deficits in stroke mice10,11. Similar neuroprotective effects of CX549 were found in our study. CX549, given after MCAo, reduced brain infarction and suppressed the expression of TNF-α. Both CX549 and AMD3100 reduced neurological deficits as seen in the Bederson's test. However, CX549, but not AMD3100, significantly improved locomotor activity in stroke rats. The difference in locomotor recovery may be attributed to the higher affinity and chemotaxis inhibition by CX549. The correlation of locomotor function with these factors requires further investigation.
CXCR4 antagonists can mobilize BM-HSCs to the peripheral blood13,16. We found that CX549 and AMD3100 significantly increased circulating CXCR4+/CD34+ and CXCR4+/CD133+ cells. CX549 was more potent than AMD3100 in mobilizing CXCR4+/CD133+ cells, suggesting CX549 was more efficient to mobilize HSCs to the peripheral blood. The mobilizing effect of CX549 was next examined through cell transplantation. CM-Dil-labeled PBMCs from the donors receiving CX549 or AMD3100 were grafted to the stroke animals. CM-Dil and CX34+ cells were found in the penumbra of the recipients. The density of CD34 immunoreactivity was significantly enhanced in the lesioned cortex in animals receiving the PBMC grafts from the CX549 or AMD3100 donors. These data suggest that CX549 mobilizes the CD34+ cells to the peripheral blood in donors, and these cells migrated to the host brain.
The homing of grafted HSCs to the lesioned site in the brain is also regulated by the CXCR4/CXCL12. Overexpressing CXCR4 in the MSC transplants increased the cell migration to the ischemic boundary and improved behavioral recovery in stroke rats 24 . AMD3100 reduced the chemotaxic effect and prevented the migration of BM-HSCs to the lesioned brain region in stroke animals 8 . Furthermore, inhibiting CXCR4 by the CXCR4-blocking antibody reduced the migration of subventricular zone neural progenitor cells to the lesioned brains 2 . In our study, CXCR4 antagonists were not applied to the stroke rats, and the PBMCs were washed before transplantation. It is likely that the SDF-1 signal for migration was not reduced, which was further supported by the enhanced migration of CD34+ cells to the penumbra after transplantation.
Stroke animals receiving transplants from the CX549 or AMD3100 donors had significant improvement in locomotor activity and reduction of neurological scores. It has been well documented that transplantation of BM-HSCs induced behavioral improvement in stroke animals18,19,21. The cells, transplanted peripherally, provided trophic factors for protection 18 . Grafted cells can be differentiated into neuron-like cells for neurorepair in the stroke brain 19 21 . In our study, behavioral recovery was found early (i.e., days 2 and 5) after transplantation. It is possible that the recovery in behavior in this early stage may be attributable to the indirect trophic responses from the HSCs.
In conclusion, we demonstrated the neuroprotective effect of a novel CXCR4 antagonist, CX549, against ischemic brain injury. CX549 reduced inflammation and neurodegeneration in the poststroke brain. Compared to AMD3100, CX549 has a higher affinity for CXCR4 and is more efficient in mobilizing HSCs and induces behavioral improvement. Our data support that CX549 may have clinical implications for the treatment of stroke.
Footnotes
Acknowledgments
This study was supported by the National Health Research Institutes and the Ministry of Science and Technology, Taiwan. CX549 has been filed for the US patent application by Shia et al. (US 2016/0083369 A1).
