Abstract
Postreplicative mismatch repair safeguards the stability of our genome. The defects in its functioning will give rise to microsatellite instability. In this study, 50 meningiomas were investigated for microsatellite instability. Two major mismatch repair genes, MLH1 and MSH2, were analyzed using microsatellite markers D1S1611 and BAT26 amplified by polymerase chain reaction and visualized by gel electrophoresis on high-resolution gels. Furthermore, genes DVL3 (D3S1262), AXIN1 (D16S3399), and CDH1 (D16S752) were also investigated for microsatellite instability. Our study revealed constant presence of microsatellite instability in meningioma patients when compared to their autologous blood DNA. Altogether 38% of meningiomas showed microsatellite instability at one microsatellite locus, 16% on two, and 13.3% on three loci. The percent of detected microsatellite instability for MSH2 gene was 14%, and for MLH1, it was 26%, for DVL3 22.9%, for AXIN1 17.8%, and for CDH1 8.3%. Since markers also allowed for the detection of loss of heterozygosity, gross deletions of MLH1 gene were found in 24% of meningiomas. Genetic changes between MLH1 and MSH2 were significantly positively correlated (p = 0.032). We also noted a positive correlation between genetic changes of MSH2 and DVL3 genes (p = 0.034). No significant associations were observed when MLH1 or MSH2 was tested against specific histopathological meningioma subtype or World Health Organization grade. However, genetic changes in DVL3 were strongly associated with anaplastic histology of meningioma (χ2 = 9.14; p = 0.01). Our study contributes to better understanding of the genetic profile of human intracranial meningiomas and suggests that meningiomas harbor defective cellular DNA mismatch repair mechanisms.
Keywords
Introduction
Being one of the most common primary intracranial tumors and accounting for ~30% of intracranial neoplasms,1,2 meningiomas are slowly growing and benign with long survival times and low rates of recurrence. They originate from the arachnoidal cap cells of the leptomeninges. Although most meningiomas are classified as World Health Organization (WHO) grade I,3–5 histologically distinct subtypes of benign meningiomas may exhibit higher risk of recurrence, 2 and some are prone to evolve into atypical and anaplastic subtypes. 6 About 5%–7% of all meningiomas shows increased mitotic activity and a higher risk for recurrence and are classified as WHO grade II (atypical). Furthermore, ~3% that display aggressive malignant behavior associated with brain invasion7–9 are classified as anaplastic meningiomas (WHO grade III) showing low survival rates of only 14.2%.
Genomic instability is a prominent characteristic of tumor cells and the result of defective DNA repair mechanisms.10,11 Microsatellite instability (MSI) is a type of genomic instability caused by malfunctioning of DNA mismatch repair (MMR).12,13 It recognizes and corrects mismatched bases that happen due to errors by DNA polymerases as they synthesize short repeats termed microsatellites.14–16 The number of repeats for any microsatellite is constant in a healthy individual but polymorphic across populations. 11 The defects in the repair of short mismatches result in changes in the number of these repetitive sequences in tumor as compared to normal autologous DNA. MMR machinery numbered eight genes in human: hMSH2 (2p21), hMSH3 (5q14.1), hMSH5 (6p21.3), hMSH6 (2p16), hMLH1 (3p21), hPMS1 (hMLH2) (2q31.1), hMLH3 (14q24.3), and hPMS2 (hMLH4) (7q22.2).17–19 Mutations of those genes generate insertions and deletions in microsatellites but also random mutations throughout the genome which creates genomic instability and contributes to neoplastic transformation and progression.20,21
The importance of MSI phenotype in human tumors is an emerging field demonstrating its involvement in the clinical course of the disease, response to therapy, and survival outcomes.11,19,22–24 The presence of MSI in colon cancer has been known for a long time.25–27 It has been shown to be especially important for Lynch syndrome which is characterized with an increased risk of colon cancer and cancers of the endometrium, ovary, stomach, small intestine, hepatobiliary tract, urinary tract, brain, and skin and is associated with germline pathogenic mutations in the MMR genes.11,23,28–31 Additionally, many sporadic cancers also harbor MSI. 32 Besides colon cancer, where mutations in the MMR genes are found in about 17% of sporadic cases, MSI is involved in other sporadic cancers too.11,24,33–36 Tumors with MSI are usually categorized as MSI-High (MSI-H), MSI-Low (MSI-L), or microsatellite stable (MSS) depending on the number of microsatellite markers showing MSI. This classification is relevant for prognostic and predictive purposes,37–40 whereas MSI-H tumors have a more positive prognosis when compared to MSI-L or MSS tumors. 10
New research indicates MSI involvement in brain tumors too.20,33,41 It has been shown that subsets of meningiomas exhibit MSI.20,41,42 Nevertheless, the frequency with which MSI is present in meningioma is still controversial.43,44 Meningiomas can demonstrate variability in the biological characteristics and clinical behavior raising many questions that need to be answered in their molecular profiles. The aim of this study is to investigate the presence of MSI in meningiomas by analyzing microsatellite markers for MLH1, MSH2, DVL3, AXIN1, and CDH1 genes and correlate genetics to clinical and histopathological findings. We decided to investigate the involvement of two major MMR genes, MLH1 and MSH2, in our group of collected intracranial meningiomas, which are the highest targeted ones in human tumors.29,37,39,45,46
Materials and methods
Tumor specimens
In all, 50 meningioma samples together with corresponding autologous blood tissue were collected with patients’ consents from the Department of Neurosurgery and “Ljudevit Jurak” Department of Pathology, University Hospital “Sisters of Mercy”, Zagreb, Croatia. Using the magnetic resonance imaging (MRI), tumor lesions were found in different cerebral regions, with the surrounding zone of perifocal edema. During the operative procedure, the tumor was maximally reduced using a microneurosurgical technique. The patients had no family history of brain tumors and did not undergo chemotherapy or radiotherapy prior to surgery. Collected tumor tissues were frozen in liquid nitrogen and were kept at −80°C. The peripheral blood samples were collected in ethylenediaminetetraacetic acid (EDTA) and processed immediately. All tumors were studied by pathologists and classified according to the WHO criteria.4,5 There were 20 meningothelial meningiomas, 6 fibrous (fibroblastic), 15 transitional (mixed), 1 psammomatous, 3 angiomatous, 2 atypical, and 3 anaplastic. Clinical and pathological data are shown in Table 1; 28 patients were female and 22 males. The age of patients varied from 32 to 79 years (mean age = 63 years, median = 66 years). The mean age at diagnosis for females was 65.93 (median = 67.00) years and for males 58.86 (median = 63.00) years.
Meningioma pathohistological subtype, demographic variables, the polymorphic status of microsatellite markers, MSI, and LOH of MLH1, MSH2, DVL3, AXIN1, and CDH1 genes.

MSI: microsatellite instability; LOH: loss of heterozygosity; HETERO: heterozygous samples; HOMO: homozygous uninformative samples; ND: not determined; I: WHO grade one; II: WHO grade two; III: WHO grade three.
Ethical approval was received from the Ethical Committees of School of Medicine University of Zagreb and University Hospital “Sisters of Mercy”, and the patients gave their informed consent.
DNA extraction
Approximately 0.5 g of tumor tissue was homogenized with 1 mL extraction buffer (10 mM Tris-HCl; 0.1 M EDTA; 0.5% sodium dodecyl sulfate, pH 8.0) and incubated with 100 mg/mL proteinase K (Sigma-Aldrich, St. Louis, MO, USA) overnight at 37°C. Phenol/chloroform extraction and ethanol precipitation followed. Blood was used to extract leukocyte DNA; 5 mL of blood was lysed with 15 mL of red blood cells lysis buffer (RCLB); 155 mM NH4Cl; 0.1 mM EDTA; 12 mM NaHCO3) and centrifuged (15 min/5000g) at 4°C. The pellet was further processed same as for DNA extraction from the tissue samples.
Polymerase chain reaction and MSI
Selected microsatellite markers D1S1611, BAT26, D3S1262, D16S3399, and D16S752 were amplified by polymerase chain reaction (PCR) in a volume of 25 µL. The optimal conditions were as follows: 5 pmol of each primer, 200–400 ng DNA, 2.5 µL PCR buffer (500 mM KCl, 100 mM Tris-HCl, pH 8.3), 2 mM MgCl2, 2.5 mM of each deoxynucleotide triphosphates (dNTP), and 5 U/µL Taq polymerase (Promega, Madison, WI, USA). PCR amplifications were carried out in an Eppendorf Mastercycler Personal thermocycler (Hamburg, Germany). Gene markers, primers, PCR product size, and chromosomal location of the marker are given in Table 2.
Microsatellite markers and primers used, PCR product sizes, and chromosomal location of the marker.

Optimal PCR conditions for D1S1611 marker for MLH1 gene were as follows. Reaction was preceded by 5-min initial denaturation at 96°C. Samples were subjected to 40 cycles of three-step amplification consisting of 30 s of denaturation at 96°C, 30 s of annealing at 58°C, and 30 s of extension at 72°C adding 1 extra second after every cycle. A final extension at 72°C was lengthened to 10 min.
Marker BAT26 was used for MSH2.29,47 Optimal PCR conditions for BAT26 marker amplification were as follows. Reaction was preceded by 5-min initial denaturation at 96°C. Samples were subjected to 40 cycles of three-step amplification consisting of 30 s of denaturation at 96°C, 30 s of annealing at 57°C, and 30 s of extension at 72°C adding 1 extra second after every cycle. A final extension at 72°C was lengthened to 10 min.
The detection of MSI of DVL3 gene was achieved by the optimization of the PCR conditions of marker D16S1262 as follows; reaction was preceded by 5-min initial denaturation at 94°C. Samples were subjected to 40 cycles of three-step amplification consisting of 30 s of denaturation at 94°C, 35 s of annealing at 56°C, and 30 s of extension at 72°C. A final extension at 72°C was lengthened to 10 min.
Optimal amplification conditions for D16S3399 polymorphic sequence-tagged site (STS) marker for the AXIN1 gene were as follows. Reaction was preceded by 3-min initial denaturation at 95°C. Samples were subjected to 35 cycles of three-step amplification consisting of 30 s of denaturation at 96°C, 30 s of annealing at 56°C, and 30 s of extension at 72°C adding 1 extra second after every cycle. A final extension at 72°C was lengthened to 10 min.
The D16S752 (GATA51G03) polymorphic region linked to the E-cadherin gene was amplified under following PCR conditions: initial denaturation, 3 min/96°C; denaturation, 30 s/96°C; annealing, 35 s/55°C; extension, 30 + 1 s/72°C; final extension, 72°C/10 min; 35 cycles.
All PCR products were analyzed on 2% agarose gels. The selection of gene markers was based on several criteria: the gene marker had to be either intragenic or close to the selected gene, the selected gene marker needed to be variable in the population to ensure enough heterozygous (informative) patients, or the pseudomonomorphic marker would be informative in the case of genetic instability; amplified markers did not exceed 300 base pairs to ensure successful amplification.
To discover MSI in meningioma patients, samples were visualized on Spreadex EL 400 Mini gels (Elchrom Scientific, AL-Labortechnik, AL-Diagnostic GmbH, Amstetten, Austria) stained with SYBR Gold (Invitrogen, Molecular Probes, Eugene, OR, USA). Samples were considered positive for MSI if additional DNA bands were observed or the existing DNA band had shifted position in comparison to bands of autologous blood tissue indicating impaired cellular DNA MMR in tumor cells. Microsatellite markers also revealed loss of heterozygosity (LOH) in several cases. LOH of the gene was considered to have occurred if one out of two alleles in heterozygous samples was missing in comparison to the alleles from autologous blood sample.
Statistical analysis
The sex, age, pathohistological subtype, WHO grade, MSI, and LOH of MLH1 MSH2, DVL3, AXIN1, and CDH1 were coded and entered into a data base. Age was coded into one of the two possible categories: a group consisting of patients ≤66 years and a group of >66 years. The reason is that 66 years is the cut-off point of the 50 percentile of the total sample. In other words, this value divides the total sample into two almost (26 vs 24 individuals) equal parts and gives us two reasonably well-defined and equal groups of patients to compare: a younger group comprising individuals younger than 66 years whose mean age is 53.8 years and an older group comprising individuals older than 66 years whose mean age is 71.8 years. The correlations between MSI and various meningioma subtypes and the analyzed demographic features were analyzed with Spearman’s rank-order correlation test. Spearman’s rank-order correlation is the nonparametric version of the Pearson product–moment correlation. Spearman’s rho correlation coefficient (ρ) measures the strength and direction of association between two ranked variables and takes a value between minus one and plus one. A positive correlation signifies that the ranks of both the variables are increasing, while a negative correlation signifies that as the rank of one variable increases, the rank of the other variable decreases. Statistical significance was set at p < 0.05. All statistical evaluations were performed with the SPSS statistical package 14.0 (SPSS Inc., Chicago, IL, USA).
Results
In this study, we investigated the involvement of MSI in histologically diverse group of intracranial meningiomas. Samples harboring MSI tumor phenotype can be visualized on high-resolution gels after electrophoresis techniques as samples with bands on different positions in comparison to bands of autologous normal tissue. We found MMR repair machinery to be targeted in 58% of our total meningioma sample tested with five MSI markers (Figure 1); 38% of investigated samples showed MSI at one microsatellite locus, 16% on two, and 13.3% on three microsatellite loci.

Spreadex gels stained with SYBR Gold showing microsatellite instability (MSI) and loss of heterozygosity (LOH) when tumor and blood DNA were compared: (a) D1S1611 microsatellite marker for MLH1 gene, (b) D16S3399 microsatellite marker for AXIN1, (c) D16S1262 microsatellite marker for DVL3, (d) microsatellite marker for CDH1 gene, (e) microsatellite marker for MSH2 gene (M: molecular DNA standards; lanes 1, 3, 5, and 7: meningiomas; lanes 2, 4, 6, and 8 autologous DNA from blood).
Each microsatellite marker employed demonstrated constant finding of relatively low frequency of MSI in meningioma when compared to autologous normal DNA extracted from blood of the same patient. We found MSI for DVL3 gene in 22.9%, for AXIN1 in 17.8%, and for CDH1 in 8.3% of investigated informative samples.
Next, we were interested to determine whether any particular MMR gene is involved and analyzed two major ones implicated in the MMR in the majority of tumors reported in the literature, namely, MSH2 and MLH1. Therefore, we chose intragenic markers: for MLH1, a marker that is in its exon 15—D3S1611 and for MSH2 monomorphic marker BAT26 within its intron 5. There are four possible alleles for D3S1611, 34 while BAT26 locus contains a sequence of 26-mononucleotide repeat of deoxyadenine. We demonstrated that genes MSH2 and MLH1 were changed in our group of collected meningiomas. The percent of detected MSI for MSH2 was 14% and for MLH1 it was 26%.
Since polymorphic microsatellite markers also allow for the detection of LOH, our meningioma sample also showed 24% of LOH of the MLH1 gene in the tumor DNA when compared to constitutive autologous DNA. LOH of DVL3 was noted in 4.17%, of AXIN1 in 6.67%, and of CDH1 in 37.5% (Figure 2).
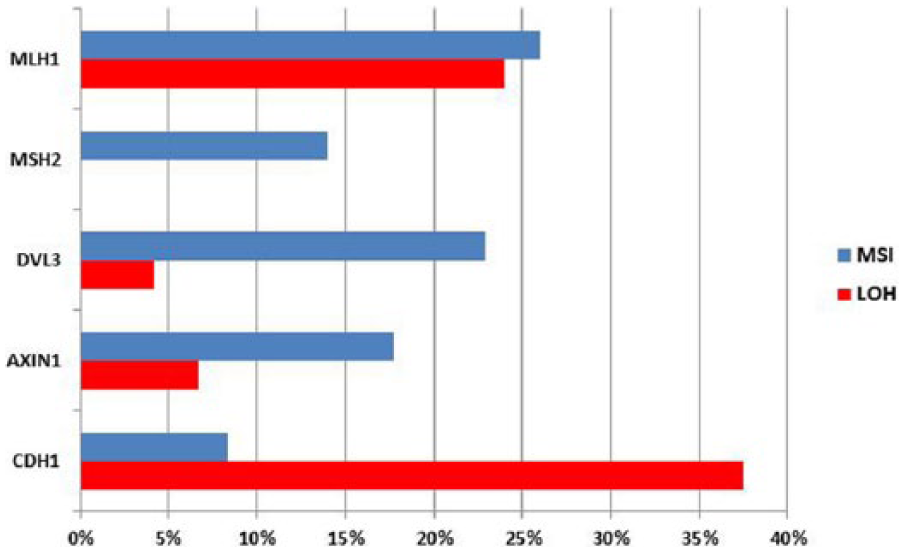
The percent of patients showing MSI and LOH on total meningioma sample.
When statistical analysis was performed on our results, we found that genetic changes on MLH1 and MSH2 were significantly positively correlated (Spearman’s rho = 0.305; p = 0.032; Table 3). In other words, patients exhibiting changes on MLH1 also harbored changes on MSH2.
Correlation between MLH1 and MSH2 genetic changes in meningioma patients.

Correlation is significant at the 0.05 level (two-tailed).
No such correlation was noted when changes on both or either MLH1 and MSH2 genes were tested against changes on either DVL3, AXIN1, and CDH1 genes (Spearman’s rho values are from −0.155 to 0.141; p values are between 0.784 and 0.308). When, however, genetic changes on MSH2 gene were analyzed independently, a strong positive correlation was noted with changes in DVL3 gene (Spearman’s rho = 0.303; p = 0.034). Next, genetic changes were correlated with clinicopathological parameters. When genetic changes on either MLH1 or MSH2 were tested against specific histopathological meningioma subtype, no significant associations were observed on any gene investigated (χ2 values ranged from 2.32 to 10.3; p values ranged from 0.112 to 0.888). Similarly, when genetic changes on either MLH1 or MSH2 were tested against two composite grade categories comprising lower (I) and higher (II and III) grades, no significant associations were also detected. The same applies to the genetic changes in DVL3, AXIN1, and CDH1 that were not associated with two grade categories. When, however, a more nuanced pathohistological grade analysis was employed including separate benign grade I, atypical grade II, and anaplastic grade II, genetic changes in DVL3 were strongly associated with anaplastic histology of meningioma (grade III; χ2 = 9.14; p = 0.01). We checked expression levels of DVL3 by immunohistochemistry and noted that in two out of three anaplastic cases, the protein was strongly expressed both in cytoplasm and nucleus.
We also investigated the association of epidemiological characteristics to MMR gene changes and the presence of MSI and LOH. When we divided our meningioma sample into two composite age categories, one comprising patients 65 years or younger (n = 23) and the other 66 years or older (n = 27), no significant correlation between age groups and genetic change on any of the five analyzed genes was noted (Spearman’s rho values from 0.003 to −0.030; p values from 0.528 to 0.982). Additionally, the distribution of the observed genetic changes was not statistically different between sexes for all investigated genes (Spearman’s rho values from 0.06 to 0.448; p values from 0.448 to 0.780).
Discussion
In this study, MSI status for D16S3399, D3S1262, and D16S752 loci was evaluated. At the same time, two major MMR genes MLH1 and MSH2 were investigated in the same group of meningioma patients. Altogether 58% of MSI cases were found. Our findings on MSI in MSH2 and MLH1 genes that exhibit frequencies of 14% and 26%, respectively, as well as the detection of LOH of MLH1 gene in 24% of cases, indicate that they are implicated in meningioma pathology. LOH of the MLH1 gene may also indicate that patients harboring gross deletions have a lost or nonfunctional protein.
There are different reports on the frequency of MSI in meningioma with different conclusions on its importance. Studies that support our findings are reported by Pykett et al., 41 who found 25% of MSI. Furthermore, Alvino et al. 48 report on 20%, Chen et al. 42 on 11.6%, Sobrido et al. 20 on 6.3%, while Zhu et al. 49 and Kirsch et al. 50 report on 2.4% and 1.8% of MSI, respectively. In contrast, several authors report a lack of MSI in meningioma.43,44 While some authors believe that MSI is unimportant in meningioma, we believe that the evidence collected by numerous investigators, including also this study, supports the hypothesis that MSI is part of the meningioma molecular profile.
Epigenetic silencing of MMR genes through miRNAs or promoter hypermethylation is also sufficiently found in meningioma. Chen et al. 42 found 18% of meningiomas with hypermethylated hMLH1 promoter. Since LOH usually functions with promoter hypermethylation of the other allele, this could explain our findings of MLH1 gene’s LOHs.
Three microsatellite markers that we analyzed are in close proximity to genes involved in Wnt signaling. Several new studies show that Wnt pathway plays important roles in meningioma.8,51–56 These studies concentrated on mutational analyses, proteomics, and so on, but did not search for MSI of Wnt signaling genes. In our previous work, we came across MSI for AXIN1 and CDH1 that in our opinion could not be neglected 57 and decided to explore this line of evidence on a larger group of meningiomas. Consequently, in this study, we found a higher percent of MSI for AXIN1 gene (17.8%), while CDH1 displayed similar frequency (8.3%). DVL3 gene was studied for the first time in meningiomas and 22.9% of MSI was revealed. Genetic changes of the DVL3 gene were positively correlated to MSH2 changes (p = 0.034) and associated with anaplastic (grade III) histology of meningioma (p = 0.01).
Our findings on the significant positive correlation between genetic changes in MLH1 and MSH2 (p = 0.032) suggest that dysfunctional MMR mechanisms could affect multiple levels of repair in certain meningioma patients. Similar findings were observed for other cancers. Studies of the expression of MutS homologs in European colorectal carcinoma patients demonstrated lower expression levels of all MMR genes. 58 Moreover, the correlation of simultaneous loss of expressions between different MMR proteins in various tumors was reported by several authors.59–61
Microsatellite marker BAT26 is located in the fifth intron of the MSH2 gene, just 3ʹ of MSH2 exon 5. Because of the monomorphic nature, BAT26 is very sensitive, reliable, and a widely used marker to establish the MSI status of human tumors.29,37,39,46 Some authors consider it to be able to determine the MSI status of tumors even without matching normal DNA.29,47,62,63 Although there are reports on the polymorphic nature of BAT26,39,64 the allelic pattern of BAT26 described for European population is homogenous, with one major allele and small ranges of normal size variation. To improve reliability and rule out the possibility of polymorphic nature of BAT26, we compared meningioma to autologous blood DNA of the same patient, so variations in size observed in tumor can be solely attributed to the MSI.
Our results on 8.3% of MSI detected with D16S752 bring new perspectives on the involvement of a new phenomenon in genomic instability field, the so called elevated microsatellite alterations at selected tetranucleotide (EMAST) repeats. 38 The role this specific form of MSI plays in a cell is still unknown and it has not been addressed by the Bethesda definitions. 26 However, it has been shown that in most investigated cancers, instability occurs at loci with (AAAG)n or (ATAG)n repeats 38 and D16S752 is a tetranucleotide (ATAG)n repeat.
No significant associations were observed when we tested MLH1 or MSH2 genes against specific histopathological meningioma subtype or WHO grade. The findings on this even distribution of genetic changes among different histopathological subtypes, as well as the finding that the changes of MLH1 and MSH2 could not be significantly associated with any specific grade, indicate that the MMR repair defects may be continuous characteristic of some meningioma cases rather than an indication of tumor progression. Furthermore, we showed that genetic alterations found in our group of meningioma patients could not be significantly attributed to any particular age group. This could also support the conclusion that changes in MMR genes are constant and do not represent a later event connected to progression.
When we systematize results obtained for the MSI spectrum in meningioma,65–67 it is obvious that the overall number of investigated sporadic cases remains rather small. Our meningioma group represents relevant sample size, and this study is the first to report on parallel changes in two major MMR genes. As far as we are aware, genetic changes on MLH1 in meningioma have not been reported previously.
We believe that our study contributes to better understanding of the genetic profile of human intracranial meningiomas and suggest that meningiomas harbor defective cellular DNA MMR mechanisms. Knowledge on MSI status could be useful for helping diagnostics, therapeutic profiling, and prognosis.
Footnotes
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Ethical approval
All procedures performed in studies involving human participants were in accordance with the ethical standards of the institutional and national research committee and with the Declaration of Helsinki. Ethical approval was received from the Ethical Committees of Medical School University of Zagreb and University Hospital “Sisters of Mercy.” Informed consent was obtained from all individual participants included in the study.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This study was funded by Croatian Science Foundation (grant no. 6625).
