Abstract
Long non-coding RNAs are emerging as new players in gene regulation, but whether long non-coding RNAs influence the expression of microRNA is unclear. The expression levels of misato family member 2, pseudogene were significantly associated with lymphatic metastasis and distal metastasis in 80 paired gastric cancer tissues using real-time quantitative reverse transcription polymerase chain reaction experiments. The effects of long non-coding RNA misato family member 2, pseudogene were assessed by overexpressing or downexpressing long non-coding RNA misato family member 2, pseudogene in gastric cancer cells. Long non-coding RNA misato family member 2, pseudogene promoted gastric cancer cell growth, colony formation, migration, and invasion in gastric cancer cells. Long non-coding RNA misato family member 2, pseudogene influenced biologic functions in gastric cancer cells via indirectly regulating the activation of miR-335. Our results reveal long non-coding RNA misato family member 2, pseudogene as an oncogenic long non-coding RNA that promotes cell growth and invasion. Therefore, long non-coding RNAs might function as key regulatory hubs in gastric cancer progression.
Introduction
Gastric cancer is a major health burden throughout the world. It is currently the most common cancer in China, responsible for about 300,000 deaths per year.1,2 Gastric cancer is commonly diagnosed at an advanced stage because of the lack of early detection strategies and is usually associated with a dismal outcome.3,4 The development of gastric cancer is a complex, multistep process involving multiple genetic and epigenetic alterations of oncogenes, tumor suppressor genes, and non-coding RNAs (ncRNAs).5–7 Some ncRNA alterations present in gastric cancer have been characterized.8,9 However, many important questions, including the mechanisms underlying gastric cancer metastasis, remain unanswered.
Long non-coding RNAs (lncRNAs) are non-protein coding transcripts longer than 200 nucleotides. A number of lncRNAs have been shown to be involved in a wide range of biological functions including RNA processing,10,11 gene transcription regulation, 1 miRNAs’ host genes, 2 modulation of apoptosis and invasion, 4 marker of cell fate, 5 and chromatin modification. 6
Altered expression of lncRNAs has been documented in human gastric cancer, prompting increased interest in their potential use as biomarkers for diagnosis and prognosis, in addition to their potential roles as therapeutic targets.8,9,12 Accumulating evidence demonstrates that lncRNAs play a pivotal role in promoting gastric cancer metastasis, for example, lncRNA H19 in gastric cancer.2,13,14 MicroRNAs (miRNAs) act as negative regulators of gene expression by inhibiting the translation or promoting the degradation of target messenger RNAs (mRNAs).15,16 Growing evidence has demonstrated that miRNAs can play a significant role in the regulation of development, differentiation, proliferation, and apoptosis in gastric cancer.17,18 It has been shown that lncRNAs may act as endogenous sponge RNAs to interact with miRNAs and influence the expression of miRNA. 19 A recent report shows that the H19 lncRNA can act as a molecular sponge for the major let-7 family of miRNAs. 13
In the study, we screened the expressing levels of lncRNA misato family member 2, pseudogene (MSTO2P) and miR-335 in both primary gastric cancer tissues and peripheral blood of gastric cancer patients. We also found that lncRNA MSTO2P dramatically promoted the ability of colony formation and cell growth in vitro. Our data suggest that lncRNA MSTO2P may participate in molecular interactions relevant to malignant transformation and metastasis in gastric cancer.
Materials and methods
Tissue and blood collection
Paired gastric cancer tissue, peripheral blood, and metastatic tissue were obtained from patients who had undergone surgical gastric cancer resection between 2006 and 2011 at the Department of General Surgery, the Affiliated Hospital of Qingdao University, China. Local or systemic treatment had not been performed in these patients prior to the operation, and the clinicopathological characteristics of the patients with gastric cancer are recorded. Tissue samples were immediately macrodissected at the time of surgery and placed directly in RNALater stabilization solution (Qiagen, Hilden, Germany). All the tissues were stored at −80°C until total RNA was extracted. The differentiation grade, pathological stage and grade, and nodal status were appraised by an experienced pathologist. Clinicopathological characteristics including tumor–node–metastasis (TNM) staging were also scored. The non-tumorous tissues were 5 cm from the edge of the tumor, contained no obvious tumor cells and were also evaluated by the pathologist. All the experiments were approved by the Research Ethics Committee of the Affiliated Hospital of Qingdao University, and written informed consent was obtained from all patients.
Cell lines
Gastric cancer cell lines NCI-N87, SNU-1, SNU-16, AGS, and KATO-III were obtained from American Type Culture Collection (Manassas, VA, USA). Gastric cancer cell lines MKN-45, MKN-28, and SGC-7901 and immortalized human gastric mucosa cell line GES-1 were preserved in our institute. Briefly, cells were grown in RPMI 1640 supplemented with 10% fetal calf serum and 2 mM
Quantitative reverse transcription polymerase chain reaction analysis
Purified total RNA was obtained from the tissue, peripheral blood, and cells, and total RNA was extracted using TRIzol solution. Reverse transcription (RT) was performed in a 20-µL reaction according to the manufacturer’s recommendations (Qiagen). Real-time quantitative reverse transcription polymerase chain reaction (qRT-PCR) analyses were performed using primers as follows: 5′-GTTCTGGATGGTTCCCCCAA-3′; 5′-TCTGCTACGTCATCGTGCTC-3′. Transcript expression levels were determined by quantifying the intensity of the PCR product normalized to U6 expression. Quantitative measurement of mRNA levels was performed using the ABI Prism 7000 (Applied Biosystems, Foster City, CA, USA). These data were analyzed using the comparative Ct method.
Databases searching
We searched the following lncRNA and microRNA databases: http://www.lncrnadb.org/, http://www.microrna.org/microrna/home.do, and http://www.mirbase.org/.
Plasmid vector construction and transfection
The MSTO2P sequences were synthesized and subcloned into the pcDNA3.1 (Invitrogen, Shanghai, China) vector. The siRNA expression vector, the pSilencer 4.1-CMV/neo plasmid (Invitrogen Corp, Carlsbad, CA, USA), was used for cloning small synthetic oligonucleotides. Stability-enhanced precursor, miR-335miRNA inhibitor, and negative control RNA-oligonucleotides were purchased from Ambion Corporation (Austin, TX, USA). NCI-N87 and SGC-7901 gastric cancer cell lines were used for overexpression and knockdown studies.
For transfection, complexes of Lipofectamine 2000 (Invitrogen) and the desired plasmid were prepared according to the manufacturer’s instructions. We obtained stably transfected clones by G418 selection (Promega, Madison, WI, USA). The level of MSTO2P and miR-335 expression after transfection was assayed by RT-PCR. A stable transfectant of the empty vector was used as a control, and these complexes were directly mixed with cells in 24-well cell culture plates at a density of 4 × 104 cells per well.
Cell growth and soft agar colony formation assay
Gastric cancer cells (2 × 103 cells) were incubated with 100 µL of culture medium in 96-multiwell plates for 1 day at 37°C in 5% CO2. The cells were transfected with the plasmid for 24, 48, 72, and 96 h. Cell number was assessed using the cell counting kit-8 (CCK-8; Dojindo, Kumamoto, Japan). Briefly, CCK-8 (10 µL) was added to each well. After 1 h of incubation at 37°C, absorbance at 450 nm was measured using the ARVO MX plate reader (PerkinElmer, Waltham, MA, USA). The number of cells was determined by the relative absorbance at 450 nm.
Gastric cancer cells were trypsinized to single-cell suspensions of 3 × 103 cells and then were plated in six-well plates in complete culture medium containing 0.3% agar layered on top of 0.6% agar. The plates were incubated at 37°C in the presence of 5% CO2 for 16 days. Colonies containing at least 50 cells were scored. The data are presented as the mean ± standard deviation of five randomly scored fields.
Migration and invasion assays
For migration and invasion assays, cell culture was performed in Transwell chambers (8 mm, 24-well format; Corning, Painted Post, NY, USA). For the invasion assay, the insert membranes were coated with diluted Matrigel (BD Biosciences, San Jose, CA, USA). Cells (1 × 105) were added to the upper chamber and cultured for 48 h. For the migration assay, the insert membranes were not coated with Matrigel but were cultured under the same conditions. Finally, the insert membranes were removed and stained with crystal violet (Beyotime, Haimen, shanghai, China), and the cells were counted under an inverted microscope and photographed.
Dual-luciferase reporter assay
Cells (1 × 105/well of a six-well plate) were transiently transfected with 1.5 µg of luciferase construct (pGL4 Luciferase Reporter Vectors) or indicated plasmids and 0.2 µg of pRL-tk (Promega) with the use of the Lipofectamine 2000. Transfection was performed with at least three different batches of each reporter plasmid. After incubation for 36–48 h, the cells were harvested with Passive Lysis Buffer (Promega), and luciferase activities of cell extracts were measured with the use of the dual-luciferase assay system (Promega) according to manufacturer’s instructions. Luciferase activity was measured and normalized for transfection efficiency with Renilla luciferase activity. Transfection was performed with at least three different batches of each reporter plasmid.
Statistical analysis
Statistical analysis was performed using SPSS 15.0 software (SPSS Inc., Chicago, USA). Data are expressed as the mean ± standard deviation from at least three separate experiments. Associations of lncRNA MSTO2P with clinical and pathological features were determined using the chi-square test or Cox proportional hazards regression model as appropriate. Kaplan–Meier survival curves were constructed to show survival differences according to lncRNA MSTO2P expression. The survival time for either overall or time to recurrence was calculated as the time from surgery until the occurrence of death and relapse. The Mann–Whitney U test was used for comparing differences in cell counts. Spearman correlation coefficients were calculated for correlation analysis. A value of p < 0.05 was considered statistically significant.
Results
Relationship of lncRNA MSTO2P expression levels and clinicopathological factors in patients with gastric cancer
We investigated the relationship between lncRNA MSTO2P expression levels in 80 paired gastric cancer tissues and clinicopathological factors in patients with gastric cancer. We found that the expression level of lncRNA MSTO2P was associated significantly with lymphatic metastasis and distal metastasis (Table 1, p < 0.05). However, lncRNA MSTO2P expression levels were not associated with gender, age, tumor size, cell differentiation, depth of cancer invasion, gross appearance, and TNM stage.
Relationship between lncRNA MSTO2P expression levels and clinicopathological factors in patients with gastric cancer.
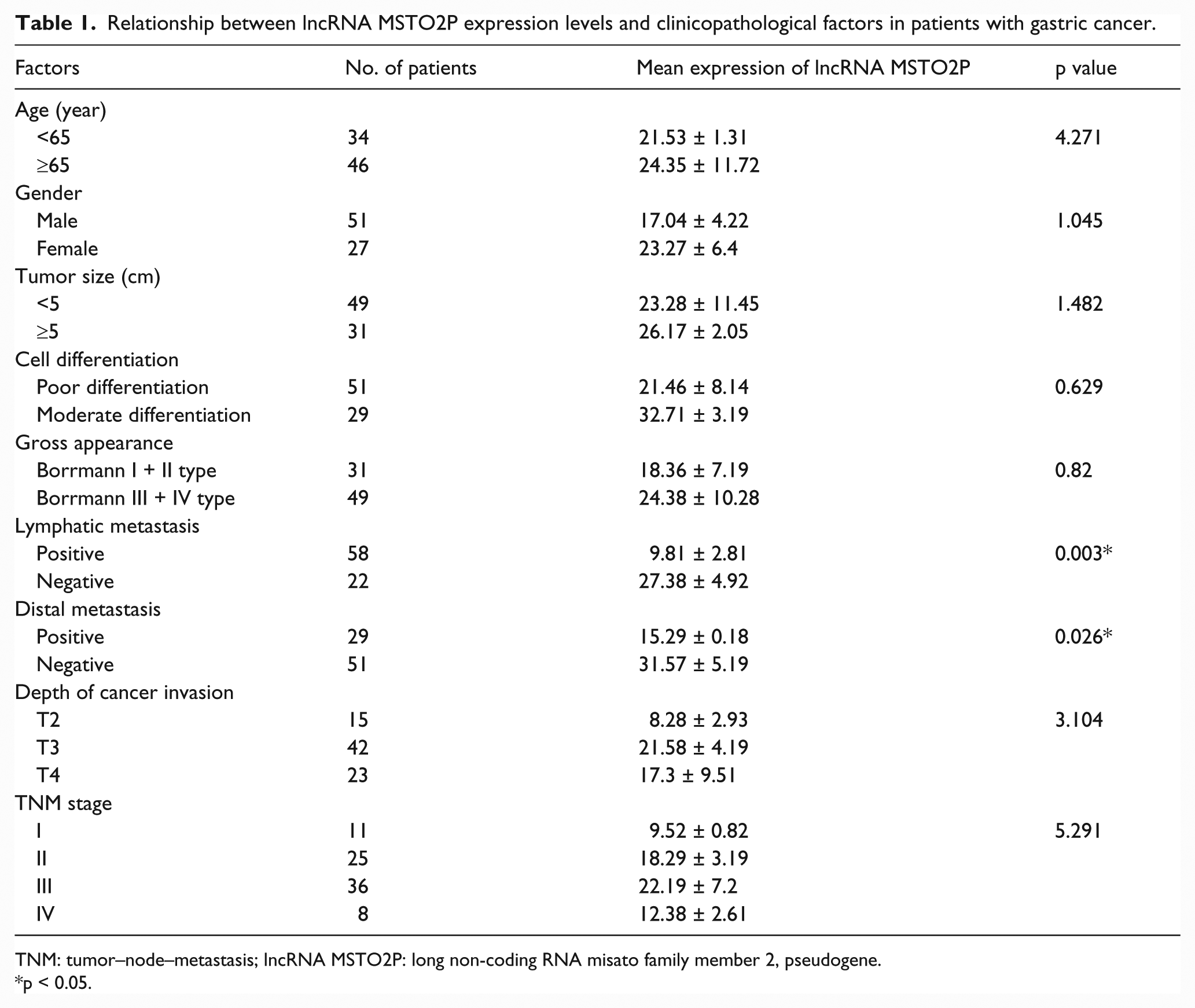
TNM: tumor–node–metastasis; lncRNA MSTO2P: long non-coding RNA misato family member 2, pseudogene.
p < 0.05.
High lncRNA MSTO2P expression was correlated with a shorter overall survival rate in gastric cancer patients
We next investigated the role of lncRNA MSTO2P in gastric cancer by examining a panel of 30 matched sets of normal gastric tissue, gastric cancer tissue, and corresponding metastatic tissue. The mean expression levels of lncRNA MSTO2P in 30 normal gastric tissue, gastric cancer tissue, and corresponding metastatic tissues were 10.65% ± 6.99%, 18.62% ± 14.52%, and 35.71% ± 17.27%, respectively. LncRNA MSTO2P expression was upregulated in metastatic tissues compared with the corresponding normal gastric tissues and gastric cancer tissues (Figure 1(a), p < 0.05). Meanwhile, we detected the expression levels of lncRNA MSTO2P in plasma from healthy patients, gastric carcinoma patients, and metastatic gastric carcinoma patients. Similar findings were obtained using RT-PCR assays. It is noteworthy that the expression levels of lncRNA MSTO2P were significantly associated with gastric cancer metastasis. Overall survival curves and time-to-recurrence (TTR) curves were plotted according to the lncRNA MSTO2P expression level using the Kaplan–Meier method. The high lncRNA MSTO2P expression group had significantly shorter overall survival and TTR than did the low lncRNA MSTO2P expression group (p < 0.05; Figure 1(b) and (c)). Taken together, these data suggest an important role for lncRNA MSTO2P in gastric cancer.
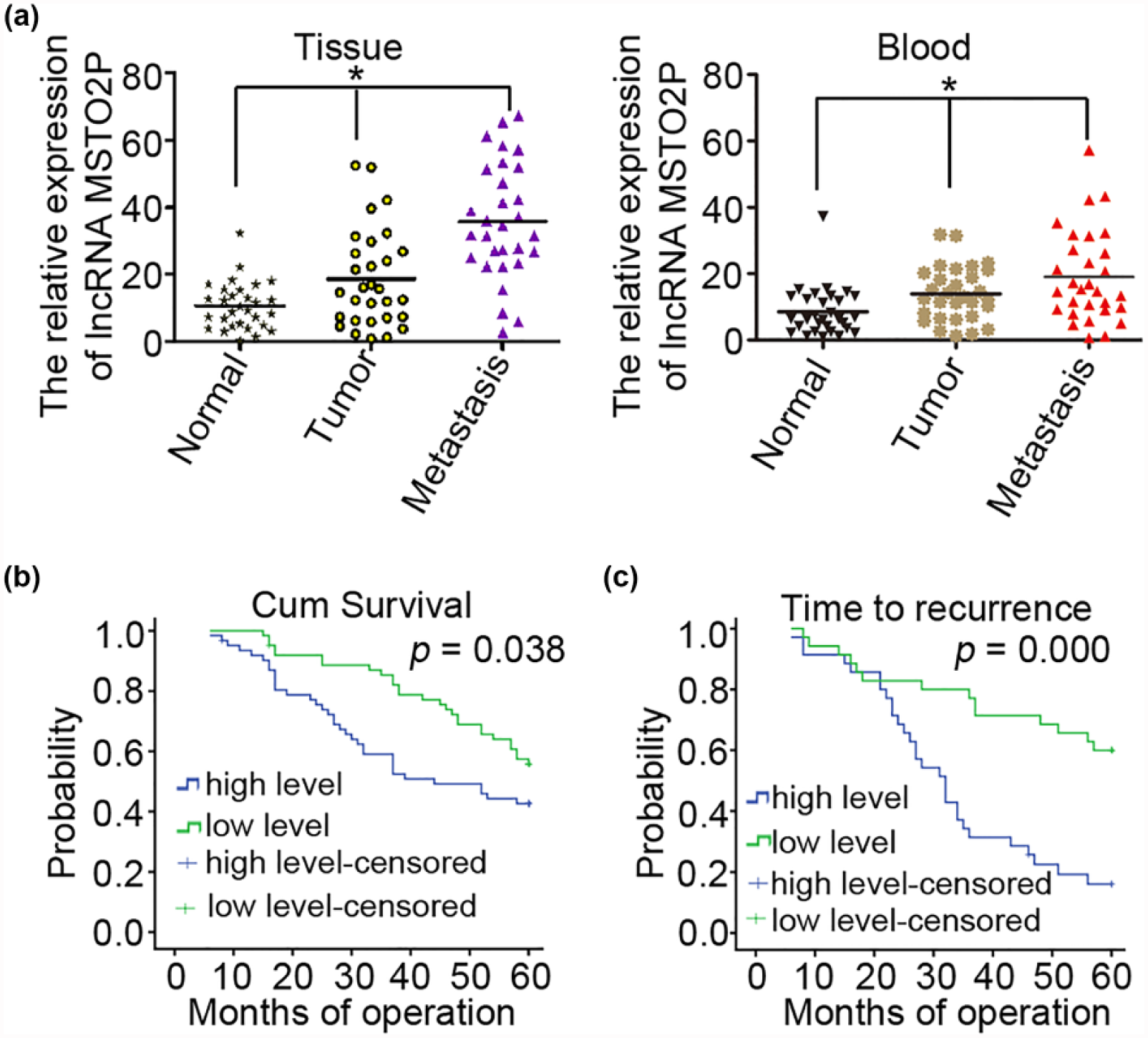
Correlation between lncRNA MSTO2P expression and prognosis of gastric cancer patients. (a) qRT-PCR analysis of lncRNA MSTO2P expression in primary tumor tissues and peripheral blood samples from 30 matched sets of normal gastric tissue, gastric cancer tissue, and corresponding metastatic tissue. The mean and standard deviation of lncRNA MSTO2P expression levels are shown. (b) Kaplan–Meier analysis of overall survival curves for patients with gastric cancer, according to the level of lncRNA MSTO2P expression. (c) Time to recurrence of the high lncRNA MSTO2P expression group was significantly shorter than that of the low lncRNA MSTO2P expression group (*p < 0.05).
Enhanced lncRNA MSTO2P expression in gastric cancer cells promoted cell growth, colony formation, migration, and invasion in vitro
We next used qRT-PCR analysis which revealed that lncRNA MSTO2P was highly expressed in eight different gastric cancer cell lines (AGS, SNU-1, MKN-45, SGC-7901, KATO-III, SNU-16, NCI-N87, and MKN-28) and 1 immortalized gastric mucosa cell line (GES-1; Figure 2(a)). To evaluate the effects of lncRNA MSTO2P on the biological behavior of gastric cancer cell lines, we chose NCI-N87 and SGC-7901 cancer cells and constructed a stable pcDNA-lncRNA MSTO2P expression vector and a siRNA MSTO2P expression vector. They were transfected gastric cancer cell lines NCI-N87 and SGC-7901 with these constructs. After transfection, QRT-PCR confirmed that different lncRNA MSTO2P expression levels influenced the expression of miR-335 (Figure 2(b), p < 0.01), compared with those observed in control transfectants. Cell-counting kit-8 assays revealed increased cell proliferation in NCI-N87 cells transfected with the MSTO2P-expressing vector, compared with control cells (Figure 2(c), left, p < 0.01). Similar findings were obtained using a colony-formation assays, in which gastric cancer cells transfected with the MSTO2P expression vector were grown on soft agar. The colony-formation rate of NCI-N87 cells was 4.67% ± 0.70% in the NCI-N87 group, 3.85% ± 0.63% in the NCI-N87/con group, and 9.45% ± 1.07% in the NCI-N87/lnc group (Figure 2(d), p < 0.005). LncRNA MSTO2P promoted the colony formation in gastric cancer cells (Figure 2(d), p < 0.05), compared with those observed in NCI-N87/con transfectants. Taken together, these results suggest that MSTO2P has a physiological role in regulating cell proliferation.
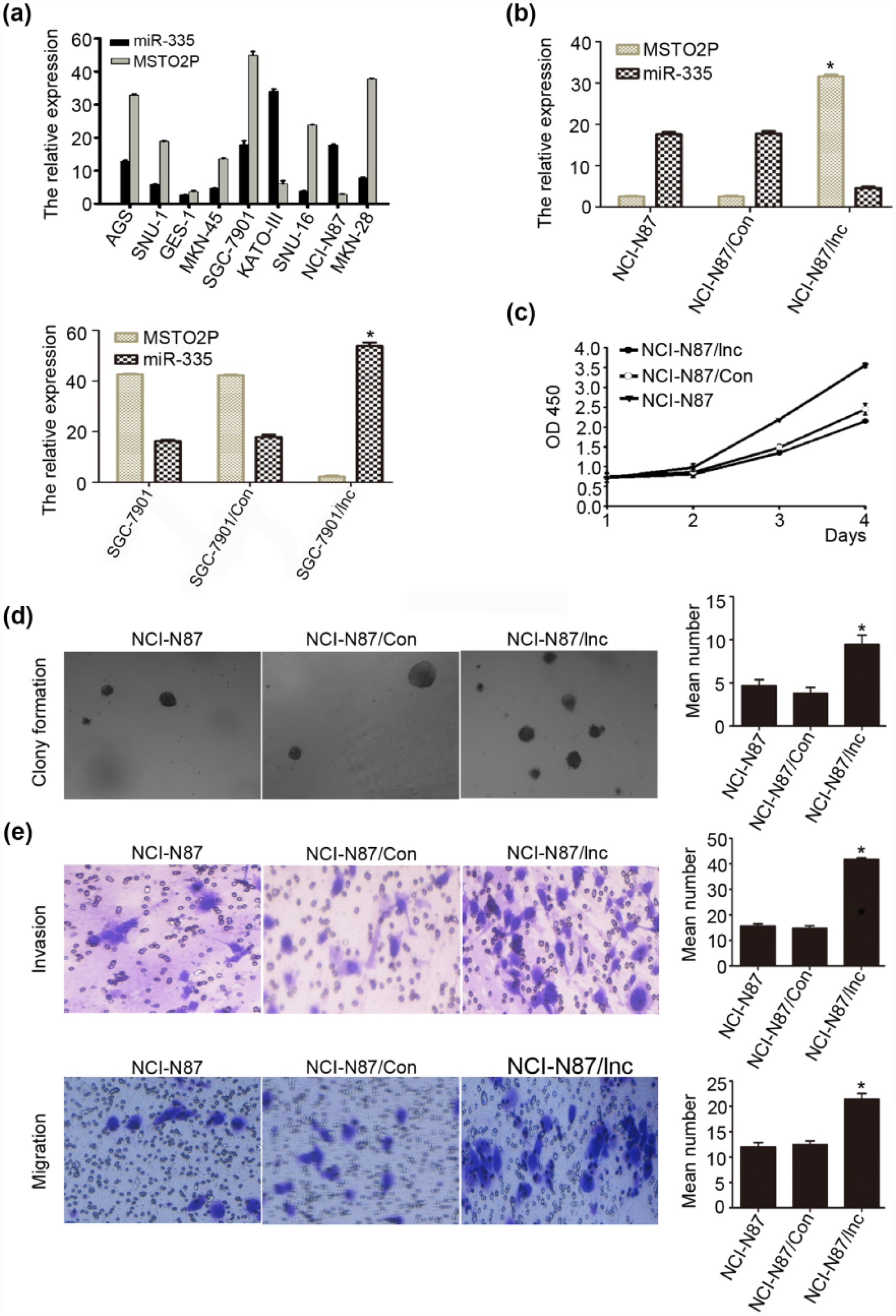
LncRNA MSTO2P promotes proliferation, colony formation, migration, and invasion in gastric cancer cells. (a) LncRNA MSTO2P and miR-335 expression levels in eight gastric cancer cell lines and GES-1 cells by qRT-PCR. (b) qRT-PCR analysis confirmed lncRNA MSTO2P overexpression in NCI-N87 cells stably transfected with lncRNA MSTO2P expression vector versus cells transfected with the vector control. (c and d) LncRNA MSTO2P overexpression promotes cell proliferation and colony formation in soft agar in NCI-N87 cells, compared with control group. (e) Migratory and invasive capacities in gastric cancer cells increased after lncRNA MSTO2P overexpression, as determined by performing Transwell-migration and Transwell-invasion assays (*p < 0.05).
We also evaluated the cell migration and invasion capacities of gastric cancer cells with modified expression of lncRNA MSTO2P using a Transwell-migration assays (Figure 2(e), upper panels) and a Transwell-invasion assays (Figure 2(e), lower panels). The migration ability of NCI-N87 cells overexpressing lncRNA MSTO2P was significantly enhanced, compared with that of control transfectants (12.52 ± 0.67 vs 21.45 ± 1.08, p < 0.005; Figure 2(e)). The number of invasive cells in the lncRNA MSTO2P-transfected sample also significantly increased compared with the control transfectants (14.85 ± 0.89 vs 41.79 ± 0.55, p < 0.001; Figure 2(e)). However, SGC-7901 cells with depleted MSTO2P did not influence cell growth and colony-formation and invasion capacities, compared with controls (Supplemental Figure 1(A) and (B)). Our results suggest that lncRNA MSTO2P promotes the migration and invasion in gastric cancer cells.
Long non-coding RNA MSTO2P regulates the expression of miR-335
Based on PubMed, lncRNA and microRNA database search, the association of lncRNA MSTO2P and miR-335 was hinted (Figure 3(a)). We evaluated the effect of lncRNA MSTO2P on miR-335 using qRT-PCR method in gastric cancer cell lines and 80 gastric cancer tissues (Figures 2(a) and 3(b)). As shown in Figure 3(b), high expression of lncRNA MSTO2P led to a significant low expression of miR-335. Alternatively, enhanced lncRNA MSTO2P expression in gastric cancer cells was also significantly reduced the expression level of miR-335 compared with the control (18.85 ± 1.86 vs 3.79 ± 0.55, p < 0.001; Figure 2(e)). However, high expression or low expression of miR-335 in a stably expressed MSTO2P NCI-N87 cells did not influence the expression of lncRNA MSTO2P, compared with controls (Supplemental Figure 1(D), upper panel). We identified the interaction between lncRNA MSTO2P and miR-335 using luciferase reporter assay. Our results showed that it was not the direct interaction between lncRNA MSTO2P and miR-335 (Supplemental Figure 1(D), lower panel). All experimental results showed that long non-coding RNA MSTO2P indirectly regulates the expression of miR-335 in gastric cancer tissues and cells.
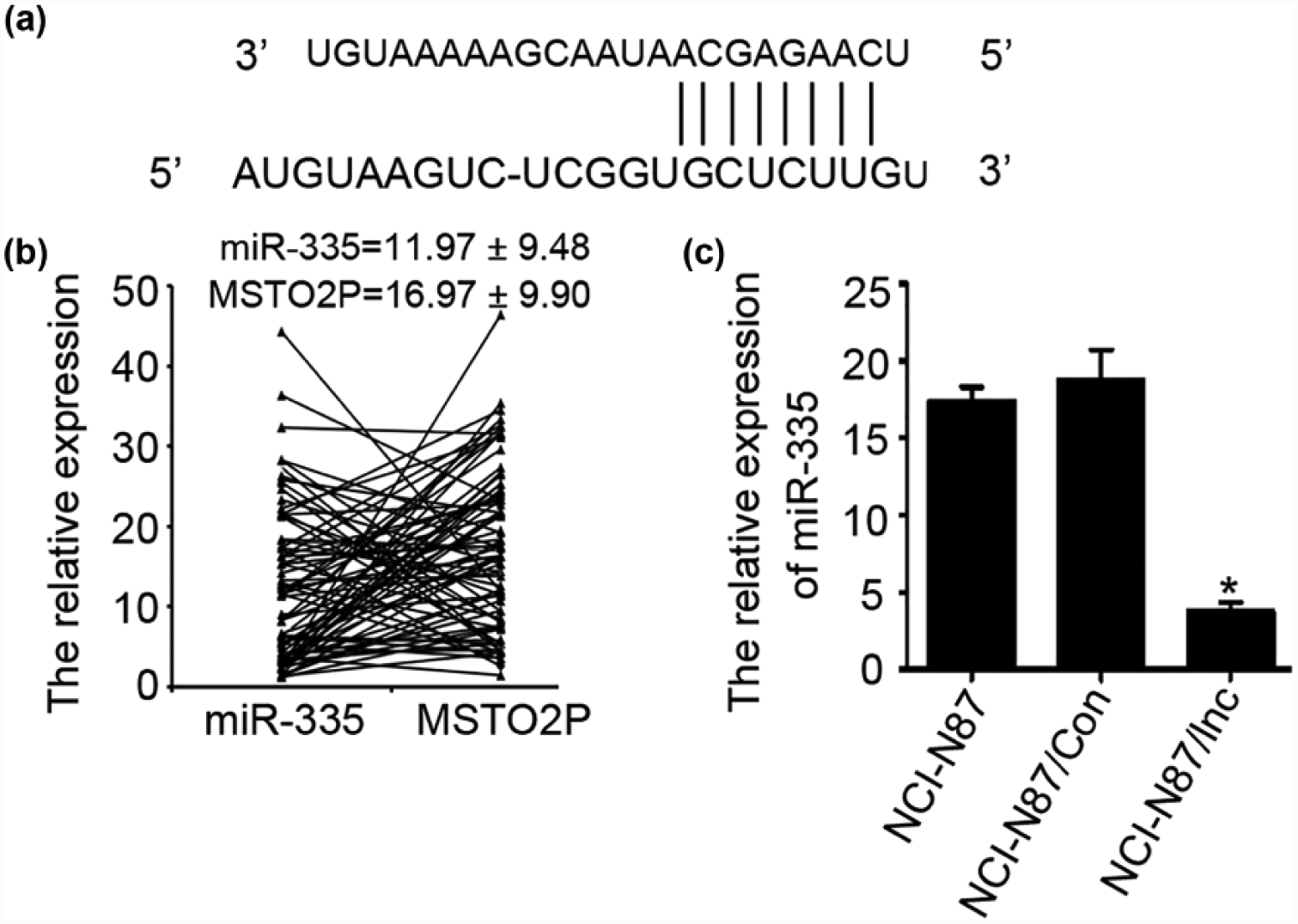
LncRNA MSTO2P regulated the expression levels of miR-335. (a) Representative nucleotide sequence matches between lncRNA MSTO2P and miRNA. Only matched nucleotides with miRNA seed sequences are indicated with vertical lines. (b) qRT-PCR analysis of lncRNA MSTO2P and miR-335 expression in 80 primary tumor tissues. The mean and standard deviation of lncRNA MSTO2P and miR-335 expression levels are shown. (c) After lncRNA MSTO2P transfection, lncRNA MSTO2P inhibited the expression of miR-335. Each bar represents the mean value ± standard deviation from three independent experiments (*p < 0.05).
Discussion
LncRNAs are transcriptional products longer than 200 nucleotides that primarily have a regulatory role rather than encode proteins.20,21 In some studies, lncRNAs play a pivotal role in many biological mechanisms, including gene imprinting, activation, and repression.22–24 At the transcriptional level, lncRNAs can bind to DNA or proteins, regulate the subcellular localization of transcription factors, and inhibit transcript elongation25–27 and are involved in diseases such as malignancies, including lung cancer, 28 renal cell carcinoma, 29 bladder cancer, 30 hepatocellular carcinoma, 31 and gastric cancers. 9 Therefore, identification of gastric cancer-specific lncRNAs is critical for understanding their role on tumorigenesis and may be important for finding novel therapeutic targets.32,33
In our study, we searched three lncRNA microarray data (Series Accession: GSE84787, GSE65917, and GSE54835) in GEO DataSets. LncRNA MSTO2P has been reported in gastric cancer.9,32 LncRNA MSTO2P belongs to gastric cancer–associated lncRNA. Prior to this study, very little was known about lncRNA MSTO2P expression in the tissues and blood of patients with gastric carcinoma. To address these questions, the expression of lncRNA MSTO2P in gastric carcinoma and its correlation with patient clinicopathological features were poorly characterized prior to this study. Therefore, we examined lncRNA MSTO2P expression levels and the clinicopathological characteristics of 80 patients with gastric cancer. Although no relationship was observed between lncRNA MSTO2P expression and patient sex and age, tumor size, cell differentiation, gross appearance, the depth of cancer invasion, site of tumor, or TNM stage, there were significant associations between lncRNA MSTO2P expression and lymphatic metastasis and distal metastasis. These results indicate that the lncRNA MSTO2P expression level might influence tumor progression during gastric cancer development. Next, lncRNA MSTO2P expression levels were examined in 30 patients with gastric carcinoma tissues. LncRNA MSTO2P showed the highest expression level and was upregulated in gastric cancer and metastatic tissues. Therefore, lncRNA MSTO2P may have an important role in the malignant transformation and the metastasis of gastric cancer.
In clinical practice, patients with gastric cancer are often not symptomatic in the early stages of the disease. Therefore, serum screening for lncRNA MSTO2P represents a promising diagnostic tool because it is easy and inexpensive. Significant lncRNA MSTO2P overexpression was detected in the peripheral blood of gastric cancer patients and gastric cancer metastatic patients, compared with that observed in healthy control subjects. We found that patients with high lncRNA MSTO2P expression had an increased risk of recurrence and a significantly reduced overall post-operative survival. Thus, the lncRNA MSTO2P expression level in gastric cancer tissues and peripheral blood may serve as a valuable prognostic indicator for patients with gastric cancer.
To better understand the function and mechanism of lncRNA MSTO2P in the metastasis of gastric cancer, our data clearly showed that lncRNA MSTO2P is highly expressed in gastric cancer tissues from patient samples and in gastric cancer cell lines. We regulated lncRNA MSTO2P expression by transfected lncRNA MSTO2P expression or siRNA vector into gastric cancer cell line and observed the influence of lncRNA MSTO2P on gastric cancer cells in vitro. From the study, CCK-8 assay results indicated that cell proliferation increased in NCI-N87 cells upon forced lncRNA MSTO2P overexpression. Tumor spreading and metastasis are influenced by tumor migration and invasion capacity, both of which were promoted by lncRNA MSTO2P in vitro. Recent studies have suggested that lncRNAs may act as endogenous sponge RNA to interact with miRNAs and influence the expression of miRNA.16,19,34 By now, the cross talk between lncRNAs and miRNAs has been described in a wide range of human carcinomas. Many research works reveal that lncRNA HOTAIR, H19, and MEG3 have played the emerging role of lncRNA–miRNA interplay in gastric cancer development and progression based on the types of lncRNAs. 34 So, we analyze lncRNA–miRNA possible interaction position by PubMed, lncRNA and microRNA bioinformatics tools. The association of lncRNA MSTO2P and miR-335 was hinted. In our study, we evaluated the effect of lncRNA MSTO2P on miR-335 in gastric cancer cell lines and gastric cancer tissues using qRT-PCR method. High expression of lncRNA MSTO2P led to a significant low expression of miR-335. Alternatively, enhanced lncRNA MSTO2P expression in gastric cancer cells was also significantly reduced the expression level of miR-335. Our results showed that miR-335 cannot influence the expression of lncRNA MSTO2P using QRT-PCR and luciferase reporter assay All our data suggest that lncRNA MSTO2P indirectly regulated the expression of miR-335 in gastric cancer tissue and cells. We will further study their mechanism in future studies. Although the detailed molecular mechanisms of lncRNA MSTO2P in gastric cancer have not been clearly clarified, for example, which possible signal pathway or downstream genes resulting in tumorigenesis can lncRNA MSTO2P effectively influence? It needs further researches in vitro in the future. But our study first illustrated that lncRNA MSTO2P could effectively influence gastric cancer cell growth and invasion. These findings might help us elucidate the cause and effects of lncRNA MSTO2P in the development and progression of gastric cancer in the future.
In summary, this study reveals that lncRNA MSTO2P was strongly expressed in gastric cancer tissues and matched peripheral blood samples. We used a vector-based overexpression method to demonstrate that lncRNA MSTO2P promoted cell proliferation, colony formation, migration, and invasion in vitro. LncRNA MSTO2P thus possesses important functions for the promotion of gastric cancer. Elevated lncRNA MSTO2P expression in the peripheral blood of gastric cancer patients suggests its potential use as a biomarker for gastric cancer. Moreover, lncRNA MSTO2P activity may serve as a potential strategy for gastric cancer treatment. More importantly, we demonstrated that lncRNA MSTO2P acts as endogenous sponge RNA and inhibits miR-361 expression. Thus, modulation of lncRNA MSTO2P and miR-335 may represent novel approaches for interventional treatment of gastric cancer. This finding may provide a new clue for the understanding of lncRNAs- and miRNAs-controlled cellular events.
Footnotes
Acknowledgements
The authors would like to thank all the members of the Central Laboratories of the Affiliated Hospital of Qingdao University for thought-provoking discussion and collegial support in this study. H.L. and H.Z. contributed equally to this work.
Compliance with ethical standards
This study was reviewed and approved by the Research Ethics Committee of the Affiliated Hospital of Qingdao University. All patients provided informed consent before surgery. All samples were handled according to the ethical and legal standards.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported in part by grants from the National Youthful Science Foundation of China (81202307 and 81201144). The funders had no role in study design, data collection and analysis, decision to publish, or preparation of the manuscript.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
