Abstract
Long non-coding RNA HOXD-AS1 (HOXD cluster antisense RNA 1) has been demonstrated to be closely associated with the progression of several tumors. However, the biological function of HOXD-AS1 and the underlying molecular mechanism in gastric cancer are still unclear. The expression of HOXD-AS1 in gastric cancer cell lines was evaluated by quantitative real-time polymerase chain reaction. The association of HOXD-AS1 expression and clinical parameters was statistically analyzed by chi-square test. Cell viability, colony formation capacity, and phosphorylation of Janus kinase 2 and signal transducer and activator of transcription 3 in treated SGC-7901 and BGC-823 cells were detected by 3-(4,5-dimethylthiazol-2-yl)-2,5-diphenyltetrazolium bromide assay, colony formation assay, and western blot analysis, respectively. The results indicated that HOXD-AS1 was significantly upregulated in gastric cancer cells and clinically involved in tumor size, invasion depth, tumor–node–metastasis stages, regional lymph nodes, lymphatic metastasis, as well as distant metastasis. HOXD-AS1 knockdown dramatically inhibited gastric cancer cell proliferation, colony formation capacity, and phosphorylation of Janus kinase 2 and signal transducer and activator of transcription 3 in vitro. In addition, HOXD-AS1 overexpression significantly promoted gastric cancer cell proliferation and colony formation capacity, whereas both Janus kinase small interfering RNAs and Janus kinase 2 inhibitor AG490 overturned these effects. Furthermore, xenograft assays confirmed the biological function of HOXD-AS1 in vivo. Taken together, our data elucidate that knockdown of HOXD-AS1 dramatically suppresses gastric cancer cell growth by inactivating the Janus kinase 2/signal transducer and activator of transcription 3 pathway in vitro and in vivo, contributing to a better understanding of gastric cancer pathogenesis and providing a possible theoretical foundation for long non-coding RNA–directed diagnosis and therapy against this disease.
Keywords
Introduction
Gastric cancer (GC) is one of the most common types of malignancies, especially in East Asia, including Japan, China, and Korea.1,2 GC represents the second leading cause of cancer-related deaths worldwide, although the incidence and mortality of GC is decreasing in developing countries.3,4 The majority of GC patients are diagnosed at advanced stages and over 700,000 people die from GC per year due to distant metastasis and malignant proliferation. 5 Despite great improvements in treatments, including chemotherapy, radiotherapy, and surgery therapy, the early diagnosis of GC still remains poor and the median 5-year survival rate is less than 30% for patients with advanced-stage GC.6,7 Thus, more research should be devoted to discover useful diagnostic biomarkers for early GC detection and accurate prediction of tumor behavior and to better understand the molecular mechanism underlying GC carcinogenesis.
The human genome sequencing project has displayed that more than 90% of the genome is protein-non-coding sequences and transcribed as non-coding RNAs (ncRNAs), playing vital regulatory roles in complex organisms. 8 Long non-coding RNA (lncRNA) is a newly discovered ncRNA composed of more than 200 nucleotides in length. LncRNA is involved in pathogenesis that contributes to disease occurrence and biological processes, including cancer cell apoptosis and metastasis as well as chemotherapy drug resistance.9–12 LncRNA has been shown to hold both pro-oncogenic and tumor-suppressive roles in the GC carcinogenesis and progression.13,14
The HOX genes are the key developmental regulators in numerous processes, including apoptosis, receptor signaling, and differentiation.15,16 Dysregulation of HOX genes is frequently related to malignancy and plays important roles in oncogenesis and tumor suppression. 16 LncRNA HOXD cluster antisense RNA 1 (HOXD-AS1) is transcribed from the HOXD cluster on human chromosome 1q31.2, which is one member of HOX gene clusters. 17 HOXD-AS1 has been demonstrated to be upregulated and closely associated with the progression and prognosis of several tumors, such as neuroblastoma, adenocarcinoma, and breast cancer. 17 However, the role and underlying molecular mechanism of HOXD-AS1 in GC are still unclear.
Janus kinase 2 (JAK2), one of the JAK family members, is a vital intracellular mediator of cytokine and hormone signaling and induces activation of downstream signaling pathway, including signal transducer and activator of transcription (STAT) cascade. 18 STAT3 is a member of STAT family and can be frequently activated by phosphorylation at tyrosine (Y) reside 705 in response to certain cytokine and growth factor in a growing number of cancers. 19 Constitutively activated STAT3 has been demonstrated to be associated with GC cell growth, metastasis, invasion, and angiogenesis. 20 Mounting evidences have illuminated that the JAK2/STAT3 pathway is an important mediator in angiogenesis and tumor growth. 21
In this study, we discovered that lncRNA HOXD-AS1 expression was significantly elevated in GC cell lines. In addition, we proved that knockdown of HOXD-AS1 repressed GC cell proliferation and colony formation abilities in vitro. Furthermore, we found that inhibition of the JAK2/STAT3 pathway was demonstrated to suppress GC cell proliferation and colony formation abilities induced by HOXD-AS1. The studies further revealed that HOXD-AS1 knockdown inhibited tumorigenesis of GC cells in vivo. Our results shed light on the role of lncRNA HOXD-AS1 in GC cells and its involvement in regulating the JAK2/STAT3 pathway, indicating the potential application of HOXD-AS1 in GC therapy.
Materials and methods
Participants and tissue samples
A total of 104 biopsy samples of gastric tumor were collected from Huaihe Hospital of Henan University. All tissues were verified by two independent clinical pathologists. The characteristics of patients with GC are presented in Table 1. The collected tissue biopsy specimens were instantly snap-frozen in liquid nitrogen and stored at −80°C for subsequent use. All procedures were performed with approval of the Ethics Committee of Huaihe Hospital of Henan University. Signed informed consents have been obtained from all the participants.
Association between HOXD-AS1 expression and clinical parameters of patients with gastric cancer.

TNM: tumor–node–metastasis.
Cell culture
The human GC cell lines (SGC-7901, BGC-823, MGC-803, and MKN-45) were obtained from the Institute of Biochemistry and Cell Biology of the Chinese Academy of Sciences (Shanghai, China). Poorly differentiated human gastric adenocarcinoma cell line AGS and normal gastric mucosa cell line GES-1 were purchased from American Type Culture Collection (ATCC, Rockville, MD, USA). All cell lines used were grown and cultured in RPMI-1640 medium (Gibco, Carlsbad, CA, USA) supplemented with 10% fetal bovine serum (FBS; Gibco), 100 U/mL of penicillin, and 100 µg/mL streptomycin (Invitrogen, Carlsbad, CA, USA) in a humidified atmosphere containing 5% CO2 at 37°C.
Cell transfection
The small interfering RNAs (siRNAs) specially targeting HOXD-AS1 (HOXD-AS1 siRNA1 and HOXD-AS1 siRNA2), siRNAs specially targeting JAK2 (JAK2 siRNA1 and JAK2 siRNA2), and scrambled siRNA (control siRNA) were synthesized by Shanghai GenePharma Co. Ltd. (Shanghai, China). The HOXD-AS1 complementary DNA (cDNA) sequence was subcloned into the pcDNA vectors (Shanghai Novobio Biotechnology, Shanghai, China) and the recombinant vectors were named as pcDNA-HOXD-AS1. An empty pcDNA vector was used as the control. SGC-7901 and BGC-823 cells were plated onto six-well plates at 37°C for 24 h and then transiently loaded with HOXD-AS1 siRNAs, JAK2 siRNAs, pcDNA-HOXD-AS1, or matched controls by Lipofectamine 2000 (Invitrogen, Grand Island, NY, USA). The subsequent experiments were carried out 48 h after transfection.
Quantitative real-time polymerase chain reaction
Total RNA was extracted from tissues and cultured cells by TRIzol reagent (TaKaRa, Dalian, China). The quantity of RNA was tested by an ultraviolet spectrophotometer at the absorbance of 260 nm and 280 nm. The ratio of optical density (OD) 260/280 between 1.7 and 2.1 suggested an adequate purity for subsequent experiments. The reverse transcription reactions were carried out using the Primer-Script one step RT-PCR kit (TaKaRa). The messenger RNA (mRNA) expression analysis of HOXD-AS1 was performed by real-time polymerase chain reaction (RT-PCR) using a standard SYBR Green PCR kit (TaKaRa) protocol on Applied Biosystems 7500 Real-Time PCR system (Applied Biosystems, Foster City, CA, USA) according to the manufacturer’s instructions. The reaction condition of RT-PCR was 95°C for 30 s, 60°C for 30 s, 40 cycles at 95°C for 30 s, 60°C for 30 s, and 72°C for 30 s. Glyceraldehyde 3-phosphate dehydrogenase (GAPDH) expression was regarded as the endogenous control in each sample. Each experiment was conducted in triplicate.
Cell viability assay
SGC-7901 and BGC-823 cells in the logarithmic growth were washed with twice phosphate-buffered saline (PBS) and seeded on a 96-well plate with six duplicate wells at a final density of 5 × 103 per well at 37°C for 24 h. Then, the cells were transfected with HOXD-AS1 siRNAs, JKA2 siRNAs, pcDNA-HOXD-AS1, or corresponding controls and treated with different concentrations of AG490 (25, 50, 100 µM). At indicated times, 20 µL of 5 mg/mL 3-(4,5-dimethylthiazol-2-yl)-2,5-diphenyltetrazolium bromide (MTT; Sigma-Aldrich, St. Louis, MO, USA) was added into each well and incubated for another 4 h at 37°C and 5% CO2. Then, the culture medium was removed and 150 µL dimethyl sulfoxide (DMSO) was added to each MTT-treated well for 10 min to dissolve the crystals. The absorbance at 490 nm was determined by a microplate reader (Molecular Devices, Sunnyvale, CA, USA) to assess cell viability. The experiment was repeated at least three times.
Colony formation assay
Transfected or non-transfected SGC-7901 and BGC-823 cells (500 cells/well) were evenly seeded on six-well plates in triplicate and kept growing for 48 h in RPMI-1640 medium with 10% FBS at 37°C. Then, the cells were treated with or without varying concentrations of AG490 (25, 50, and 100 µM) for another 48 h. After 14 days, the cells were fixed with 4% paraformaldehyde (Sigma-Aldrich) for 30 min, stained with Giemsa (Sigma-Aldrich) for 20 min, and photographed under an inverted fluorescence microscope (IX51, Olympus, Tokyo, Japan) equipped with an Olympus Qcolor 3 digital camera (Olympus). The numbers of colony of more than 50 cells were analyzed by an inverted microscope (Nikon, Inc., Garden City, NY, USA).
Western blot analysis
The total proteins from treated cells and biopsy samples of gastric tumor were extracted with radioimmunoprecipitation assay (RIPA) lysis buffer (Pierce, Rockford, IL, USA). The contents of the extracted proteins were quantified by the BCA Protein Assay Reagent Kit (Pierce). For analysis of protein expression, equal amounts of protein (40 µg) from each sample were run on 12% Sodium dodecyl sulfate–polyacrylamide gel electrophoresis (SDS-PAGE) and electro-transferred to polyvinylidene difluoride (PVDF) membranes (EMD Millipore, Billerica, MA, USA). Following blocking with 5% bovine serum albumin for 2 h at 37°C, the membranes were probed with the primary antibody against phosphorylated JAK2 (Try1007/1008), JAK2, phosphorylated STAT3 (Try705), STAT3 (Cell Signaling Technology, Beverly, MA, USA), and β-actin (CWBio, Beijing, China) at a concentration of 1:1000 overnight at 4°C. After being washed three times with Tris buffered saline with Tween 20 (TBST) buffer, the horseradish peroxidase–goat anti-rabbit antibodies were added at 1:3000 dilutions and incubated with the membranes at room temperature for 1 h. After being washed again, the protein bands were visualized and quantified using Electrochemiluminescence Plus Detection system (EMD Millipore). β-actin was considered as an internal control.
Tumor xenografts in nude mice
All animal experiments were approved by the Institutional Animal Care and Use Committee of Huaihe Hospital of Henan University. Female BALB/c nude mice (6 weeks old) purchased from the Model Animal Research Center of Nanjing University (Nanjing, China) were randomly divided into two groups and acclimatized for 4 days under pathogen-free conditions. A volume of 3 × 106 SGC-7901 cells transfected with HOXD-AS1 siRNA1 or control siRNA were prepared and subcutaneously inoculated into the right side of the groin of nude mice in a volume of 0.1 mL PBS. Seven days later, size of tumor xenografts was estimated for every 7 days using a caliper. The tumor volumes were calculated by the equation: tumor volume (mm3) = (length (mm) × width (mm)2)π/6. After 21 days, the tumor-bearing mice were sacrificed and weighted. Subsequently, the expressions of HOXD-AS1, p-JAK2 (Try1007/1008), JAK2, p-STAT3 (Try705), and STAT3 in excised tumor samples were detected by quantitative RT-PCR (qRT-PCR) and western blot analysis.
Statistical analysis
Statistical analyses were performed using SPSS 20.0 software package (SPSS, Chicago, IL, USA). All data are shown as mean ± standard deviation (SD) from at least three independent experiments. Statistical significance was estimated with Student’s t test between two groups and one-way analysis of variance (ANOVA) for three and more groups. The association between HOXD-AS1 expression and clinical parameters was analyzed using chi-square test. p < 0.05 was chosen to represent statistical significance.
Results
Association between HOXD-AS1 expression and clinical parameters of GC patients
To study the role of HOXD-AS1 in GC progression, we checked the expression of HOXD-AS1 in 104 GC tissues and evaluated the association between HOXD-AS1 expression and clinical parameters of GC patients (Table 1). Statistical analysis showed that HOXD-AS1 expression in GC patients was dramatically correlated with tumor size, invasion depth, tumor–node–metastasis (TNM) stages, regional lymph nodes, lymphatic metastasis, as well as distant metastasis (p < 0.05). However, there were no significant association between HOXD-AS1 expression and the rest of clinical parameter factors (age, gender, histologic differentiation; p > 0.05).
HOXD-AS1 knockdown suppresses GC cell growth in vitro
To further identify the biological significance of HOXD-AS1 in the tumorigenesis of GC, we investigated HOXD-AS1 expression in several GC cell lines by qRT-PCR. As displayed in Figure 1(a), the expression of HOXD-AS1 was obviously upregulated in all five GC cell lines (SGC-7901, BGC-823, MGC-803, MKN-45, and AGS) compared with that in normal gastric mucosa cell line GES-1, especially in SGC-7901 and BGC-823 cells. Consequently, SGC-7901 and BGC-823 cells were selected for the following experiments. In consideration of high expression of HOXD-AS in GC cell lines, siRNA-mediated knockdown of HOXD-AS1 in SGC-7901 and BGC-823 cells was performed to investigate the impact of HOXD-AS1 on cell viability and colony formation ability. As shown in Figure 1(b) and (c), HOXD-AS1 siRNA1 and HOXD-AS1 siRNA2 significantly downregulated HOXD-AS1 expression in SGC-7901 and BGC-823 cells compared with control siRNA group, confirming the knockdown efficiency of siRNAs. Besides, a significant decrease in cell viability at 48 and 72 h was observed in HOXD-AS1 siRNA–treated SGC-7901 (Figure 1(d)) and BGC-823 (Figure 1(e)) cells compared with the control siRNA–transfected cells. Furthermore, HOXD-AS1 knockdown remarkably reduced colony numbers of SGC-7901 (Figure 1(f)) and BGC-823 (Figure 1(g)) cells compared with control siRNA group. These data suggested that the HOXD-AS1 knockdown suppresses GC cell growth in vitro.
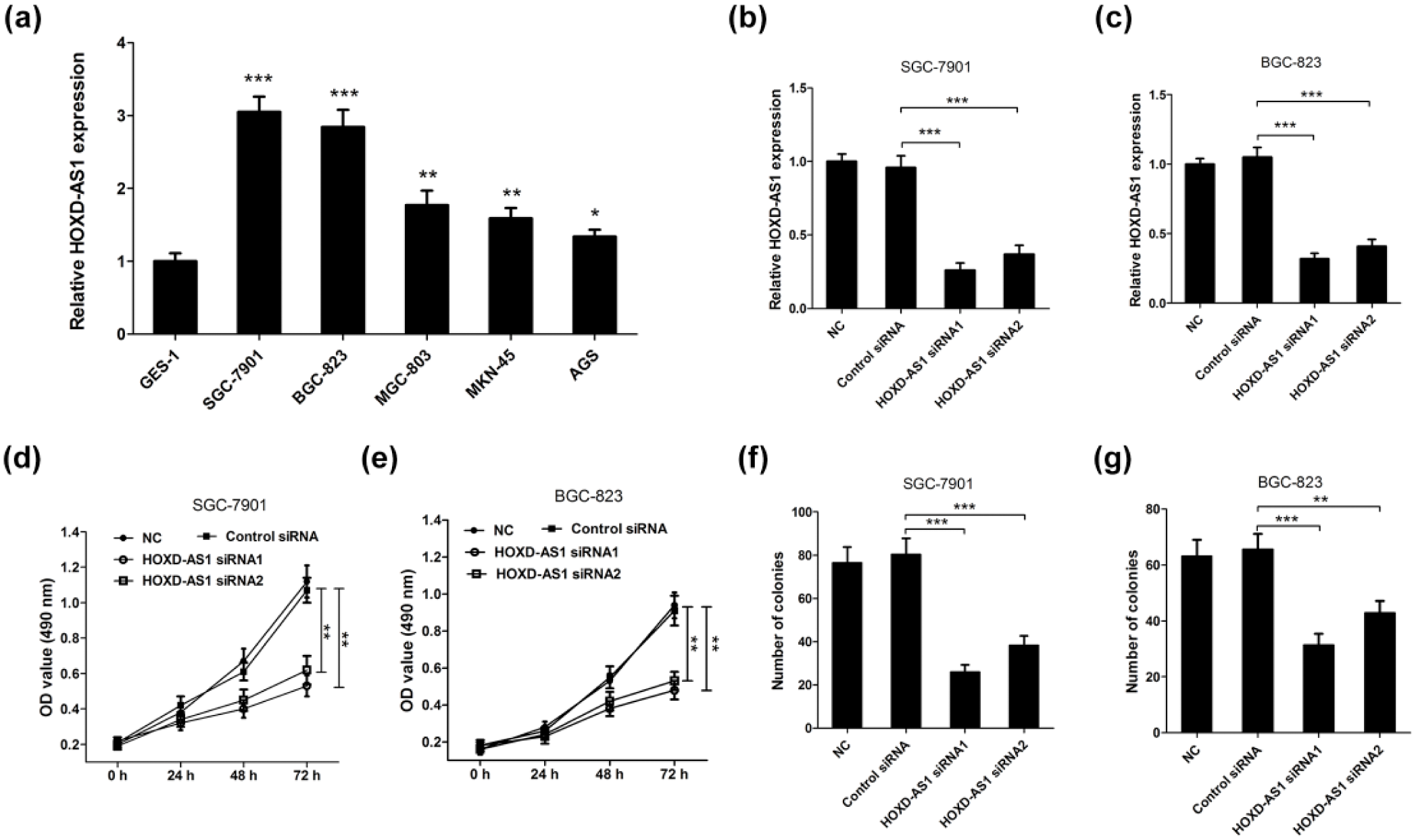
The relative expression of HOXD-AS1 in GC cell lines and its role in GC cell growth in vitro. (a) The relative expression of HOXD-AS1 in GC cell lines (SGC-7901, BGC-823, MGC-803, MKN-45, and AGS) and normal gastric mucosa cell line GES-1 was evaluated by qRT-PCR analysis. The expression of HOXD-AS1 in (b) SGC-7901 and (c) BGC-823 cells transfected with HOXD-AS1 siRNA1, HOXD-AS1 siRNA2, or control siRNA was determined by qRT-PCR, and GAPDH was used as the endogenous control. Cell viability in HOXD-AS1 siRNA–treated (d) SGC-7901 and (e) BGC-823 cells was examined by MTT assay at 24, 48, and 72 h post transfection. Colony formation ability in HOXD-AS1 siRNA–treated (f) SGC-7901 and (g) BGC-823 cells was assessed by colony formation assay. Data are presented as the mean ± SD (n = 3; *p < 0.05, **p < 0.01, and ***p < 0.001).
HOXD-AS1 knockdown suppresses the JAK/STAT pathway in GC cells
To investigate the effect of HOXD-AS1 knockdown on the JAK/STAT pathway in GC cells, the protein levels of p-JAK2 (Try1007/1008), JAK2, p-STAT3 (Try705), and STAT3 in HOXD-AS1 siRNA–transfected SGC-7901 and BGC-823 cells were measured by western blot analysis. The results revealed that HOXD-AS1 knockdown markedly decreased the levels of p-JAK2 (Try1007/1008) and p-STAT3 (Try705) in SGC-7901 (Figure 2(a)) and BGC-823 (Figure 2(b)) cells, suggesting that HOXD-AS1 knockdown dramatically inhibited the JAK/STAT pathway in GC cells in vitro. To verify this, the levels of p-JAK2, p-STAT3, and HOXD-AS1 in 15 randomly selected biopsy samples from GC patients were determined, and the results showed a significant positive correlation between p-JAK2 and HOXD-AS1 (Pearson’s correlation, r = 0.7680, p = 0.0008; Figure 2(c)) and p-STAT3 and HOXD-AS1 (Pearson’s correlation, r = 0.7836, p = 0.0005; Figure 2(d)).
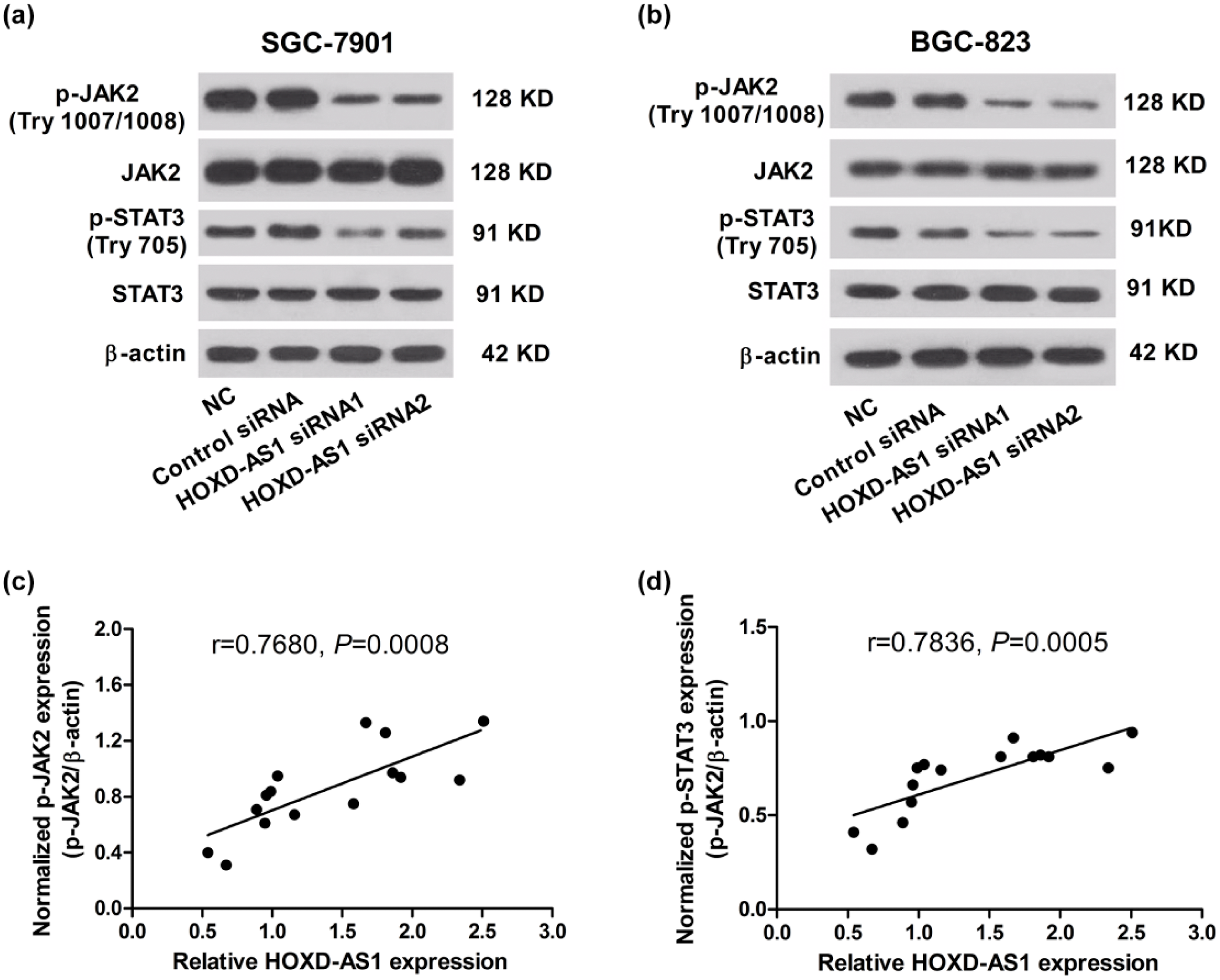
HOXD-AS1 knockdown repressed the activation of JAK/STAT pathway in GC cells. SGC-7901 and BGC-823 cells were transfected with HOXD-AS1 siRNA1, HOXD-AS1 siRNA2, or control siRNA and maintained for 48 h. Western blot analysis was performed to detect the expression of p-JAK2 (Try1007/1008), JAK2, p-STAT3 (Try705), and STAT3 in (a) SGC-7901 (b) and BGC-823 cells. β-actin was regarded as the internal control. Data are presented as the mean ± SD (n = 3). Correlations between (c) p-JAK2 and HOXD-AS1 levels, and (d) p-STAT3 and HOXD-AS1 levels was analyzed using chi-square test (n = 15).
HOXD-AS1 promotes GC cell growth by activating the JAK2/STAT3 pathway
JAK siRNAs and JAK2 inhibitor AG490 were used to further confirm the underlying mechanism of HOXD-AS1 knockdown on GC cell growth. As expected, JAK siRNA1 and JAK siRNA2 strikingly inhibited the phosphorylation of JAK2 and STAT3 but did not affect the levels of JAK2 and STAT3 in SGC-7901 cells (Figure 3(a)). Besides, AG490 significantly decreased the levels of p-JAK2 and p-STAT3 in BGC-823 cells in concentration-dependent manners but no obvious change of JAK2 and STAT3 levels was observed (Figure 3(b)). Furthermore, a significant decrease of cell viability rates at 48 h was observed in JAK2 knockdown SGC-7901 cells compared with control siRNA group (Figure 3(c)), and AG490 treatment also led to a strong inhibition on cell viability at 48 h in concentration-dependent manners in BGC-823 cells (Figure 3(d)). Consistently, the colony formation abilities were dramatically reduced in SGC-7901 cells transfected with JAK siRNA1 or JAK siRNA2 compared with control siRNA group (Figure 3(e)). Also, AG490 treatment showed a significant suppression on colony formation abilities of BGC-823 cells in concentration-dependent manners (Figure 3(f)).
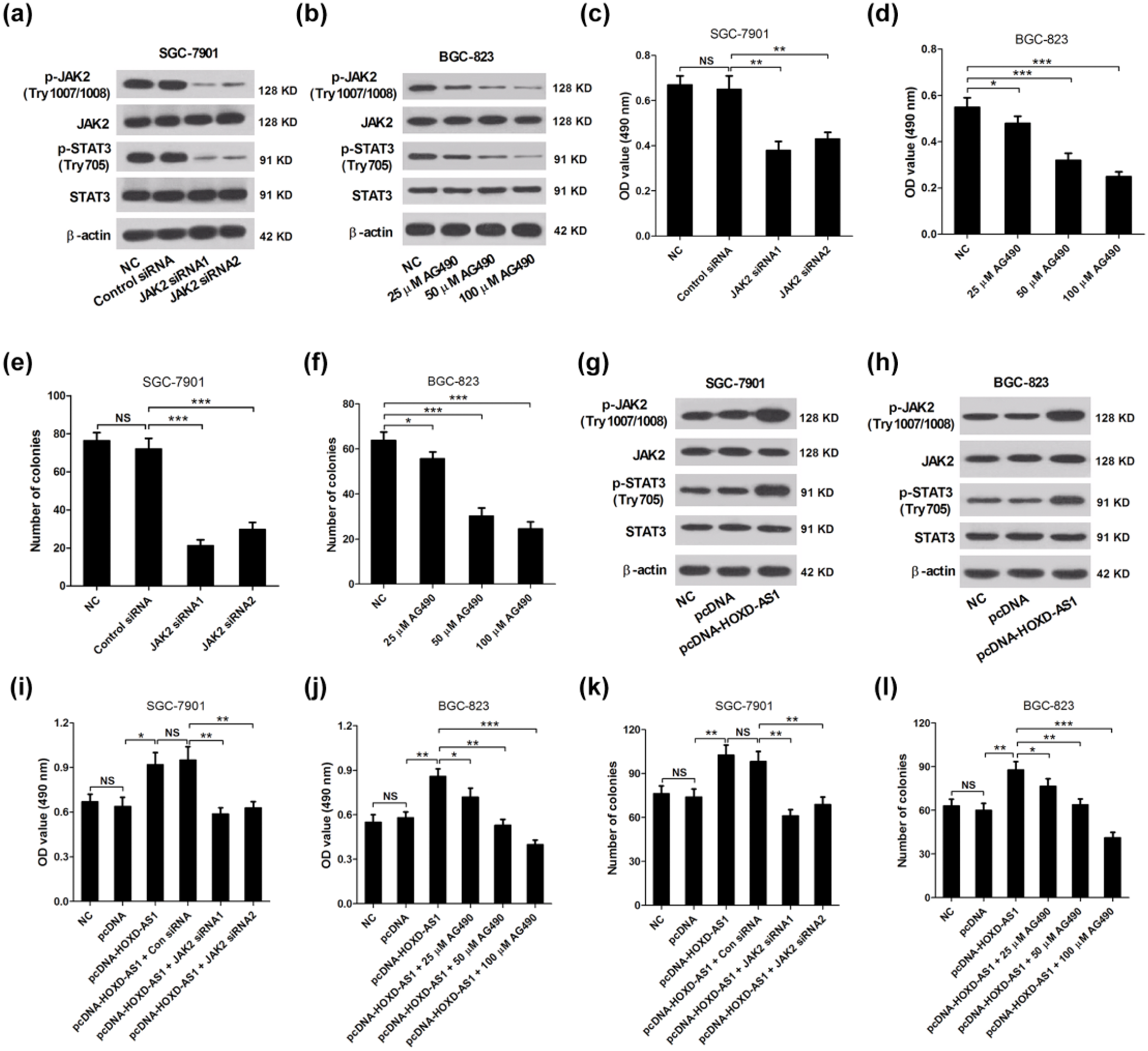
JAK siRNAs and the JAK2 inhibitor AG490 significantly overturned the promotion of GC cell growth mediated by overexpression of HOXD-AS1. (a) Western blot analysis was performed to determine the expression of p-JAK2 (Try1007/1008), JAK2, p-STAT3 (Try705), and STAT3 in SGC-7901 cells transfected with JAK siRNA1, JAK siRNA2, or control siRNA. (b) Western blot analysis was performed to determine the expression of p-JAK2 (Try1007/1008), JAK2, p-STAT3 (Try705), and STAT3 in BGC-823 cells treated with 25, 50, and 100 µM AG490 for 48 h. MTT assay was carried out to assess cell viability at 48 h in (c) SGC-7901 cells transfected with JAK2 siRNA1, JAK2 siRNA2, or control siRNA and (d) in BGC-823 cells treated with 25, 50, and 100 µM AG490 for 48 h. Colony formation assay was used to examine the colony formation abilities in (e) SGC-7901 cells transfected with JAK2 siRNA1, JAK2 siRNA2, or control siRNA and (f) in BGC-823 cells treated with 25, 50, and 100 µM AG490 for 48 h. The expression levels of p-JAK2 (Try1007/1008), JAK2, p-STAT3 (Try705), and STAT3 in (g) SGC-7901 and (h) BGC-823 cells transfected with pcDNA-HOXD-AS1 or pcDNA. (i) Cell viability at 48 h was evaluated by MTT assay in SGC-7901 cells transfected with pcDNA, pcDNA-HOXD-AS1, pcDNA-HOXD-AS1 + JAK2 siRNA1, pcDNA-HOXD-AS1 + JAK2 siRNA2, or pcDNA-HOXD-AS1 + control siRNA. (j) Cell viability at 48 h was detected by MTT assay in BGC-823 cells treated with pcDNA, pcDNA-HOXD-AS1, pcDNA-HOXD-AS1 + 25 µM AG490, pcDNA-HOXD-AS1 + 50 µM AG490, or pcDNA-HOXD-AS1 + 100 µM AG490. (k) Colony formation abilities were estimated by colony formation assay in SGC-7901 cells transfected with pcDNA, pcDNA-HOXD-AS1, pcDNA-HOXD-AS1 + JAK2 siRNA1, pcDNA-HOXD-AS1 + JAK2 siRNA2, or pcDNA-HOXD-AS1 + control siRNA. (l) Colony formation abilities were estimated by colony formation assay in BGC-823 cells treated with pcDNA, pcDNA-HOXD-AS1, pcDNA-HOXD-AS1 + 25 µM AG490, pcDNA-HOXD-AS1 + 50 µM AG490, or pcDNA-HOXD-AS1 + 100 µM AG490. Data are presented as the mean ± SD (n = 3; *p < 0.05, **p < 0.01, and ***p < 0.001).
In addition, overexpression of HOXD-AS1 by pcDNA-HOXD-AS1 contributed to an improvement in phosphorylation of JAK2 and STAT3 in SGC-7901 (Figure 3(g)) and BGC-823 (Figure 3(g)) cells. Compared with the cells transfected with pcDNA, overexpression of HOXD-AS1 by pcDNA-HOXD-AS1 significantly promoted cell viability at 48 h in SGC-7901 (Figure 3(i)) and BGC-823 (Figure 3(j)) cells, which was conspicuously relieved by JAK knockdown or AG490 in concentration-dependent manners. Besides, overexpression of HOXD-AS1 by pcDNA-HOXD-AS1 obviously improved colony formation abilities of SGC-7901 (Figure 3(k)) and BGC-823 (Figure 3(l)) cells, which was dramatically reversed by JAK knockdown or AG490 in concentration-dependent manners. All the results indicated that HOXD-AS1 promoted GC cell growth by activating the JAK/STAT pathway.
HOXD-AS1 knockdown repressed GC cell growth by inactivating the JAK2/STAT3 pathway in vivo
SGC-7901 cells transfected with HOXD-AS1 siRNA1 or control siRNA were subcutaneously injected into nude mice for xenografts to further examine the tumor-suppressing effect of HOXD-AS1 siRNA in GC in vivo. The results of in vivo xenograft assays showed that HOXD-AS1 knockdown significantly inhibited xenograft tumor volume at the indicated time (Figure 4(a)) and xenograft tumor weight (Figure 4(b)) in comparison with that of control siRNA group. Besides, the expressions of HOXD-AS1 (Figure 4(c)) and the phosphorylation of JAK2 and STAT3 (Figure 4(d)) in excised tumor samples were significantly reduced by HOXD-AS1 knockdown. These data indicated that HOXD-AS1 knockdown obviously repressed GC cell growth by inactivating the JAK2/STAT3 pathway in vivo.

Effect of HOXD-AS1 knockdown on GC cell growth in vivo and its potential mechanism. SGC-7901 cells transfected with HOXD-AS1 siRNA1 or control siRNA were subcutaneously injected into nude mice for xenografts. (a) Tumor volume of in vivo xenograft experiments was calculated for every 7 days from day 7 of injection. (b) Tumor weight of in vivo xenograft experiments was detected at 21 days after injection. (c) The expression of HOXD-AS1 in excised tumor samples was determined by qRT-PCR. (d) The levels of p-JAK2 (Try1007/1008), JAK2, p-STAT3 (Try705), and STAT3 in excised tumor samples were evaluated by western blot analysis. Data are presented as the mean ± SD (n = 6; *p < 0.05 and ***p < 0.001).
Discussion
GC is one of the most aggressive gastrointestinal malignancies globally. Despite remarkable advances in GC have been made, including oncogenes or tumor suppressor gene mutation, aberrant protein expression, and identification of cancer stem cell, the early diagnosis and treatment of GC still remains unfavorable. 22 The poor prognosis of GC poses a serious challenge to the survival of patients with GC. 23 Therefore, an improved understanding of the molecular mechanisms associated with GC pathogenesis and development is urgently needed to identify useful indicators and develop novel effective therapeutic strategies for GC.
Due to the rapid advancement of whole-genome and transcriptome-sequencing technologies, an increasing number of lncRNAs are found to play important roles in regulating complicated biological processes of GC, such as cell proliferation, metastasis, and apoptosis. 24 For example, Li et al. 25 reported that lncRNA CASC2 inhibited GC cell growth in vitro and in vivo by inactivation of mitogen-activated protein kinase (MAPK) pathway. Chen et al. 26 found that lncRNA XIST (X-inactive specific transcript) regulated GC progression by functioning as a molecular sponge of miR-101 to modulate EZH2 (enhancer of zeste homolog 2) expression. Zhou et al. 27 indicated that the upregulation of lncRNA ZFAS1 is involved in the regulation of epithelial-to-mesenchymal transition (EMT) in GC progression. In addition, HOXD-AS1 has been recently demonstrated to be involved in the development of various cancers. For example, Li et al. 17 reported that HOXD-AS1 overexpression was related to the bladder cancer by suppressing the phenotypes and activating endogenous cancer-related molecular pathway. Yarmishyn et al. 15 found that HOXD-AS1 was a marker of neuroblastoma progression via integrative analysis of non-coding transcriptome. In this study, we investigated the clinical significance of HOXD-AS1 expression in GC and found that the high expression of HOXD-AS1 was clinically associated with tumor size, invasion depth, TNM stages, regional lymph nodes, lymphatic metastasis, as well as distant metastasis of GC patients. Besides, the expression levels of HOXD-AS1 were elevated in GC cells compared to normal gastric mucosa cells. Furthermore, siRNA-mediated HOXD-AS1 knockdown was performed in GC SGC-7901 and BGC-823 cells to explore the role of HOXD-AS1 in GC progression. The results indicated that HOXD-AS1 knockdown strikingly inhibited GC cell proliferation and colony formation ability in vitro, suggesting that HOXD-AS1 may play an oncogenic role in GC progression.
The JAK2/STAT3 pathway plays a crucial role in cell proliferation, differentiation, and apoptosis. Recent studies have indicated that the abnormality of JAK2/STAT3 pathway has been consistently implicated in numerous cancers, including GC. 21 For example, Zhou et al. 28 reported that miR-133b and miR-135a promoted apoptosis by JAK2/STAT3 signaling pathway in human renal carcinoma cells. Sulaiman et al. 29 revealed that an azaspirane derivative inhibited breast cancer cell growth, metastasis, invasion, and induced apoptosis by inhibiting the JAK2/STAT3 pathway. Judd et al. 30 reported that JAK inhibitor WP1066 inhibited GC cell growth and reduced inflammation in vitro and in vivo by inhibiting STAT2 phosphorylation. This study showed that HOXD-AS1 knockdown dramatically suppressed the phosphorylation of JAK2 and STAT3, while overexpression of HOXD-AS1 remarkably promoted the phosphorylation of JAK2 and STAT3 in GC cells. The studies further revealed that both JAK2 knockdown and JAK2 inhibitor AG490 significantly inhibited GC cell proliferation and colony formation abilities. Importantly, JAK2 knockdown and JAK2 inhibitor AG490 also, obviously, inhibited the promotion of GC cell proliferation and colony formation abilities mediated by HOXD-AS1 overexpression, indicating that HOXD-AS1 promoted GC cell growth by activating the JAK2/STAT3 pathway in vitro. Furthermore, in vivo xenograft assays confirmed that knockdown of HOXD-AS1 significantly inhibited GC cell growth by inactivating the JAK2/STAT3 pathway in vivo.
In conclusion, this study first investigated the biological function of HOXD-AS1 in GC progression, including cell proliferation and colony formation assay, and these data demonstrated that HOXD-AS1 was significantly upregulated in GC cells and may serve as an effective biomarker for GC. In addition, knockdown of HOXD-AS1 dramatically suppressed GC cell growth by inactivating the JAK2/STAT3 pathway in vitro and in vivo, indicating that HOXD-AS1 may be a novel prognostic factor and a potential therapeutic target for patients with GC.
Footnotes
Acknowledgements
L.Z. and J.C. contributed equally to this work.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Ethical approval
All procedures involving human participants were performed with approval of the Ethics Committee of Huaihe Hospital of Henan University and all the patients signed informed consents. All animal experiments were approved by the Institutional Animal Care and Use Committee of Huaihe Hospital of Henan University.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
