Abstract
MicroRNA 183 (miR-183) was identified to be downregulated in nasopharyngeal carcinoma spheroids and served as a tumor suppressor in nasopharyngeal carcinoma. However, the regulatory mechanism of miR-183 and its role in cisplatin (DDP) resistance in nasopharyngeal carcinoma cells are still unclear. The expression of miR-183 and metastasis-associated protein 1 at messenger RNA and protein levels in nasopharyngeal carcinoma tissues and cells was evaluated using quantitative reverse transcription real-time polymerase chain reaction and western blotting, respectively. CNE1 and CNE2 cells were transfected with miR-183 mimic, miR-183 inhibitor, pcDNA–metastasis-associated protein 1, or respective controls. The effects of miR-183 and metastasis-associated protein 1 overexpression on cell proliferation, invasion, and DDP-induced apoptosis were detected by 3-(4,5-dimethylthiazol-2-yl)-2,5-diphenyltetrazolium bromide assay, Transwell invasion assay, and flow cytometry analysis, respectively. Luciferase reporter assay was performed to explore whether miR-183 directly targeted metastasis-associated protein 1. Xenograft tumor experiment was applied to confirm the biological function of miR-183 in vivo. MiR-183 was downregulated in nasopharyngeal carcinoma tissues and cells and negatively correlated with metastasis-associated protein 1 expression. Ectopic expression of miR-183 markedly suppressed cell proliferation and invasion and strikingly enhanced DDP-induced apoptosis in nasopharyngeal carcinoma cells, whereas metastasis-associated protein 1 overexpression partially reversed these effects. Luciferase reporter assay demonstrated that metastasis-associated protein 1 was a direct target of miR-183. MiR-183 negatively regulated the expression of metastasis-associated protein 1 at both the messenger RNA and protein levels. Xenograft tumor experiment indicated that miR-183 overexpression repressed tumor growth and improved DDP-induced cytotoxicity in nasopharyngeal carcinoma cells in vivo. MiR-183 overexpression inhibited tumorigenesis and enhanced DDP-induced cytotoxicity by targeting metastasis-associated protein 1 in nasopharyngeal carcinoma, contributing to the development of novel therapeutic approaches for the treatment of clinical nasopharyngeal carcinoma patients.
Introduction
Nasopharyngeal carcinoma (NPC) is a highly invasive and metastatic malignancy of the head and neck in southern China and South East Asia, with the incidence rate of approximately 10–50/105 individuals per year.1,2 Although the contemporary therapeutic strategies are effective for patients with early-stage disease, patients with advanced NPC often undergo treatment failure due to distant metastasis. 3 A significant number of patients with NPC show local recurrence, cervical lymph node metastasis, as well as post-treatment distant metastasis, and thus, the clinical prognosis for these patients is still generally poor. 4 Cisplatin (DDP) is one of the most effective and commonly used chemotherapeutic agents against various human cancers including NPC. 5 However, the efficacy of DDP-based chemotherapy is limited due to dose-related cytotoxicity and DDP resistance, which are becoming the major obstacles for the effective treatment of NPC. 6 Therefore, it is crucial to identify valuable novel biomarkers for NPC early diagnosis and better understand the molecular mechanisms underlying the pathogenesis, invasion, and metastasis of NPC.
MicroRNAs (miRNAs) are an evolutionarily conserved family of endogenous small non-coding RNAs with 22–25 nucleotides. They play crucial roles in post-transcriptionally inhibiting gene expression through directly binding to complementary target sequences in the 3′ untranslated region (UTR) of their target messenger RNAs (mRNAs), mediating translation repression and/or mRNA degradation. 7 Accumulating evidence has demonstrated that miRNAs can serve as either oncogenes or tumor suppressors to regulate a wide range of biological processes, such as cell proliferation, differentiation, apoptosis, and motility.8,9 Abnormal expression or dysfunction of miRNAs has been demonstrated to be involved in the initiation and progression of most tumor types including NPC.10,11 For example, Mao et al. 12 discovered that overexpression of miR-205 promoted tumorigenesis and tumor progression in NPC through increasing cell proliferation, migration, and invasion and decreasing radiation-induced apoptosis by activating AKT signaling. Chen et al. 13 demonstrated that the tumor-suppressive effects of miR-1 in NPC were confirmed by the suppression of tumor growth and metastasis, which were mediated chiefly by repressing K-ras expression. Xu et al. 14 reported that miR-124-3p expression was downregulated in NPC and overexpression of miR-124-3p promoted apoptosis and inhibited cell proliferation, migration, and invasion of NPC cells by targeting signal transducer and activator of transcription 3 (STAT3). MiR-183, located on human chromosome 7q32.2, was demonstrated to play an oncogenic or tumor-suppressive role in the development and metastasis of various malignant tumors. 15 In a previous study, miR-183 was identified to be downregulated in NPC spheroids and served as a tumor-suppressive miRNA in NPC by suppressing cell growth and tumor sphere formation. 16 However, the underlying mechanism of miR-183 in NPC tumorigenesis remains to be elucidated.
A growing body of evidence has indicated that metastasis-associated protein 1 (MTA1), a component of the nucleosome remodeling and deacetylase (NuRD) complex, is associated with tumor progression and metastasis in multiple tumors including NPC.17,18 In particular, a previous study showed that MTA1 overexpression was significantly correlated with poor survival in NPC and served as a novel biomarker for assessing the metastatic potential of NPC.19,20 In addition, a recent study provided direct evidence that MTA1 promoted DDP resistance in NPC cells. 21
In this study, we aimed to analyze the expression of miR-183 and MTA1 in NPC tissues and cells; explore the role of miR-183 in NPC cell proliferation, invasion, as well as DDP-induced apoptosis; and investigate the relationship between miR-183 and MTA1 in NPC cells.
Materials and methods
Patients and samples
In total, 29 primary fresh frozen NPC biopsies from patients (11 women and 18 men; age range: 45–87 years; median age: 73 years) who were newly histologically diagnosed with NPC and undergone standard surgical procedures and 17 normal fresh nasopharyngeal epithelium tissues from individuals (9 women and 8 men; age range: 43–85 years; median age: 70 years) with chronic nasopharyngitis were collected at the Department of Otorhinolaryngology, Huaihe Hospital of Henan University from January 2012 to December 2014. All fresh samples were immediately snap-frozen in liquid nitrogen and preserved at −80°C until use. The experimental protocols were approved by the Institutional Research Ethics Committee, and written informed consent was obtained from all the individuals participating in the study.
Cell lines and culture
Human Epstein–Barr virus (EBV)-positive NPC cell line, C666-1, and EBV-negative NPC cell line, CNE1 (high differentiation), CNE2 (low differentiation), HONE1 (low differentiation), and 5-8F (high tumorigenesis and metastasis), obtained from American Type Culture Collection (ATCC, Manassas, VA, USA) were cultured in RPMI-1640 medium (Invitrogen, Carlsbad, CA, USA) supplemented with 10% fetal bovine serum (FBS; Gibco, Grand Island, NY, USA) and 1% penicillin/streptomycin solution. The human immortalized nasopharyngeal epithelial cell line NP69 (ATCC) was cultured in keratinocyte serum–free medium (KSFM; Invitrogen) supplemented with epidermal growth factor (Invitrogen). All cells were maintained in a humidified incubator containing 5% CO2 at 37°C.
Cell transfection
MiR-183 mimic (miR-183), miRNA control (miR-control), miR-183 inhibitor, pcDNA-MTA1, and pcDNA empty control (scrambled control) were all purchased from GenePharma Co., Ltd (Shanghai, China). Briefly, CNE1 and CNE2 cells (1.5 × 105) were seeded and maintained on six-well plates containing complete medium without antibiotics at least 24 h prior to transfection. Then, the cells were transiently transfected with miR-183, miR-183 inhibitor, pcDNA-MTA1, miR-183 + pcDNA-MTA1, or matched scrambled controls using Lipofectamine 2000 (Invitrogen) according to the manufacturer’s instructions. Cells were collected at 48 h post-transfection for further experiments.
RNA isolation and quantitative reverse transcription real-time polymerase chain reaction
Total RNA from tissues and cell lines was isolated using TRIzol® reagent (Invitrogen), and RNA concentration was quantified by NanoVue™ Plus (Thermo Fisher Scientific, Loughborough, UK). For detection of miR-183 and MTA1 mRNA expression, 1 µL of total RNA was reverse transcribed into complementary DNA (cDNA) using TaqMan miRNA Reverse Transcription Kit (Applied Biosystems, Foster City, CA, USA) and PrimerScript™ RT Kit (TaKaRa, Shiga, Japan), respectively. Real-time polymerase chain reaction (PCR) was performed to examine the expression of miR-183 and MTA1 mRNA using TaqMan Human MiRNA Assay Kit (Applied Biosystems) and SYBR Green detection system (Thermo Fisher Scientific, Waltham, MA, USA) on a iCycler iQ5 Detection system (Bio-Rad Laboratories, Hercules, CA, USA), respectively. Cycling conditions were as follows: 95°C for 10 min, followed by 45 cycles of 95°C for 15 s, 60°C for 15 s, and 72°C for 10 s. The expression of miR-183 was normalized to small nuclear RNA U6 and MTA1 mRNA was normalized to β-actin. The results were calculated using the 2−ΔΔCt method as described previously. 22
3-(4,5-Dimethylthiazol-2-yl)-2,5-diphenyltetrazolium bromide assay
Cell viability was determined using 3-(4,5-dimethylthiazol-2-yl)-2,5-diphenyltetrazolium bromide (MTT) assay following transfection. Briefly, at 48 h following transfection of NPC cells with miR-183, miR-control, miR-183 + pcDNA-MTA1, or miR-183 + scrambled control, cells were plated onto 96-well plates (3 × 103 cells/well) and incubated for 24, 48, and 72 h. Subsequently, the culture medium was removed and 10 µL of sterile MTT (5 mg/mL; Sigma, St. Louis, MO, USA) was added to each well. Following incubation for another 4 h at 37°C, the MTT solution was removed and 150 µL of dimethyl sulfoxide (DMSO; Sigma) was added to dissolve the formazan crystals on a shaker for 10 min. The absorbance at 570 nm was determined on a model 680 microplate reader (Bio-Rad Laboratories).
Apoptosis analysis by flow cytometry
For the cell apoptosis assay, the transfected cells were treated with DDP (20 µM; Sigma) for 48 h. Then, the cells were harvested and washed with 400 µL of binding buffer. Samples were stained with 100 µL of Annexin V–fluorescein isothiocyanate (BD Biosciences, San Jose, CA, USA) and propidium iodide (BD Biosciences) in the dark for 15 min at room temperature. The percentage of apoptotic cells was measured on a flow cytometer (EPICS Xl-4; Beckman–Coulter, CA, USA). Each experiment was performed at least three times.
Western blotting
Proteins were extracted from NPC tissues and cells using modified radioimmunoprecipitation assay (RIPA) buffer (Beyotime, Haimen, China). Protein concentration was quantified by a bicinchoninic acid (BCA) protein assay kit (Beyotime). Subsequently, a total protein of 50 µg was separated by 10% sodium dodecyl sulfate–polyacrylamide gel electrophoresis (SDS-PAGE) and electroblotted onto a nitrocellulose membrane (GE Healthcare Life Sciences, Chalfont, UK). After blocking the membrane with 5% non-fat milk in phosphate-buffered saline (PBS), the membrane was probed with the primary antibody for MTA1 (1:1000; sc-17773; Santa Cruz Biotechnology, Santa Cruz, CA, USA) and β-actin (1:1000; sc-47778; Santa Cruz Biotechnology) and incubated overnight at 4°C. The membranes were then incubated with a horseradish peroxidase (HRP)-conjugated secondary antibody (1:1000; sc-2379; Santa Cruz Biotechnology). Immunoreactive bands were finally visualized by Chemiluminescent ECL Reagent Kit (Millipore, Bedford, MA, USA) and quantified using ImageJ analysis software (GE Healthcare, Milwaukee, WI, USA). β-actin was used as the internal control.
Luciferase reporter assay
Online bioinformatics softwares TargetScan 23 and microRNA.org 24 were used to predict the interaction probability between miR-183 and MTA1. To generate the luciferase reporter constructs, the 3′-UTR sequences of MTA1 mRNA region containing the wild-type or mutant miR-183 binding sites were synthesized and cloned into the Firefly luciferase–expressing vector psiCHECK™ (Promega, Madison, WI, USA), namely, psiCHECK-MTA1-3′UTR-wt and psiCHECK-MTA1-3′UTR-mut. NPC cells were seeded onto 24-well plates one day prior to transfection and then cotransfected with wild-type or mutant reporter plasmid vectors, along with the Renilla luciferase–expressing vectors pRL-TK (Promega), and miR-183 or miR-control using Lipofectamine 2000 (Invitrogen). Luciferase activity was performed using the Dual Luciferase Reporter Assay (Promega) at 48 h post-transfection. Firefly luciferase values were normalized to Renilla luciferase activity.
Cell invasion assay
The transfected cells (2 × 104/well) were resuspended in 100 µL of serum-free RPMI-1640 medium and plated onto the upper chamber (24-well insert; pore size: 8 µm; Corning Costar, Corning, NY, USA) pre-coated with Matrigel (1:5; 50 µL/well; BD Biosciences). The bottom chambers were filled with 500 µL of RPMI-1640 medium containing 10% FBS. After 24 h of incubation in a humidified chamber containing 5% CO2 at 37°C, the non-invading cells on the upper surface of the filter were removed by wiping with a wet cotton swab and the invasive cells on the lower surface of the filter were fixed with methanol and stained with 0.1% crystal violet (Sigma) for 15 min. Then, the stained inserts were slowly washed in a beaker of water and dried in the air, photographed, and counted under a microscope (magnification: 200×) for each filter in five random optical fields.
Xenograft mouse model
The animal experiments were approved by Huaihe Hospital of Henan University. Male BALB/c nude mice (6 weeks old; weighing 16–19 g; Chinese Academy of Sciences, Shanghai, China) were housed in special pathogen-free (SPF) condition under a 12-h dark/light cycle at constant humidity and temperature for 1 week. To evaluate the tumor growth in vivo, 200 µL of cell suspension from 5 × 106 CNE1 cells were subcutaneously injected into the left inguinal of nude mice. When the xenograft volumes reached 80 mm3 (defined as day 0), all mice were divided into four groups (n = 5/group): miR-183 group, miR-control group, miR-183 + DDP group, and miR-control + DDP group. Mice received intratumoral injection with either miR-183 or miR-control at a dose of 1 nmol per mouse daily or together with DDP (3.0 mg/kg body weight) thrice a week through intraperitoneal injection. Tumor volume was measured every 5 days and calculated by the following formula: Volume = (Length × Width2) × 0.5. Mice were euthanized by cervical dislocation on day 30 and tumors were excised and weighted.
Statistical analysis
All results were showed as mean ± standard deviation (SD) from at least three independent experiments and analyzed using SPSS 17.0 statistical software package (SPSS, Inc., Chicago, IL, USA). The correlation between miR-183 and MTA1 mRNA expression was tested with a two-tailed Pearson’s correlation analysis. Statistical differences were assessed using the student’s t test and one-way analysis of variance (ANOVA). Data were considered to be statistically significant at p < 0.05.
Results
MiR-183 expression was downregulated and MTA1 was upregulated in human NPC tissues
The expression of miR-183 in NPC tissues was examined by quantitative reverse transcription real-time PCR (qRT-PCR). As shown in Figure 1(a), the expression of miR-183 in NPC tissues was significantly lower than that in normal tissues. In addition, the expression of MTA1 at mRNA and protein levels was further evaluated by qRT-PCR and western blotting, respectively. Our data exhibited that MTA1 expression was conspicuously elevated at both mRNA (Figure 1(b)) and protein (Figure 1(c)) levels in NPC tissues compared with that in normal tissues. More notably, a negative correlation between miR-183 and MTA1 mRNA expression in NPC tissues was found (Figure 1(d)). These results showed that miR-183 was dramatically downregulated and MTA1 was markedly upregulated in human NPC tissues.
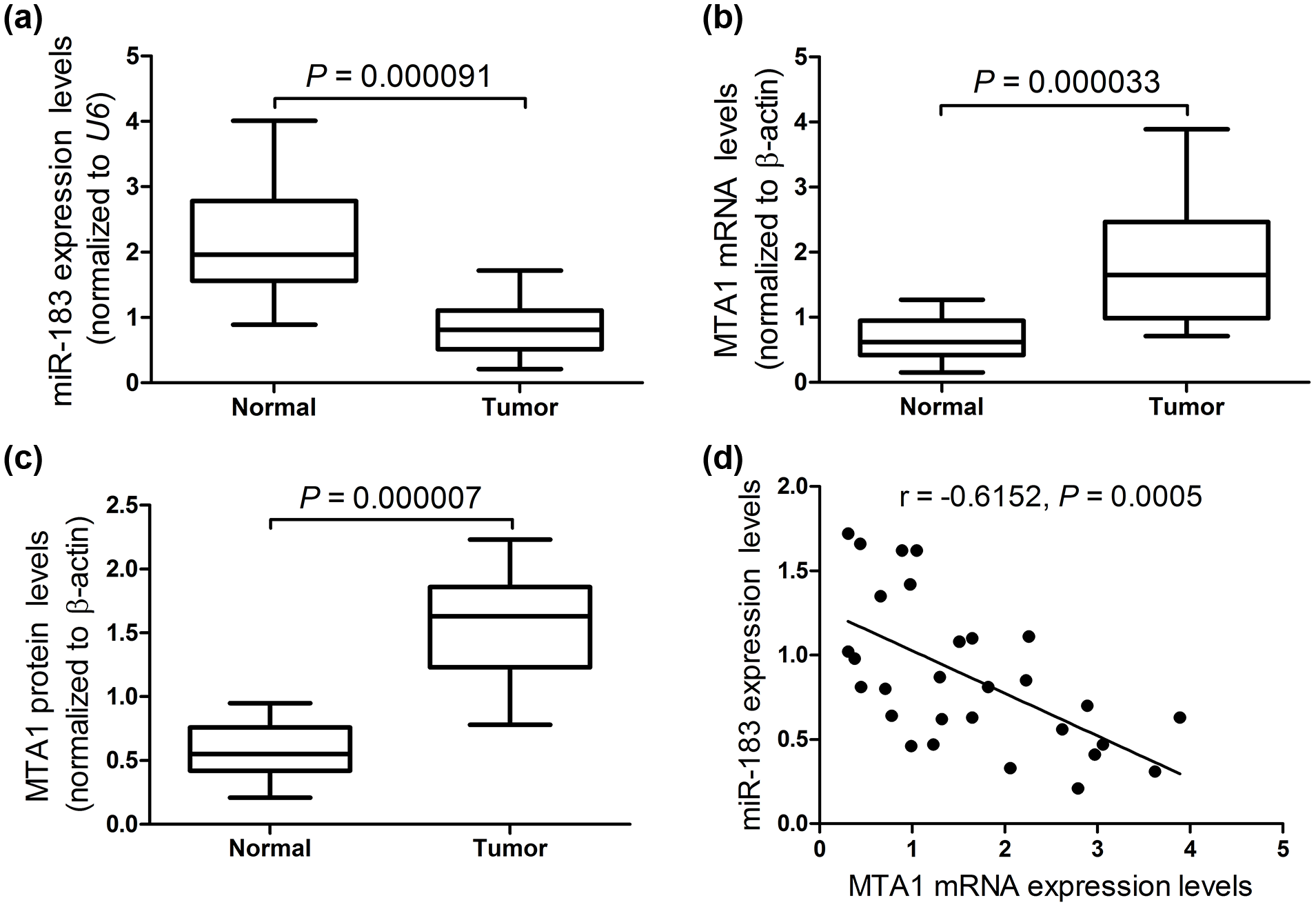
MiR-183 was downregulated and MTA1 was upregulated in human NPC tissues. qRT-PCR was performed to detect the expression of (a) MiR-183 and (b) MTA1 mRNA in 29 NPC tissues and 17 normal fresh nasopharyngeal epithelium tissues. (c) Western blotting was carried out to assess the level of MTA1 protein in 29 NPC tissues and 17 normal fresh nasopharyngeal epithelium tissues. (d) A negative correlation between miR-183 and MTA1 mRNA expression in NPC tissues was found.
MiR-183 was downregulated and MTA1 was upregulated in NPC cells
The expression of miR-183 and MTA1 in NPC cells were further investigated. As demonstrated by qRT-PCR, all NPC cell lines expressed lower levels of miR-183 than that in NP69 cells, especially in CNE1 and CNE2 cells (Figure 2(a)). Meanwhile, the expression levels of MTA1 at both the mRNA and protein levels in all NPC cell lines were exceptionally higher than that in NP69 cells (Figure 2(b)–(d)), especially in CNE1 and CNE2 cells. Therefore, CNE1 and CNE2 cells were chosen for further experiments.

MiR-183 was downregulated and MTA1 was upregulated in NPC cells. (a) The expression of miR-183 in NPC cell lines (HONE1, C666-1, 5-8E, CNE1, and CNE2) and human immortalized nasopharyngeal epithelial cell line NP69 was determined by qRT-PCR. The expression levels of MTA1 at (b) mRNA and (c and d) protein levels were detected by qRT-PCR and western blotting, respectively (*p < 0.05).
MiR-183 overexpression suppressed tumorigenesis and improved DDP-induced cytotoxicity in NPC cells
To confirm the biological function of miR-183 in the development and progression of NPC, we transiently transfected miR-183 or miR-control into CNE1 and CNE2 cells and performed MTT assay, cell invasion assay, and flow cytometry analysis. As shown in Figure 3(a), ectopic expression of miR-183 led to a significant inhibition on cell proliferation at 48 and 72 h in NPC cells. Besides, cell invasion assay results demonstrated that miR-183 overexpression remarkably repressed CNE1 and CNE2 cells invasion with respect to miR-control group (Figure 3(b)). By flow cytometry analysis, exogenous miR-183 dramatically improved apoptotic rates of CNE1 and CNE2 cells compared with miR-control group (Figure 3(c) and (d)). Importantly, miR-183 overexpression further markedly enhanced DDP-induced apoptosis of CNE1 and CNE2 cells, suggesting that miR-183 overexpression improved DDP-induced cytotoxicity in NPC cells. Taken together, we could conclude that miR-183 overexpression suppressed tumorigenesis and improved DDP-induced cytotoxicity in NPC cells.

MiR-183 overexpression suppressed tumorigenesis and improved DDP-induced cytotoxicity in NPC cells. CNE1 and CNE2 cells were transfected with miR-183 or miR-control and cultured for 48 h. (a) The effect of miR-183 on cell proliferation at 24, 48, and 72 h was explored by MTT assay in transfected CNE1 and CNE2 cells. (b) Cell invasion assay was employed to detect cell invasiveness in transfected CNE1 and CNE2 cells. The apoptotic rates of transfected (c) CNE1 and (d) CNE2 cells were determined by flow cytometry (*p < 0.05).
MTA1 was a direct target of miR-183 in NPC
To elucidate the molecular mechanism of miR-183 in NPC, online bioinformatics softwares, TargetScan and microRNA.org, were used to predict the potential targets of miR-183. Based on bioinformatics analyses, MTA1 was identified as a potential target for miR-183. The complementary sequences of miR-183 were found in the 3′-UTR of MTA1 mRNA (Figure 4(a)). To verify the direct target association of miR-183 and MTA1, luciferase reporter assay was carried out in CNE1 and CNE2 cells. As shown in Figure 4(b), cotransfection of CNE1 and CNE2 cells with psiCHECK-MTA1-3′UTR-wt and miR-183 resulted in a marked decrease of luciferase activity compared with miR-control group. However, no obvious inhibitory effects on luciferase activity of MTA1 mutant reporter vectors were observed. To further explore whether miR-183 could regulate the expression of MTA1, qRT-PCR and western blotting were performed to detect the expression of MTA1 at mRNA and protein levels in CNE1 and CNE2 cells transfected with miR-183, miR-183 inhibitor, or miR-control. The qRT-PCR and western blotting results showed that miR-183 overexpression strikingly reduced the expression of MTA1 at mRNA and protein levels in both the CNE1 and CNE2 cells, whereas miR-183 inhibitor remarkably exerted an opposite effect on MTA1 expression levels (Figure 4(c) and (d)), indicating that miR-183 could negatively regulate MTA1 expression. These findings indicated that MTA1 was a direct target of miR-183.

MTA1 was a direct target of miR-183. (a) Fragments of the MTA1 mRNA 3′-UTR containing the wild type or mutant potential binding sites of miR-183. (b) The luciferase activities of the indicated CNE1 and CNE2 cells by luciferase reporter assay. The expression of MTA1 at (c) mRNA and (d) protein levels was assessed by qRT-PCR and western blotting in CNE1 and CNE2 cells transfected with miR-183, miR-183 inhibitor, or miR-control (*p < 0.05; NC: non-transfected cells).
MiR-183 overexpression suppressed tumorigenesis and improved DDP-induced cytotoxicity by targeting MTA1 in NPC cells
Based on the regulatory effect of miR-183 on MTA1 expression, we further investigated the effect of the interaction between miR-183 and MTA1 on tumorigenesis and DDP-induced cytotoxicity in NPC cells. As demonstrated by MTT assay, miR-183 overexpression significantly suppressed cell viability in both CNE1 (Figure 5(a)) and CNE2 (Figure 5(b)) cells compared with miR-control group, while MTA1 overexpression by pcDNA-MTA1 dramatically reversed this effect. Meanwhile, cell invasion was markedly inhibited by miR-183 overexpression in both CNE1 (Figure 5(c)) and CNE2 (Figure 5(d)) cells, whereas MTA1 overexpression significantly recuperated miR-183-induced cell invasion suppression. In addition, flow cytometry analyses results revealed that miR-183 overexpression conspicuously promoted DDP-induced apoptosis in both CNE1 (Figure 5(e)) and CNE2 (Figure 5(f)) cells. However, MTA1 overexpression dramatically restrained the promotion on DDP-induced apoptosis by miR-183. Taken together, these results suggested that miR-183 overexpression suppressed tumorigenesis and improved DDP-induced cytotoxicity by targeting MTA1 in NPC cells.

MiR-183 overexpression suppressed tumorigenesis and improved DDP-induced cytotoxicity by targeting MTA1 in NPC cells. CNE1 and CNE2 cells were cotransfected with miR-183 or miR-control and pcDNA-MTA1 or scrambled control and incubated for 48 h. MTT assay was used to detect cell viability in transfected (a) CNE1 and (b) CNE2 cells. Cell invasion assay was performed to analyze cell invasiveness in transfected (c) CNE1 and (d) CNE2 cells. Flow cytometry was carried out to determine cell apoptosis in transfected (e) CNE1 and (f) CNE2 cells treated with 20 µM DDP for 48 h (*p < 0.05).
MiR-183 overexpression inhibits tumorigenesis and improved DDP-induced cytotoxicity in NPC cells in vivo
We constructed a nude mice xenograft model to explore the effect of miR-183 overexpression on tumorigenesis and DDP-induced cytotoxicity in NPC cells in vivo. As compared with miR-control group, miR-183 overexpression significantly inhibited tumor growth, with the smaller size (Figure 6(a)), suggesting that miR-183 overexpression inhibited tumorigenesis of NPC cells in vivo. Moreover, combination of miR-183 and DDP treatment led to a slower tumor growth than that in tumors treated with DDP and miR-control, indicating that miR-183 overexpression markedly exacerbated DDP-induced cytotoxicity in NPC cells in vivo. Furthermore, miR-183 overexpression also remarkably reduced tumor weight in contrast to miR-control group, while combination of miR-183 and DDP significantly exacerbated this effect (Figure 6(b)). These data demonstrated that miR-183 overexpression inhibited tumorigenesis and improved DDP-induced cytotoxicity in NPC cells in vivo.
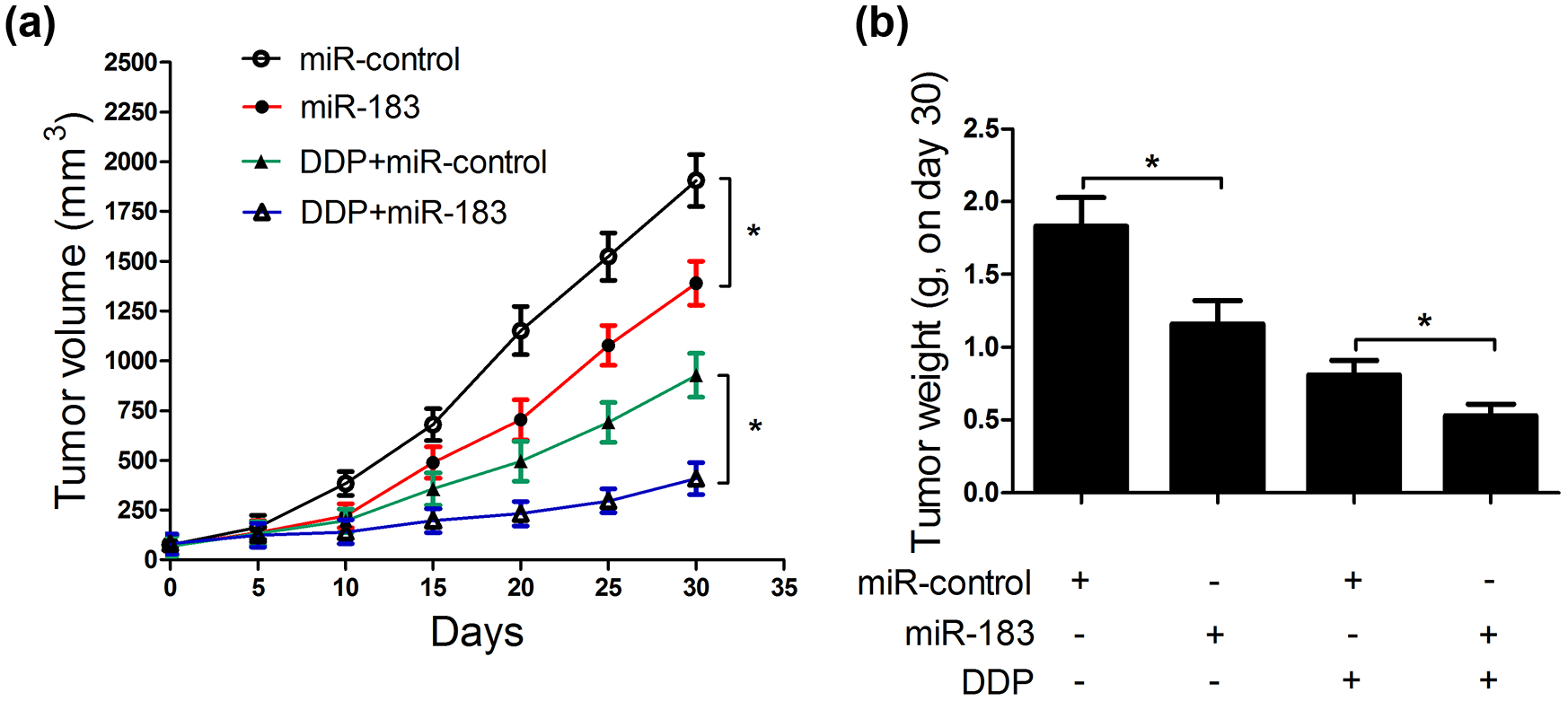
MiR-183 overexpression improved DDP-induced cytotoxicity in NPC cells in vivo. CNE1 cells were subcutaneously injected into the left inguinal of nude mice. When the xenograft volumes reached 80 mm3, mice were injected with either miR-183 or miR-control or together with DDP. (a) Tumor volume in various groups was measured every 5 days. (b) Mice were euthanized by cervical dislocation on day 30 and tumors were excised and weighted (*p < 0.05).
Discussion
In this study, we identified that miR-183 was significantly downregulated and MTA1 was dramatically upregulated in NPC tissues and cells. Notably, a significantly negative correlation between miR-183 and MTA1 mRNA expression was observed in NPC tissues. Besides, ectopic expression of miR-183 restrained tumorigenesis and promoted DDP-induced cytotoxicity in vitro and in vivo. By using luciferase reporter assay and qRT-PCR analyses, miR-183 was demonstrated to directly target MTA1 and negatively regulate its expression. Our results further demonstrated that MTA1 overexpression partially alleviated the suppressive effect of miR-183 on tumorigenesis and abolished the promotion effect of miR-183 on DDP-induced apoptosis in NPC cells, revealing that miR-183 overexpression restrained tumorigenesis and promoted DDP-induced cytotoxicity by targeting MTA1.
Recently, increasing evidence suggests that miRNAs play a crucial role in maintaining normal cellular function and dysregulation of miRNA expression is associated with tumorigenesis and tumor progression.25,26 It has been suggested that miR-183 is a member of an evolutionarily conserved miRNA cluster (miR-183, miR-96, and miR-182) and dysregulated in several types of human tumors, such as breast cancer, osteosarcoma, and hepatocellular carcinoma.27–29 Recent studies showed that miR-183 plays an oncogenic or tumor-suppressive role in the development and metastasis of various types of human tumors. 30 For example, Wang et al. 31 reported that miR-183 was upregulated in astrocytoma tissues and glioblastoma cell lines and promoted glioma cell proliferation and invasion by targeting neurofilament light polypeptide (NEFL). Qiu et al. 32 uncovered that miR-183 was upregulated in renal tissues and functioned as an oncogene by promoting cell proliferation, migration, and invasion by targeting protein phosphatase 2A in renal cancer cells. Cao et al. 30 revealed that miR-183 expression was significantly decreased in gastric cancer tissues compared with its normal tissues and acted as a tumor suppressor in gastric cancer by downregulating ezrin expression. In our study, we identified that miR-183 was significantly downregulated in NPC tissues and cells compared with the normal nasopharyngeal epithelium tissues and NP69 cells. In line with the previous studies,16,30 we showed that ectopic expression of miR-183 suppressed NPC cell proliferation and invasion and induced apoptosis in vitro and repressed tumor growth in vivo, suggesting the tumor-suppressive role of miR-183 in NPC cells.
DDP is widely used as the main chemotherapeutic agent for treating NPC in the clinic. 33 However, DDP resistance subsequently develops during NPC treatment, thus leading to local recurrences and distant metastases in 30%–40% of patients with advanced-stage NPC. 34 Therefore, it is essential to develop new approaches to improve sensitivity to DDP in NPC cells. Increasing evidence has suggested that miRNAs are implicated in the development of chemoresistance of human cancers, mainly through regulating drug resistance–related pathways in cancers.35,36 Zhu et al. 37 reported that overexpression of miR-218 inhibited cell viability, migration, and invasion and increased chemosensitivity to DDP treatment by targeting branched-chain amino acid transaminase 1 (BCAT1) in prostate cancer. Li et al. 38 exhibited that miR-326 overexpression reversed DDP chemoresistance in lung adenocarcinoma cells in vivo and in vitro by targeting specificity protein 1 (SP1). Wu et al. 39 found that the downregulated miR-489 promoted cell growth and reduced the sensitivity of ovarian cancer cells to DDP by targeting protein kinase-b3 (Akt3). In this study, we demonstrated that miR-183 overexpression conspicuously promoted DDP-induced apoptosis in NPC cells, indicating that miR-183 overexpression improved DDP-induced cytotoxicity in NPC cells. In addition, MTA1 was confirmed to be upregulated in NPC tissues and cells and negatively correlated with miR-183 expression. More interestingly, MTA1 was identified as a direct target of miR-183, as demonstrated by luciferase reporter assay and qRT-PCR and western blotting. Further mechanistic analyses revealed that MTA1 overexpression partially overturned miR-183-induced suppression on cell proliferation and invasion and promotion on DDP-induced cytotoxicity in NPC cells, suggesting that miR-183 exerted its biological function in NPC cells by directly targeting MTA1. Consistently, a previous study reported that MTA1 expression was enhanced in drug-resistant NPC cells and played a critical role in promoting DDP resistance in NPC cells. 21 However, there were two limitations in our study: First, no information about tumor–node–metastasis (TNM) stages of the NPC samples was provided and gene expression varies depending on the stage; second, we did not use recent technologies like laser capture microdissection to enrich populations of cells from heterogeneous tissues and the tissues might not be necessarily homogeneous.
In conclusion, we demonstrated that miR-183 was downregulated in NPC tissues and cells and negatively correlated with MTA1 expression. Furthermore, we found that miR-183 overexpression significantly inhibited tumorigenesis and improved DDP-induced cytotoxicity in NPC cells by targeting MTA1, which contributed to novel therapeutic approaches for the treatment of clinical NPC patients.
Footnotes
Acknowledgements
G.W. and S.W. contributed equally to this work.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Ethical approval
Ethical approval was obtained by Huaihe Hospital of Henan University.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
