Abstract
This study aimed to investigate the effects of long non-coding RNA ROR (regulator of reprogramming) on cisplatin (DDP) resistance in patients with non-small-cell lung cancer by regulating PI3K/Akt/mTOR signaling pathway. Human cisplatin-resistant A549/DDP cell lines were selected and divided into control group, negative control group, si-ROR group, ROR over-expression group, Wortmannin group, and ROR over-expression + Wortmannin group. MTT assay was used to determine the optimum inhibitory concentration of DDP. Quantitative real-time polymerase chain reaction and western blotting were applied to detect expressions of long non-coding RNA ROR, PI3K, Akt, and mTOR. Colony-forming assay, scratch test, Transwell assay, and flow cytometry were conducted to detect cell proliferation, migration, invasion, and apoptosis, respectively. Tumor-formation assay was performed to detect the growth of transplanted tumors. Long non-coding RNA ROR expression was high in human A549/DDP cell lines. Compared with the control and negative control groups, the mRNA and protein expressions of PI3K, Akt, mTOR, and bcl-2 decreased, whereas the mRNA and protein expression of bax and the sensitivity of cells to DDP significantly increased. Cell proliferation, migration, and invasion abilities decreased in the si-ROR and Wortmannin groups. In comparison with control and negative control groups, the mRNA and protein expressions of PI3K, Akt, mTOR, and bcl-2 increased, whereas the mRNA and protein expressions of bax decreased, the sensitivity of cells to DDP significantly increased, and cell proliferation, migration, and invasion abilities decreased in the ROR over-expression group. For nude mice in tumor-formation assay, compared with control and negative control groups, the tumor weight was found to be lighter (1.03 ± 0.15) g, the protein expressions of PI3K, Akt, mTOR, and bcl-2 decreased, and the protein expression of bax increased in the si-ROR group. Long non-coding RNA ROR may affect the sensitivity of lung adenocarcinoma cells to DDP by targeting PI3K/Akt/mTOR signaling pathway.
Keywords
Introduction
Non-small-cell lung cancer (NSCLC) serves as the most common sub-type of lung cancer, representing more than 80% of all lung tumors, and about 35% of patients with NSCLC are at the locally advanced non-metastatic disease stage at present.1–4 NSCLC consists of three histological sub-types, including adenocarcinoma, large cell carcinoma, and squamous cell carcinoma. 5 Although some progress has been made in treating lung cancer, the survival rate of lung cancer patients remains quite poor. 6 Also, the prognosis of early-stage NSCLC after surgery is troubling, as the recurrence rate may reach 35%–50%. 7 Furthermore, patients with NSCLC also encounter complications with pervasive and in situ malignant changes, and statistics also demonstrate that smoking is the main risk factor of lung cancer, responsible in more than 80% of cases.8,9 The definition of tumor sub-types plays a crucial role in treatment selection for patients with NSCLC, and treatment regimens should also consider other factors including age, histology, molecular pathology, and comorbidities. 9 Cisplatin (DDP) is one of the classic drugs used during lung cancer chemotherapy which may inhibit DNA replication and harm cell membrane structure. 10 DDP resistance in lung cancer cells is common, so it is important to explore the mechanisms of DDP resistance and provide useful methods to reverse DDP resistance for NSCLC treatment. 11
Approximately 20,000 human protein-coding genes have been identified and most other genes are transcribed into non-coding RNAs (ncRNAs). 12 NcRNAs may be generally divided into two types according to the arbitrary length cutoff, in which those under 200 nucleotides are usually described as short or small ncRNAs, and those greater than 200 bases are known as long non-coding RNAs (lncRNAs). 13 LncRNAs are involved in almost every step in the life cycle of the gene, and regulate the different functions, including gene expression at different levels, chromatin modification, transcription, and post-transcriptional processing. 14 LncRNAs are often found in disease tissues and are characterized by higher developmental stage specificity, offering ways to target and point to specific features for lncRNAs in development and diseases. 15 As an important player in reprogramming differentiated cells into pluripotent stem cells, the lncRNA ROR (regulator of reprogramming) gene is 2.6 kb in length consisting of four exons.16,17 LncRNA ROR has been reported to be involved in many kinds of human cancers and its over-expression in gallbladder carcinoma could predict and promote tumor cell proliferation, migration, and invasion. 18 LncRNA ROR may be used to enhance the invasive ability of female breast cancer by inhibiting the expression of miR-145. 19 Also, lncRNAs screening is beneficial in the diagnosis, sub-classification, and the personalized treatment of NSCLC. 20 Besides this, the PI3K/Akt/mTOR pathway may be activated in NSCLC, and LncRNA ROR has been proven to suppress angiogenesis via the PI3K/Akt/mTOR signaling pathway in human gliomas.21,22 Therefore, our study aims to explore the inner effect mechanisms of lncRNA ROR on DDP resistance in patients with NSCLC by regulating PI3K/Akt/mTOR pathways, which could provide a treatment target for NSCLC.
Materials and methods
Cell culture
The A549 lung adenocarcinoma cell line and the A549/DDP cisplatin-resistant lung adenocarcinoma cell line were purchased from the Cancer Institute of Chinese Academy of Sciences. A549/DDP cells were cultured in RPMI-1640 culture medium containing 10% calf serum and 1% penicillin–streptomycin under conditions of 37°C and 5% CO2. After digestion and passage with 0.25% trypsin, culture medium was changed every 12 h. Cells were then passaged and cryopreserved when covering about 80% of the culture flask. During the culture process, A549/DDP cells were kept in 4-µg/mL DDP to maintain resistance. All cells used for experiments were in the logarithmic growth phase.
MTT assay
A549 cells were obtained in the logarithmic growth phase and suspended with cell growth solution. The cell density was adjusted to 1.0 × 104 cells/mL, and cells were evenly spread in 96-well plates with 100 µL in each well. Cell-culture medium was discarded when cells were adherent to the surface after 24 h of culture, and cells were transferred to different groups (as explained in the following section) of short hairpin RNA (shRNA), respectively, with three duplicated wells. After 24 h of culture, media with different concentrations of DDP (0, 0.5, 1, 2, 4, and 8 µg/mL; Sigma, USA) were used to culture cells for 48 h. Each well was then added 10 µL MTT (3-(4,5-dimethylthiazol-2-yl)-2,5-diphenyltetrazolium bromide; Sigma) and incubated for 4 h before extracting culture solution from 96-well plates and adding 100-µL dimethyl sulfoxide (DMSO; Sigma) in each well away from light. After 15 min of oscillation and dissolution with the flat plate oscillator, a microplate reader was used for detection and the test was repeated for 3 times. Optical density (OD) values of each well were detected under 570-nm wavelength to calculate the inhibition rate of cells. The calculation formula was as follows: cell inhibition rate (%) = 1 − (OD value of experimental group/OD value of normal group).
Cell transfection and grouping
Long intergenic non-protein coding RNA (lincRNA)-ROR shRNA, pCDNA 3.1-LincRNA-ROR, and negative control (NC) shRNA were synthesized by GenePharma Company. The cells were divided into control group (without treatment), NC group (transfected with unrelated sequence), si-ROR group (transfected with LincRNA-ROR shRNA), ROR over-expression group (transfected with pCDNA 3.1-LincRNA-ROR), Wortmannin group (inhibitor Wortmannin, specifically inhibiting the catalytic activity of the PI3Kp110 subunit, thus blocking the activation of the PI3K/Akt/mTOR pathway), and ROR over-expression + Wortmannin group. When cells grew by 30%–50%, complex of transfection reagent and shRNA was prepared according to the instruction of Lipofectamine 2000 Transfection Kit (Invitrogen, USA). The complex was mixed and added into the culture medium to transfect NC shRNA with no interference effect as the control group. After transfection, this was cultured for 24 h in incubator before replacing the medium with RPMI-1640 complete medium containing serum and antibodies. After another 48 h of culture, cells were collected for later experiments.
Quantitative real-time polymerase chain reaction
The total RNA was extracted with Trizol reagent, then the purity and concentration of RNA were detected by ultraviolet (UV) spectrophotometer, and RNA integrity was observed by agarose gel electrophoresis. Tissue samples were ground into homogenate with liquid nitrogen and the total RNA extraction was performed by adding 1-mL Trizol reagent to each 50–100 mg sample tissues. Sample RNA was reversed to cDNA according to the instructions of reverse transcription kit and the total system volume was 10 µL. Then, 65 µL diethyl pyrocarbonate (DEPC)-treated water was added to the obtained cDNA for dilution and complete mixing. Quantitative real-time polymerase chain reaction (qRT-PCR) was performed using ABI 7500 Real-Time PCR System (Applied Biosystems, Foster City, California, USA). Each PCR reaction mixture contained 5-µL SsoFast EvaGreen Supermix (2×), 0.5-µL forward primer (10 µM), 0.5-µL reverse primer (10 µM), and 4-µL cDNA (Bio-Rad Laboratories, Inc., USA). PCR amplification conditions included a total of 40 cycles of pre-degeneration at 95°C for 1 min, degeneration at 95°C for 30 s, annealing at 58°C for 30 s, and finally extending at 58°C–95°C to drawing solubility curve. qRT-PCR primer was synthesized by Beijing Genomics Institute (Table 1). Glyceraldehyde 3-phosphate dehydrogenase (GAPDH) was the internal reference, 2−ΔΔCt was used to calculate the relative expression of target gene, and the results were statistically analyzed.
qRT-PCR primer sequences.

Linc-ROR: long intergenic non-protein coding RNA–regulator of reprogramming; mTOR: mechanistic target of rapamycin; bcl-2: B-cell lymphoma-2; bax: bcl-2-associated X protein; GAPDH: glyceraldehyde-3-phosphate dehydrogenase.
Western blotting
After cell collection, adherent cells were digested by trypsin. Next, cells were centrifuged and washed twice with pre-cooled phosphate-buffered saline (PBS). Then, the cells were added with cell lysates and placed on ice for 30 min. Centrifugation was performed at 12,000 r/min for 20 min at 4°C for full centrifugation of cell fluid. The supernatant was extracted and transferred to EP tube. Bicinchoninic acid assay (BCA) was used to detect the protein concentration and each group was adjusted for identical protein concentration. Then, 5× sodium dodecyl sulfate (SDS) loading buffer was added for 5 min denaturation at 95°C. SDS-PAGE (polyacrylamide gel electrophoresis) electrophoresis was conducted before transferring membrane, followed by the addition of 5% skim milk for closure overnight at 4°C. Following this, after membrane washing with Tris-HCl buffer solution-Tween (TBST), the following primary antibodies were added respectively: PI3K, Akt, mTOR, p-PI3K, p-Akt, p-mTOR, bcl-2, and bax, and internal reference GAPDH (all were purchased from CST Company, USA). After overnight incubation, membranes were washed with TBST and the marked secondary antibody horseradish peroxidase (HRP) was added to incubate for 1 h at 37°C. Then, the membrane was washed with TBST, and HRP-enhanced chemiluminescence (ECL) was added for development. Films were washed with pure water after development and dried for scanning and recording.
Colony-forming assay
A549/DDP cells of logarithmic growth phase were obtained for 0.25% trypsin digestion and divided into single cells after repeated pipetting before suspending in RPMI-1640 culture medium containing 10% fetal bovine serum (FBS). The cell suspensions were diluted by gradient dilutions. Cells of each group were inoculated in 10-mL culture dish at 37°C with gradient densities of 50, 100, and 200 cells/dish, and then evenly mixed. They were placed into 5% CO2 incubator at 37°C and saturated humidity. After 12, 24, 48, and 72 h of culture, the supernatant was, respectively, discarded, carefully washed twice with PBS, followed by the addition of 5-mL of 4% paraformaldehyde for fixed 15 min, then the fixative solution was discarded, and 10 min of crystal violet staining was conducted. This was followed by washing twice with PBS, removal of staining solution, and air drying. The culture dish was inverted with a transparent film (with grid) on its bottom to count the colonies with more than 10 cells under low-power microscope. The colony-forming efficiency was calculated, and the experiment was repeated three times. Colony-forming efficiency = (number of colonies/number of inoculated cells) × 100%.
Scratch test
A549/DDP cells of each group were seeded in 96-well plates and cultured until cells overspread the plate. Spearhead of transferpettor was used to draw vertical scratches on the plate, and an optical microscope was used to observe and record the relative distance of scratches. The cells were cultured for another 48 h in RPMI-1640 culture medium containing 5% serum and then observed for their migration under optical microscope. Scratches were photographed and their relative distance was recorded. The experiment was repeated for three times.
Transwell assay
Cells in each group were digested with 0.25% trypsin containing ethylenediaminetetraacetic acid (EDTA) and centrifuged before diluting with RPMI-1640 culture medium to 5 × 108/L. The Transwell chamber member of each group was evenly coated with 50 µL of 1 g/L Matrigel at 37°C for 30 min so that Matrigel covered all microspores. Diluted A549/DDP cells were seeded on the upper chamber of Transwell, and 0.5-mL RPMI-1640 medium containing 10% FBS was added into the lower chamber before being cultured in an incubator of 37°C and 5% CO2 for 48 h. After the experiment, 95% ethanol was used to fix the cell membrane and cotton swabs were used to wipe cells whose upper surface did not penetrate. Then, 10 min of crystal violet staining was conducted before washing with water. The number of cells penetrating Matrigel was recorded as the number of invasive cells. The experiment was repeated three times.
Flow cytometry
Cells were digested with trypsin containing no EDTA and centrifuged for 5 min at 2000 r/min before washing twice with pre-cooled PBS. Cells were collected by centrifugation, and 1 × 106 cells were transferred into Eppendorf (EP) tubes. Into each EP tube, 500-µL binding buffer, 5-µL propidium iodide (PI), and 5-µL Annexin V (Jiangsu KeyGEN BioTECH Corp., China) were added. After adding staining material, the EP tube was incubated for 10–15 min away from light. Cell suspension was filtered with 400-mesh cell strainer to remove cell masses and placed on ice. Within 30 min, flow cytometry detection was conducted to analyze cell apoptosis. The experiment was repeated three times.
Tumor-formation assay
BALB/C male nude mice of 4 weeks were purchased from the Animal Care Facility of our university, and all operations were carried out in accordance with the animal ethics code of our university. Cultured A549/DDP cells were divided into the control group (without treatment), the NC group, and the si-ROR group (transfected with si-ROR). About 24 h after transfection, cells were digested with trypsin, washed two times with PBS, re-suspended in serum-free 1640 medium, and counted. In each of the three groups, 1.5 × 107 cells were suspended in 0.1-mL serum-free 1640 medium and then mixed with 0.1-mL extracellular matrix (ECM) gel before injecting subcutaneously into the back of nude mice. When the tumor appeared, cis-dichlorodiammineplatinum (DDP) treatment was applied via intra-peritoneal injection (3.0 mg/kg, three times a week). The volume of subcutaneous tumor was calculated every week according to the following formula: V = (0.4 × long diameter × short diameter2)/2. No animal toxicity appeared and mice were euthanized after 4 weeks.
Immunohistochemistry assay
Immunohistochemical streptavidin–peroxidase (SP) staining was used to detect the expressions of bax and bcl-2 proteins in the subcutaneous tumor tissues of mice. Rabbit-anti-mouse bax and bcl-2 polyclonal antibodies were purchased from Bioworld Technology Company. Sample paraffin sections were dewaxed in water and heated with microwave to repair antigens. Heating was suspended for 5 min after boiling and repeated once more before cooling to the room temperature. Sections were washed with PBS and then added with normal goat serum. The primary antibody was added, and sections were preserved at 4°C overnight. When the sections were rewarmed to room temperature, secondary antibody solution EnVision (Beijing ZSGB BioTECH Corp., China) was added at 37°C for 30 min. After washing with PBS, horseradish labeled solution was added for incubation. Diaminobezidin (DAB; Beijing ZSGB BioTECH Corp.) was used for color development for 5–10 min and staining time was adjusted under the microscope. After 1 min of hematoxylin staining, sections were mounted and observed under microscope. The positive expressions of bax and bcl-2 were observed in the cytoplasm, showing yellow or brown color. Three to five high-power fields (×400) with number of tumor cells greater than 500 were selected randomly, and the proportion of positive cells was counted as the relative expression of the target protein.
Statistical analysis
Statistical analysis was conducted with SPSS 21.0 (SPSS, Chicago, IL, USA). The experiment was repeated three times to calculate mean value and standard deviation (SD). Measurement data were presented as mean ± SD. Differences between groups were analyzed using t-test. Comparisons among multiple groups were analyzed using one-way and two-way analyses of variance (ANOVAs). p < 0.05 was considered statistically significant.
Results
Expressions of lincRNA-ROR in A549 lung adenocarcinoma cell line compared with A549/DDP cisplatin-resistant lung adenocarcinoma cell line
qRT-PCR detection showed that the expression of lincRNA-ROR significantly increased in the A549/DDP cisplatin-resistant lung adenocarcinoma cell line compared with the A549 lung adenocarcinoma cell line (p < 0.05) (Figure 1).

Expression of lincRNA-ROR in lung adenocarcinoma cells.
Comparison of DDP sensitivity of transfected cells in each group
MTT detection showed that within the mass concentration range of 0–8 µg/mL, the increase in cisplatin concentration enhanced the inhibitory effect on cell proliferation (Figure 2). In concentrations of 0.5, 1, 2, 4, and 8 µg/mL, the inhibition rates of cell proliferation were (2.25 ± 0.21)%, (9.77 ± 1.32)%, (32.74 ± 2.07)%, (62.61 ± 2.78)%, and (68.08 ± 2.95)%, respectively. There was no significant difference in inhibition rate of cell proliferation between concentrations of 4 and 8 µg/mL. Therefore, 4 µg/mL is selected as the cisplatin concentration in the follow-up experiment.

Effects of DDP on cell proliferation of A549 cells under different concentrations.
Comparison of ROR, PI3K, Akt, mTOR, bax, and bcl-2 mRNA expressions among six groups
There was no obvious change in the mRNA expressions of ROR, PI3K, Akt, mTOR, bax, and bcl-2 in the control group and the NC group, and no significant difference was found between the two groups (p > 0.05). Compared with control group, the mRNA expression of ROR in the si-ROR group was decreased, the Wortmannin group had no significant change, and those in the ROR over-expression group and the ROR over-expression + Wortmannin group significantly increased (p < 0.05). Also, the mRNA expressions of PI3K, Akt, and mTOR in the si-ROR and Wortmannin groups significantly decreased and those in the ROR over-expression group significantly increased (all p < 0.05), whereas those in the ROR over-expression + Wortmannin group showed no significant change (p > 0.05) (Figure 3).

Comparison of ROR, PI3K, Akt, mTOR, bax, and bcl-2 mRNA expression among six groups.
Comparison of apoptosis-related protein and autophagy-related protein expressions among six groups
No change was found in the expressions of apoptosis-related proteins and autophagy-related proteins in the control group or NC group, and there was no significant difference between the two groups (p > 0.05). Compared with control group, the p-Akt, p-mTOR, p-PI3K, and bcl-2 expressions in si-ROR group and Wortmannin group had significantly decreased, whereas the bax expression had significantly increased (p < 0.05). The p-Akt, p-mTOR, p-PI3K, and bcl-2 expressions in the ROR over-expression group all increased, whereas the bax expression decreased (p < 0.05). There were no significant changes in the expression of the related proteins in the ROR over-expression + Wortmannin group (p > 0.05) (Figure 4(a) and (b)).

Comparison of apoptosis-related protein and autophagy-related protein expression among six groups: (a) protein expressions of apoptosis-related proteins and autophagy-related proteins in each group and (b) protein maps of groups with different treatment.
Comparison of cell proliferation ability among six groups
The results of the colony-forming assay indicated that when compared with the control group, the si-ROR group and the Wortmannin group enhanced the inhibitory effect of DDP on lung adenocarcinoma and decreased proliferation ability of A549/DDP cells at the same time (p < 0.05), and there was no significant difference between these two groups (p > 0.05). The inhibitory effect of DDP on lung adenocarcinoma in the ROR over-expression group had weakened and the proliferation ability of A549/DDP cells had increased at the same time (p < 0.05). The ROR over-expression + Wortmannin group showed no significant change in proliferation ability of A549/DDP cells (p > 0.05) (Figure 5).

Colony-forming efficiencies of lung adenocarcinoma cells at different times after DDP treatment among six groups.
Comparison of cell migration and invasion abilities among six groups
Compared to the control group, the abilities of migration and invasion of cells in si-ROR group and Wortmannin group decreased (p < 0.05), with no significant difference between the two groups (p > 0.05). Cell migration and invasion abilities in the ROR over-expression group increased compared with those in the control group (p < 0.05). There was no significant change in cell migration and invasion abilities of cells in the ROR over-expression + Wortmannin group (p > 0.05) (Figure 6(a) and (b)).

Comparison of cell migration and invasion ability among six groups: (a) number of migrated cells, (b) number of invasive cells, (c) migration diagram, and (d) invasion diagram.
Comparison of cell apoptosis among six groups
The results of the flow cytometry demonstrated that apoptosis occurred in cells in control group and NC group due to the action of DDP, but the apoptosis rate was low and DDP’s effect on apoptosis was not significant (p > 0.05), and there was no significant difference between the two groups (p > 0.05) (Figure 7(a)). Compared with the control group, the si-ROR and Wortmannin groups significantly enhanced the apoptotic effect of DDP on the lung adenocarcinoma cells (p < 0.05). The apoptotic effect of DDP on lung adenocarcinoma cells in the ROR over-expression group weakened (p < 0.05). Also, the ROR over-expression + Wortmannin group showed no significant change in the apoptotic effect of DDP on lung adenocarcinoma cells (p > 0.05) (Figure 7(b)).
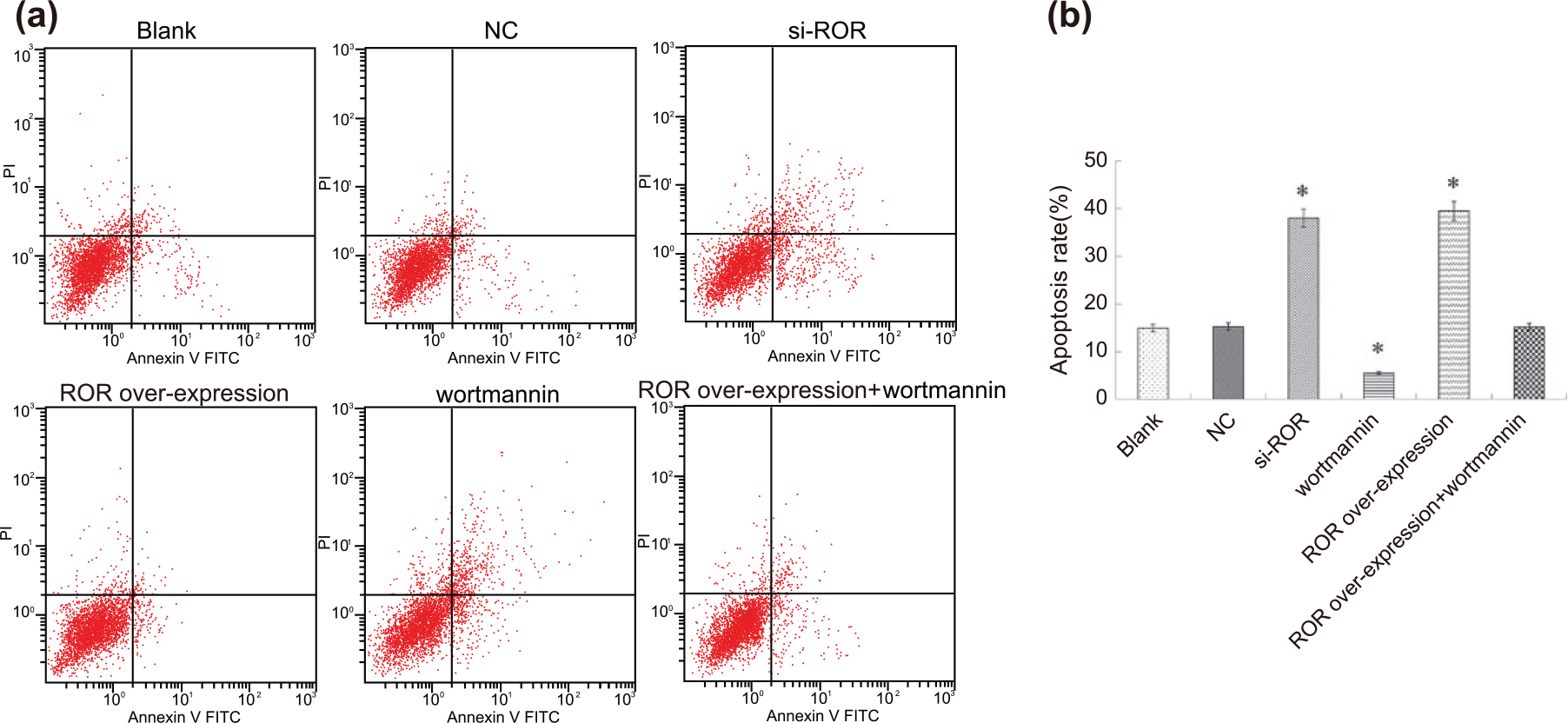
Comparison of cell apoptosis among six groups: (a) flow cytometry diagram and (b) apoptosis rate.
Comparison of growth of transplanted tumor in nude mice among six groups
After 4 weeks of growth, transplanted tumor was clearly observed in mice. The mice were euthanized and their tumors stripped for weighing. The weights of tumors in the control, NC, and si-ROR groups were (3.65 ± 0.28), (3.55 ± 0.37), and (1.03 ± 0.15) g, respectively (Figure 8).

Tumor weights of nude mice among control, NC, and si-ROR groups.
Effects of different treatments on the expressions of related genes in the PI3K/Akt/mTOR signaling pathway of transplanted tumor
In order to further investigate the mechanism of PI3K/Akt/mTOR signaling pathway on attacking tumors in different chemotherapies, we studied the expressions of related genes of transplanted tumors after different inactivation treatment. The results showed that the si-ROR group had significantly lower expressions of Akt, mTOR, and PI3K than the control group (p < 0.05) (Figure 9).

Expressions of related genes in the PI3K/Akt/mTOR signaling pathway of transplanted tumors among the control, NC, and si-ROR groups.
Comparison of bcl-2 and bax expression in transplanted tumors among three groups
Compared with the control group, the positive product of bcl-2 decreased and the positive product of bax increased in the si-ROR group (Figure 10(a)). When inactivated ROR is combined with cisplatin, the effect of PI3K/Akt/mTOR signaling pathway is more obvious, its apoptotic function significantly increased, expression of bax significantly increased, and expression of bcl-2 significantly down-regulated (p < 0.05) (Figure 10(b)).

Expressions of bax and bcl-2 in transplanted tumors among the control, NC, and si-ROR groups: (a) immunohistochemical results of expressions of bax and bcl-2 of transplanted tumors and (b) bax and bcl-2 expression of transplanted tumors.
Discussion
NSCLC accounts for approximately 85% of all lung cancer cases and is a common human cancer with considerably high morbidity and mortality. 20 Aberrant activation of PI3K/Akt/mTOR pathway may be one of the mechanisms of targeted therapy resistance in NSCLC patients. 21 One of the reasons for the failure of chemotherapy in the treatment of lung cancer is drug resistance in tumor cells, so it is urgent to overcome DDP resistance. 23 Therefore, the use of PI3K/Akt/mTOR pathway inhibitors on DDP may hold promise in the treatment of NSCLC.
Our study has demonstrated that the expression of lncRNA ROR in human A549/DDP cell line significantly increased. MTT detection showed that within the mass concentration range of 0–8 µg/mL, the inhibitory effect on cell proliferation is enhanced with the increase in the concentration, in which the optimum concentration of DDP was 4 µg/mL. Inactivation of ROR could significantly reduce bcl-2 expression and increase the expression of bax. LncRNAs are an important component of tumor biology, and their dysregulated expression in cancer suggests that disease progression served as an independent predictor for patient outcomes. 24 As such, lncRNA may be a new marker of cisplatin resistance for NSCLC patients. 25 Previous research has proven that lncRNA ROR expression increased in breast tumors to suggest a possible therapeutic target for aggressive and metastatic breast cancers. 26 Chemotherapy with DDP is superior to the best supportive treatment for this advanced and currently incurable disease. 27 Previous studies have shown that DDP can inhibit relevant cell proliferation and siRNA Stat1 expression in Hela cells, which regulates cell proliferation, and this is increased with higher DDP concentrations.28,29 LncRNAs have important functions in regulating gene expression in pulmonary fibrosis, and lncRNA ROR is involved in various key roles in DNA damage, in which knockdown of lncRNA ROR could decrease some siRNAs.30,31 Expression of the proto-oncogene bcl-2 is connected with tumor progression, and bcl-2 knockdown could represent a new method for human tumor treatment. 32 Bcl-2 and bax are the key mediators in relaying the cell death signaling and finally causing apoptotic cell death. 33 Moreover, lncRNA H19 knockdown has been proven to increase caspase-3, caspase-9, and bax expression and reduce bcl-2, which is consistent with our study findings. 34 Also, inactivation of lncRNA ANRIL could inhibit cell proliferation and increase cell apoptosis with reduced expression of bcl-2 and increased expressions of bax again in line with our research. 35
In addition, our study has indicated that lncRNA ROR can inhibit the PI3K/Akt/mTOR signaling pathway so that inactivation of ROR or inhibition of PI3K/Akt/mTOR signaling pathway could increase the DDP sensitivity of NSCLC so as to promote cell apoptosis, and inhibit cell proliferation, migration, invasion, and tumor growth. PI3K/Akt/mTOR pathway has various cell functions, including cell proliferation, survival, differentiation, and intrusion, and previous literature has proven that lncRNA highly upregulated in liver cancer (HULC) silencing could reduce the phosphorylation of ERK, Akt, and mTOR, which implies that lncRNA ROR could inhibit PI3K/Akt/mTOR pathway in this way. 21 DDP could be associated with the DNA of tumor cells to produce chain and inter chain cross-linking, as well as DNA protein cross link so as to inhibit the expression of DNA repair genes, inhibit cell proliferation, and promote cell apoptosis. 36 LncRNAs have been shown to be involved in tumor genesis and chemotherapy drug resistance, and lncRNA MEG3 may regulate DDP resistance of human lung adenocarcinoma, the knockdown of which has been demonstrated to promote A549/DDP cell apoptosis in vitro. 25 Studies have shown that lncRNA IRAIN can control cell proliferation in NSCLC, and knockdown of IRAIN could inhibit proliferation of A549 cells, which is in line with our study result. 37 Moreover, previous research has proven that lncRNA ROR can promote proliferation, migration, and invasion of colon cancer cells, suggesting that lncRNA ROR has similar functions to NSCLC. 18 LncMALAT-1 transcript may also induce migration of NSCLCs and stimulates tumor growth and invasion in vivo, so lncMALAT-1 could be a prognostic marker in squamous cell carcinoma of the lung. 38 Also, study has found that over-expression of lncRNA ROR indicates a poor outcome in gallbladder cancer patients, showing that inactivation of lncRNA ROR may inhibit the tumor growth. 18
In conclusion, lncRNA ROR may affect drug treatment of NSCLC cells by regulating PI3K/Akt/mTOR signaling pathway, in which silencing lncRNA ROR can improve the sensitivity of NSCLC to DDP. However, the research on lncRNA in NSCLC is only a small part of the mystery, and more biological functions and specific molecular mechanisms remain in the unknown phase. Therefore, more research should be undertaken to provide better reference for the clinical treatment of NSCLC.
Footnotes
Acknowledgements
The authors wish to acknowledge the assistance of the reviewers of this article. H.S. and J.P. are the first co-authors.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
