Abstract
Cancer is the leading cause of death and exhausts human and economic resources for treatment and protection. Zinc oxide nanoparticles play an effective role in tumor treatment but with some cautions, such as overexpression of cytochrome P450, hepatic overload, and the mammalian target of rapamycin pathway resistance. Although lanthanides have antitumor activity, their use is limited. Therefore, the current study aims to improve the effectiveness of zinc oxide nanoparticle via doping with lanthanides, such as samarium. In vitro study revealed that samarium doped with zinc oxide showed more antitumor activity than the other lanthanides, and the antitumor activity depends on the concentration of samarium in the nanocomposite. The in vivo experiment on mice bearing Ehrlich solid tumor revealed that intramuscular injection of samarium/zinc oxide downregulates the expressions of CXCR4 and PI3K/Akt/mammalian target of rapamycin pathway in respect to Ehrlich solid tumor group. Regarding the apoptotic biomarkers, samarium/zinc oxide upregulates the apoptotic biomarker; Bax accompanied with the mitotic catastrophe which was indicated by cell cycle arrest in G2 phase. Moreover, samarium:zinc oxide nanoparticles exhibited minimum toxicity which was indicated by suppressed activities of cytochrome P450 and hepatic enzymes, including alanine transaminase and aspartate transaminase. In addition, the histopathological finding, as well as immunophenotyping results, appreciated the biochemical finding. Therefore, samarium:zinc oxide might be offered a new approach to improve the effectiveness of zinc oxide nanoparticles along with lower toxic effect. Also, samarium:zinc oxide nanoparticles can be a candidate as a new antitumor compound to detect its mode of action.
Introduction
Cancer is widely spread all over the world and threats the life, so that cancer therapy is a hot topic that several means of researches have been carried out to reach suitable pathways to treat cancer, 1 and many chemotherapeutic agents were developed showing great anticancer effect with high risk of side effects and resistance. 2 The applications of nanoparticles (NPs) are growing up in many fields, especially in the medical field to enhance the activity and minimize the toxicity. 3 In spite of involvement of zinc oxide NPs (ZnO NPs) in drug delivery, industry of pharmaceutics, biomedical studies, cosmetics, and food industries, 4 ZnO NPs exhibit toxic effects due to the increase in free zinc ions-inducing oxidative damage and inflammatory response. 5 In general, Zinc ion has some disadvantages, such as elevation of cytochrome P450 as well as liver dysfunction. 6 In addition, some researchers who are interested in lanthanides (rare earth elements), especially in nanoform, try to utilize them in imaging, diagnosis, and treatment. 7 Lanthanides exhibit antitumor effect, such as NPs of CeO2 (nanoceria) which can inhibit the intermediates of reactive oxygen. 8
Some lanthanide complexes show higher antitumor activity than the ligand (liriodenine) but the samarium complex shows the higher anticancer activity than the rest of tested lanthanide complexes. 9 Also, samarium was complexed with (–)-epigallocatechin-3-gallate (EGCG), green tea polyphenols, to enhance its antitumor effect on Cutaneous melanoma. 10 In addition, GdCl3 downregulates the expression of CXCR4 11 which is a chemokine acts as a transducer of the mitogen-activated protein kinase (MAPK) and the mammalian target of rapamycin (mTOR) pathway to increase the proliferation, durability, and migration of tumor cells. 12 The mTOR is kept by phosphoinositide 3-kinase (PI3K) which takes a central function in several signaling pathways, such as cytoskeleton organization of the cells, proliferation, autophagy, and cell survival metabolism. As well as a cross talk between PI3K and CD95 keeps the normal balance of cell proliferation and CD95 inhibition leading to apoptosis 13 where CD95 is considered as a survival factor in some tissues that help in hepatic tissue regeneration 14 and overexpressed in many tumor tissues as pulmonary cancer 15 colon carcinoma 16 and pancreatic cancer. 17 Hence, the aim of this study is, carried out some modifications on ZnO NPs, to improve antitumor activity and minimize its toxicity by doping with some lanthanides.
Materials and methods
Chemicals
Samarium nitrate and chemicals used in this work were purchased from Sigma–Aldrich.
Preparation procedure of Ln3+:ZnO NPs and characterization
Synthesis and characterization were done according to Saif et al. 18 and Ibrahim and Asal, 19 where the typical synthesis of NPs of lanthanides doped ZnO (Ln3+ = Eu3+, Gd3+, and Sm3+) using precipitation method. The synthesis started with addition of 2 g of Zn (acetate)2 into 100 mL of distilled water, then stirred for half an hour. A concentration of 0.015 mol of lanthanide nitrate was added and further subjected to 10-min stirring. The adjustment of pH range (pH = 10–11) of the above solutions was carried out by 15 mL of NaOH solution (4M) as a precipitation of white crystals appear during 1-h stirring at room temperature. The precipitates were separated and washed with deionized water till being neutral. The dryness of the products took place at 80°C in oven. After complete dryness, calcination took place at 450°C for half an hour because any increase in the calcination temperature leads to increase in the crystal size. 20 Meanwhile, pure ZnO was also prepared in the same way without doping. Also, x mol Sm3+:ZnO samples were also prepared with the previous method at different Sm3+ ion concentrations (x = 0.00, 0.015, 0.025, and 0.05 mol).
Characterization
X-ray diffraction (XRD) measurements were made with a Philips diffractometer equipped with a CuKα1 radiation (
Ehrlich Ascites Carcinoma cell line
The in vivo and in vitro experiments were carried out using Ehrlich ascites carcinoma (EAC) cell line which was supplied by Egyptian National Cancer Institute (NCI).
Animals
Swiss albino female mice weighing 22–25 g were purchased from the breeding animal house unit of the Nile Company for pharmaceutical drugs, Cairo, Egypt. The animals’ diet was a commercial standard pellet diet and tap water ad libitum. Animal treatment was in accordance with the National Institute of Health Guide for Animal, as approved by Institutional Animal Care and Use Committee (IACUC).
Antitumor activity, inhibition concentration (IC50), and selection of the most active lanthanide element
Screening the antitumor activity of Ln3+ doped ZnO NPs (Ln3+ = Eu3+, Sm3+, and Gd3+) was carried out using EAC cell line. 21 Briefly, the tumor cells were suspended in saline at concentration (2.5 × 105 cells/mL) then, serial dilutions of NPs were prepared to provide four concentrations 25,50, 100, and 200 μg/mL dimethyl sulfoxide (DMSO). The previous concentrations were added to the suspension of tumor cells and incubated at 37°C for 2 h. The samples were stained with trypan blue. The dead cells can absorb the trypan blue and stained with blue color while the viable cells appear with a clear cytoplasm. The samples were placed on hemocytometer to count the dead and viable cells in two different samples of each concentration. The percentage of dead cells was calculated by dividing the number of dead cells by the total number of the cells and multiplying the result by 100.
Toxicity
Total 50 of mice (10 animals per airy cage) were kept under environmental controlled conditions at 25°C as well as 12 h of darkness, 12 h of light with free supply of diet and water. Intramuscular injections of five concentrations were carried out for the determination of LD50 in the mice. After the injection, the number of alive and dead animals was counted 24 h later to calculate the mortality percentage to determine median lethal dose (LD50) as follows 22


The dose selected for treatment is 1/5 of the LD50.
Effect of ZnO on liver function enzymes
A group of six mice was injected intramuscularly with ZnO suspended in saline (45 mg/kg b. wt.) three times per week for 3 weeks to estimate liver function enzymes.
Side effects of samarium nitrate
A group of six mice was injected intramuscularly with samarium nitrate solution in saline (45 mg/kg b. wt.) three times per week for 3 weeks to notice the side effects of samarium nitrate.
Tumor transplantation
The cells of EAC are hematogenous and have metastatic property. EAC cells were taken from a mouse with ascites and suspended in sterile saline solution then, tested by trypan blue to examine the viability of cells. Dilution of Ehrlich solid tumor (EC) cell suspension to a concentration of (2 × 106 cells/ 0.2 mL) was prepared. The adjusted suspensions were harvested in mice by intramuscular injection in femoral zone and left for 1 week to allow solid tumor growth. 23
Toxicity of ZnO and samarium nitrate
Two groups of six mice each were used to estimate the toxicity of ZnO and samarium salt. The first group was injected intramuscularly with ZnO suspended in saline (45 mg/kg b. wt.) and the second group was injected with samarium nitrate solution in saline (45 mg/kg b. wt.) for 3 weeks day after day, and the side effects and liver function enzymes were tested.
Experimental design
A total of 40 female Swiss albino mice (20–25 g) were randomly divided into four equal groups, each of 10 mice as follows:

Tumor size
Vernier calipers were used to measure the diameters of the solid tumor, then the size of the tumor was calculated as follows 24

where “length” is the greatest diameter longitudinally and “width” is the greatest diameter transversely.
The tumor size was recorded to each mouse in EC and EC + Sm3+:ZnO groups. The mean value of different tumor sizes is calculated for each group, then the standard deviation (SD) is calculated for each group. The values of tumor size were expressed as follows: mean value (SD).
Collection of samples
At the end of the experiment, all animals were subjected to urethane anesthesia 24 h later the last treatment dose. Samples of blood, sera, and tumor tissues were collected and kept at −20°C, and samples from tumor tissues were kept in formal saline for histopathological examination.
Western blotting analysis of PI3K, Akt, mTOR, and β-catenin proteins
Tissues of EC and normal muscles were treated with TRIzol reagent (Invitrogen) to extract the proteins.
25
Levels of proteins were estimated by Bradford
26
method. Separation of 20 mg of proteins takes place with 10% SDS-PAGE (sodium dodecyl sulfate–polyacrylamide
Detection of CXCR4, Bcl2, and Bax gene expression in EC solid tumor
The expression of mRNA of chemokine (CXCR4), anti-apoptotic Bcl2 and apoptotic Bax genes were carried out by extraction of RNA and cDNA synthesis. First, isolation of total RNA from 50 mg of EC tissues by TRIzol reagent. 25 Then 1 mg RNA is used as a template for the synthesis of standard cDNA by reverse transcriptase. The primers are mentioned in Table 1. Quantitative real-time polymerase chain reaction (qPCR) RT-PCRs is done according to Pfaffl 27 for mRNA expression of Nrf2 and COX-2 relatively and the average normalization by control b-actin.
Quantitative real-time PCR primers.

Determination of CD95 in tissue homogenate
The levels of CD95 of tumor and normal cells were measured by flow cytometer (Becton Dickinson, CA, USA) computerized with Dean and Jett 28 mathematical analysis.
Evaluation of cell cycle
Cell cycle and apoptosis were analyzed by flow cytometer. 29 Briefly, the EC tissues were cut and fixed with 70% ethanol. Then incubation with RNase A (50 μg/mL) was taken place overnight, and the staining was done by propidium iodide to be ready for computerized flow cytometric analysis with a program for Dean and Jett 28 mathematical analysis.
Estimation of P450
Cytochrome P450 level in tissues was measured by ELISA kit for mice (MyBioSource, USA).
Biochemical investigation of liver function enzymes
The enzymes of liver activity aspartate transaminase (AST) and alanine transaminase (ALT) in serum were estimated colorimetrically 30 using assay kit (Quimica Clinica Aplicada S.A., Spain).
Histopathological study
Samples of EC and normal thigh muscles tissues in formal saline were washed with water, and serial dilutions of alcohols were used for dehydration. Xylene was used for clearance, and the samples were immersed in paraffin and placed in oven at 56° for 24 h. The blocks of paraffin tissues were sectioned by microtone at 4-µm thickness. The sections were fixed on glass slides, then the paraffin was removed and hematoxylin and eosin stain was used for routine staining. Light electric microscope was used for examination. 31
Statistical analysis
Data were collected and calculated as mean and standard deviation SE. The data were calculated by the statistical software (SPSS) using one-way ANOVA. The significance was set between mean values of the groups and expressed at
Results
Characterization of Ln3+:ZnO
The XRD patterns of pure and 0.015 mol Ln3+:ZnO were shown in Figure 1. The incorporation of samarium appeared as slight shift in the maxima of diffraction peak toward higher 2θ values with respect to pure ZnO where 2θ for pure ZnO = 36.090° and 2θ for lanthanides = 36.217° This means that oxygen vacancies were formed because of lanthanide ions introduction. 32 The ionic radius of Sm3+ is 0.95 Å, Eu3+ is 0.94 Å, and Gd3+ is 0.93 Å which are larger than ZnO 0.6 Å, so that the ions of lanthanides do not incorporate the ZnO lattice but ZnO substitutes ions of lanthanides in their oxides lattice on the interface of Ln3+:ZnO. Lanthanide effectively resides into the interstitial sites of Ln3+:ZnO and oxygen affinities of the lanthanide interstitially located. 33

XRD patterns of 0.015 mol Ln3+ doped ZnO NPs (Ln3+ = Eu3+; Sm3+ and Gd3+).
The TEM image shows that the 0.015 mol Eu3+:ZnO characterized with high performed nanorod and sheet morphologies with low porosity structure (Figure 2(a)). The concentration of 0.015 mol Sm+3:ZnO shows high porous sheet with average pore diameter 12 nm. A minor rod shape was also observed with average 60 nm diameters and 130 nm length (Figure 2(b)). Figure 2(d) shows nanosheets with porosity for 0.015 Gd+3:ZnO.

TEM images of prepared particles. (a) Pure ZnO has relatively nonporous sheets in microscale. (b) Eu3+:ZnO nanoscale rods and sheets with low porous structure. (c) Sm3+:ZnO highly porous nanosheets with high porosity and minor nanospherical shapes. (d) Gd3+:ZnO nanomaterials.
Antitumor experiments (IC50)
Antitumor activity of different Ln3+ doped ZnO NPs (Ln3+ = Eu3+, Sm3+ and Gd3+) is examined against EAC cell line. The NPs of Sm3+:ZnO show the highest activity where 95% of tumor cells died. Eu3+:ZnO and Gd3+:ZnO show very weak activities against EAC cell line (10% and 5%, respectively) as mentioned in (Table 2).
Antitumor activity of different Ln3+ doped ZnO NPs (Ln3+ = Eu3+, Sm3+, and Gd3+) against EAC cell line.
The NP Sm3+:ZnO was selected to study the antitumor activity at different concentrations to select the most active concentration against EAC cell line.
Selection of the most active concentration of Sm3+:ZnO NP
The NP Sm3+:ZnO was selected because its higher antitumor activity against EAC cell line than other lanthanides as shown in Table 2. Series of concentration of Sm doped ZnO NP were tested as antitumor to select the most active concentration of Sm:Zn against EAC cell line (Table 3).
Antitumor activity of different concentrations of Sm3+ doped ZnO NPs against EAC cell line.

IC50: Inhibition concentration of a substance that inhibit 50% of the tumor cells.
The previous Table 3 reveals that the ZnO particles have very weak antitumor activity against EAC cell line while different concentrations Sm3+:ZnO show different antitumor activities which increased as the concentration of Sm3+ increased until Sm3+ reaches 0.015. Any increase in the concentrations of Sm3+ higher than 0.015 show the same activity (i.e. the antitumor activity is almost steady in spite of increasing concentration of Sm from 0.015 to 0.025, so that the concentration (0.015) of Sm3+:ZnO was selected to start the in vivo studies.
Determination of LD50 of Sm3+:ZnO (0.015) in mice
The selected nanosample of Sm3+:ZnO was suspended in saline to prepare five different concentrations (150, 200, 250, 300, and 350 mg/kg body weight). Each concentration was injected intramuscularly in a group of 10 mice, then the number of dead mice was observed. Table 4 mentions the LD50 of the selected sample (235.2 mg/kg body weight). In addition, Sm3+:Zn NPs show low toxicity that it can be injected intramuscular in mice at dose 45 mg/kg b. wt. with no mortality.
Median lethal dose (LD50) of Sm3+:ZnO.

Average of increasing factor ≈ 1.26.
Average of proportionate distance ≈ 0.67.
Log LD50 = Log LD next below 50% + (Log increasing factor x proportionate distance).
LD50 (mg/kg b. wt.) = 235.2.
Toxicity of ZnO and samarium nitrate
The intramuscular injection of ZnO in mice leads to significant increase in hepatic enzymes ALT and AST which means hepatic dysfunction (Table 5).
The effect of ZnO on the liver function enzymes.

ALT: alanine transaminase; SD: standard deviation; AST: aspartate transaminase.
Side effects of samarium nitrate
The most common and clear side effect of intramuscular injection of samarium nitrate was ataxia and muscular spasms which appear half an hour after the injection of samarium nitrate dose, so that the Sm salt of nitrate cannot be injected solely, it should be tested in vivo in form of Sm doped ZnO NPs because ataxia is not noticed in case of doping.
In vivo treatment of EC in mice
Mice were treated with a selected dose (approx. = 45 mg/kg b.wt.) which is one-fifth of LD50. The tumor EC tumor size was monitored weekly. It is clear that the tumor was produced 7 days after inoculation with a mean size of 459.2 (36.04) mm 3 . The tumor size increased by the time and reached 1586 (414) mm 3 on the Day 28.
Treatment of mice bearing tumor (Group 3) by intramuscular injection of Sm3+:ZnO with (45 mg/kg b.wt.) reduced the tumor size from 459.2 (36) mm 3 on the Day 7 to 332.9 (55.2) mm 3 on the Day 28 as shown in Figure 3(a) and (b) and Table 6.

Tumor size treated and untreated mice. (a) Represents the tumor size of treated and untreated mice monitored during the experiment weekly. (b) Photographs of treated untreated tumors EC group and EC + Sm:ZnO group at the end of experiment.
The tumor size in treated and untreated EC groups.

EC: Ehrlich solid tumor (untreated group); EC + Sm3+:ZnO (treated group); SD: standard deviation.
The values are expressed in mm3.
Histopathological examination of tumor
Histopathological examination by light microscope of EC tumor shows compact and aggregated tumor cells involved in the thigh muscles and spread as polygonal, round, and pleomorphic cells with hyperchromatic irregular nuclei (Figure 4).

Photomicrograph represents subcutaneous EC in mice. (a) Normal thigh muscle bundles of control group. (b) Control solid Ehrlich carcinoma within the muscular tissues. (c) Solid Ehrlich carcinoma in mice treated with Sm3+:Zn showing remnants of tumor cells and necrotic areas. (d) Thigh muscle bundles of normal mice treated with Sm3+:Zn.
Treatment of female mice bearing EC tumor by Sm3+:ZnO yields wide destruction of tumor tissue appeared as dead and necrotic cells. The normal mice treated with Sm3+:Zn Figure 4(d) shows normal muscle bundles with respect to control group (4A).
Ehrlich carcinoma is a type of cancer cells undifferentiated, hyperdiploid originally, that can be transplanted easily with no regression and rapid proliferation with short malignancy life span and no antigen for transplanted cells. The high speed growth and undifferentiated EC tissues are similar to human cancer and sensitive to chemotherapy. 34
Western blotting of PI3K, Akt, and mTOR proteins
The presence of EC in mice leads to upregulation of PI3K and mTOR significantly (

(a) Western plot analysis showing the effect of Sm3+:ZnO NPs and EC on the expression of PI3K, Akt, and mTOR in mice. (b) Effect of Sm3+:ZnO NPs and EC on the expression of PI3K, Akt, and mTOR in mice. Values are expressed as mean ± SD. *
Detection of CXCR4 relative gene expression
As the EC was developed in mice, the CXCR4 was upregulated significantly (

Effect of Sm3+:ZnO NPs and EC on the expression of CXCR4 in mice. Values are expressed as mean ± SD. *
Detection of Bcl2 and Bax relative gene expression
Figure 7 shows a disturbance in the levels of Bcl2 and Bax upon development of EC in mice where the Bcl2 increased in significantly (

Effect of Sm+3:ZnO NPs and EC on the Bax and Bcl2 expression in mice. Values are expressed as mean ± SD. *
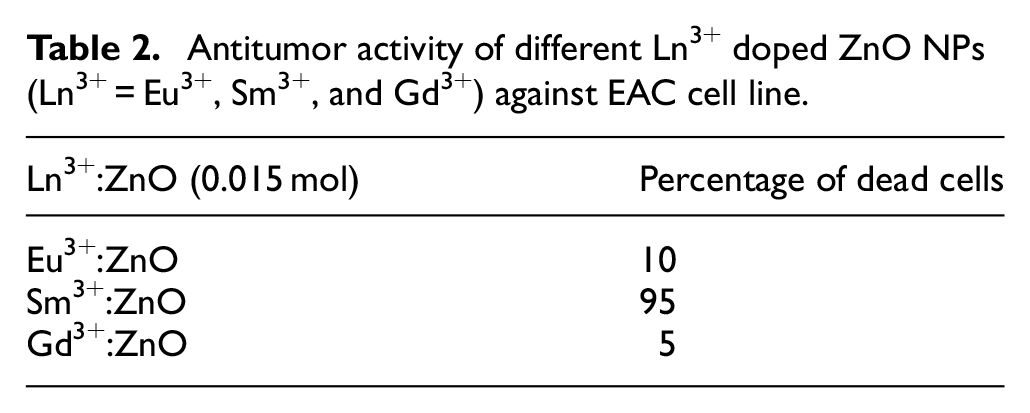
Determination of CD95 in tissue homogenate
The expression of CD95 was elevated in the EC group (
Effect of Sm3+:ZnO NPs and EC on the expression of CD95 in mice. Values are expressed as mean ± SD. *
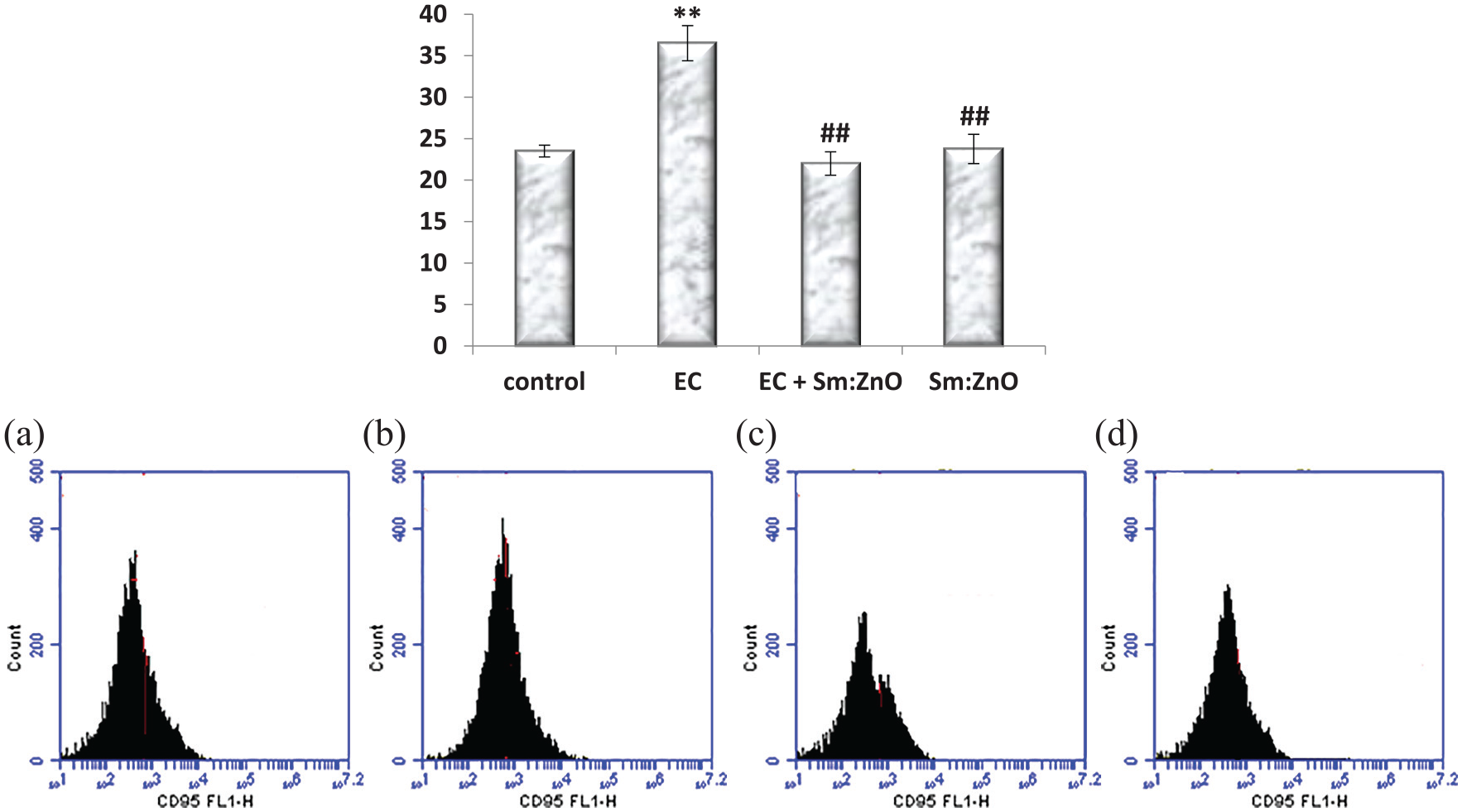
Evaluation of apoptosis and cell cycle analysis by flow cytometry
Cell cycle analysis by flow cytometry of EC shows arrest in the cell cycle at the G2 phase and increased the cell population in the sub-G1 phase upon the NPs treatment (Figure 9).

Flow cytometric charts of the EC cell cycle in mice and effect of NPs treatments in the tumor cell cycle.
Estimation of P450
The level of P450 was increased due to the growth of EC (

Effect of Sm3+:ZnO NPs and EC on the level of P450 in mice. Values are expressed as mean ± SD. *
Biochemical investigation
The liver function enzymes (ALT and AST) were elevated in the transplanted EC mice with significant value (

Changes of ALT and AST enzymes in mice treated with Sm3+:ZnO NPs and nontreated EC bearing mice. Values are expressed as mean ± SD. *
Discussion
Involvement of lanthanides in NPs synthesis is a modern approach in cancer therapy. The efficient permeability of NPs to the cells is due to the advantage of their small size to facilitate their distribution and activity. Also, the smaller size of NPs has higher ability to penetrate and accumulate at tumor tissues. The NPs sustain at the cells with long half-life time because their ability to avoid the capture by the system reticuloendothelial. 35 In this study, ZnO particles are present in microscale, so that ZnO shows weak antitumor activity against EAC cell line because the antitumor activity is observed for NPs ranged from 10 to 100 nm. 36
The NPs were modified by doping with some lanthanide elements to achieve higher antitumor activity to ZnO NPs with lower toxicity.
Lanthanide elements’ participation in cancer treatment is limited in spite of their antitumor activity which were pointed to them in some researches, such as the activity of cerium oxide NPs, 37 lanthanum chloride, 38 and gadolinium. 39 In addition, samarium complexes were tested as antitumor agents and showed higher activity than the ligand-natural alkaloid liriodenine 40 and other lanthanides’ complexes with coumarin-3-carboxylic acid 41 but samarium has not yet fully described from a biochemical pathway point of view.
In addition, treatment with samarium nitrate leads to some side effects, such as ataxia and muscular spasms, which appeared during injection of it in mice and this result was supported by Bruce et al. 42 So that the use of doped Sm is better than treatment with Sm salts to avoid the side effects of Sm.
In the current study, gadolinium, samarium, and europium lanthanide elements were doped ZnO in a constant concentration of 0.015 mol and their antitumor activity was tested in vitro. The samarium Sm3+:ZnO NPs showed the highest antitumor activity against EAC cell line (Table 2). Then different concentrations of Sm3+ element doped ZnO NPs were synthesized and evaluated as cytotoxic agents against EAC cell line to select the suitable concentration for further study. Where, the in vitro antitumor activity of ZnO NPs was increased upon doping with samarium in a concentration-dependent manner till the concentration 0.015 mol with IC50 = 24 µg/mL and any further increase in concentration of Sm3+ up to 0.015 mol, which shows the same activity.
The antitumor effect of ZnO is mTOR-dependent pathway, where the activation of mTOR pathway is well defined in many types of human cancers. So, the mTOR pathway is paid a great attention in cancer therapy. In this study, ZnO shows antitumor activity; this may be related to the efficacy of Zn ions released from ZnO particles to inhibit mTOR activity. This effect may be related to the inhibition of mTOR pathway. It is documented before that ZnO particles induce macrophage autophagy and apoptosis through de-phosphorylation of Akt that leads to PI3K/Akt/mTOR pathway inhibition. 43 Despite, the important role of mTOR, its inhibition solely is not enough that cancer cells can develop resistance to this pathway, so that targeting CXCR4 improves the therapeutic ability and avoids the resistance or enhances the efficacy of mTOR inhibitors. 44 In the current work, a significant decrease in the volume of solid tumor in mice treated with Sm3+:ZnO NPs relative to the tumor bearing group was found. The significantly enhanced antitumor effect accompanied by samarium addition is related to the great ability of samarium to change the cell membrane permeability 45 where the size of Sm ions has high affinity for Ca2+ binding sites where the lanthanides are suitable ions for cationic substitution because their ionic radii are similar to Ca2+ and geometrical coordination as polyhedron. 46
The mTOR pathway has a central role that facilitates the cross talk between many factors to control the cancer cells that it can transfer signals from a receptor, such as CXCR4, to affect other far factors, such as CD95 and Bcl2, to induce death of cancer cells, where the mTOR pathway can be transduced by CXCR4 which induce PI3-kinase phosphorylation leading to progression and spreading of cancer. 12
It was found that the addition of metallic elements improves the antitumor effect via targeting CXCR4 where the affinity of the antitumor ligand (plerixafor) toward the CXCR4 was increased upon complexation with copper, 47 Tc, 48 and Ga 49 complexation. In addition, Aghanejad et al., 50 mentioned that attacking CXCR4 receptor specifically is an important step in cancer cure that downregulates the subsequent pathways, such as mTOR, that they tested the complex of 153Sm-tetraaza macrocycles and found that Sm increases the affinity of complex toward CXCR4 receptor. Also, they studied the biodistribution of 153Sm- tetraaza macrocycles complex in wild-type rats and found that the major sites of accumulation were spleen and lungs which is the highest concentration of CXCR4 receptors. It is found that the expression of CXCR4 increases in several types of tumors and it is necessary for the cancer cell survival. 51 In this work, the addition of samarium increases the activity of the of ZnO NP in a concentration-dependent manner against Ehrlich Carcinoma EC cell line. This is may be due to the affinity of the samarium to the CXCR4 receptor which can downregulate the mTOR pathway more efficiently.
In addition, mTOR pathway can stimulate one of the most important receptors which is CD95. 52 First, CD95 (Fas/APO-1) was known as a receptor for inducing apoptosis 53 but the researches who depended on stimulation of it to obligate the cancer cells to the fate of apoptosis were reached disappointed results because CD95 does not only induce apoptosis but also has dual complex properties (apoptotic and nonapoptotic), but its nonapoptotic property is dominant 54 and inhibition of CD95 is more effective than its initiation for treatment of cancer and inflammatory disorders.
Overexpression of CXCR4 activates the phosphorylation of the PI3K which subsequently mediates the increase in CD95 that enhances the invasion of tumor 55 and acts as an effective factor for prosurvival of stem cell.
Moreover, the overexpression of CD95 is related to cancer cell as a survival factor with no growing defect or death signs in embryonic normal cells of CD95 L knockout mice. 56 The downreglulation of CD95 leads to kill cancer cells in an operation known as death induction by CD95 elimination (DICE) 57 and tested the elimination of CD95 in many cancer cells (~40 cell lines) and they found that knockdown of CD95 led to killing of cancer cells via DICE.
DICE has some subsequent events, such as cells swelling, and dilated vacuoles are appeared according to autophagy, as well as ROS yield. These events are followed by diminished cell viability and subsequent activation of caspases as well as increase in permeability of outer mitochondrial membrane leading to cell death. DICE looks like necrosis due to mitotic catastrophe. 58 In this study, necrosis appeared in the histopathology of the treated tumor with Sm3+:ZnO as an antitumor NP that can induce apoptosis of tumor cells.
The presented data of this study revealed that the increase in the CD95 is associated with increase in the levels of Bcl2 and decrease in Bax. This is may be due to the effect of CD95 to enhance the antiapoptotic protein Bcl2 and decrease Bax to avoid death of the tumor cells. 58 In addition, the decrement of the level of CD95 is accompanied by downregulation of PI3K, Akt, and mTOR as well as inhibition of Bcl2 and elevation in Bax upon treatment with Sm3+:ZnO NPs leading to apoptosis as noticed in the pathological finding as well as cell cycle arrest in phase G2. This relation was supported by Dan et al. 59 whose data revealed that the induction of PI3K/Akt/mTOR pathway via phosphorylation of Akt leads to downregulation of Bax and stabilization of Bcl2 antiapoptotic protein to allow the cancer cell to avoid apoptosis.
Some researchers carried out important steps to detect the mode of action of lanthanides in general. Lanthanides were found to be selective inhibitors for cytochrome P450 60 while the samarium atom has a specific site and interact with two charged side chains of the P450 system. 61 This is may be the reason of decrease in the level of the P450 upon treatment with Sm3+:ZnO NPs in this study. In addition, Li et al., 62 confirmed that Sm has the ability to aggregate in the mitochondria via a competitive interaction with Ca+2 receptors in the biological membranes.
Many studies about ZnO revealed that the treatment with ZnO increases the level of P450 leading to more inflammation and injury. 6 Also, the cytochrome P450 is highly expressed in renal cancers and enhances the achievement of carcinogenicity cell and drug resistance, 63 and CYP2J2 is a member of the cytochrome P450 system which induces the proliferation and decreases the death of cancer cells; therefore, the inhibition of cytochrome P450 is a modern strategy to treat the carcinogenicity. 64
One of the most important limitations of treatment with ZnO was mentioned by Tang et al., 6 who found histopathological changes and injury were increased in the liver upon ZnO uptake in a dose-dependent manner due to the elevation of P450 in such organs. Addition of samarium does not alter the hepatic enzymes and keeps them within the normal ranges in this work. This finding is supported by Weilin et al. 65 who treated Sprague Dawley rats with samarium nitrate and recorded meaningful increase in the liver weight. In addition, the treatment of rats with samarium salts exhibits no hispathological changes in liver 66 and keeps the liver function enzymes within normal ranges. Also, the complexation of samarium 153Sm with 1,1′-[1,4-Phenylenebis (methylene)]bis-1,4,8,11-tetraazacyclotetradecane (AMD 3100; Plerixafor) does not alter the hepatic function 10 days after the injection. 50
In conclusion, Sm+3 doped ZnO NPs enhance the antitumor effect of ZnO via increasing the affinity toward CXCR4 receptor that controls the carcinogenicity of CD95 through PI3K/Akt/mTOR pathway to enhance apoptosis by DICE and G2 cell cycle arrest. Moreover, addition of samarium to ZnO offers a chance to avoid cancer cell resistance via downregulation of P450 and prevent hepatotoxicity. The NPs Sm+3/ZnO could be a promising antitumor agent but more researches must be planned to study its biochemical action and cross talk between several pathways in vivo in more details.
Footnotes
Acknowledgements
The authors thank Dr Adel M Bakeer, Professor of Pathology, Faculty of Veterinary Medicine, Cairo University, Egypt, for his support.
Approval
Animal dealing conditions and treatment were guided as per the National Institute of Health Guide for Animal and approved by the Institutional Animal Care and Use Committee (IACUC). 67
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
