Abstract
Breast cancer is the most commonly diagnosed cancer among women in Turkey and worldwide. It is considered a heterogeneous disease and has different subtypes. Moreover, breast cancer has different molecular characteristics, behaviors, and responses to treatment. Advances in the understanding of the molecular mechanisms implicated in breast cancer progression have led to the identification of many potential therapeutic gene targets, such as Breast Cancer 1/2, phosphatidylinositol 3-kinase catalytic subunit alpha, and tumor protein 53. The aim of this review is to summarize the roles of phosphatidylinositol 3-kinase regulatory subunit 1 (alpha) (alias p85α) and phosphatase and tensin homolog in breast cancer progression and the molecular mechanisms involved. Phosphatase and tensin homolog is a tumor suppressor gene and protein. Phosphatase and tensin homolog antagonizes the phosphatidylinositol 3-kinase/AKT signaling pathway that plays a key role in cell growth, differentiation, and survival. Loss of phosphatase and tensin homolog expression, detected in about 20%–30% of cases, is known to be one of the most common tumor changes leading to phosphatidylinositol 3-kinase pathway activation in breast cancer. Instead, the regulatory subunit p85α is a significant component of the phosphatidylinositol 3-kinase pathway, and it has been proposed that a reduction in p85α protein would lead to decreased negative regulation of phosphatidylinositol 3-kinase and hyperactivation of the phosphatidylinositol 3-kinase pathway. Phosphatidylinositol 3-kinase regulatory subunit 1 protein has also been reported to be a positive regulator of phosphatase and tensin homolog via the stabilization of this protein. A functional genetic alteration of phosphatidylinositol 3-kinase regulatory subunit 1 that results in reduced p85α protein expression and increased insulin receptor substrate 1 binding would lead to enhanced phosphatidylinositol 3-kinase signaling and hence cancer development. Phosphatidylinositol 3-kinase regulatory subunit 1 underexpression was observed in 61.8% of breast cancer samples. Therefore, expression/alternations of phosphatidylinositol 3-kinase regulatory subunit 1 and phosphatase and tensin homolog genes have crucial roles for breast cancer progression. This review will summarize the biological roles of phosphatidylinositol 3-kinase regulatory subunit 1 and phosphatase and tensin homolog in breast cancer, with an emphasis on recent findings and the potential of phosphatidylinositol 3-kinase regulatory subunit 1 and phosphatase and tensin homolog as a therapeutic target for breast cancer therapy.
Keywords
Introduction
Breast cancer (BCa) is the most diagnosed cancer in women worldwide and occurring with the highest frequency in industrial countries (the United States, Australia, and the European Union (EU)). Moreover, 131,000 of the 464,000 women who developed BCa in Europe in 2012 died as a result of this diagnosis. 1 The best-known causes of BCa are alterations in important genes, such as Breast Cancer 1/2 (BRCA1/BRCA2), tumor protein 53 (TP53), phosphoinositide 3-kinase catalytic subunit alpha (PIK3CA), Sal-like protein 4 (SALL4), phosphatase and tensin homolog (PTEN), and phosphatidylinositol 3-kinase (PI3K) regulatory subunit 1 (PI3KR1) (alpha). In this review, we aimed to share new information and recent studies about PIK3R1 and PTEN in applications (about mutation, expression, or methylation status) of BCa. PTEN is a critical tumor suppressor whose dysregulation leads to metabolic disease and cancer. Interestingly, the loss of PTEN expression, observed in about 20%–30% of cases, is known to be one of the most frequent and important tumor changes leading to PI3K pathway activation in BCa. 2 In addition, germ-line mutations of PTEN are the cause of PTEN hamartoma tumor syndromes (e.g. Cowden syndrome, Bannayan–Riley–Ruvalcaba syndrome, and Proteus-like syndrome) with an increased risk for the advancement of cancers. 3 PI3K is a heterodimeric molecule comprised of a regulatory subunit and a catalytic subunit. PI3K regulatory subunit 1 (alpha) (PIK3R1, alias p85α) and PI3K catalytic subunit alpha (PIK3CA, alias p110α) have been extensively studied, which are encoded by PIK3R1 and PIK3CA genes, respectively. PIK3R1 can take part as an oncogene, and knockouts of various isoforms of p85 subunits have shown that p85 may endogenously function as a tumor suppressor. 4 A study by Yan et al. 5 demonstrated the regulation of p85α expression in BCa and its potential role on prognosis predication. The protein p85α is crucial for stabilization and membrane recruitment of the p110α subunit of PI3K. 6 Loss of the p85α protein leads to downstream PI3K pathway activation.4,7–10 Therefore, the impact of p85α downregulation on pathway signaling could be caused by the loss of the inhibitory effect of p85α on p110α and PI3K pathway activity.9,11 p85α protein found a positive regulator of PTEN via stabilization of this protein. However, the importance of the PI3K/AKT pathway in BCa is well known, and the function of PIK3R1 in BCa has not been extensively studied. PTEN and PIK3R1 underexpressions have opposite effects on patient outcomes and could become valuable prognostic and predictive factors of BCa. If expression/alternations of PIK3R1 and PTEN genes increase or restore, this may be a novel treatment strategy for BCa. Therefore, some inhibitors have been developed for PIK3R1 (isoproterenol, 3-methyladenine, XL147, and Bi2536) and PTEN (bpV (HOpic), bpV (pic), bpV (bipy) trihydrate, VO-OHpic trihydrate, and SF1670). In this review, we focus on PIK3R1 and PTEN and possible targeted therapies in BCa.
Methodology of data mining for PIK3R1 and PTEN in cancer
We comprehensively reviewed electronic databases including PubMed. The keywords “PIK3R1,” “PTEN,” “PIK3R1 and cancer,” “PTEN and cancer,” “PIK3R1 and carcinoma,” “PTEN and carcinoma,” “PIK3R1 and breast cancer,” “PTEN and breast cancer,” “PIK3R1 and breast carcinoma,” “PTEN and breast carcinoma,” and “PIK3R1 and PTEN and breast cancer” were used for relative studies searching without any language restriction. There were about “PIK3R1” 301 studies, “PTEN” 12,524 studies, “PIK3R1 and cancer” 164 studies, “PTEN and cancer” 8924 studies, “PIK3R1 and carcinoma” 47 studies, “PTEN and carcinoma” 2564 studies, “PIK3R1 and breast cancer” 29 studies, “PTEN and breast cancer” 1388 studies, “PIK3R1 and breast carcinoma” 19 studies, “PTEN and breast carcinoma” 115 studies, and “PIK3R1 and PTEN and breast cancer” 9 studies identified from PubMed. But, about PIK3R1 and PTEN and BCa review research was detected in any of the studies.
Structure, location, and function of genes
PIK3R1 gene
Aberrations of components of the PI3K pathway are collectively the most common activating events in cancers. The PI3K pathway is commonly activated in human BCa and leads to proliferation and migration5,12 (Figure 1). The PI3K family of enzymes involves class I, II, and III, with only class I being involved in human cancer.13–16 Class IA PI3K consists of a catalytic subunit (p110α as a key subunit) and a regulatory subunit (p85α as a key subunit decoded by PIK3R1).15–17 Also, p85 stabilizes p110 and suppresses its catalytic activities. 18 PIK3R1 encodes the 85 kD regulatory subunit. The PIK3R1 gene is localized on 5q13.1. PIK3R1 encodes p85α, p55α, and p50α. Moreover, these isoforms have two carboxyl (C) terminal Src homology 2 (SH2) domains flanking a p110-binding domain (iSH2). Other isoforms differ at their N-terminus and contain a unique p55α or p50α and p50γ. In addition, unique to the larger isoforms are the N-terminal SH3 domain, GTPase-activating protein (break point cluster region-homology) (GAP (BH)) domain, and two proline-rich regions. p85α selects binding proteins and regulates phosphorylation. 19 It binds to activated (phosphorylated) protein tyrosine kinases (Tyr kinases), through its SH2 domain, and acts as an adaptor, mediating the association of the p110 catalytic unit to the plasma membrane. It is required for the insulin-stimulated increase in glucose uptake and glycogen synthesis in insulin-sensitive tissues. It plays a significant role in signaling in response to fibroblast growth factor receptors (FGFRs), KIT ligand/stem cell factor (KITLG/SCF), KIT, platelet-derived growth factor receptor alpha and beta (PDGFRA and B). In addition, it plays a role in integrin beta-2 (ITGB2) signaling.20–22 It modulates the cellular response to endoplasmic reticulum (ER) stress by promoting nuclear translocation of X-box-binding protein 1 (XBP1) isoform 2 in an ER stress- and/or insulin-dependent manner during metabolic overloading in the liver and hence plays a role in glucose tolerance improvement. 23

PI3K pathway (RTK: receptor tyrosine kinases; miR: microRNA; SH2: Src homology 2, PIP2: phosphatidylinositol 4,5-bisphosphate or PtdIns(4,5)P2; PIP3: phosphatidylinositol (3,4,5)-trisphosphate or PtdIns(3,4,5)P3; PTEN: phosphatase and tensin homolog; PIK3R1: PI3K regulatory subunit 1 (alpha); PIK3CA: PI3K catalytic subunit alpha; ERK: extracellular signal–regulated kinase).
PIK3R1 is also frequently mutated; indeed, PIK3R1 is the 11th most commonly mutated gene across cancer lineages in The Cancer Genome Atlas database 24 (Figure 2). Previously characterized PIK3R1 mutations exclusively target PI3K pathway activation, but the PIK3R1 mutation frequency is about 1.79% (Catalogue of Somatic Mutations in Cancer (COSMIC)). Many studies have identified crucial roles for PIK3R1 in human carcinogenesis. 5 PIK3R1 expression has been powerfully associated with breast, renal cell carcinoma, pancreatic carcinoma, and hepatocellular carcinoma (HCC). Other than these, mutations affecting PIK3R1 have caused agammaglobulinemia 7, autosomal recessive (AGM7) 25 and SHORT syndrome (SHORTS). 26

Structure of PIK3R1 gene and occurrence of mutations in exons.
PTEN gene
PTEN is a capable and pleiotropic tumor suppressor gene. PTEN is localized on 10q23.3 (Figure 3). The structure of PTEN contains an amino (N)-terminal phosphatase domain that can dephosphorylate phosphotyrosine, phosphoserine, and phosphothreonine within highly acidic substrates, however, with weak catalytic activity (Figure 3). Moreover, the PTEN structure shows a phosphatase domain that is similar to protein phosphatases. However, it has an enlarged active site significant for the accommodation of the phosphoinositide substrate. 27 PTEN also has a C2 domain and ties phospholipid membranes in vitro. Moreover, the phosphatase and C2 domains correlate across a wide interface, suggesting that the C2 domain may productively position the catalytic domain on the membrane. 28 Chalhoub and Baker 29 suggested that a multitude of posttranslational modifications can modulate PTEN activity. However, in general, PTEN is a relatively stable protein, and the physiological stimuli that induce its turnover, phosphorylation, or dephosphorylation are unknown. We know that reductions in the level or activity of the PTEN protein can have deep consequences for tumor incidence, penetrance, and aggressiveness. Moreover, PTEN is a dual protein and lipid phosphatase that is frequently mutated in many human malignancies. 30 A loss of PTEN reduces the dephosphorylation of phosphoinositide 3,4,5-trisphosphate (PIP3), which leads PI3K to phosphorylate phosphatidylinositol 4,5 bisphosphate (PIP2) and improves the levels of PIP3. PIP3 stimulation increases cell proliferation and cell migration, cell survival, and cell size by inducing downstream proteins as AKT31,32 (Figure 1). Furthermore, PTEN loss may reduce p53 levels via AKT-mediated phosphorylation of murine double minute 2 (MDM2), which rises MDM2 nuclear translocation and subsequent ubiquitination and degradation of p53.33,34 Finally, PTEN may also influence MDM2 promoter activity. It also plays a crucial role in the response of human cancer cells to oncoprotein-targeting agents. PTEN is also a negative regulator of PI3K/AKT signaling.

Structure of PTEN gene and occurrence of mutations in exons (PBD: a phosphatidylinositol-4,5-bisphosphate (PtdIns(4,5)P2)-binding domain).
PTEN loss has been revealed in 13%–86% of human epidermal growth factor receptor 2 (HER2)-positive BCa.35,36 The mutation and inactivation of PTEN lead to uncontrolled cell proliferation and survival and subsequently increases tumor formation. Many studies have shown that PTEN has a significant role in DNA damage responses. In addition, BRCA1 functionally assists with PTEN and might be a needed stoppage in the progress of several tumors. As a tumor suppressor molecule, PTEN appears to improve the prognosis of several cancers. In short, the absence of PTEN worsens prognosis in the early stages of cancer.37,38 Likewise, germ-line mutations of PTEN are the reason for PTEN hamartoma tumor syndromes (e.g. Cowden syndrome, Bannayan–Riley–Ruvalcaba syndrome, and Proteus-like syndrome), increasing the risk for the progress of cancers. 3 Loss-of-function mutations of PTEN as well as hypermethylation and silencing of the PTEN promoter are found in a variety of human cancers. Mutation of PTEN frequency was detected as 4% (COSMIC). PTEN belongs to a class of “gatekeeper” tumor suppressor genes together with p53, Rb, and APC. 39 Moreover, high frequency of inactivation in somatic cancer, PTEN is ranked the second most mutated tumor suppressor gene after p53. 40
As a summary, it is implied that the products of the PTEN and PIK3R1 genes function as negative regulators of PI3K activity, either by controlling the levels of PIP3, which mediates AKT phosphorylation, or by directly increasing PI3K activity in a study. 41 Thus, these genes may have crucial roles in cancer progression.
PIK3R1 and PTEN status in human cancers
Recently, a lot of studies have showed that PIK3R1 and PTEN expression is decreased or lost in human cancers, consistent with its role as a tumor suppressor gene. In this part, we will summarize the current studies on the expression, mutation, and methylation of PIK3R1 and PTEN in different human malignancies because PIK3R1 and PTEN are deregulated in the majority of human cancers.
In particular, numerous genetic and epigenetic changes have been detected in these genes in tumor samples. Therefore, these genes have shown oncogenic or tumor suppressor effects in cancer. MicroRNA (miR)-885-5p and PIK3R1 are the best indicators for the classification of laryngeal cancer tissue and normal mucosa. 42 Cai et al. 43 showed that exogenous miR-542-3p repressed glioblastoma cell invasion through targeting AKT1, integrin-linked kinase, and PIK3R1. The findings of one study implied that PIK3R1: rs3756668 (A > G) was associated with longer overall survival (OS) in limited disease-small-cell lung cancer (LD-SCLC) patients after chemoradiotherapy. 44 Cheung and Mills 45 found that targeting therapeutic liabilities are stimulated by PIK3R1 mutations for cancer treatment. Chang et al. 46 reported that the 21 genes comprised of eight tumor suppressor candidates (ATM, MSH2, PIK3R1, PTCH1, PTEN, TET2, TP53, and TSC1) and 13 oncogene candidates (ALK, BCL9, CTNNB1, ERBB2, FGFR2, FLT3, HNF1A, KIT, MTOR, PDGFRA, PPP2R1A, PTPN11, and SF3B1). They detected a high frequency of mutations in PTEN (50%) in the endometrial cancer-related molecular pathway (Table 1). Cybula et al. 42 reported that (Table 2) PIK3R1 was downregulated in laryngeal cancer. Wu et al. 47 showed that the tendency of changes in the expression of 12 selected differentially expressed genes (DEGs) (five downregulated genes, PIK3R1, RARB, HGF, MAPK11, and SESN1, and seven upregulated genes, PAK1, E2F1, CCNE1, EGF, CDC25A, PTTG1, and UHRF1) in quantitative real-time polymerase chain reaction (qRT-PCR) was consistent with the expression profiling data in lung adenocarcinoma tissues and corresponding nontumor tissues. Liu et al. 48 reported that DEGs such as AKT1, TNF SYK, and PIK3R1 were the frequent genes included in the important pathways. Moreover, these genes were identified to play a vital role in sorafenib-treated HCC. According to the results of a study, mutant forms of p85α (PIK3R1) may play an oncogenic role in bladder cancer via a loss in the ability to regulate p110α and changed function of the BH domain. 49
Alterations of PIK3R1 and PTEN genes in different cancers.
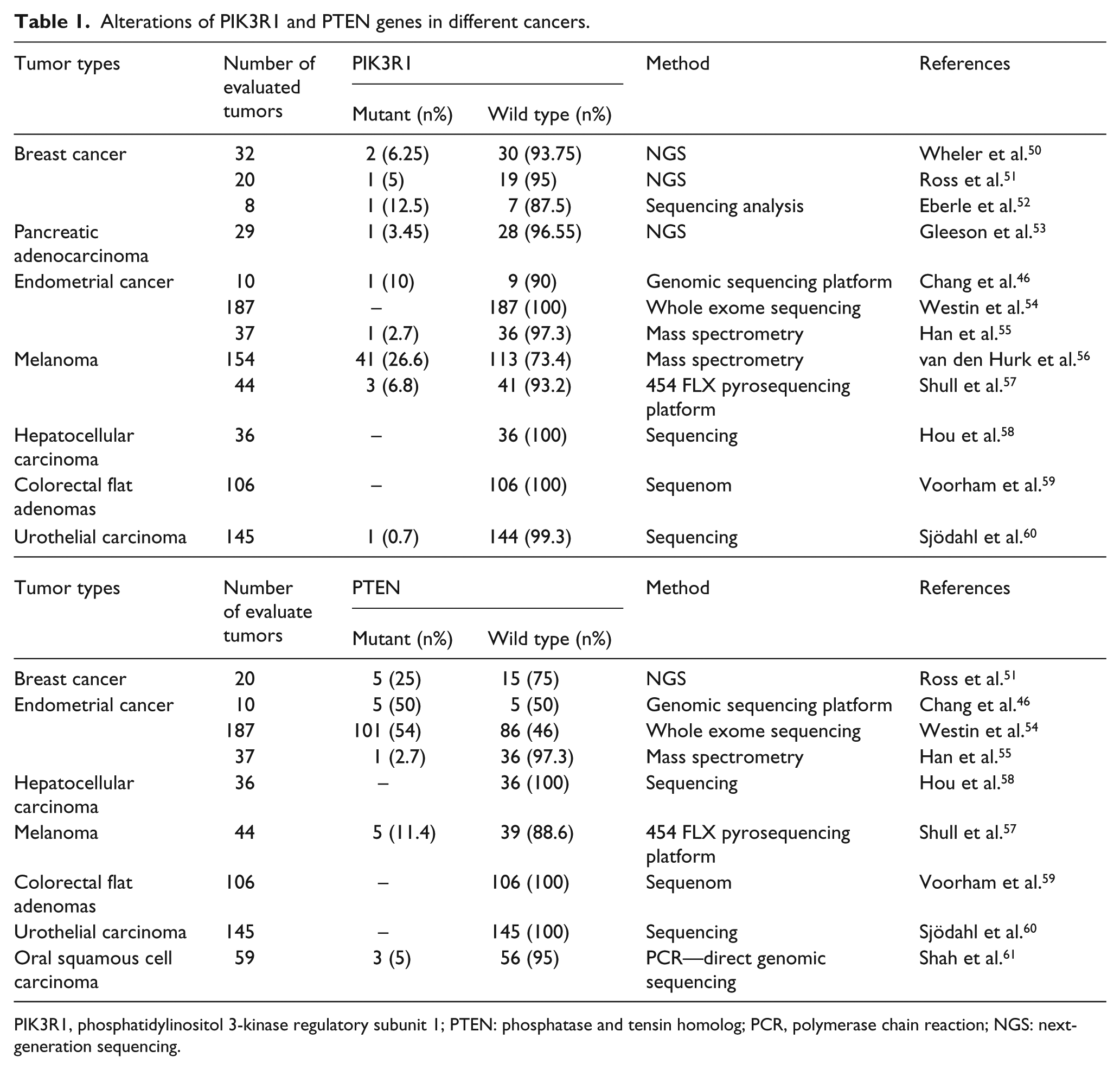
PIK3R1, phosphatidylinositol 3-kinase regulatory subunit 1; PTEN: phosphatase and tensin homolog; PCR, polymerase chain reaction; NGS: next-generation sequencing.
The expression status of PIK3R1 and PTEN in different cancers.

PIK3R1, phosphatidylinositol 3-kinase regulatory subunit 1; PTEN: phosphatase and tensin homolog; RT-PCR: real-time polymerase chain reaction; qRT-PCR: quantitative real-time polymerase chain reaction; NSCLC, non-small-cell lung cancer; SCC, squamous cell carcinoma; IHC: immunohistochemistry.
Otherwise, Xia et al. 79 showed that tramadol repressed proliferation, migration, and invasion of human lung adenocarcinoma cells through the elevation of PTEN and inactivation of PI3K/AKT signaling. Reduced PTEN expression may be a marker for poor prognosis in patients with lung cancer. 80 Qiao and Hong 81 reported that FER1L4 revealed downregulation in endometrial carcinoma (EC) tissues and cells. Moreover, it regulated PTEN expression and AKT signaling, which might explain its inhibition of cell proliferation. Shah et al. 61 detected 5% somatic mutational frequency in the PTEN gene. However, mutations of PTEN were found to be absent in the coding region. PTEN was verified as a direct downstream target of miR-92a in nasopharyngeal carcinoma (NPC) cells, and changes in miR-92a expression regulated PTEN/AKT pathway in NPC cells. 82 Xin et al. 83 reported that miR-214 may advance peritoneal metastasis of gastric cancer (GC) cells via downregulation of PTEN, thus leading to the progression of GC. Zhu et al. 84 showed that suberoylanilide hydroxamic acid (SAHA; vorinostat) stimulated the loss of the tumor suppressor PTEN gene, causing thyroid carcinogenesis.
Preclinical and clinical studies have been done on PIK3R1 and PTEN to find an effective and practical personalized therapy for other cancers. Yu et al. 85 reported that inhibition by ARQ 092 and ARQ 751 was more dominant in cancer cell lines containing PIK3CA/PIK3R1 mutations compared to those with wild-type (wt)-PIK3CA/PIK3R1 or PTEN mutations. Moreover, they implied that for both ARQ 092 and ARQ 751, PIK3CA/PIK3R1 and AKT1-E17K mutations can potentially be used as predictive biomarkers in clinical studies. Oliveira et al. 86 showed that synthetic miR-29a mimics into Jurkat cells revealed the downregulation of several predicted targets of some important genes (CDK6, PXDN, MCL1, PIK3R1, and CXXC6), including targets with roles in active and passive DNA demethylation. Maliqueo et al. 87 showed that estradiol stimulated estrous cycle changes but reduced circulating luteinizing hormone (LH) and the ovarian expression of Adipor1, FOXO3, and PIK3R1. Toste et al. 8 demonstrated that p85α expression is a factor of chemosensitivity in human pancreatic ductal adenocarcinoma (PDAC). In addition, they contributed novel evidence that miR-21 can affect PI3K-AKT signaling via its direct regulation of p85α. Wang et al. 88 reported that miR-29b repressed downstream effectors of PIK3R1 and AKT3. In addition, the knockdown of PIK3R1 or AKT3 repressed α-SMA and collagen I and provoked apoptosis in both hematopoietic stem cells (HSCs) and mice.
Role of PIK3R1 and PTEN genes in Bca
PI3K is a heterodimer and contain of a p110α catalytic subunit encoded by the PIK3CA gene.9,89 Furthermore, a p85 regulatory subunit alpha is encoded by the PIK3R1 gene. PIK3CA and PIK3R1 have crucial roles in BCa. Especially, PIK3R1 underexpression might possibly lead to PI3K pathway activation and confer tumor development and progression (Taniguchi et al. 7 ). PIK3R1 gene has a tumor suppressor role because PI3K subunit p85α regulates and stabilizes p110α.7,9 On the other hand, PTEN is a dual lipid/protein phosphatase that adjusts cell cycle progression, survival, cell growth, angiogenesis, and genomic stability through PI3K/AKT signaling pathway.30,90 Loss of PTEN expression, observed in about 20%–30% of cases, is known to be one of the most common tumor alteration leading to PI3K pathway activation in BCa. 2 PTEN also deleted on chromosome 10, as a tumor suppressor gene, is crucial for the development of both familial and sporadic BCas. PTEN holds significant processes such as translation, cell cycle progression, and apoptosis by arresting the activation of AKT/protein kinase B (PKB). 4
PIK3R1 and PTEN alterations and clinical outcomes in BCa subtypes
Description and refinement of molecular tumor subtypes, improvement of predictive biomarkers along, and rational treatment strategies are significant on the therapeutic potential of PI3K pathway–directed therapies in BCa. PI3K pathway activity is elevated in over half of the luminal subtypes, in one of two HER2/Neu subtypes and three of three basal subtypes. Particularly striking is the very high level of PI3K pathway activation observed in luminal B subtype 6, suggesting that mutations in several genes may cooperate to hyperactivate the pathway in these tumors. 91 Therefore, last decade Therefore, last decade in the PI3K pathway have evaluated the correlation for the expression or alterations of PIK3R1 and PTEN in the molecular subtypes of breast carcinoma.
Cizkova et al. 64 showed that by combining PIK3CA mutation and PIK3R1 expression status, four prognostic groups were described with considerably different metastasis-free survival (p = 0.00046). In their study, Cox multivariate regression analysis indicated that the prognostic importance of PIK3R1 underexpression was proven in the total population (p = 0.0013) and in BCa subgroups. They showed greater PIK3R1 underexpression in the HR (−) ERBB2 (−) tumor type (88.4%) than HR (+) ERBB2 (+) (59.3%).
PTEN loss was inversely correlated with estrogen receptor (ER) and androgen receptor (AR) expression, although PTEN loss was still found in both ER (+) and ER (−) tumors as well as AR (+) and AR (−) cancers. 92 Azeez et al. 93 showed that PTEN is a cooperating partner for TOB-1. Furthermore, PTEN may regulate the downstream expression of cell cycle control protein p27 via multiple downstream signaling pathways of progesterone through a progesterone receptor (PR). Shore et al. 94 determined that luminal-specific PTEN loss outcomes had distinct effects on epithelial homeostasis and mammary tumor formation. In addition, luminal PTEN loss increased the proliferation of HER2 (−) cells, thereby reducing the percentage of HER2 (+) cells. André et al. 95 revealed that patients with HER2-positive improved BCa having tumors with PIK3CA mutations, PTEN loss, or hyperactive PI3K pathway could acquire progression-free survival (PFS) benefit from everolimus.
According to one study, PIK3CA and PTEN mutations were more frequently discerned in patients with greater expression of ER, PR, and AR. 92 Liu et al. 96 suggested that combined PTEN-p53 mutations enhanced the formation of claudin-low, triple-negative-BCa (TN-BCa) that displayed hyperactivated AKT signaling and more mesenchymal features compared to PTEN or p53 single-mutant tumors.
Li et al. 97 reported that a low expression of PTEN and a high expression of epidermal growth factor receptor (EGFR) and p53 are detected in TN-BCa. Also, it may synergistically contribute to the pathogenesis of TN-BCa. Dębska-Szmich et al. 98 proved that HER3 overexpression, PTEN, and p-HER2 positivity in tumor cells of HER2 (+) patients are associated with more a progressive clinical stage of BCa. Maggi and Weber 99 showed PTEN expression levels at the nexus of luminal B-BCa and revealed that patients with PTEN-low ER (+) tumors might help from combined endocrine and PI3K pathway therapies.
Clinical trials in BCa progression
Preclinical data indicate that PTEN is a regulator of PI3K/AKT signaling, tumor growth, and invasion and PIK3R1 represses growth, invasiveness, and metastatic properties. PI3K pathway aberrations have a distinct role in the pathogenesis of different BCa subtypes. The specific aberration present may have implications for the selection of PI3K-targeted therapies in HER2 (+) BCa. 2 Chagpar et al. 100 demonstrated that p85α binds directly to PTEN via the p85α-SH3-BH domains. Cells expressing a synthetic mutant of p85α that stopped the p85α–PTEN interaction exhibited increased AKT activation following stimulation by growth factors. Chagpar et al. 100 thus pointed that p85α can bind to PTEN and improve PTEN activity.
PIK3R1 has been found to be considerably downregulated in MDA-MB-231 cells. 12 Moreover, the Michigan Cancer Foundation-7 (MCF-7)–invasive clone is related to MCF-7 cells, thereby possibly contributing to metastasis progress. The findings of one study showed that p85α (PIK3R1) downregulation was an independent prognostic marker in BCa. 64 PI3K is important for BCa and is well known, but the function of PIK3R1 in BCa is not clear. PIK3R1 also has tumor suppressor properties through the negative regulation of growth factor signaling. 7 Richardson et al. 101 and Uchino et al. 12 showed that PIK3R1 expression was drastically reduced by 18% in BCa tissues (Table 2). Reduced PIK3R1 expression has also been connected with reduced survival in BCa patients. 64 The influence of PIK3R1 downregulation on pathway signaling could be caused by the loss of the inhibitory effect of PIK3R1 on p110α and the PI3K pathway activity.9,11 PIK3R1 protein is a positive regulator of PTEN via stabilization of this protein.102,103 PIK3R1 downregulation may be a prognostic marker for inferior clinical stage. According to many studies, there is a relation between PIK3R1 downregulation and an inferior prognosis not only in BCa 64 but also in pancreatic cancers,8,104 hepatocellular cancers, 7 neuroblastoma, 67 and lung cancers. 105 Another finding showed that PIK3CA mutations were related to better metastasis-free survival. Furthermore, PIK3R1 underexpression has been associated with poorer metastasis-free survival.
The PI3K pathway can be constitutively activated by genomic aberrations in cancer. 106 Common alterations include 107 activating mutations or/and amplification of the catalytic subunit alpha (PIK3CA),20,108 the loss of PTEN, 31 and the mutation and/or amplification of AKT, 109 a serine/threonine-specific protein kinase. These alterations are sufficient to induce tumorigenesis in preclinical models.107,110 Reduced PTEN function collaborates with MYC and HER2 activation in conferring an aggressive phenotype to cancer cells. Olasz et al. 111 reported that loss of PTEN enhances the autocrine fibroblast growth factor (FGF) signaling, promoting cell proliferation. FGF-2 and FGFR1 can be potential targets in PTEN-deficient BCa. Lebok et al. 112 showed that PTEN deletion may occur in a relevant fraction of BCa. In addition, PTEN deletion is connected to aggressive tumor behavior.
According to one study, 76 there was an inverse significant relationship between the likelihood of death and PTEN gene expression (p < 0.01). These findings indicate that the lack of PTEN gene expression can indicate a poor prognosis for BCa. Yang et al. 113 showed that PTEN negativity was significantly related to an adverse prognosis in terms of OS in BCa. De Amicis et al. 114 implied that PTEN is a key target of Bergapten action in BCa cells for the stimulation of autophagy. Beg et al. 115 showed that a loss of PTEN protein expression occurs at a high frequency in Middle Eastern patients with BCa. PTEN inactivation may lead to the aggressive behavior of tumor cells by inducing tumor cell proliferation. Okutur et al. 116 reported that a trend toward statistical significance for longer OS was found for PTEN (+) patients (p = 0.058).
Siddiqui et al. 117 reported promoter methylation as a mechanism partially responsible for PTEN silencing in sporadic BCa. Moreover, methylation and the expression loss of PTEN demonstrated promising potential as candidate biomarkers of risk evaluation in subcategorized breast tumors with critical pathologic parameters. Kuang and Nie 118 reported that miR-21 can promote the proliferation and invasion of BCa cell lines, and its downstream target genes include PDCD-4, FasL, PTEN, RhoB, Maspin, TIMP3, and RECK.
Current clinical strategies and targeting PIK3R1 and PTEN-defined BCa
The identification and validation of molecular pathways underlying BCa may lead to the development of effective therapeutics. PI3K pathway may be important to specific marker for cancer progression and a potential drug target, which can be used for treatment of BCa. We think PIK3R1 and PTEN genes of PI3K as a potential target for the development of diagnostic and prognostic biomarkers for BCa in the future.
The PIK3R1 expression state (low/high) and loss of PTEN protein expression are common in BCa and strongly correlated with clinical pathological data (as ER (+/−), lymph node, and metastasis). Ou et al. 119 showed that RNA interference (RNAi) screens regulated in combination with rapamycin in multiple BCa cell lines identified six genes—AURKB, PLK1, PIK3R1, MAPK12, PRKD2, and PTK6—that when silenced, each enhanced the sensitivity of multiple BCa lines to rapamycin. Yan et al. 5 reported that miR-21 knockdown represses cell growth, migration, and invasion partly by inhibiting PI3K/AKT activation via directly targeting PIK3R1 and reversing epithelial–mesenchymal transition (EMT) in BCa. Moreover, p85α downregulation described a specific subgroup of BCa with a shorter 5-year disease-free survival (DFS) and OS, which may need more aggressive treatment. Díaz et al. 120 reported evidence implicating p85α (also known as PIK3R1), a Rab5 GTPase-GAP, in CAV1-dependent effects, by showing that CAV1 recruits p85α, precluding p85α-mediated Rab5 inactivation and increasing cell migration. Hsieh et al. 121 showed that when lunasin/aspirin therapy use, its potent pro-apoptotic effect is at least partially achieved through modulating the extrinsic apoptosis-dependent pathway. Moreover, synergistic downregulatory effects were detected for ERBB2, AKT1, PIK3R1, FOS, and JUN signaling genes. Their amplification is responsible for BCa cell growth and resistance to apoptosis.
On the other hand, in the literature, clinical strategies and trials have been reported on PTEN in BCa. Liu et al.
122
showed that the expression levels of LC3 and phosphorylated AKT (p-AKT) were reduced in the combined groups more than in the gefitinib alone group, while the p-PTEN, caspase-3, and caspase-9 levels were increased. Furthermore, autophagy inhibitor may improve the sensitivity to gefitinib in MDA-MB-468 and MDA-MB-231 cells by activating the PTEN/P13K/AKT pathway. Sun et al.
123
suggested that MEOX1 is a clinically relevant novel target in breast cancer stem cells (BCSCs) and mesenchymal-like cancer cells in PTEN-deficient trastuzumab-resistant BCa. Jahanafrooz et al.
124
showed that PTEN messenger RNA (mRNA) in T47D and P21 and P27 mRNAs in MCF-7 were not affected by silibinin. Gogas et al.
125
showed that MYC gene copies, centromere status, and PI3K activation (PIK3CA mutations; PTEN and phospho-mechanistic target of rapamycin (mTOR) protein expression) may adversely impact trastuzumab-treated metastatic breast cancer (mBC) patients’ outcomes and seem worthy validating in larger series. Islamian et al.
126
reported that doxorubicin (DOX) and 2-deoxy-
Epigenetic mechanisms are important for BCa progression and treatment applications. Bahena-Ocampo et al. 133 reported that miR-10b expression in BCa stem cells helped self-renewal through negative PTEN regulation and maintained AKT activation. Kuang and Nie 118 reported that miR-21 can stimulate the proliferation and invasion of BCa cell lines. Moreover, downstream target genes involve PDCD-4, FasL, PTEN, RhoB, Maspin, TIMP3, and RECK. Li et al. 134 suggested that miR-221/222 enhances BCa growth, migration, and invasion while enhancing the self-renewal of BCSCs. This is likely achieved through targeting the PTEN/AKT pathway. Li et al. 135 showed that GAS5 represses cancer proliferation by acting as a molecular sponge for miR-21, leading to the derepression of PTEN, the endogenous target of miR-21. Ma et al. 136 showed that miR-487a directly targeted the MAGI2 included in the stability of PTEN. According to their findings, the downregulation of miR-487a increased the expression of p-PTEN and PTEN and diminished the expression of p-AKT. Wang et al. 137 showed that the introduction of PTEN complementary DNA (cDNA) lacking the 3′ untranslated region (3′UTR) significantly repressed the miR-214-induced activation of the PI3K/AKT signaling pathway and reversed the protective effects of miR-214 on cell survival and resistance to apoptosis. Yu et al. 138 showed that miR-21 organized the function of autophagy and apoptosis by targeting PTEN. They revealed that the silencing of miR-21 enhanced the sensitivity of ER (+) BCa cells to tamoxifen (TAM) or fulvestrant (FUL) by increasing autophagic cell death. De la Parra et al. 139 demonstrated that with reduced cell viability, miR-155 is downregulated, whereas pro-apoptotic and anticell proliferative miR-155 targets, FOXO3, PTEN, casein kinase, and p27, are upregulated in MDA-MB-435 and Hs578t cells in response to genistein treatment. Zhang and Yan 140 showed that cantharidin (CTD) stimulated a rise in the protein expression levels of miR-106b-93 target genes, p21 and PTEN. Wu et al. 141 reported that PTEN, a direct target gene of miR-21, was downregulated in gemcitabine-resistant BCa cells and restoration of PTEN expression blocked miR-21-stimulated EMT and gemcitabine resistance.
Conclusion and future directions and perspectives
Many BCa researchers aim to investigate new biomarkers as potential diagnostic tools that could help to clinically control this type of cancer. We know that these biomarkers may be genetic mutations (e.g. PIK3CA, TP53, MED12, and CDH1), polymorphisms, RNA levels (as SALL4, IGFBPs, PTEN, and PIK3R1), protein levels, and telomere length or telomerase activity indicative of the degree of the cancer’s advance and prognosis, or a patient’s tendency to develop cancer. Interestingly, these genes may be known or potential therapeutic targets themselves or may affect a drug’s efficacy. These effects may improve drug resistance or sensitivity.
BCa is the most frequent cancer and the second leading cause of cancer-related deaths among women worldwide. While patients are frequently diagnosed in the early and remediable stages, the treatment of metastatic BCa remains a major clinical challenge. Today, there are combinations of chemotherapy with new targeting agents and molecular gene therapy that are helpful in advancing patient survival. We know that novel treatment strategies, such as molecular gene therapy, are required to improve clinical applications in large populations. In this review, we discussed on the biology of PIK3R1 and PTEN in an attempt to determine their roles in BCa and therapeutic strategies to target these genes, as PIK3R1 and PTEN play important roles in breast carcinogenesis. Today, there are several inhibitors for PIK3R1 (W1628, W3144, and D2926)142,143 and PTEN (bpV (phen), bpV (pic), VO-OHpic, and SF1670). 144 Schmid et al. 145 reported the substantially greater potency of bpV (phen), bpV (pic), and bpV (HOpic) against PTEN than against the classical protein tyrosine phosphatases (PTPs), protein tyrosine phosphatase 1B (PTP1B), and protein tyrosine phosphatase B (PTPb). Moreover, they detected a bpV-mediated enhancement of insulin-stimulated AKT activation that was observed in cells expressing PTEN (NIH3T3 fibroblasts), but not in a PTEN null cell line (UM-UC-3 bladder cancer). 145 In particular, many researchers have implied that this stimulated significant interest in these compounds as PTEN inhibitors.146,147 SF1126 is a small molecule conjugate containing a pan-PI3K inhibitor. It selectively inhibits all PI3K class IA isoforms and other key members of the PI3K superfamily (as DNA-dependent protein kinase (DNA-PK) and mTOR). A major factor in tumor resistance to approved chemotherapy agents is thought to be the activation of the PI3K/PTEN pathway. 148 Also some other inhibitors have been development for PIK3R1 as isoproterenol, XL147, and SF1126 (as shown in Table 3). When we saw the mechanism of these inhibitors, for example, isoproterenol binds to activated (phosphorylated) protein-Tyr kinases, through its SH2 domain, and acts as an adaptor, mediating the association of the p110 catalytic unit to the plasma membrane. This is essential for the insulin-stimulated increase in glucose uptake and glycogen synthesis in insulin-sensitive tissues. 148 XL147 is a small molecule that selectively inhibits the activity of phosphoinositide-3 kinase (PI3K). Activation of PI3K is a frequent event in human tumors, promoting tumor cell growth, survival, and resistance to chemotherapy and radiotherapy. When PI3K is inactivated, it inhibits growth and induces apoptosis (programmed cell death) in tumor cells. 148 On the other hand, PTEN was related to clinical response gefitinib or erlotinib. PTEN deficiency improved the sensitivity of the cells to proliferation arrest by erlotinib. 149 In the future, these genes can be targeted to develop new inhibitor(s) and new drug(s) or new therapeutic strategy. In the near future, as part of an ongoing project, we will detect PIK3CA and TP53 gene mutations with high-resolution melting (HRM) analyses and will introduce an association between SALL4, PTEN, and PIK3R1 expression levels in BCa. 150 Despite studies showing PTEN and PIK3R1 deregulation in different cancers, the roles of PTEN and PIK3R1 in BCa have not been fully explained. This review article summarizes PIK3R1 and PTEN as potential biomarkers and used for BCa diagnosis, prognosis, and treatment.
Inhibitors for PIK3R1 and PTEN genes.

PIK3R1, phosphatidylinositol 3-kinase regulatory subunit 1; PTEN: phosphatase and tensin homolog; PIK3CA: phosphoinositide 3-kinase catalytic subunit alpha; 3-MA: 3-methyladenine; PI3K: phosphatidylinositol 3-kinase.
Based on trials that were listed in www.selleckchem.com and www.tocris.com (2 January 2017).
Footnotes
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
This work was supported by grants (SAG-C-DRP-100216-0040 to M.A.) from the Research Foundation of Marmara University (BAPKO).
