Abstract
MicroRNAs are small, endogenous, and non-coding RNAs that play important regulatory roles in multiple biological processes in cancers. Recent evidence has indicated that miR-19a participates in the cancer tumorigenic progression. However, the functional roles of miR-19a in cancer stem cells are still unclear. As the cancer stem cells are considered to be responsible for the tumor recurrence and treatment failure in osteosarcoma, the aim of this study is to investigate the molecular mechanism of miR-19a underlying osteosarcoma tumorigenesis. In this study, we observed significant upregulation of miR-19a in osteosarcoma patients’ tumor tissues as well as the osteosarcoma cell lines in vitro. We showed that knockdown of miR-19a by its antisense oligonucleotide (anti-miR-19a) significantly decreased the population of cancer stem cells in osteosarcoma cell lines. Furthermore, we found the miR-19a regulated the cell proliferation, migration, and viability in the human osteosarcoma–cancer stem cells. The gene of phosphatase and tensin homolog deleted on chromosome 10, which is an important tumor suppressor, was found to be directly regulated by miR-19a in human osteosarcoma–cancer stem cells. We demonstrated that knockdown of miR-19a increased the expression of phosphatase and tensin homolog deleted on chromosome 10. As the anti-miR-19a inhibited the phosphatidylinositol 3-kinase/AKT pathway and induced apoptosis of human osteosarcoma–cancer stem cells, the phosphatase and tensin homolog deleted on chromosome 10 small interfering RNA inhibited the effect of it. Meanwhile, the phosphatase and tensin homolog deleted on chromosome 10 small interfering RNA also abolished the effect of anti-miR-19a on inhibiting the cell proliferation, migration, and viability in the human osteosarcoma–cancer stem cells. In conclusion, our findings demonstrated that dysregulation of miR-19a plays critical roles in the osteosarcoma stem cells, at least in part via targeting the phosphatase and tensin homolog deleted on chromosome 10. Knockdown of miR-19a may represent a potential strategy for the osteosarcoma treatment.
Keywords
Introduction
Osteosarcoma (OS) represents one of the most commonly diagnosed malignant bone cancer affecting children and adolescents. 1 Due to the efforts of extensive researches, molecular mechanisms underlying OS have been clarified gradually. However, the targets for OS therapy remain largely insufficient. 2 Recently, studies indicated that the cancer stem cells (CSCs), which are a small subpopulation of tumor cells owning the multilineage differentiation potential and the ability of self-renewal, are believed to have a tumor-initiating capacity and play crucial roles in cancer relapse and metastasis.3–5 Therefore, the CSC-targeted therapy has been considered as new strategies to improve the efficiency of cancer treatment. 6
CD133 is a pentaspan transmembrane glycoprotein encoded by the prominin 1 (PROM1) gene in humans. 7 Previous studies have identified the CD133-positive phenotype as the molecular marker in many tumors such as breast cancer, lung cancer, hepatocellular carcinoma (HCC), and OS.8–11 The CD133-positive OS cells are reported to display the stem-like probabilities of tumorigenicity, proliferation, and self-renewal. 12 Therefore, CD133 may serve as a specific marker of OS stem cells that may be the potential target for therapy.
MicroRNAs (miRNAs), a family of small, non-coding RNA molecules that regulate gene expression by binding to the target messenger RNAs (mRNAs) at the 3′-untranslated region (3′-UTR), play important roles in cancer initiation and progression.13,14 Increasing evidence showed that miRNAs are usually dysregulated in various cancers, leading to the therapeutic resistance and the promotion of cell proliferation, metastasis, and survival.15,16 However, the dysregulation of miRNAs in CSCs is still largely unclear. In this study, we showed that the miR-19a was dysregulated in the OS stem cells. Our data provide new insights into the role of miR-19a in regulating the cell growth and apoptosis of OS stem cells by targeting phosphatase and tensin homolog deleted on chromosome 10 (PTEN).
Materials and methods
Cell culture
OS cells lines of HOS, MG-63, and Saos-2 were purchased from American Type Culture Collection (USA). Cells were cultured in Dulbecco’s Modified Eagle’s Medium (DMEM) basic medium (Gibco, USA) supplemented with 10% heat-inactivated fetal bovine serum (FBS; Gibco) at 37°C in a humidified 5% CO2 incubator.
Serum samples
A total of 25 pairs of primary OS tissue samples and paracancerous tissues were collected from The First Affiliated Hospital of Shantou University Medical College. For the use of these clinical materials for research purposes, prior consent from patients and approval from the Ethics Committee of The First Affiliated Hospital of Shantou University Medical College were obtained.
Quantitative real-time polymerase chain reaction
Expression levels of miR-19a and PTEN in tissue samples and cell lines were measured using the quantitative real-time polymerase chain reaction (qRT-PCR). Briefly, total RNAs in tissues and cell lines were extracted using the TRIzol reagent (Invitrogen, USA) according to the manufacturer’s instructions. For miR-19a detection, stem-loop miRNA qRT-PCR Primer Sets (one RT primer and a pair of quantitative PCR (qPCR) primers for each set) specific for miR-19a were designed by RiboBio Co., Ltd. (China). The reverse transcription reaction for miR-19a and PTEN was performed using the PrimeScript One Step miRNA cDNA Synthesis Kit (TaKaRa, China), and the qPCR reaction was performed using the SYBR® Premix Ex Taq™ II (TaKaRa) according to the manufacturer’s instruction. Expression levels of miR-19a and PTEN were determined using the 2−ΔΔCT analysis method 17 and normalized to the endogenous U6 small nuclear RNA (snRNA) and glyceraldehyde-3-phosphate dehydrogenase (GAPDH), respectively.
Transfection of RNA oligonucleotides
Negative control oligonucleotides (NCO), miR-19a mimics (miR-19a), antisense-miR-19a (anti-miR-19a), and PTEN small interfering RNA (siRNA) were purchased from RiboBio Co. Ltd. For transfection, cells were plated at 30%–50% confluence. After 24 h, 50 pmol/mL RNA oligonucleotides were transfected using the Lipofectamine™ 2000 reagent (Invitrogen, USA) according to the manufacturer’s instructions.
Identification of the CSCs population
CD133 was considered as the maker of OS stem cells. Briefly, cells were collected and washed twice before staining with anti-CD133-FITC antibody (Miltenyi Biotec, Germany) for 20 min at room temperature. The population of CD133-positive cells were analyzed and sorted as the OS stem cells using the fluorescent-activated cell sorting equipment (Beckman Coulter, USA).
Cell viability and proliferation assays
After the HOS cells were plated and transfected with RNA oligonucleotides, the cell viability was then measured by 3-(4,5-dimethylthiazol-2-yl)-2,5-diphenyltetrazolium bromide (MTT) assay as previously described. 18 To determine the cell proliferation, cancer cells were seeded onto six-well plate. During the last 6 h of treatment, the 3H thymidine was added into the culture medium, and then, the 3H thymidine incorporation assay was performed to determine the cell proliferation.
Cell migration assay in vitro
HOS cells were transfected with RNA oligonucleotides using Lipofectamine 2000 in 24-well plates; 48 h after transfection, cell migration assay was performed using Transwell chambers (Millipore, Massachusetts, USA). Briefly, cells (5 × 104) were seeded in the upper compartment filled with DMEM without FBS, but the lower compartment was filled with DMEM supplemented with 10% FBS. After 4 hours of cell migration, the migratory cells on the bottom surface of the filters were fixed, stained with crystal violet, and then counted under a phase-contrast microscope.
Luciferase reporter assay
A fragment of PTEN 3′-UTR was cloned into pGL3 Luciferase Reporter Vectors (Promega, USA; wild type). Site-directed mutagenesis of the miR-19a binding site in PTEN 3′-UTR was performed using the site-directed mutagenesis kit (TaKaRa; mutation). For luciferase reporter assay, 50 pmol/mL miR-19a mimics (or inhibitors), 2 µg/mL firefly luciferase reporters, and 100 ng/mL Renilla luciferase pRL-TK vector (Promega) were co-transfected into the HOS-CSCs. After 48 h, luciferase activities were measured using the Dual-Luciferase Reporter Assay System (Promega) according to the manufacturer’s instructions. Firefly luciferase was normalized to the Renilla luciferase.
Mitochondria isolation
To evaluate the protein level of cytochrome c and second mitochondria-derived activator of caspase/direct inhibitor of apoptosis protein (IAP) binding protein with low pI (Smac/DIABLO) in cytoplasm and mitochondria of HOS-CSCs, the mitochondria in cells were isolated using Mitochondria/Cytosol Fraction Kit (BioVision, USA) according to the manufacturer’s guidance. Subsequently, western blot analysis was performed to detect the released cytochrome c and Smac/DIABLO.
Western blot analysis
Total proteins from the OS cells were extracted using radioimmunoprecipitation assay (RIPA) buffer (Cell Signaling Technology, USA). A volume of 50 µg of the extracted proteins was separated by 10% sodium dodecyl sulfate polyacrylamide gel electrophoresis (SDS-PAGE), and the proteins were transferred to polyvinylidene difluoride (PVDF) membranes (Millipore, USA) by electroblotting. The membranes were then blocked with 5% non-fat milk and incubated overnight at 4°C with various antibodies followed by incubation of horseradish peroxidase–conjugated antibody (Santa Cruz Biotechnology). Signals were detected using enhanced chemiluminescence (Pierce, USA) and X-ray film according to the manufacturer’s guidance.
Detection of apoptosis
An amount of 50 pmol/mL of RNA oligonucleotides was transfected into the HOS-CSCs. After 48 h, cells were collected and washed twice followed by staining with PI and Annexin-V (Sigma-Aldrich, USA) for 20 min at temperature. The apoptosis was then detected on the flow cytometry according to their manufacturer’s guidance.
Statistical analysis
All data are represented as the mean ± standard deviation (SD) and carried out by at least three independent experiments. Statistical analysis was performed by Student’s t-test using SPSS 14.0 software. Values of p < 0.05 were considered significant.
Results
MiR-19a is overexpressed in OS stem cells
To investigate the role of miR-19a in OS, its expression levels in OS tissue samples and paracancerous tissues are detected using qRT-PCR analysis. We found that expression level of miR-19a was 6.54-fold higher in the OS tissues than their paracancerous tissues (Figure 1(a)). To investigate the role of miR-19a in OS stem cells, we separated stem cells and non–stem cells from HOS, MG-63, and Saos-2 OS cell lines using CD133 antibody on flow cytometry. The isolation efficiency was shown in Figure 1(b). Subsequently, we evaluated the expression of miR-19a in the OS cell lines in vitro. We observed an upregulation of miR-19a in the HOS, MG-63, and Saos-2 OS cell lines compared with the OS paracancerous cells. More importantly, as compared to the non-CSCs of HOS, MG-63, and Saos-2, the CSCs of these OS cell lines expressed significantly higher levels of miR-19a (Figure 1(c)). These results suggested that miR-19a may act as an onco-miRNA in OS, especially in the CSCs of OS.

MiR-19a is overexpressed in osteosarcoma stem cells. (a) Expression levels of miR-19a in the OS tissue samples and paracancerous tissues are detected using qRT-PCR analysis (n = 25; **p < 0.01 vs the paracancerous tissues). (b) Isolation efficiency of OS stem cells and OS non–stem cells was evaluated by flow cytometry analysis using CD133 antibody. (c) QRT-PCR analysis was performed to detect the expression of miR-19a in the OS paracancerous cells, non-CSCs, and CSCs of HOS, MG-63, and Saos-2 OS cell lines. U6 snRNA was used as the internal control (*p < 0.05 vs OS paracancerous cells; #p < 0.05 vs non-CSCs of OS cell lines).
Knockdown of miR-19a decreases the population of CSCs in OS cells
As we observed a significant upregulation of miR-19a in the CSCs of OS, we next investigated the role of miR-19a in regulating the population of OS. We therefore knockdown the expression of miR-19a in the entire population of HOS, MG-63, and Saos-2 OS cells using the antisense-miR-19a (anti-miR-19a). As shown in Figure 2(a), transfection with anti-miR-19a significantly decreased the expression level of miR-19a in all of these OS cells. Interestingly, the flow cytometric analysis showed that transfection with miR-19a increased the proportion of CD133-positive CSCs populations by 65%–72% in HOS, MG-63, and Saos-2 cell lines (Figure 2(b)). It is suggested that knockdown of miR-19a could suppress population of CSCs in OS cells.

Knockdown of miR-19a suppressed the population of CSCs in OS cells. (a) QRT-PCR analysis was performed to detect the expression of miR-19a in HOS, MG-63, and Saos-2 osteosarcoma cell lines after they were transfected with anti-miR-19a for 48 h (*p < 0.05 vs NCO group). (b) Flow cytometry analysis was performed to evaluate the CD133+ cancer stem cell population in HOS, MG-63, and Saos-2 OS cell lines after they were transfected with anti-miR-19a for 48 h (*p < 0.05).
MiR-19a regulates the cell proliferation, migration, and survival in HOS-CSCs and HOS-non-CSCs
To further investigate the potential role of miR-19a in regulating the biological processes in OS stem cells, we performed several experiments to detect the cell proliferation, migration, and survival. We observed that overexpression of miR-19a increased the cell proliferation of HOS-CSCs by 21.7% and HOS-non-CSCs by 10.2%. Similarly, overexpression of miR-19a increased the cell migration of HOS-CSCs by 27.6% and HOS-non-CSCs by 11.8%. In contrast, knockdown of miR-19a expression decreased the cell proliferation of HOS-CSCs by 53.5% and HOS-non-CSCs by 28.4%. Similarly, knockdown of miR-19a decreased the cell migration of HOS-CSCs by 48.7% and HOS-non-CSCs by 25.9% (Figure 3(a) and (b)). More importantly, according to our date of cell viability assays, we observed that the inhibition of miR-19a decreased the cell viability of HOS-CSCs by 40.4% and HOS-non-CSCs by 24.2% (Figure 3(c)). Therefore, we demonstrated that miR-19a acts as a novel onco-miRNA to promote the cell proliferation, migration, and survival in OS cells. Furthermore, OS stem cells were more sensitive to anti-miR-19a-dependent inhibition of cell proliferation, migration, and survival compared with the OS non-stem cells.
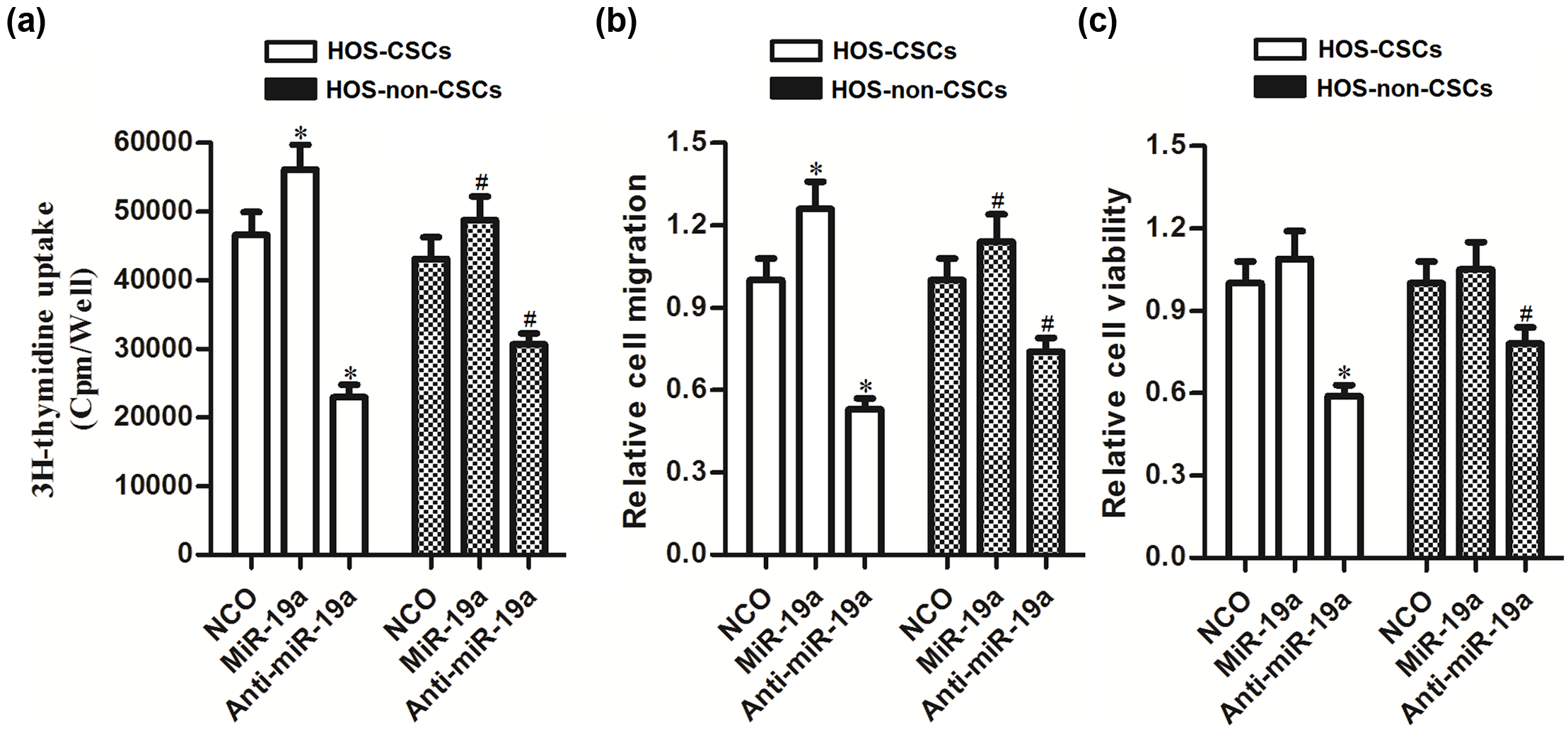
MiR-19a acts as a novel onco-miRNA to promote the cell proliferation, migration, and survival in HOS-CSCs and HOS-non-CSCs. (a) HOS-CSCs and HOS-non-CSCs were transfected with miR-19a mimics or the anti-oligonucleotides; 48 h after transfection, cell proliferation was evaluated using 3H thymidine incorporation assay (*p < 0.05 vs NCO group in HOS-CSCs; #p < 0.05 vs NCO group in HOS-non-CSCs). (b) Cell migration was evaluated using Transwell assay 48 h after transfection with miR-19a mimics or the anti-oligonucleotides. Relative cell migration was normalized to the cells transfected with NCO (*p < 0.05 vs NCO group in HOS-CSCs; #p < 0.05 vs NCO group in HOS-non-CSCs). (c) Cells were transfected with miR-19a mimics or the anti-oligonucleotides; 48 h after transfection, MTT assay was performed to evaluate the cell viability. Relative cell viability was normalized to the cells transfected with NCO (*p < 0.05 vs NCO group in HOS-CSCs; #p < 0.05 vs NCO group in HOS-non-CSCs).
PTEN is the target of miR-19a in OS stem cells
To understand the mechanism by which miR-19a regulates the cell viability of OS stem cells, the public database of TargetScan was used to search the potential target of miR-19a. It showed that the PTEN mRNA 3′-UTR contained highly conserved sequences which were targeted by miR-19a (Figure 4(a)). Furthermore, as the miR-19a was upregulated in HOS-CSCs (Figure 1(c)), we observed that the expression level of PTEN was significantly downregulated in HOS-CSCs compared with the HOS-non-CSCs (Figure 4(b)). Therefore, these results showed a negative correlation between the miR-19a and PTEN. To confirm the relationship between miR-19a and PTEN in OS, we cloned the PTEN 3′-UTR sequence into the pGL3 Luciferase Reporter Vectors. As shown in Figure 4(c), miR-19a mimics decreased the luciferase activity by 60.3%, and miR-19a inhibitors increased the luciferase activity by 36.4% specifically in the wild-type PTEN 3′-UTR group in HOS-CSCs, while in mutant 3′-UTR and empty control groups, this activity was abrogated. In accordance with this, the miR-19a mimics or inhibitors also regulated the expression of PTEN at the mRNA level and protein level in the HOS-CSCs (Figure 4(d)). Taken together, we demonstrated that the PTEN gene is an actual target of miR-19a in HOS-CSCs.

PTEN is the target of miR-19a in HOS-CSCs. (a) The putative miR-19a binding sites on 3′-UTR of PTEN mRNA was predicted by the TargetScan database. (b) The expression of PTEN at mRNA level and protein level in HOS-CSCs and HOS-non-CSCs was measured by qRT-PCR and western blot, respectively (*p < 0.05 vs HOS-non-CSCs). (c) The luciferase activity in HOS-CSCs was measured using Dual-Luciferase Reporter Assay System according to the manufacturer’s instruction (*p < 0.05 vs NCO group). (d) After transfection with miR-19a mimics or inhibitors for 48 h, the expression of PTEN at mRNA level and protein level in HOS-CSCs was measured by qRT-PCR and western blot, respectively (*p < 0.05 vs NCO group).
Knockdown of miR-19a inhibited the viability of OS stem cells through PTEN pathway
To explore the role of PTEN in the anti-miR-19a-induced cell death in OS stem cells, we transfected the HOS-CSCs with anti-miR-19a and PTEN siRNA. We observed that the PTEN siRNA could abolish the effect of anti-miR-19a on upregulating the PTEN expression level in OS stem cells (Figure 5(a)). We then found that the knockdown of PTEN significantly inhibited the anti-miR-19a-induced cell death in OS stem cells (Figure 5(b)). PTEN was the effector of anti-miR-19a in OS stem cells.
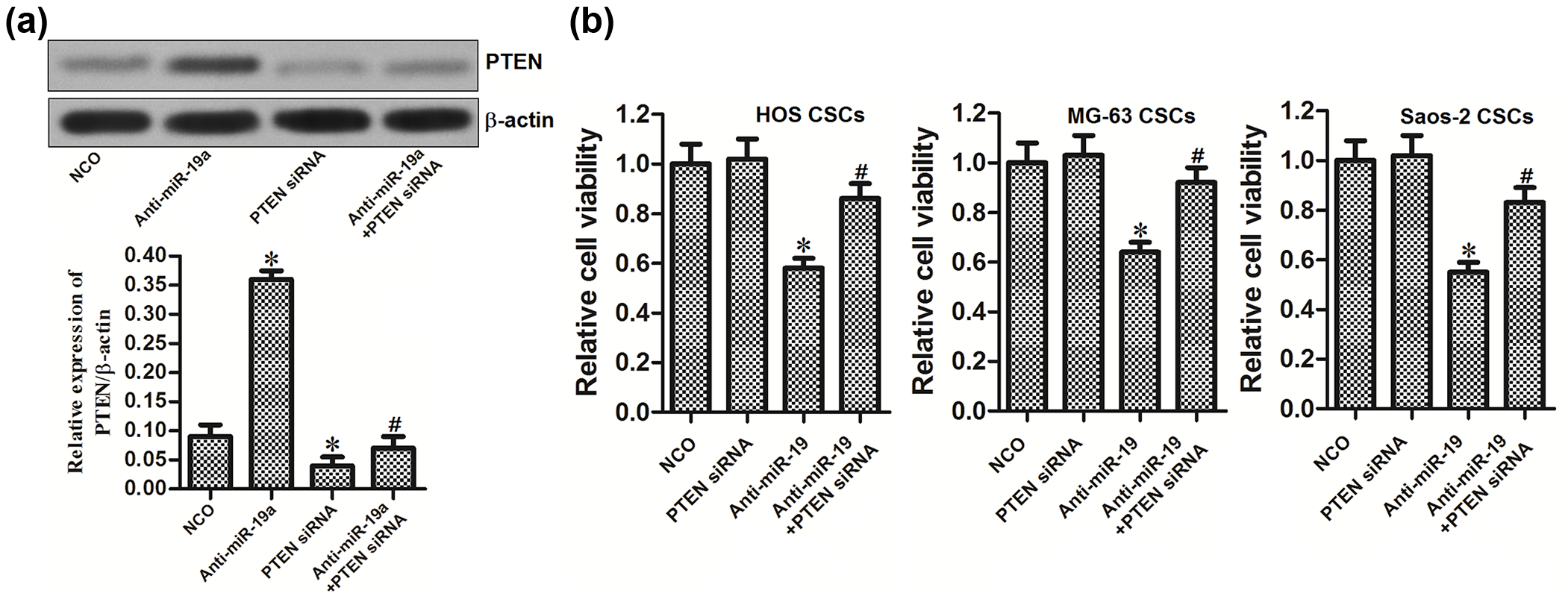
PTEN pathway is essential for the anti-miR-19a-induced cell death in HOS-CSCs. (a) Western blot analysis was performed to detect the expression of PTEN in HOS-CSCs after they were transfected with anti-miR-19a and PTEN siRNA for 48 h. (b) MTT assay was performed to evaluate the viability of HOS-CSCs, MG-63-CSCs, and Saos-2-CSCs after they were transfected with anti-miR-19a and PTEN siRNA for 48 h (*p < 0.05 vs NCO group; #p < 0.05 vs anti-miR-19a group).
Anti-miR-19a induced the apoptosis of OS stem cells through PTEN-PI3K/AKT pathway
PTEN is reported to be the suppressor of phosphatidylinositol 3-kinase (PI3K)/AKT pathway. 19 As shown in Figure 6(a), we found that transfection with anti-miR-19a significantly inhibited the phosphorylation of PI3K and AKT via upregulation of PTEN. Due to the PTEN-induced inactivation of PI3K/AKT pathway, anti-miR-19a induced the cell apoptosis in HOS-CSCs (Figure 6(b)). Accompanied by apoptosis, mitochondria-derived apoptotic inducers such as cytochrome c and Smac/DIABLO were released into the cytoplasm (Figure 6(c)). Additionally, cleavage of caspases 9, 7, and 3 was also induced by anti-miR-19a transfection (Figure 6(d)). Taken together, we demonstrate that anti-miR-19a induced the apoptosis of HOS-CSCs through the PTEN-PI3K/AKT pathway.

Anti-miR-19a induced the apoptosis of HOS-CSCs by inhibiting the PI3K/AKT pathway. (a) Western blot analysis was performed to detect the phosphorylation of PI3K and AKT. (b) Flow cytometry analysis was performed to measure the apoptosis of HOS-CSCs after they were transfected with anti-miR-19a and PTEN siRNA for 48 h (*p < 0.05 vs NCO group; #p < 0.05 vs anti-miR-19a group). (c) Western blot analysis was performed to measure the release of cytochrome c and Smac/DIABLO from mitochondria into cytoplasm. (d) Western blot analysis was performed to evaluate the cleavage of caspases 9, 7, and 3 in HOS-CSCs after they were transfected with anti-miR-19a and PTEN siRNA for 48 h.
Upregulation of PTEN is essential in the anti-miR-19a-inhibited cell proliferation and migration in OS stem cells
We next investigated the role of anti-miR-19a/PTEN pathway in regulating the cell proliferation and migration of OS stem cells. As shown in Figure 7(a), although the anti-miR-19a significantly inhibited the proliferation of OS stem cells, transfection with PTEN siRNA obviously impaired the effect of anti-miR-19a. Similarly, the results of cell migration assay indicated that knockdown of PTEN promoted the cell migration in the anti-miR-19a-treated OS stem cells (Figure 7(b)). We therefore demonstrated that anti-miR-19a inhibited the cell proliferation and migration of OS stem cells through PTEN pathway.
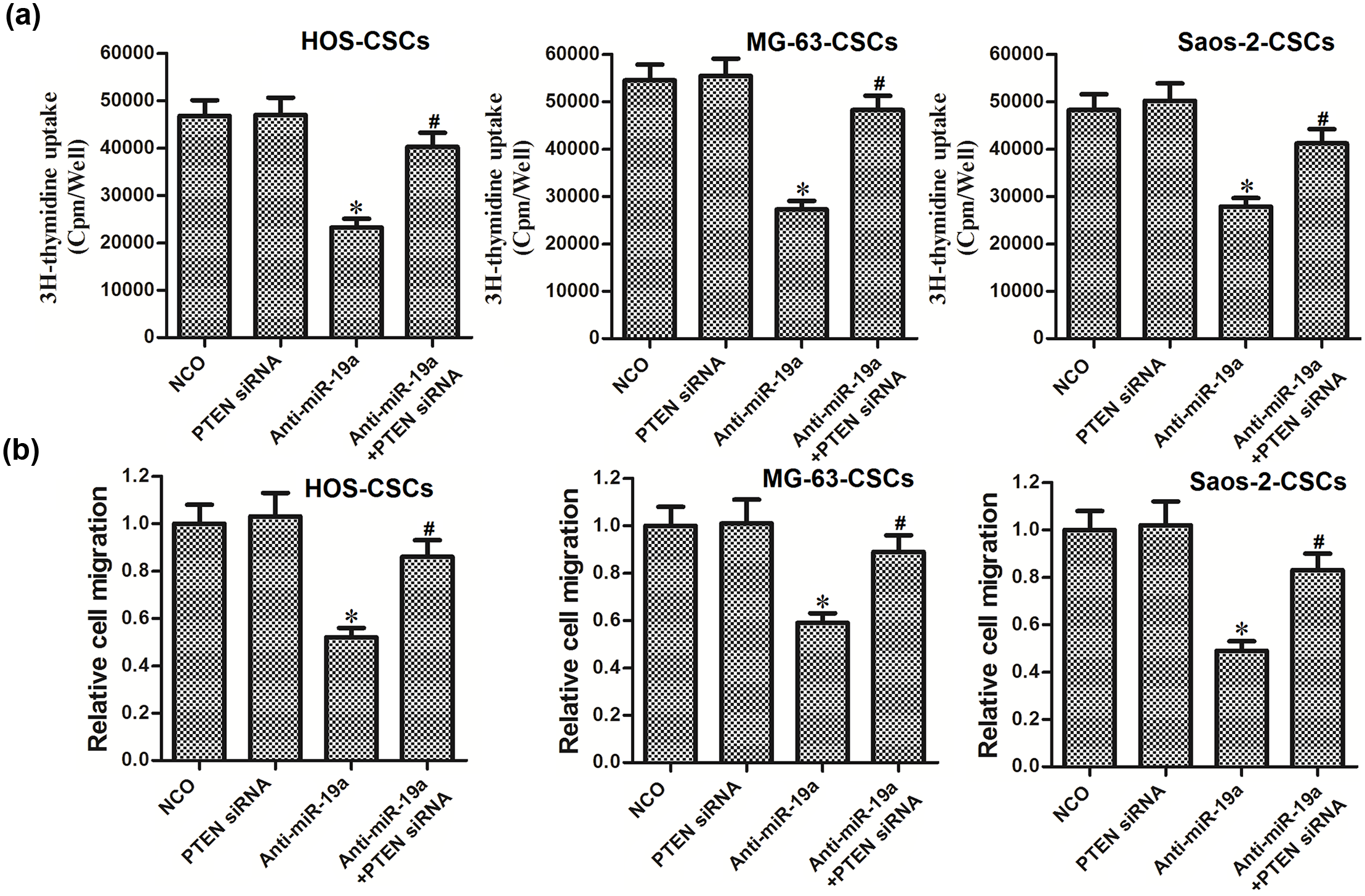
Upregulation of PTEN is essential in the anti-miR-19a-inhibited cell proliferation and migration in HOS-CSCs. (a) HOS-CSCs, MG-63-CSCs, and Saos-2-CSCs were transfected with miR-19a anti-oligonucleotides and PTEN siRNA. Cell proliferation was evaluated using 3H thymidine incorporation assay 48 h after transfection (*p < 0.05 vs NCO group; #p < 0.05 vs anti-miR-19a group). (b) Cell migration was evaluated using Transwell assay 48 h after transfection with miR-19a anti-oligonucleotides and PTEN siRNA. Relative cell migration was normalized to the cells transfected with NCO (*p < 0.05 vs NCO group; #p < 0.05 vs anti-miR-19a group).
Discussion
CSCs are a small subpopulation of cancer cells that obtain the ability of self-renewal, multilineage differentiation potential, and tumorigenicity. 4 Due to their ability of tumor initiation and self-renewal, CSCs are believed to be responsible for the cancer relapse and metastasis.5,20 Therefore, CSCs have been considered as a promising target for cancer therapy. Removing the CSCs is believed to improve the survival of cancer patients. 21 Clinical therapy approaches of OS include surgery, chemotherapy, and radiationtherapy.22,23 Although these treatments may improve the survival of cancer patients for a while, OS stem cells were usually identified to develop the resistance to chemotherapy.24,25 Therefore, the poor prognosis of OS is associated with the CSCs.
MiR-19a has been reported to act as an onco-miRNA in several cancers. For example, miR-19a is overexpressed in HCC cells. Ectopic expression of miR-19a dramatically induced the proliferation and cell growth of HCC cells by targeting tissue inhibitors of metalloproteinase-2 (TIMP-2), whereas inhibition of miR-19a reduced these effects. 26 Tumor necrosis factor-α (TNF-α) upregulated the expression of miR-19a, and the miR-19a was required for TNF-α-induced epithelial-to-mesenchymal transition (EMT) and metastasis in colorectal cancer cells. 27 For lung cancer patients, high levels of miR-19a were associated with poor prognosis of lung cancer. MiR-19a promoted proliferation and metastasis of lung cancer cells by targeting MAX dimerization protein 1 (MXD1). 28 It is clear that miR-19a promotes cancer occurrence and development. However, the role of miR-19a in CSCs, especially in the OS stem cells, remains unclear.
In accordance with previous studies, we found that the expression level of miR-19a was upregulated in OS patients’ tumor tissues and OS cell lines in vitro. More importantly, we discovered that the miR-19a was dramatically upregulated in the OS stem cells compared to the non-CSCs. Knockdown of miR-19a by its anti-oligonucleotides significantly reduced the population of CSCs in these OS cell lines. We therefore demonstrate that miR-19a plays important roles in OS stem cells. The data of 3H thymidine assay, Transwell penetration test, and MTT assay prove that miR-19a regulates the cell proliferation, migration, and survival in OS stem cells.
PTEN is well known as a tumor suppressor in multiple cancers. Expression of PTEN is usually downregulated in various types of human cancer. Subtle changes in PTEN expression may cause critical outcomes in tumor initiation and development in vivo.29,30 Loss of PTEN induces superactivation of PI3K/AKT pathway, which leads to cell survival and apoptosis resistance.31,32 Recently, several studies have indicated that PTEN is regulated by miRNAs in various types of cancer. MiRNAs/PTEN axis has been proved to regulate the cell growth and apoptosis in multiple cancers.33,34
In this study, we demonstrate that the gene of PTEN, which is downregulated in OS stem cells, is the target of miR-19a. Our data prove that knockdown of miR-19a increases the expression of PTEN in OS stem cells. Furthermore, miR-19a antagonists induce caspase-dependent apoptosis and inhibit cell proliferation and migration through the PTEN-PI3K/AKT pathway.
In summary, this study reveals an oncogenic function of miR-19a in OS cells, especially in the CSCs. Notably, knockdown of miR-19a inhibits cell proliferation, migration, and survival in OS stem cells through the PTEN pathway. Therefore, miR-19a/PTEN axis may be the potential target for the treatment of OS.
Footnotes
Compliance with ethical standards
The study was approved by Ethic Committee of The First Affiliated Hospital of Shantou University Medical College.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by 2015 Shantou City Science and Technology Planning Project (Grant No. 114).
