Abstract
Objectives
The objective of this study was to investigate the therapeutic effect and mechanism of Chang-An-Kang on ulcerative colitis rats.
Materials and Methods
The rat model of ulcerative colitis induced by 6% glacial acetic acid was established. The corresponding therapeutic drugs were given 24 h after the model was established. The pathological morphology of colon tissue was observed after 21 days of continuous administration. The levels of interleukin-6 (IL-6), interleukin-7 (IL-7), interleukin-10 (IL-10), and tumor necrosis factor-alpha (TNF-α) were measured by ELISA kit. The expression levels of NF-κB and STAT3 proteins in colon tissue were detected by IHC.
Results
The results showed that the changes in the pathological and biochemical indexes of the rat colon tissue indicate that the model was successfully established. Chang-An-Kang could significantly reduce rat colon tissue damage, decrease the content of IL-6, IL-7, and TNF-α, and increase the content of IL-10. The NF-κB and STAT3 protein expression levels were decreased.
Conclusion
By increasing the content of anti-inflammatory factor IL-10, decreasing the content of pro-inflammatory factors IL-6 and TNF-α, and inhibiting the expression of NF-κB and STAT3 proteins, Chang-An-Kang can reduce inflammatory infiltration. It has an obvious therapeutic effect on acetic acid ulcerative colitis in rats.
Introduction
Ulcerative colitis (UC) is a chronic inflammatory disease of the rectum and colon with unknown etiology. It is usually bloody diarrhea or purulent blood stool. It may have paroxysmal spastic colic in the left lower abdomen or lower abdomen, occasionally nausea, vomiting, epigastric discomfort, fever, and other symptoms. 1 At present, there is no effective medicine prevention and treatment for the etiology of UC. The clinical treatment of UC is still based on salicylic acid and sulfasalazine. However, the high recurrence rate limits its widespread use in UC patients.2, 3
Chang-An-Kang is composed of Pulsatilla chinensis, Coptisine, Phellodendron amurense, Atractylodes macrocephala, Astragalus membranaceus, Poria cocos, Panax notoginseng, and Halloysitum rubrum. It has the effect of clearing heat, promoting diuresis, and invigorating spleen. 4 Clinical application of Chang-An-Kang has significant efficacy in the treatment of UC, mainly by eliminating intestinal inflammation, improving intestinal microcirculation, and accelerating the healing of ulcer surface,5, 6 but its potential mechanism is still unclear. This study explored the potential mechanism of Chang-An-Kang on UC based on the NF-κB/STAT3 signaling pathway by establishing a UC rat model and provided an experimental basis for the clinical treatment of UC.
Materials and Methods
Experimental Animal
A total of 40 SPF male Wistar rats (200 ± 20g) were provided by the experimental animal center of Heilongjiang Academy of Traditional Chinese Medicine Sciences (license number: SYXK (H) 2016-008).
Experimental Drug
Drug
Chang-An-Kang is composed of P. chinensis, Coptisine, P. amurense, A. macrocephala, A. membranaceus, P. cocos, P. notoginseng, and H. rubrum. It is provided by the Heilongjiang Academy of Traditional Chinese Medicine.
Preparation of Drug
Chang-An-Kang is composed of P. chinensis (6 g), Coptisine (10 g), P. amurense (10 g), A. macrocephala (10 g), A. membranaceus (20 g), P. cocos (15 g), P. notoginseng (5 g), and H. rubrum (15 g). After soaking, the herbs were decocted and extracted twice. After filtration, the extract is combined and concentrated in a rotary evaporator solution (2 g/mL). After cooling, the extract was transferred into a sterilized glass vial and stored in a refrigerator at 4℃ for later use.
Positive Control Drug
Sulfasalazine Tablets (SASP), Shanghai Sanwei Pharmaceutical Co., Ltd., Number: 22201203.
Experimental Reagents
The reagents used were hematoxylin-eosin (HE) staining kit (Biyuntian Biotechnology Co., Ltd.), IL-6, IL-7, IL-10, TNF-α ELISA kit (Nanjing Jiancheng Institute of Bioengineering), STAT3 Rabbit Monoclonal Antibody (Biyuntian Biotechnology Co., Ltd.), NF-kB Rabbit Monoclonal Antibody (Biyuntian Biotechnology Co., Ltd.), and SABC-POD kit (Wuhan Boster Biological Technology Co., Ltd.).
Method
Establish UC Model
The rats were raised under conventional conditions in the laboratory. Rats were randomly divided into blank control group (BC), model control group (MC), SASP group, and Chang-An-Kang group according to body weight. Except for the rats in the BC group, the rats in each group were given 10% MgSO4 solution by gavage, 3 mL/kg, twice/day for 3 days, and then fasted for 24 h without water deprivation. The rats were anesthetized with 2% sodium pentobarbital intraperitoneally and then fixed. The stomach needle was inserted about 8 cm from the anus (colon), and 2 mL of 6% glacial acetic acid was slowly injected into the stomach with a syringe. The rats in the BC group were injected with the same volume of normal saline. Then 0.3 mL air was injected into the anus of rats for 1.5 h.7, 8 After natural wakefulness, the rats in the BC and MC groups were given normal saline by gavage (10 mL/kg, once/day). The rats in the SASP group were given sulfasalazine tablets (0.5 g/kg, once/day). 9 The rats in the Chang-An-Kang group were given experimental drugs (5.98 mL/kg, twice/day). 10 After continuous administration for 21 days, during the trial, rats in each group were free to feed and drink during the experiment.
Index Detection
Colonic Histomorphology
Preparation of colon tissue paraffin sections: Rats were fasted for 24 h after the last administration. After the rats were anesthetized and killed, the whole colon (2–10 cm away from the anus) was quickly cut off. The intestinal contents were removed with normal saline. After washing with PBS solution twice, the colon was fixed in 10% formaldehyde solution. After 24 h, the colon was dehydrated in an ethanol gradient and embedded in paraffin. The thickness of the paraffin section was 5 µm. After drying, the colon was stored in the section box for use.
Dewaxing process of dry paraffin sections: It was soaked in xylene Ⅰ for 15 min, xylene Ⅱ for 15 min, 100% ethanol Ⅰ for 15 min, 100% ethanol Ⅱ for 15 min, 95% ethanol Ⅰ for 5 min, 95% ethanol Ⅱ for 5 min, 80% ethanol for 5 min, 30% ethanol for 5 min, and deionized water for 10 min, then stained in hematoxylin for 4 min, washed with 1% hydrochloric acid alcohol for 5 s, washed with weak ammonia water twice, and washed with water for 10 min. After staining, the effect was observed under a microscope. Eosin staining was performed for 12 min, and then it was soaked in 80% ethanol for 5 s, 95% ethanol for 5 s, 100% ethanol Ⅰ for 5 min, 100% ethanol Ⅱ for 10 min, xylene Ⅰ for 10 min, and xylene Ⅱ for 10 min. The slides were mounted with neutral gum and observed under a microscope.
Contents of IL-6, IL-7, IL-10, and TNF-α in Colonic Tissue
Notably, 10 mg of rat colon tissue was taken, the tissue was homogenized and centrifuged at 4°C for 15 min, and the supernatant at 3000 rpm/min was taken. The contents of IL-6, IL-7, IL-10, and TNF-α in rat colon tissue were detected according to the kit steps.
Detection of NF-κB and STAT3 Protein Expression in Colon Tissue by IHC
The pathological colon tissues (0.5 × 0.5 cm2) were taken, fixed in 10% formaldehyde solution, and prepared into paraffin sections. Three view fields were randomly selected under a 20× microscope. The positive cell area (Area) and integrated optical density (IOD) of NF-κB and STAT3 proteins staining in colonic tissue were analyzed by the Image-pro plus image softening method. The expression of NF-κB and STAT3 proteins in colonic tissue was semi-quantitatively analyzed.
The expression process of NF-κB protein in colon tissue was detected by IHC: The dried paraffin sections were dewaxed with xylene Ⅰ for 15 min, xylene Ⅱ for 15 min, 100% ethanol Ⅰ for 15 min, 100% ethanol Ⅱ for 15 min, 95% ethanol Ⅰ for 5 min, 95% ethanol Ⅱ for 5 min, 80% ethanol for 5 min, 30% ethanol for 5 min, and deionized water for 10 min. The deparaffinized sections were washed with PBS solution for 3 min for three times, the residual PBS was removed by suction, non-immune serum solution was added, and they were incubated for 10 min (room temperature). The blocking solution was sucked. Sufficient antibody was added with a concentration of 1:500 to the sections, and they were incubated overnight at 4℃ in a wet box. They were washed with PBS for 3 min for three times, the residual PBS was removed by suction, enough biotin-labeled goat anti-rabbit was added with a concentration of 1:1000, and they were incubated for 10 min (room temperature). They were washed with PBS for 3 min for three times, residual PBS was removed by suction, streptavidin-peroxidase was added, and they were incubated for 10 min (room temperature). They were washed with PBS for 3 min for three times, the residual PBS was absorbed, DAB chromogenic solution was added in the dark, they were rinsed with deionized water, the staining degree was observed under the microscope, and the nucleus was stained with hematoxylin for 2 min, washed with weak ammonia blue and deionized water, dehydrated with gradient ethanol and xylene according to the reverse order of dewaxing, and mounted in neutral resin after drying under the microscope observation photos.
The IHC method was used to detect the expression of STAT3 protein in colon tissue, which was the same as that of NF-κB protein.
Statistics
The experimental data were expressed as x^-±s. The t-test was used to analyze the significant difference between the two groups (p < 0.01).
Results
Effects on the Pathological Morphology of Colon Tissue
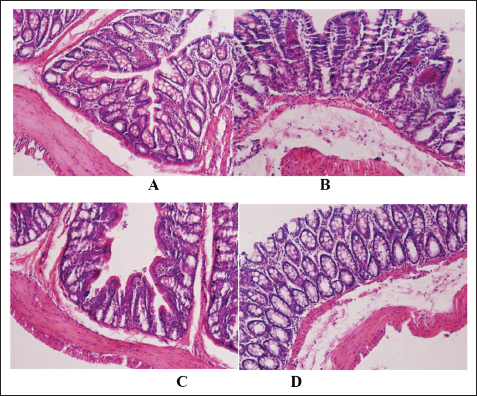
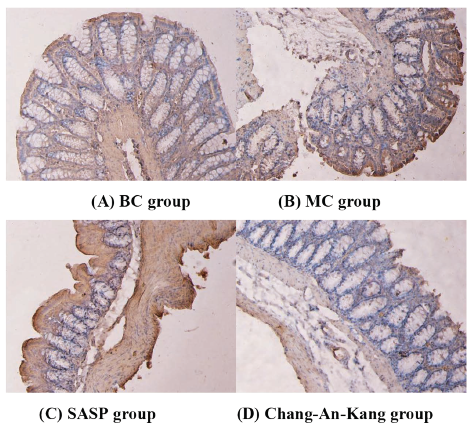
Compared with rats in the BC group, rats in the MC group showed significant colon tissue edema and severe damage. SASP and Chang-An-Kang had varying degrees of improvement effects on the above indicators, and Chang-An-Kang had a more significant improvement effect. Under the light microscope, the mucosal epithelial cells of the colon tissue of the BC group rats were intact, the glands were arranged neatly, and the structure was clear. The mucosal epithelium of the colon tissue in the MC group of rats was detached, damaged, and erosive, with inflammatory granulocyte infiltration and cellulose-like exudation, accompanied by necrosis and ulceration. The mucosal epithelial shedding of the colon in SASP group rats was reduced, and inflammatory granulocytes were reduced. The colon tissue of rats in the Chang-An-Kang group showed a reduction in intestinal mucosal epithelial detachment and defect, repair of epithelial hyperplasia of the surrounding intestinal mucosa, significant reduction of the ulcer area, and proliferation of granulation tissue, showing significant improvement. The results are shown in Figure 1A–D.
Effects on the Pathological Morphology of Colon Tissue. (A) BC group, (B) MC group, (C) SASP group, and (D) Chang-An-Kang group.
Levels of IL-6, IL-7, IL-10, and TNF-α in Colon Tissue
Compared with the BC group, the contents of IL-6, TNF-α, and IL-7 in the colon tissue of the MC group were significantly increased (p < 0.01), and the contents of IL-10 were significantly decreased (p < 0.01). SASP and Chang-An-Kang had different effects on the above indexes, and Chang-An-Kang had more obvious effects. The results are shown in Table 1.
Effects of Chang-An-Kang Oral Administration on IL-6, IL-7, IL-10, and TNF-α in UC rats (x^-±s).
Expression of NF-κB and STAT3 Proteins in Colon Issue
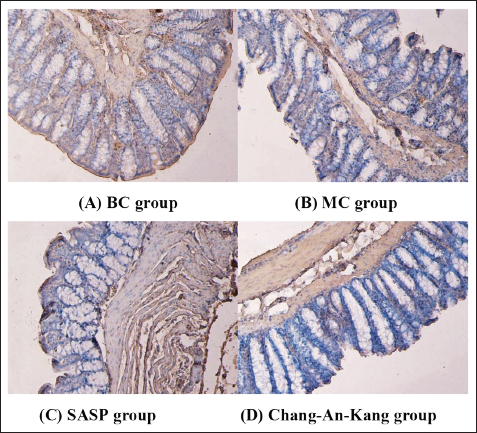
Compared with the BC group, the expression of NF-κB protein in the MC group increased, which showed that the Area of positive cells and the IOD of NF-κB protein were increased (p < 0.05). SASP and Chang-An-Kang decreased the expression of NF-κB protein, which showed that Area and IOD decreased. Chang-An-Kang had a significant effect on reducing Area (p < 0.01) and IOD (p < 0.05). SASP could decrease Area (p < 0.05) and IOD, but the difference was not significant. The results are shown in Figure 2A–D and Table 2.
Expression of NF-κB Protein in Colon Tissue. (A) BC group, (B) MC group, (C) SASP group, and (D) Chang-An-Kang group.
Expression of Chang-An-Kang Oral Administration on the Levels of NF-κB and STAT3 in UC rats (x^-±s).
Compared with the BC group, the expression of STAT3 protein in the colon of the MC group was decreased, which showed that STAT3 protein Area (p < 0.05) and IOD were decreased, but there was no significant difference between the MC and BC groups. SASP could increase the expression of STAT3 protein and Area (p < 0.01) and IOD (p < 0.05). The expression of STAT3 protein was decreased in Chang-An-Kang, which showed a significant decrease in Area and IOD (p < 0.01). The results are shown in Figure 3A–D and Table 2.
Expression of STAT3 Protein in Colon Tissue. (A) BC group, (B) MC group, (C) SASP group, and (D) Chang-An-Kang group.
Discussion
The occurrence and development of UC is a very complex biological phenomenon involving multiple genes and multiple factors. The study of the NF-κB/STAT3 signaling pathway provides a large number of favorable and reliable evidence for the pathogenesis of UC. 11 Intervening the key factors of the NF-κB/STAT3 signaling pathway and then blocking the excessive inflammatory response of UC has become a hot spot in the treatment of UC. 12
During UC inflammation, activation of both NF-κB and STAT3 is stimulated by various extracellular signals, and regulation of NF-κB involves target gene expression of tumor proliferation, apoptosis, infiltration, metastasis, and angiogenesis. NF-κB target genes, such as IL-6, are also important activators of STAT3.13, 14 STAT3 can directly affect P65, a member of the NF-κB family, to keep it in the nucleus and inhibit its negative feedback regulatory pathway.15, 16 Recent studies have shown that NF-κB is involved in the regulation of cytokines such as TNF-α, IL-6, IL-10, and IL-17 at the transcriptional level.17, 18 IL-6 is a multifunctional, multipotent glycosylated protein factor that binds specifically to the target cell surface receptor, participates in the local inflammatory response, and also regulates the immune response. 19 Activation of NF-κB via the STAT-3 pathway induces polarized expression of cell adhesion molecule (ICAM-1), and phosphorylated STAT dissociates from the receptor when the cell is stimulated by the signal. they enter the nucleus in the form of homogenous or heterodimerization and multimerization, bind specifically to DNA target sequences, regulate the expression of corresponding genes, and complete the whole process of cytokine-mediated signal transduction. 20 The role of IL-6 in acute inflammatory response is mainly manifested as pro-inflammatory effect on various cells, induction of acute response protein in liver tissue, promotion of electrolyte secretion of intestinal epithelial cells, increase of permeability of endothelial cells, and migration of neutrophils to inflammatory sites to cause lesions. 21 IL-10 is an anti-inflammatory factor mainly produced by activated macrophages and monocytes, and TNF-α is a multifunctional cytokine secreted by mono-nuclear macrophages and activated T lymphocytes, which has the effect of promoting inflammatory response and cellular immune regulation. 22 IL-10 can reduce inflammatory mediator TNF-α and inhibit antigen presentation and plays an important role in maintaining a normal intestinal non-inflammatory immune state and preventing and controlling excessive immune response under the biological activity of B cells and T cells.23, 24 Studies have shown that mice develop spontaneous UC due to the absence of IL-10, a decrease in the anti-inflammatory factor IL-10, and an increase in the pro-inflammatory factors IL-6 and TNF-α. 25 IL-17 can induce the expression of innate immune inflammatory cytokines, acute response protein, granulocyte-macrophage stimulating factor (G-CSF), and prostaglandin E2 and also has a synergistic effect with TNF-α to enhance its inflammatory effect. 26 IL-17 induces the expression of several genes associated with inflammation, including IL-6, cell adhesion molecules, and so on.27–;29 Studies have shown that the expression of IL-17 in intestinal mucosal tissue and serum of active UC patients is significantly higher than that of healthy people, and it gradually increases with the increase of disease activity. 30
In this study, we investigated the effect of Chang-An-Kang on the NF-κB/STAT3 signaling pathway and revealed that Chang-An-Kang may inhibit the abnormal activation of STAT3 and NF-κB signaling pathway in the treatment of UC, to maintain the dynamic equilibrium of pro-and anti-inflammatory factors, so as to control and alleviate the inflammation of colonic mucosa, protect the colonic mucosa, and provide a theoretical basis for clinical application.
Footnotes
Authors’ Contributions
LZ conceived ideas, completed the experiments, and implemented the animal experiment. WG collected and arranged the data. CR and LY revised the article and contributed to supportive work. CX designed and funded the experiments.
Availability of Data and Material
All relevant data are contained within the article: The original contributions presented in the study are included in the article/supplementary material, and further inquiries can be directed to the corresponding author/s.
Consent for Publication
Written informed consent for publication of their clinical details and/or clinical images was obtained from the patient/parent/guardian/ relative of the patient. A copy of the consent form is available for review by the Editor of this journal.
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship and/or publication of this article.
Funding
This work was partially supported by the Nature Science Foundation of Heilongjiang Province of China (Grant number: LH2023H077).
Statement of Ethics Approval and Consent to Participate
This study was reviewed by the Ethics Committee of the Heilongjiang Academy of Chinese Medicine Sciences (No. 2015-64), and the experiments were conducted by the Chinese Government’s “Guidelines on the Care and Use of Laboratory Animals” to minimize their suffering.
