Abstract
Our study aimed to investigate the protective effects and potential mechanisms of pterostilbene on rats with ulcerative colitis (UC). We established 2,4,6-trinitrobenzenesulfonic acid (TNBS) induced colitis rat model. Rats were randomly divided into three groups, including control group, model group, and pterostilbene group (30 mg/kg). Disease activity index (DAI) including body weight, stool consistency, and gross bleeding was measured. The concentration of superoxide dismutases (SODs), glutathione superoxide (GSH-px), malondialdehyde (MDA), and methylpropanediol (MPO) in serum were detected by enzyme-linked immunosorbent assay (ELISA). The levels of interleukin-1 beta (IL-1β), IL-17, IL-6, and tumor necrosis factor–alpha (TNF-α) in serum were also analyzed by ELISA kits. Histological evaluations of colons were conducted. The levels of peroxisome-proliferator-activated receptor–γ (PPAR-γ), nuclear factor-κB (NF-κB), ZO-1, and Occludin were analyzed by immunohistochemistry. Compared with model group, pterostilbene notably suppressed the production of TNF-α, IL-17, IL-1β, IL-6, MDA and MPO in serum, and markedly increased the SOD and GSH-Px activity in serum. Pterostilbene significantly attenuated macroscopic damage and histological injury, when compared with model rats. Furthermore, pterostilbene also markedly activated the expression of PPAR-γ, ZO-1, and Occludin, and suppressed the expression of NF-κB. The protective effects of pterostilbene might be associated with suppression of NF-κB and activation of PPAR-γ. Pterostilbene might be a promising therapeutic agent for colitis treatment.
Introduction
Ulcerative colitis (UC) is an inflammatory autoimmune disease which affects millions of people worldwide and is characterized by chronic uncontrolled inflammation of intestinal mucosa. 1 Many studies have shown that pro-inflammatory mediators were overexpressed in the pathological process of colitis, including reactive oxygen species (ROS), neutrophil infiltration, and cytokines. 2 Although the current treatments are effective, many complications are caused, including high blood pressure, fever, and cramps. 3 Therefore, it is necessary to find a new drug with low side effects for the treatment of UC.
Many drugs with antioxidant and anti-inflammatory have been reported to treat UC. Brusatol ameliorates 2,4,6-trinitrobenzenesulfonic acid (TNBS)–induced experimental colitis in rats via inhibiting NF-κB pathway and recombinant NLR family, pyrin domain containing protein 3 (NLRP3) inflammasome. 4 Indigo naturalis improved colonic mucosal injuries and inflammation in rats with dextran sodium sulfate induced UC. 5 Geniposide ameliorates TNBS-induced experimental colitis in rats via reducing inflammatory cytokine release and restoring impaired intestinal barrier function. 6 These studies showed that many medicines ameliorated UC by inhibiting inflammation and oxidant stress. However, there is little research about the effect of pterostilbene on UC, which has received considerable attention in the treatment of inflammatory diseases.
Pterostilbene is a major bioactive constituent of sandalwood and blueberries, 7 which is widely used in traditional Chinese medicine. It has become increasingly popular due to the function of health benefits, including anti-inflammatory, 8 antidiabetes, 9 and cardioprotective actions. 10 Pterostilbene could inhibit the secretion of tumor necrosis factor–alpha (TNF-α) via suppressing the nuclear factor-κB (NF-κB) activation. 11 Moreover, a recent study showed that pterostilbene decreased cardiac oxidative stress and inflammation via activation of adenosine monophosphate-activated protein kinase (AMPK)/nuclear factor erythroid2-related factor (Nrf2)/heme oxygenase1 (HO-1) pathway in fructose-fed diabetic rats. 12 Studies also proved that pterostilbene improved acute kidney injury in septic mice by activating the Nrf2 antioxidative defense system. 13 Pterostilbene also suppresses ovarian cancer growth via induction of apoptosis and blockade of cell cycle progression involving inhibition of the STAT3 pathway. 14 A study revealed that pterostilbene protected against acute renal ischemia reperfusion injury via the Toll-like receptor 4 (TLR4)/NF-κB signaling pathway. 15 However, the effect of pterostilbene against UC has not been explored and whether pterostilbene is effective to treat UC is unclear.
In the study, we hypothesize that pterostilbene may protect against UC through regulating NF-κB pathway and activating the expression of peroxisome-proliferator-activated receptor–γ (PPAR-γ). We used TNBS-induced UC animal model to explore.
Materials and methods
Animals
In total, 30 adult female Sprague-Dawley (SD) rats, weighing 200 to 220 g, were purchased from Jinan Peng Yue Experimental Animal Breeding Co., Ltd. (SCXK(Lu)20140007, China). The rats were raised in standard environment, temperature 20 to 26°C, daily temperature ⩽4°C, humidity 40% to 70%, and 12 h light/dark cycle with free access to water. The feed is specially designed for specific pathogen free (SPF) grade rats and produced by Jiangsu Synergetic Medical Bioengineering Co., Ltd. (20180620). All animal experiments were approved by the Institutional Animal Care and Use Committee of Ganzhou People’s Hospital.
Animal groups and model
Rats were randomly divided into three groups, including control group, model group, and pterostilbene-treated group. The colitis model was established according to the previous study. 4 The rats were fasted for 48 h and anesthetized with 10% chloral hydrate (0.3 mL/100 g, Kai Tong Chemical Reagent Co., Ltd., Tianjin, China) intraperitoneally. The colon of rats was treated with TNBS (25 mg/kg, Sigma-Aldrich, St. Louis, MO, USA) dissolved in 50% ethanol (v/v) using a silicone tube (external diameter 2 mm) and inserted 8 cm deep into the rectum. After resuscitation, the rats were treated with intragastric administration once a day by pterostilbene (30 mg/kg, Sigma-Aldrich). The control group was treated with normal saline with the same method.
Evaluation of disease activity index
The disease activity index (DAI) was calculated during the period of experiment. The DAI was described by combining the score of weight loss, stool consistency, and stool bleeding. DAI = (weight loss index + stool + bleeding)/3. The diagnostic criteria for DAI are described in Table 1.
The diagnostic criteria for DAI.
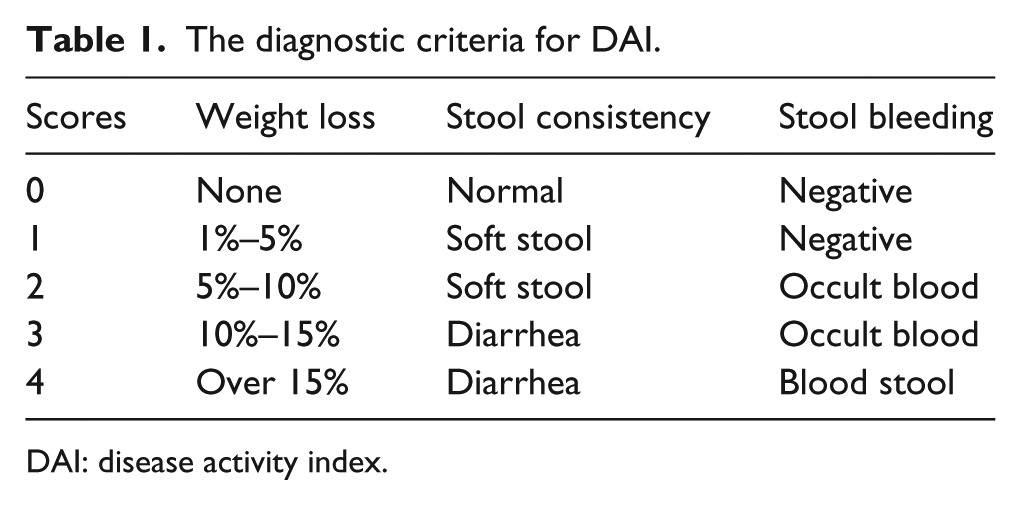
DAI: disease activity index.
Samples collection
All rats were anesthetized by intraperitoneal injection of 10% chloral hydrate (0.3 mL/100 g) on day 21. The 5 mL blood was collected from abdominal aorta and standed for 2 h. After that, the blood was centrifuged with 3000 r/min at 4°C for 15 min. The serum was separated and frozen at −80°C. The terminal colons (long distance from anus 10 cm) were collected and stored at −80°C for analysis.
Enzyme-linked immunosorbent assay
The levels of superoxide dismutases (SODs), glutathione superoxide (GSH-px), malondialdehyde (MDA), and methylpropanediol (MPO) in serum were determined by the enzyme-linked immunosorbent assay (ELISA) kits (Mibio, Shanghai, China). The levels of interleukin-1 beta (IL-1β), IL-17, IL-6, and TNF-α in serum were also analyzed by ELISA kits.
Histopathological examination
The colon tissues sized 1 mm3 were fixed in 4% neutral formaldehyde overnight at room temperature and washed by serial graded ethanol for 40 min. Subsequently, the tissues processed and embedded in paraffin, and then sectioned at 5 µm. Hematoxylin and eosin (HE) staining was performed following the manufacturer’s instructions (Baso, Zhuhai, China). The images were captured at 40× and 400× magnification. Histological scoring was described as the previous report. 16
Immunohistochemical staining
The colon sections (5 µm) were dewaxed and hydrated by serial graded ethanol. After that, the sections were washed with phosphate-buffered saline (PBS, Baso) three times, 5 min each. Antigen was heated to 95°C by a microwave (SLEE, Mainz, Germany) and cooled to room temperature for three times. Primary antibodies, PPAR-γ, NF-κB, ZO-1, and Occludin (1:1000, Cell Signaling Technology, Danvers, MA, USA), were incubated at 4°C overnight. Then, the sections were incubated at room temperature for 30 min before washing with phosphate buffered saline tween (PBST). After washing, the sections were incubated with horseradish peroxidase labeled secondary antibody (1:400) at 37°C for 30 min. The chromogen was used diaminobenzidine (DAB) Horseradish Peroxidase Color Development Kit (Beyotime Biotechnology, Shanghai, China). Positive expression areas were observed under microscope (Sigma-Aldrich). The images were captured at 40× magnification.
Statistical analysis
All data were expressed as the mean ± standard deviation (SD). Statistical analysis was conducted using SPSS software (version 19.0). The differences between groups were analyzed by one-way analysis of variance and Dunnett’s multiple comparison test. A value of P < 0.05 was considered statistically significant difference.
Results
General observation
During the course of the research, there was no obvious changes in control rats. However, rats in model group appeared weight loss, diarrhea, and purulent stools, along with lack of vitality and shaggy hair. After treatment of pterostilbene, the body weight recovered obviously from the 10th day (Figure 1(a)). Meanwhile, the state of rats improved markedly including hair color, weight, and stool. The result of DAI was consistent with the pathological symptoms (Figure 1(b)). The DAI scores in model group were obviously higher than that in control group (P < 0.01). After treatment of 20 days, the DAI scores were significantly decreased (P < 0.05).

Effects of pterostilbene on body weight change (a) and disease activity index (b) of TNBS-induced colitis in rats. Data are presented as mean ± SD (n = 10). Compared with control group, *P < 0.05, **P < 0.01. Compared with model group, #P < 0.05, ##P < 0.01.
Pterostilbene decreased the levels of inflammatory cytokines in serum
The expression of inflammatory factors was analyzed in Figure 2. Compared with the control group, the levels of TNF-α, IL-17, IL-6, and IL-β were obviously increased in model group (P < 0.05). After the treatment of pterostilbene, the contents of inflammatory cytokines in serum were significantly declined, but still higher than those in control group (P < 0.05).
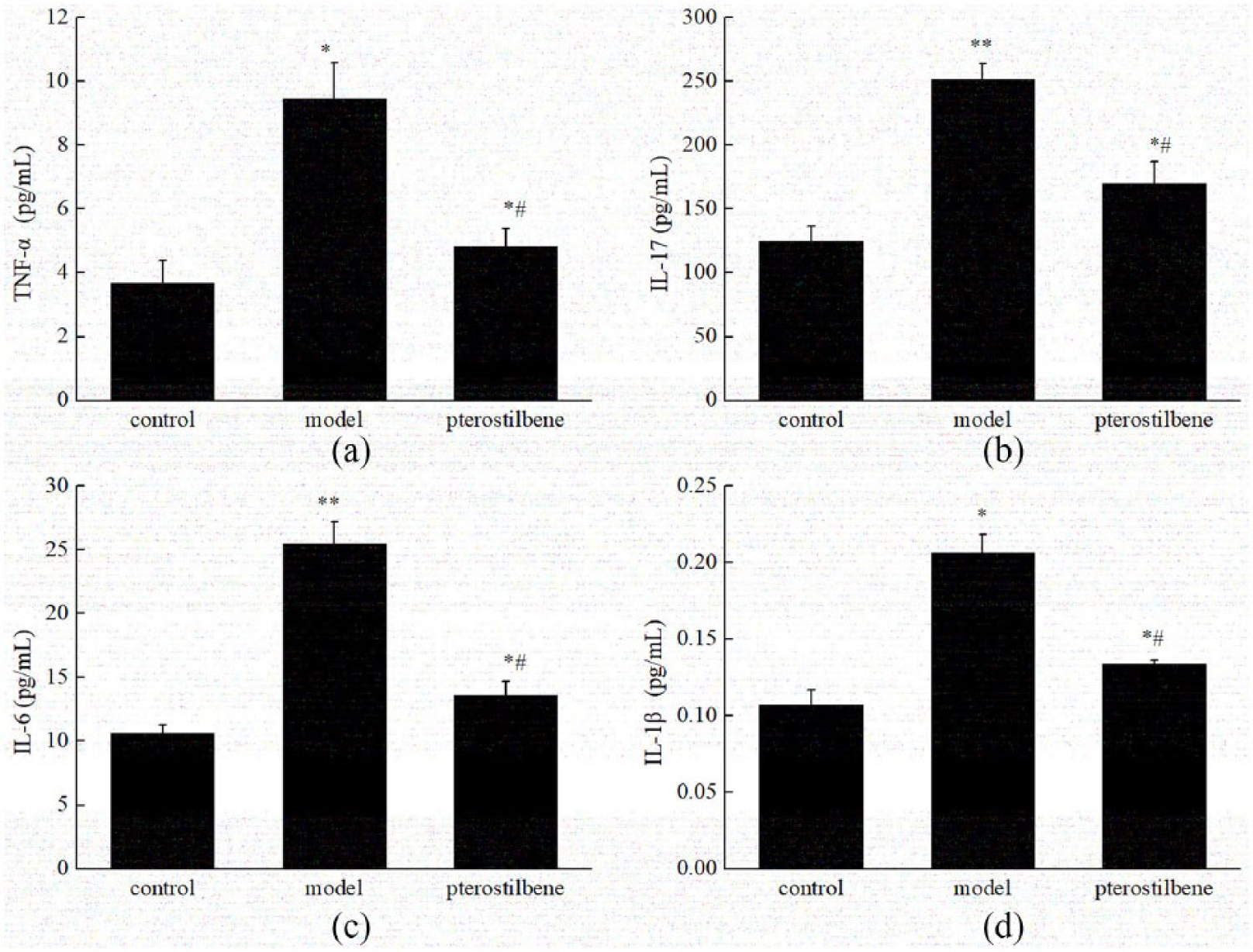
Pterostilbene decreased the expression of (a) TNF-α, (b) IL-17, (c) IL-6, and (d) IL-1β in serum. Data are presented as mean ± SD (n = 10). Compared with control group, *P < 0.05, **P < 0.01. Compared with model group, #P < 0.05.
Pterostilbene improved the oxidative stress in serum
The levels of MDA, MPO, SOD, and GSH-px in serum were measured by ELISA (Figure 3). Compared with the control group, we found that the levels of MDA and MPO in serum were remarkably increased, while the levels of SOD and GSH-px were notably decreased in model group (P < 0.05). However, the treatment of pterostilbene was obviously relieved the oxidative stress by reducing the levels of MDA and MPO, and enhancing the levels of SOD and GSH-px (P < 0.05).

Effects of pterostilbene on the expression of (a) MDA, (b) MPO, (c) SOD, and (d) GSH-px in serum. Data are presented as mean ± SD (n = 10). Compared with control group, *P < 0.05, **P < 0.01. Compared with model group, #P < 0.05, ##P < 0.01.
Histopathology changes in colon
In control group, the colonic mucosa of the rats was intact, and the glands were arranged neatly without inflammatory cell infiltration (Figure 4(a)). In model group, the colonic mucosa was severely destroyed, and the glands were disordered or absent. The cells were highly edema and hyperemia (Figure 4(a)). The damage was notably ameliorated after pterostilbene treatment (Figure 4(a)). The scores of histopathology were shown in Figure 4(b). The score was significantly higher in model group than that in control group, and the score was lower in treatment group than that in model group (P < 0.05).

Pterostilbene ameliorated the colon damage. (a) HE staining slices from colorectal sections, magnification 40×. (b) HE staining slices from colorectal sections, magnification 400×. (c) Histopathological scores. Data are presented as mean ± SD (n = 6). Compared with control group, *P < 0.05, **P < 0.01. Compared with model group, #P < 0.05, ##P < 0.01.
Pterostilbene inhibited the expression of NF-κB and activated the expression of PPAR-γ
The expressions of NF-κB and PPAR-γ were measured in colon by immunohistochemical analysis (Figure 5). The expression of NF-κB in colon tissue of control group was almost not expressed, but the positive result of model group was obvious. The positive expression of NF-κB in the treatment group was significantly lower than that in model group (P < 0.05). The positive expression of PPAR-γ was obvious in control group but decreased in model group (P < 0.05). After treatment of pterostilbene, the expression of PPAR-γ was markedly increased (P < 0.05). The expressions trend of ZO-1 and Occludin were consistent with that of PPAR-γ (Figure 6). Those levels were significantly reduced in model group compared with the control group, and the expression was increased in treatment group (P < 0.05).

Effects of pterostilbene on the expression of NF-κB and PPAR-γ in colon. The method is immunohistochemical staining (magnification 40×), and integrated optical density (IOD) is analyzed by Image-Pro Plus software. Data are presented as mean ± SD (n = 6). Compared with control group, *P < 0.05, **P < 0.01. Compared with model group, #P < 0.05, ##P < 0.01.

Effects of pterostilbene on the expression of Occludin and ZO-1 in colon. The method is immunohistochemical staining (magnification 40×), and integrated optical density (IOD) is analyzed by Image-Pro Plus software. Data are presented as mean ± SD (n = 6). Compared with control group, *P < 0.05, **P < 0.01. Compared with model group, #P < 0.05, ##P < 0.01.
Discussion
A lot of previous reports have demonstrated the effect of pterostilbene on different diseases through regulating NF-κB. For example, pterostilbene inhibited the high-fat-induced atherosclerosis inflammation via NF-κB signaling pathway. 11 To explore the effect of pterostilbene on the expression of NF-κB in TNBS-induced colitis rats, we made this research. Consistent with the previous study, we found that pterostilbene decreased the expression of NF-κB in colitis. As we all know, NF-κB represents a critical role in the regulation of inflammatory expression by activating the transcriptional capacity. 17 The inflammatory cytokines play major roles during the infection, host defense, and inflammation pathological development, 12 , 13 which induce the phosphorylation of IκB by IκB kinase (Iκκ) that allows NF-κB translocation into the cell nucleus, activating transcription of genes encoding proteins involved in inflammation responses. 18 Many researches showed that pterostilbene depressed inflammatory response via regulating the NF-κB pathway. 11 , 15 In our study, we also found pterostilbene could decrease the secretions of pro-inflammatory molecules, including TNF-α, IL-17, IL-6, and IL-β, which provided a proof that pterostilbene inhibited the inflammation in colitis rats through NF-κB pathway.
As is known to us, the pro-inflammatory cytokines cause oxidative stress by promoting the release of ROS from cells. 19 SOD and GSH are crucial antioxidant enzymes for preventing the formation of free radicals. 1 MDA and MPO reflected the oxidative stress in renal failure, inflammatory vascular disease. 1 , 20 , 21 In order to investigate the role of pterostilbene on oxidative stress in colitis, we detected the levels of SOD, GSH, MDA, and MPO. The results showed pterostilbene treatment significantly decreased the levels of SOD and GSH, while obviously increased the levels of MDA and MPO. The results were similar to these studies that pterostilbene could improve the levels of oxidative stress. 12 , 13
PPAR-γ plays an important role in anti-inflammatory reaction. It has been reported that PPAR-γ could inhibit the release of TNF-α and IL-6. 6 Previous studies have reported that PPAR-γ could inhibit the expression of NF-κB phosphorylation in cells. 20 In our study, consistent with the report, the expression of PPAR-γ declined in colitis tissue. The proteins of Occludin and ZO-1 are essential for the control of endothelial permeability, the main proteins involved in the tight junction complex. 22 The expressions of Occludin and ZO-1 were found to take part in the intestinal barrier function. 23 Our results indicated that levels of Occludin and ZO-1 were decreased in colitis tissue, which indicated that the intestinal barrier function was destroyed. The treatment of pterostilbene ameliorated intestinal epithelial barrier dysfunction in TNBS-treated rats.
In summary, pterostilbene not only inhibited the ROS production but also enhanced the antioxidant ability. These effects may be through mediating NF-κB pathway and PPAR-γ. Our research lays a foundation for the development of pterostilbene as a potential candidate for treating UC. However, safety assessment in humans needs to be conducted, and optimization of treatment protocol of pterostilbene in clinical trials needs further research.
These results demonstrated that pterostilbene treatment effectively ameliorated TNBS-induced colitis in rats. The mechanism may be connected with the suppression of NF-κB and the activation of PPAR-γ. The study indicates that pterostilbene is a promising therapeutic drug for UC.
Footnotes
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
