Abstract
Background:
Inflammatory bowel disease is a chronic immunoinflammatory disease of the gastrointestinal tract. Piperine, an alkaloid, has been reported to possess antioxidant, anti-inflammatory, antiapoptotic, and antiulcer potential.
Aim:
To elucidate the plausible mechanisms of action of piperine on experimental trinitrobenzenesufonic acid (TNBS)-induced colitis by assessing various biochemical, molecular, histological, and ultrastructural modifications.
Methods:
Colitis was induced in male Sprague–Dawley rats via intrarectal instillation of TNBS. Then, the rats were treated with piperine (10, 20, and 40 mg/kg, p.o.) for 14 days.
Results:
TNBS induced significant (p < 0.05) colonic damage, which was assessed by disease activity index, macroscopic score, and stool consistency. The administration of piperine (20 and 40 mg/kg) significantly inhibited (p < 0.05) these damages. Treatments with piperine (20 and 40 mg/kg) notably inhibited (p < 0.05) the TNBS-induced elevation of oxido-nitrosative stress (superoxide dismutase, glutathione, malondialdehyde, and nitric oxide), 5-hydroxytryptamine, and hydroxyproline content in the colon. Furthermore, colonic inducible nitric oxide synthase (iNOs), tumor necrosis factor-alpha (TNF-α), interleukin (IL)-1β, IL-6, interferon-gamma, and cyclooxygenase-2 (COX-2) messenger RNA (mRNA) expressions were upregulated after TNBS instillation and piperine (20 and 40 mg/kg) significantly attenuated (p < 0.05) these elevated mRNA expressions. TNBS decreased the expressions of tight junction (TJ) protein (claudin-1, occludin, and zonula occludens-1 (ZO-1)) and increased the expressions of proapoptotic (caspase-1) protein. These expressions were markedly inhibited (p < 0.05) by piperine treatment. Histological and ultrastructural studies of transmission electron microscopy suggested that piperine significantly ameliorated (p < 0.05) TNBS-induced colonic aberrations.
Conclusion:
Piperine ameliorated the progression of TNBS-induced colitis by modulating the nuclear factor of kappa light polypeptide gene enhancer in B-cells inhibitor-alpha/nuclear factor-kappa B signaling pathway, thus inhibiting the overexpression of proinflammatory cytokines (TNF-α and IL’s), COX-2, iNOs, oxido-nitrosative stress, and proapoptotic proteins (caspase-1) that may improve the expression of TJ protein (claudin-1, occludin, and ZO-1).
Introduction
Inflammatory bowel disease (IBD) is a complex, chronic, and relapsing immunoinflammatory disease characterized by gastrointestinal inflammation, unintentional weight loss, fatigue, reduced appetite, diarrhea, ulceration in the mucosal membrane, mucous stool, rectal bleeding, and abdominal cramping. 1 Idiopathic IBD exhibits in two major forms, which include Crohn’s disease and ulcerative colitis (UC). 2 In UC, the inflammatory ulcerations are only associated with the colon and the rectum. The pathogenesis of UC involves various genetic and environmental factors, such as alterations in the immune system or in the intestinal microbial flora. 3 UC not only negatively affects the quality of life of the patient but also increases colon cancer risk. Therefore, it is one of the major public health concerns that commonly affect individuals between 15 years and 35 years of age. A recent study suggested that in UC patients, the cumulative risk of colon cancer can increase from 8% to 18%. 4
Even though the pathogenesis of IBD still remains unclear, a number of evidences suggest that elevated levels of reactive oxygen species (ROS) in immune cells generate oxidative stress, which can be responsible for the initiation and the progression of UC. 5 –7 Increased oxidative stress and influx of inflammatory response cause alterations in permeability of intestinal mucosal barrier via the destruction of tight junctions (TJs). 8 Inflammatory cytokines, including tumor necrosis factor-alpha (TNF-α) and interleukin’s (IL’s), recruit and activate neutrophils and macrophages, which, in turn, elevate the adaptive immune response. 9 Additionally, overproduction of reactive nitrogen species (RNS) such as nitric oxide (NO) by inducible nitric oxide synthase (iNOs) in chronic colitis acts in synergy with cyclooxygenase-2 (COX-2) to induce cellular degeneration in the inflamed tissue. 10 –12
The current treatment regimen for UC includes anti-inflammatory agents such as 5-aminosalicylates (sulfasalazine and mesalamine) and corticosteroids (prednisone and hydrocortisone) and immunosuppressant such as azathioprine, mercaptopurine, cyclosporine, and infliximab. 13 These agents focus on the inhibition of the exacerbated immunoinflammatory response and long-term association of such drugs may result in significant adverse effects such as nausea, headache, abdominal pain, diarrhea, acute pancreatitis, and renal failure. Because of the immense expense of the regimen and its undesirable side effects, the clinical implication of such agents in the treatment of UC is limited. Despite significant progress in research, there is still a need for safer and more efficient therapeutic agents for IBD management. Various animal models have been developed to evaluate new therapeutic moieties from natural origin for the treatment of IBD. Intrarectal instillation of 2,4,6-trinitrobenzenesufonic acid (TNBS) is a well-established and widely used method of inducing colitis in rat models. It effectively mimics most of the clinicopathological features of human UC, including ulcerations, necrosis, and thickening of the bowel wall. 14 –16 The researchers reported that TNBS acts by interacting with high molecular weight tissue proteins, which activate Th1 and Th2 inflammatory responses, causing the release of various proinflammatory cytokines (TNF-α and ILs). 17
Recently, much attention has been focused on the application of functional foods in various disease managements. 18 Black pepper (Piper nigrum) is widely used as a functional food and has a broad range of applications, such as food additive, cosmetic, and traditional medicine. Piperine (1-(5-[1,3-benzodioxol-5-yl]-1-oxo-2,4-pentadienyl) piperidine) is a naturally occurring alkaloid present abundantly in black pepper, thus becoming an integral part of the human daily diet. 19 Literature suggests that piperine is associated with an array of beneficial effects in the prevention and treatment of various diseases including airways disorders, gastric and duodenal ulcer, and mood and cognitive disorders. 19 Previous studies have also reported that piperine has numerous biomedical properties, exhibiting antioxidative, anti-inflammatory, antiasthmatic, anti-acute pancreatitic, anti-arthritic, antihyperlipidemic, anticancer, antidiabetic, and antiulcer potentials. 20 It has been demonstrated that piperine exerts its antiulcer potentials by reducing oxidative stress, NO, and TNF-α along with toll-like receptor 4 in mice. 21 Furthermore, Liang et al. reported that piperine inhibited bacterial infection via inhibition of IL-1β levels, exerting its antidiarrheal effect. 22 Furthermore, it has been reported that piperine exerts its anti-inflammatory effects via inhibition of COX-2 and nuclear factor-kappa B (NF-κB) in murine macrophages. 23 However, no studies have evaluated its possible role in TNBS-induced UC yet. This study aims to elucidate the plausible mechanisms of action of piperine on experimental TNBS-induced colitis by assessing various biochemical, molecular, histological, and ultrastructural modification.
Materials and method
Animals
The care and treatments of the Sprague–Dawley rats (180–200 g) were completed based on the procedures recommended by the International Animal Ethics Committee. The animals were obtained from Xi’an Jiaotong University Animal Center and were maintained at 24 ± 1°C, with a relative humidity of 45–55% and 12-h dark/12-h light cycle. The animals had free access to standard pellet chow and water throughout the experimental protocol, and they were maintained in pathogen-free conditions. All experiments were carried out between 09.00 h and 17.00 h. This study was conducted strictly according to the recommendations outlined in the Guide for the Care and Use of Laboratory Animals of the National Institutes of Health and the ARRIVE (Animal Research: Reporting of In Vivo Experiments) guidelines (http://www.nc3rs.org/ARRIVE). All experimental protocols were approved by the First Affiliated Hospital, Xi’an Jiaotong University (China, Protocol: XJTULAC201811). All animals involved in this experiment were treated in accordance with the guidelines, and they were approved by the animal care and use committee of the First Affiliated Hospital, Xi’an Jiaotong University.
Chemical and kits
Piperine (purity ≥97%, CAS No.: 94-62-2, synonyms: (E,E)-5-(3,4-methylenedioxyphenyl)-2,4-pentadienoylpiperidide, and 1-piperoylpiperidine) and TNBS were purchased from Sigma Chemical Co. (St Louis, Missouri, USA). Sulfasalazine was obtained as a gift sample from Symed Pharmaceutical Pvt. Ltd., Hyderabad, Telangana, India. Total RNA extraction kit and one-step reverse transcription-polymerase chain reaction (RT-PCR) kit were purchased from MP Biomedicals India Private Limited, Mumbai, Maharashtra, India. The primary antibodies of claudin-1, occludin, caspase-1, zonula occludens-1 (ZO-1 or TJ protein-1), phospho NF-κB (p-NF-κB), and nuclear factor of kappa light polypeptide gene enhancer in B-cells inhibitor-alpha (p-IκBα) were purchased from Abcam, Cambridge, Massachusetts, USA.
Induction of colitis and drug treatment schedule
Colitis was induced according to the procedure described by Li et al.
24
Rats were lightly anesthetized with ether following a 24 h fast, and then TNBS, dissolved in 50% ethanol, was instilled into the colon of the animals (100 mg in a volume of 0.25 mL) using a medical-grade polyurethane catheter for enteral feeding (external diameter 2 mm). It is inserted 8 cm into the anus. Rats in the normal group received an injection of physiological saline instead of the TNBS solution. Rats were randomly allocated into the following groups with 18 rats in each group:
Group I: Normal animals: Received 0.25 mL (once, intrarectally) of physiological saline and 1 mL of 1% aqueous dimethyl sulfoxide (DMSO) solution for 14 days.
Group II: TNBS control animals: Received 0.25 mL (once, intrarectally) of TNBS (100 mg/kg) and 1 ml of 1% aqueous DMSO solution for 14 days.
Group III: 5-Aminosalicylic acid (5-ASA) (500 mg/kg)-treated animals: Received 0.25 mL (once, intrarectally) of TNBS (100 mg/kg) and were treated with 5-ASA (500 mg/kg, p.o.) for 14 days.
Group IV: Piperine (10 mg/kg)-treated animals: Received 0.25 mL (once, intrarectally) of TNBS (100 mg/kg) and were treated with piperine (10 mg/kg, p.o.) for 14 days.
Group V: Piperine (20 mg/kg)-treated animals: Received 0.25 mL (once, intrarectally) of TNBS (100 mg/kg) and were treated with piperine (20 mg/kg, p.o.) for 14 days.
Group VI: Piperine (40 mg/kg)-treated animals: Received 0.25 mL (once, intrarectally) of TNBS (100 mg/kg) and were treated with piperine (40 mg/kg, p.o.) for 14 days.
Group VII: Per se animals: Received ethanol (50%) in a volume of 0.25 mL (once, intrarectally) and were treated with piperine (40 mg/kg, p.o.) for 14 days.
Piperine was administered to rats in three different dosages (10, 20, and 40 mg/kg) for 14 days, whereas 5-ASA was given in a dose of 500 mg/kg/day orally to the rats. 25,26 On the 15th day, rats were anesthetized with ether for about 30–40 s. Blood was drawn using a retro-orbital puncture. Each blood sample was collected into a separate vial to determine serum parameters. After blood sample collection, the animals were killed through cervical dislocation. The colon was excised and colonic damage was assessed and frozen immediately in liquid nitrogen and stored at −80°C for further biochemical, histopathological, and ultrastructural studies.
Assessment of colonic damage, ulcer area, and ulcer index
The severity of colitis was evaluated by an independent observer who was blinded from the treatments. For each animal, a distal 10 cm portion of the colon was removed, cut longitudinally, briefly cleaned in physiological saline to remove fecal residues, and weighed. Macroscopic inflammation scores were assigned based on clinical features of the colon. The presence of disease activity index (DAI), macroscopic score, and stool consistency (score 0–4) were evaluated according to the previously reported methods. 1,16 Ulcer area and ulcer index were evaluated according to previously reported methods. 1,16
Biochemical assays
For colon homogenization, tissue segments were mixed with 0.1 M phosphate buffer and homogenized on ice for 60 s at 10,000 r/min in a homogenizer (Remi Equipment Pvt Ltd, Remi Motors Ltd, Mumbai, Maharashtra, India). The supernatant of tissue homogenates was employed to estimate superoxide dismutase (SOD), reduced glutathione (GSH), lipid peroxidation (malondialdehyde (MDA) content), and NO (NO content) as described previously. 1,16 The colonic 5-hydroxytryptamine (5-HT) and hydroxyproline content were determined according to a previously described method. 1,16
Determination of colonic iNOs, TNF-α, IL-1β, IL-6, IL-10, IFN-γ, COX-2, and LTB4 mRNA expressions by RT-PCR
The levels of colonic iNOs, TNF-α, IL-1β, IL-6, IL-10, interferon gamma (IFN-γ), COX-2, and leukotriene B4 (LTB4) messenger RNA (mRNA) expressions were analyzed in colon tissue using quantitative RT–PCR following the manufacturer’s instructions. PCR was performed using 1× forward and reverse primers and 2.5 U Taq polymerase (MP Biomedicals India Private Limited). Amplification of β-actin served as a control for sample loading and integrity.
Western blot procedure
Colon tissue was dissected and sonicated in Tissue Protein Extraction Reagent (Thermo Fisher Scientific, New York, USA). The lysates were centrifuged at 10,000 × g for 10 min at 4°C. Protein concentrations were determined using a bicinchoninic acid assay kit (Beyotime Shanghai, China) on ice for 30 min. Equal amount of extracted protein samples (50 μg) were separated by 10% sodium dodecyl sulfate-polyacrylamide gel electrophoresis and transferred onto polyvinylidene difluoride membranes. The membranes were blocked with 5% nonfat dry milk at 37°C for 1 h and incubated overnight at 4°C with the primary antibodies that recognized claudin-1, occludin, caspase-1, ZO-1, p-NF-κB and p-IκBα, and glyceraldehyde 3-phosphate dehydrogenase (GAPDH). Anti-rabbit horseradish-linked IgG was used as the secondary antibody, incubated for 37°C for 2 h. Protein bands were visualized using the Chemiluminescent kit (Bio-Rad Laboratories, Camarillo, CA). GAPDH served as the loading control.
Histological examination
The histological examination was performed using light compound microscope with Zeiss intravital microscopy setup (Zeiss Axioscope A1, Carl Zeiss MicroImaging, Jena, Germany). The dissected colon tissue specimens were fixed in 10% formaldehyde, processed routinely for paraffin embedding. Histopathological analysis of hematoxylin and eosin stain was carried out on day 15 and each successive field was individually assessed for the severity of interstitial fibrosis using the semiquantitative grading system as described previously. 27 Microscopic scoring was performed by an experienced histologist, blinded of the treatment groups.
Electron microscopic analysis
For ultrastructural studies, the colon samples were fixed with 2.5% glutaraldehyde in 0.1 M phosphate buffer, pH 7.4, for 18 h on day 28. The colon was dissected into small pieces and postfixed for 1.5 h in 1% osmium tetroxide dissolved in 0.1 M phosphate buffer (pH 7.4), then dehydrated through a series of graded ethanol solutions and embedded in Araldite (epoxy resin). Ultrathin sections were cut, stained with uranyl acetate and lead nitrate, mounted on copper grids, and examined under a transmission electron microscope (H-7000 Hitachi, Tokyo, Japan).
Statistical analysis
Data are expressed as mean ± standard error mean. Data analysis was performed using GraphPad Prism 5.0 software (GraphPad, San Diego, California, USA). Data were analyzed using one-way analysis of variance (ANOVA) followed by Tukey’s multiple range post hoc analysis. Data from macroscopical score, stool consistency, and histological score were analyzed using nonparametric one-way ANOVA followed by Kruskal–Wallis test for post hoc analysis. A value of p < 0.05 was considered to be statistically significant.
Results
Effect of piperine on TNBS-induced alterations in body weight, colon weight to length ratio, ulcer area, and ulcer index of rats
There were no significant changes in body weight, colon weight to length ratio, ulcer area, and ulcer index of normal rats as compared to per se treated rats. Intrarectal instillation of TNBS resulted in a significant decrease (p < 0.05) of body weight and a significant increase (p < 0.05) in colon weight to length ratio, ulcer area, and ulcer index in TNBS control rats in comparison to normal rats. Administration of 5-ASA (500 mg) significantly ameliorated (p < 0.05) TNBS induced alterations in body weight, colon weight to length ratio, ulcer area, and ulcer index in comparison to TNBS control rats. Treatments with piperine (20 and 40 mg/kg) also significantly increased (p < 0.05) body weight and significantly decreased (p < 0.05) colon weight to length ratio, ulcer area, and ulcer index in comparison to TNBS control rats. However, TNBS induced colon weight to length ratio and ulcer index increase were more significantly attenuated (p < 0.05) by 5-ASA (500 mg) in comparison to piperine treatment. The percentage inhibition of the formation of ulcer by 5-ASA (500 mg) was 87.77%, whereas piperine (10, 20, and 40 mg/kg) exhibited 11.29, 41.58, and 66.31% inhibition of ulcer formation (Table 1 and Figure 1).

Morphological representation of colons from (a) normal-, (b) TNBS control-, (c) 5-ASA (500 mg/kg), (d) piperine (20 mg/kg), (e) piperine (40 mg/kg), and (f) per se treated rats. TNBS: trinitrobenzenesufonic acid; 5-ASA: 5-aminosalicylic acid
Effect of piperine on TNBS-induced alterations in body weight, colon weight, colon weight to length ratio, ulcer area, ulcer index, DAI, macroscopic score, and stool consistency score in rats.a
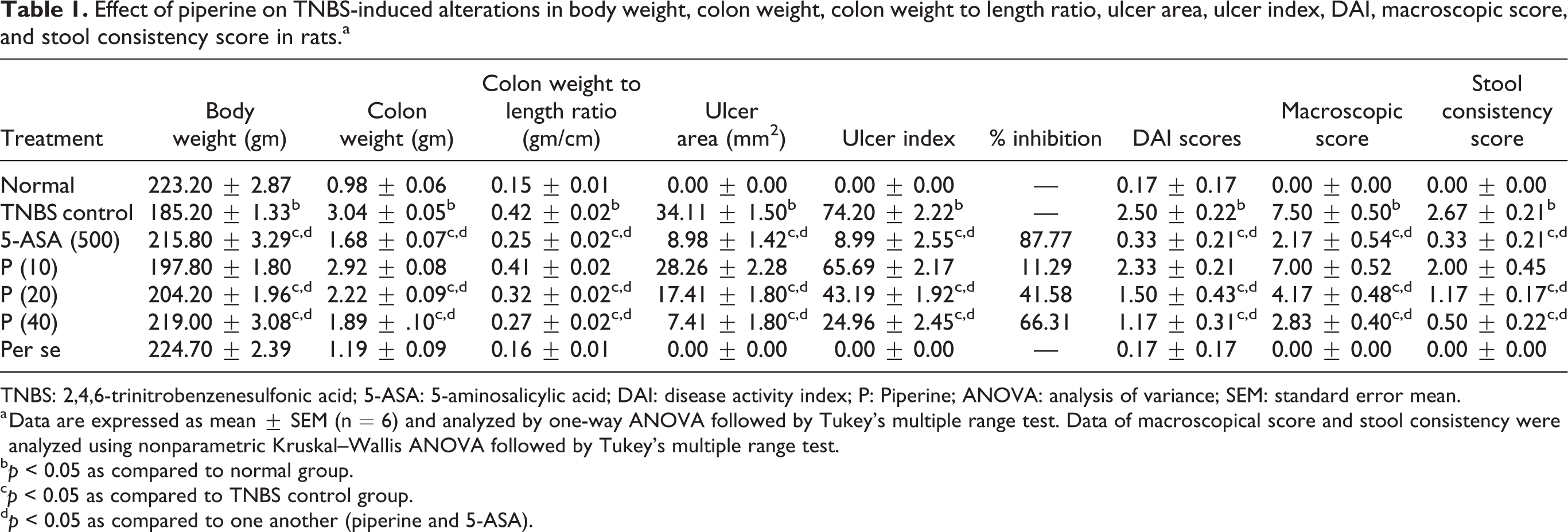
TNBS: 2,4,6-trinitrobenzenesulfonic acid; 5-ASA: 5-aminosalicylic acid; DAI: disease activity index; P: Piperine; ANOVA: analysis of variance; SEM: standard error mean.
a Data are expressed as mean ± SEM (n = 6) and analyzed by one-way ANOVA followed by Tukey’s multiple range test. Data of macroscopical score and stool consistency were analyzed using nonparametric Kruskal–Wallis ANOVA followed by Tukey’s multiple range test.
b p < 0.05 as compared to normal group.
c p < 0.05 as compared to TNBS control group.
d p < 0.05 as compared to one another (piperine and 5-ASA).
Effects of piperine on TNBS-induced alterations within the DAI, macroscopic score, and stool consistency score of rats
DAI, macroscopic score, and stool consistency score did not differ significantly in normal rats compared to that of the per se treated rats. However, DAI, macroscopic score, and stool consistency score increased significantly (p < 0.05) after intrarectal instillation of TNBS in control rats in comparison to those of the normal rats. Administration of 5-ASA (500 mg) significantly decreased (p < 0.05) these scores in comparison to that of the TNBS control rats. When compared with TNBS control rats, DAI, macroscopic score, and stool consistency score decreased significantly (p < 0.05) in piperine (20 and 40 mg/kg) treatments. However, TNBS-induced increase in DAI, macroscopic score, and stool consistency score was more significantly attenuated (p < 0.05) in 5-ASA (500 mg) in comparison to those under piperine treatment (Table 1 and Figure 1).
Effect of piperine on TNBS-induced alterations of colonic oxido-nitrosative stress of rats
Intrarectal instillation of TNBS resulted in a significant increase (p < 0.05) in oxido-nitrosative stress in colon tissues of TNBS control rats compared to that of the normal rats. However, administration of 5-ASA (500 mg) significantly decreased (p < 0.05) colonic SOD and GSH levels and it significantly increased (p < 0.05) colonic MDA and NO levels in comparison to those of the TNBS control rats. Treatment with piperine (20 and 40 mg/kg) also significantly attenuated (p < 0.05) colonic oxido-nitrosative stress in comparison to that of the TNBS control rats. TNBS-induced elevation in oxido-nitrosative stress in the colon was more significantly decreased (p < 0.05) in 5-ASA (500 mg) treatment in comparison to those under piperine treatment. There was no significant change in colonic SOD, GSH, MDA, and NO levels of normal rats as compared to those of the per se treated rats (Table 2).
Effect of piperine on TNBS-induced alterations in colonic SOD, GSH, MDA, NO, hydroxyproline, and 5-HT in rats.a

TNBS: 2,4,6-trinitrobenzenesulfonic acid; 5-ASA: 5-aminosalicylic acid; P: piperine; SOD: superoxide dismutase; GSH: glutathione; MDA: malondialdehyde; NO: nitric oxide; HP: hydroxyproline and 5-HT: 5-hydroxytryptamine; ANOVA: analysis of variance.
a Data are expressed as mean ± SEM (n = 6) and analyzed by one-way ANOVA followed by Tukey’s multiple range test.
b p < 0.05 as compared to normal group.
c p < 0.05 as compared to TNBS control group.
d p < 0.05 as compared to one another (piperine and 5-ASA).
Effect of piperine on TNBS-induced alterations of colonic hydroxyproline and 5-hydroxytryptamine in rats
Intrarectal instillation of TNBS resulted in a significant increase (p < 0.05) in colonic HP and 5-HT in TNBS control rats in comparison to those of the normal rats. The administration of piperine (20 and 40 mg/kg) also significantly decreased (p < 0.05) colonic HP and 5-HT levels when compared to those of the TNBS control rats. However, treatment with 5-ASA (500 mg) failed to produce any significant reductions in TNBS-induced increase in HP and 5-HT levels in the colon in comparison to that of the TNBS control rats (Table 2).
Effect of piperine on TNBS-induced alterations of colonic TNF-α, IL-1β, IL-6, and IL-10 mRNA expressions of rats
There were no significant differences in colonic TNF-α, IL-1β, IL-6, and IL-10 mRNA expression within normal rats compared to those of the per se treated rats. However, intrarectal instillation of TNBS resulted in significant upregulation (p < 0.05) of colonic TNF-α, IL-1β, and IL-6. It also significantly downregulated (p < 0.05) colonic IL-10 mRNA expression in TNBS control rats in comparison to those of the normal rats. The administration of 5-ASA (500 mg) significantly inhibited (p < 0.05) TNBS-induced alterations of colonic TNF-α, IL-1β, IL-6, and IL-10 mRNA expressions in comparison to those of the TNBS control rats. Treatments with piperine (20 and 40 mg/kg) also significantly downregulated (p < 0.05) colonic TNF-α, IL-1β, and IL-6. It significantly upregulated (p < 0.05) colonic IL-10 mRNA expression in comparison to those of the TNBS control rats (Table 3).
Effect of piperine on TNBS-induced alterations in colonic iNOs, TNF-α, IL-1β, IL-6, IL-10, IFN-γ, COX-2, and LTB4 mRNA expressions in rats.a

TNBS: 2,4,6-trinitrobenzenesulfonic acid; 5-ASA: 5-aminosalicylic acid; P: piperine; iNOs: inducible nitric oxide synthase, TNF-α: tumor necrosis factor-alpha, IL’s: interleukins, IFN-γ: interferon-gamma, COX-2: cyclooxygenase-2 and LTB4: leukotriene B4.
a Data are expressed as mean ± SEM (n = 6) and analyzed by one-way ANOVA followed by Tukey’s multiple range test.
b p < 0.05 as compared to normal group.
c p < 0.05 as compared to TNBS control group.
d p < 0.05 as compared to one another (piperine and 5-ASA).
Effects of piperine on TNBS-induced alterations of colonic iNOs, IFN-γ, COX-2, and LTB4 mRNA expressions in rats
Colonic iNOs, IFN-γ, COX-2, and LTB4 mRNA expressions were significantly upregulated (p < 0.05) after the administration of TNBS in control rats in comparison to those of the normal rats. However, treatments with piperine (20 and 40 mg/kg) significantly inhibited (p < 0.05) TNBS-induced upregulation of colonic iNOs, IFN-γ, and COX-2 mRNA in comparison to those of the TNBS control rats. Administration of 5-ASA (500 mg) significantly downregulated (p < 0.05) iNOs and COX-2 in comparison to those within the TNBS control rats. However, when compared with those of the TNBS control rats, upregulation of LTB4 mRNA expression after intrarectal TNBS instillation was not significantly attenuated by 5-ASA (500 mg) or piperine treatment. 5-ASA (500 mg) also did not produce any significant downregulation in IFN-γ mRNA expression in comparison to that within the TNBS control rats (Table 3).
Effect of piperine on TNBS-induced alterations of colonic claudin-1, occludin, and ZO-1 protein expression in rats
There were no significant differences in colonic claudin-1, occludin, and ZO-1 protein expressions between normal rats and per se treated rats. Intrarectal instillation of TNBS resulted in significant downregulation (p < 0.05) of colonic claudin-1, occludin, and ZO-1 protein expressions in TNBS control rats in comparison to those of the normal rats. The administration of 5-ASA (500 mg) significantly inhibited (p < 0.05) TNBS-induced downregulation of colonic claudin-1, occludin, and ZO-1 protein expression in comparison to those in the TNBS control rats. Treatment with piperine (20 and 40 mg/kg) also significantly upregulated (p < 0.05) colonic claudin-1, occludin, and ZO-1 protein expression compared to those in the TNBS control rats (Figure 2).

Effect of piperine on TNBS-induced alterations in colonic (a) claudin-1, (b) occluding, (c) ZO-1, (b) caspase-1, (e) NF-κB, and (f) IκBα protein expression in rats. Data are expressed as mean ± SEM (n = 6) and analyzed by one-way ANOVA followed by Tukey’s multiple range test. *p < 0.05 as compared to TNBS control group, # p < 0.05 as compared to normal group, and $ p < 0.05 as compared to one another (piperine and 5-ASA). Representative protein expression of normal (lane 1), TNBS control (lane 2), 5-ASA (500 mg/kg) (lane 3), piperine (10 mg/kg) (lane 4), piperine (20 mg/kg) (lane 5), piperine (40 mg/kg) (lane 6), and per se (lane 7) treated rats. TNBS: 2,4,6-trinitrobenzenesulfonic acid; 5-ASA: 5-aminosalicylic acid; P: piperine; ZO-1: zonula occludens-1 or TJ protein-1, p-NF-κB: phospho nuclear factor kappa B; p-IκBα: nuclear factor of kappa light polypeptide gene enhancer in B-cells inhibitor-alpha; GAPDH: dlyceraldehyde 3-phosphate dehydrogenase; ANOVA: analysis of variance; SEM: standard error mean; TJ: tight junction.
Effect of piperine on TNBS-induced alterations of colonic caspase-1, NF-κB, and IκBα protein expression in rats
Intrarectal instillation of TNBS resulted in significant upregulation (p < 0.05) of colonic caspase-1, NF-κB, and IκBα protein expression in TNBS control rats when compared to those in normal rats. TNBS-induced upregulation of caspase-1, NF-κB, and IκBα protein expression in the colon was significantly downregulated (p < 0.05) by the administration of 5-ASA (500 mg) in comparison to those in the TNBS control rats. Treatment with piperine (20 and 40 mg/kg) also significantly attenuated (p < 0.05) the upregulated colonic caspase-1, NF-κB, and IκBα protein expression in comparison to those in the TNBS control rats. There was no significant difference in colonic caspase-1, NF-κB, and IκBα protein expression in normal and per se treated rats. (Figure 2).
Effects of piperine on TNBS-induced alterations of histopathology in colon tissues of rats
Figure 3(a) and (f) depicts the normal structure of colon tissue, showing the presence of mild inflammatory cells with edema. Intrarectal instillation of TNBS resulted in a significant increase (p < 0.05) in inflammatory infiltration, adhesion score, wall thickening, and destruction score within TNBS control rats (Figure 3(b)) in comparison to those within the normal rats. However, administration of 5-ASA (500 mg) significantly attenuated (p < 0.05) TNBS-induced histological alterations (Figure 3(c)) when compared with TNBS control rats. In comparison to the TNBS control rats, the administration of piperine (20 and 40 mg/kg) also significantly decreased (p < 0.05) inflammatory infiltration, adhesion score, wall thickening, and destruction score (Figure 3(d), (e), and (g)).

Effect of piperine on TNBS-induced alterations in colon histopathology. Photomicrograph of sections of colon tissue from (a) normal, (b) TNBS control, (c) 5-ASA (500 mg/kg), (d) piperine (20 mg/kg), (e) piperine (40 mg/kg), and (f) per se treated rats stained with H&E stain. (g) The quantitative representation of histological score. Data are expressed as mean ± SEM (n = 3) and analyzed by one-way ANOVA followed by Kruskal–Wallis test was applied for post hoc analysis. *p < 0.05 as compared to TNBS control group, # p < 0.05 as compared to normal group, and $ p < 0.05 as compared to one another (piperine and 5-ASA). TNBS: 2,4,6-trinitrobenzenesulfonic acid; 5-ASA: 5-aminosalicylic acid; P: piperine; ANOVA: analysis of variance; SEM: standard error mean; H&E: hematoxylin and eosin. Red arrow indicated inflammatory infiltration and yellow arrow indicated necrosis. Images (×40 magnification) are typical and are representative of each study group.
Effect of piperine on TNBS-induced alterations of colonic ultrastructural changes in rats
Transmission electron microscopic examination of the epithelium from normal and per se treated colon tissue showed intact brush border with closed TJs between adjacent enterocytes (Figure 4(a) and (f)). It showed the presence of plentiful mitochondria with regular oval nuclei and numerous microvilli. However, intrarectal TNBS instillation results in increased distance between adjacent enterocytes due to increased deposition of the extracellular matrix, resulting in loosening and abnormal cell junctions (Figure 4(b)). In the colon tissue of TNBS control rats, the mitochondria were swollen, nuclei were elongated, and few microvilli were present. Ultrathin sections of colon tissue from 5-ASA- and piperine-treated (20 and 40 mg/kg) groups showed normal cell junction with the presence of numerous microvilli on the luminal surface of columnar cells (Figure 4(c) to (e)). Mitochondria and endoplasmic reticulum with basal nuclei were normal. These findings suggest that the protective potential against TNBS-induced UC can be achieved with 500 mg/kg of 5-ASA, whereas piperine requires only 20 and 40 mg/kg.

Effect of piperine on TNBS-induced alterations in alterations in colonic ultrastructure of rats (n = 3). Photomicrographs of sections of colon tissue from (a) normal (×12,530), (b) TNBS control (×12,530), (c) 5-ASA (500 mg/kg) (×14,320) (d) piperine (20 mg/kg) (×12,530), (e) piperine (40 mg/kg) (×21,480), and (f) per se (×21,480) treated rats. TNBS: 2,4,6-trinitrobenzenesulfonic acid; 5-ASA: 5-aminosalicylic acid.
Discussion
UC is a chronic relapsing disorder of the gastrointestinal system, mainly characterized by the presence of inflammation and ulceration in the colonic mucosa. 28 In this present investigation, we administered TNBS intrarectally in rats, which was able to exhibit significant similarities with human UC, manifesting cachexia, inflammatory influx, and ulceration. 29 Our study reveals that oral administration of piperine significantly ameliorated TNBS-induced UC by inhibiting several mechanisms such as elevation in oxidative stress (SOD, GSH, MDA, and NO), release of inflammatory mediator (COX-2) and proinflammatory cytokines (TNF-α, ILs, IFN-γ, NF-κB, and p-IκBα) as well as apoptosis-related factors (caspase-1). This prevents the destruction of colonic TJ proteins (claudin-1, occluding, and ZO-1), restoring the integrity of the intestinal barrier.
Changes in body weight are strongly associated with health status. The severity and extent of the inflammatory response can be reflected from the wet weight of the inflamed colon. In this investigation, intrarectal administration of TNBS caused elevated immune inflammatory response, shown as decreased body weight but an increase in colon wet weight. Administration of piperine significantly inhibited alterations of body weight and colon weight originally induced by TNBS, which may be caused by its modulatory potential in immune inflammation. Furthermore, DAI, macroscopic score, and stool consistency are indirect markers of UC. 29 Rats treated with piperine showed decreased ulcer area and ulcer index, showing piperine’s healing potential. Results of the present investigation corroborate with the findings of previous researchers where administration of plant alkaloids such as piperine ameliorates chemical-induced ulcers. 26
Numerous researchers have shown that NF-κB plays a significant role in the occurrence of UC by activating the release of proinflammatory cytokines. 30,31 In the resting cells, transcription factor NF-κB remains in inactive due to the presence of IκB-α, the inhibitory protein. However, phosphorylation of IκB-α causes its ubiquitination and degradation, which, in turn, activates NF-κB and its translocation to the nucleus via its RelA/p50 and RelA/p65 heterodimers. 30 This translocation leads to inflammation in the mucosa, cell proliferation, and transcription of various inflammatory mediators including iNOs, TNF-α, IL’s, and COX-2. Clinically, higher levels of NF-κB in epithelial cells have been associated with the inflamed colon in IBD patients. 32 Thus, most research focus on the development of therapeutic moieties with NF-κB inhibitory potential to regulate the inflammatory release for the management of IBD. In agreement with a previous researcher, 33 elevated level of phosphorylated p-NF-κB protein expression was documented in the colons from our TNBS control rats. However, results of the present investigation suggest that piperine exhibits its potential mediation via inhibition of NF-κB p65 phosphorylation, which may further inhibit the production of inflammatory cytokine and chemokine. Additionally, 5-ASA also inhibited IκB-α phosphorylation, which further downregulated NF-κB activity. The results of the present study are in line with findings of previous researchers.
5-HT (serotonin) is an important neurotransmitter that is vital in gastrointestinal signaling and in immunoinflammatory response. 34 Generally, 5-HT is stored in enterochromaffin (EC) cells and increased levels of intestinal cytokines stimulate the EC cells to release 5-HT. Released 5-HT further activates intestinal serotonergic receptors, which causes alterations in gut functions. 35 Furthermore, IBD patients suffering from diarrhea were also reported to be associated with higher levels of mucosal 5-HT, which results in visceral hypersensitivity and altered gastrointestinal motility. 35 Therefore, these evidences suggest that 5-HT is crucial in the pathogenesis of IBD. 34,35 In the present investigation, TNBS-induced colitis rats exhibited enhanced cytokine secretions with an increased serotonin secretion in the inflamed colon which may be resulted from the perturbation of the mast cells. Wu et al. reported that piperine ameliorates stress-induced irritable bowel syndrome by inhibiting the elevated expression of colonic 5-HT and findings of the present investigation are in accordance. 36
Researchers have demonstrated that COX-2 and iNOs are other important inflammatory mediators that play central roles in the progress of intestinal damage. 23,33 In IBD, overproduction of RNS such as NO causes the upregulation of iNOs expression in the intestinal mucosa, which, in turn, causes alternations of intestinal hypomotility and tissue integrity. 37 During the early phase of proinflammatory response, COX-2 has been reported to express explicitly as a response to cytokines such as TNF-α. Researchers also suggested that the role of COX-2 in various cellular mechanisms includes angiogenesis, proliferation, and apoptosis. 38 –40 Thus, the synergic action of iNOs with COX-2 further promotes an inflammatory response. In agreement with previous studies, results of the present investigation showed that piperine treatment significantly decreased the elevated expression of iNOs and COX-2 after TNBS administration, thus diminishing the inflammatory damage. 41
Claudin-1, occludin, and ZO-1 are important TJ proteins involved in processes such as TJ permeability proliferation and differentiation in the intestinal epithelium. 8 The epithelium plays a vital role in the inhibition of intestinal inflammation and the maintenance of its health. Recent studies suggest that the tightness of TJ is depended on the number, composition, and mixing ratio of claudins, whereas occludin plays a significant role in the assembly and maintenance of TJ. 42,43 Structural modification to these colonic TJ proteins due to elevated oxidative stress or inflammatory response result in downregulation of claudin-1, occludin, and ZO-1 expressions. This leads to an increase in intestinal permeability and destruction of the intestinal barrier, which are important pathogenetic factors in IBD. 44 Data of this present investigation suggest that intrarectal instillation of TNBS causes an increase in the production of ROS, resulting in decreased expression of claudin-1, occludin, and ZO-1 in TNBS control rats. However, these changes could be counteracted using piperine treatments, suggesting piperine’s protective role against intestinal barrier damage. In addition, ultrastructural analyses of colon tissue supported this notion as well, where the surfaces of the enterocytes in the mucosal epithelial layer were degraded and TJs gaps between two neighboring enterocytes were loosened in the TNBS control rats. In contrast, piperine administration reduced these aberrations.
Apoptosis has been suggested as an important pathway in the induction of mucosal erosion and ulceration in TNBS-induced UC. 45 The intrinsic pathway of mitochondria or extrinsic pathway of the death receptor may lead to apoptosis. Among various pathways of apoptosis, caspases play a major role in the apoptotic signal transduction and its activation leads to cell death. 46 Studies have reported that trauma induced in the colon from various agents such TNBS result in exaggerated levels of IL-1β, which causes activation of caspase-1. 47 Furthermore, Bax, a Bcl-2 protein family member, has the ability to release mitochondrial cytochrome c by increasing its membrane permeability. 27,48 Increased levels of cytochrome c further stimulate the activation of caspases, which, in turn, induce apoptosis. 49 Luo et al. showed that TNBS induces intestinal apoptosis in rats through activation of caspases pathway but the findings of our present investigation are inconsistent with this report. 31 Piperine has previously been documented to inhibit ATP-induced apoptosis via suppression of the caspase-1 signaling pathways in J774A.1 cells. 22 The present data are in agreement with this previous study as our present study also showed that administration of piperine significantly inhibited elevated levels of caspase-1 expression in TNBS-induced experimental colitis.
The decisive role of mucosal epithelial barrier integrity in intestinal function has been well reported. However, mediators such as inflammatory cytokines, apoptotic gene (caspases), and oxidative stress alter the structure of mucosal TJ proteins. 43,44 Histopathological and ultrastructural evaluation of colon tissue from TNBS control rats reveals infiltration inflammatory cell in colonic tissue, goblet cells hyperplasia, and the loosening intercellular gaps between adjacent epithelial cells. Piperine inhibits this release of mediator and colonic aberrations, which, in turn, restore the integrity of the mucosal epithelial barrier and decrease damage during colitis.
Currently, 5-ASA is implicated as an effective treatment strategy for the management of UC, but it is associated with several side effect. Additionally, due to its higher absorption in the upper gastrointestinal tract, it remains less effective in the local colonic region. 50 Nowadays, plant-based derived function food has received much success in the prevention and treatment of various maladies. Studies with isolated molecules from plant origins such as curcumin, HMPL-004 (Andrographis paniculate) omega-3 fatty acids, as well as herbs including aloe vera, Boswellia serrata, and Artemisia absinthium have proved their potential in the management of clinical IBD. 51 Piperine now also received much attention in the prevention and progression of cancer which may signify its importance in the future for the management of IBD.
Conclusion
The results of the present investigation demonstrated that piperine ameliorates progression of TNBS-induced colitis through modulation of the IκB-α/NF-κB signaling pathway, thus inhibiting the overexpression of proinflammatory cytokines (TNF-α and IL’s), COX-2, iNOs, and oxido-nitrosative stress along with apoptosis (caspase-1) that may improve expression of TJ protein (claudin-1, occludin, and ZO-1). Therefore, piperine as a functional food from an herbal origin may prove to be an alternative treatment strategy for the management of UC.
Footnotes
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was funded by the China postdoctoral Science Foundation (Guo Gang, No. 226536) and the Key Research and Development Program Foundation of Shaanxi Province (No. 2019KW-066 and No. 2019KW-067).
